-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
Top novinky
ReklamaSingle Strand Annealing Plays a Major Role in RecA-Independent Recombination between Repeated Sequences in the Radioresistant Bacterium
Deinococcus radiodurans is known for its exceptional ability to tolerate exposure to DNA damaging agents and, in particular, to very high doses of ionizing radiation. This exceptional radioresistance results from many features including efficient DNA double strand break repair. Here, we examine genome stability in D. radiodurans before and after exposure to ionizing radiation. Rearrangements between repeated sequences are a major source of genome instability and can be deleterious to the organism. Thus, we measured the frequency of recombination between direct repeats separated by intervening sequences of various lengths in the presence or absence of radiation-induced DNA double strand breaks. Strikingly, we showed that the frequency of deletions was as high in strains devoid of the RecA, RecF or RecO proteins as in wild type bacteria, suggesting a very efficient RecA-independent process able to generate genome rearrangements. Our results suggest that single strand annealing may play a major role in genome instability in the absence of homologous recombination.
Published in the journal: . PLoS Genet 11(10): e32767. doi:10.1371/journal.pgen.1005636
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1005636Summary
Deinococcus radiodurans is known for its exceptional ability to tolerate exposure to DNA damaging agents and, in particular, to very high doses of ionizing radiation. This exceptional radioresistance results from many features including efficient DNA double strand break repair. Here, we examine genome stability in D. radiodurans before and after exposure to ionizing radiation. Rearrangements between repeated sequences are a major source of genome instability and can be deleterious to the organism. Thus, we measured the frequency of recombination between direct repeats separated by intervening sequences of various lengths in the presence or absence of radiation-induced DNA double strand breaks. Strikingly, we showed that the frequency of deletions was as high in strains devoid of the RecA, RecF or RecO proteins as in wild type bacteria, suggesting a very efficient RecA-independent process able to generate genome rearrangements. Our results suggest that single strand annealing may play a major role in genome instability in the absence of homologous recombination.
Introduction
The extreme resistance of the bacterium D. radiodurans to DNA-fragmenting treatments, such as ionizing radiation or desiccation, is correlated with the ability to reconstruct a functional genome from hundreds of chromosomal fragments. The rapid reconstitution of an intact genome is thought to occur through an extended synthesis-dependent strand annealing process (ESDSA) followed by DNA recombination [1,2]. During ESDSA, chromosomal fragments with overlapping regions are used both as primers and templates for a massive synthesis of single-stranded DNA extensions. Newly synthesized complementary single stranded DNA extensions appear to anneal so that contiguous DNA fragments are joined together forming long linear intermediates. These intermediates require RecA-dependent homologous recombination to mature into reconstituted circular chromosomes representing DNA patchworks of numerous double-stranded DNA blocks synthesized before irradiation connected by DNA blocks synthesized after irradiation.
We have recently shown that the Deinococcal RecF, RecO, RecR proteins, by their ability to load RecA onto its single-stranded DNA substrate, play a crucial role in DNA double strand break repair via ESDSA and recombinational repair pathways [3]. Mutant ΔuvrD bacteria showed a markedly decreased radioresistance, an increased latent period in the kinetics of DNA double strand break repair and a slow rate of fragment assembly correlated with a slow rate of DNA synthesis, suggesting that UvrD helicase might be involved in the processing of double stranded DNA ends and/or in the DNA synthesis step of ESDSA [3].
More recently, it was proposed that a single strand annealing (SSA) process participates in an early step of DNA double strand break repair by facilitating the accurate assembly of small fragments to generate suitable substrates for subsequent ESDSA-promoted genome reconstitution [4]. The DdrB protein was previously shown to exhibit in vitro properties akin to those of SSB protein [5] but also to promote annealing of single stranded DNA [6]. The DdrB protein, recruited early after irradiation into the nucleoid, was also shown to be involved in the slow DNA double strand break repair observed in cells devoid of the RecA protein, and thus to play a major role in RecA-independent DNA double strand break repair through SSA [4,6].
Rearrangements between repeated sequences are a major source of genome instability and can be deleterious to the organism. These rearrangements can result in deletion or duplication of genetic material flanked by direct repeats. In Escherichia coli, recombination between directly repeated sequences occurs via both RecA-independent and RecA-dependent mechanisms, depending on the size of the repeats and of the intervening sequences between the repeated sequences [7–9]. Insertion of a sizable DNA sequence in between the repeated sequences substantially increased the RecA dependence, suggesting that increasing the distance separating the homologous regions preferentially inhibits the RecA-independent recombination in E. coli [9,10]. In E. coli, RecA-independent rearrangements between short repeats, such as deletions, are stimulated by mutations that affect the DNA polymerase or other proteins involved in DNA replication [11–13] leading to the proposal that these events occur during DNA replication by a mechanism involving mispairing of the newly synthetized DNA strand with an alternative complementary template sequence located nearby [7,12] (for review, see [14,15]). An alternate mechanism for RecA-independent deletion events involves DNA breakage, exonucleolytic erosion of the DNA ends and single strand annealing (SSA) of exposed complementary single stranded DNA [14]. A single strand annealing mechanism has also been proposed for RecA-independent deletions associated with a restart of collapsed replication forks [7,11,12,14,16]. Here, we measured the frequency of recombination between direct repeats separated by intervening sequences of various lengths restoring a functional tetA gene in the presence or absence of radiation-induced DNA double strand breaks in D. radiodurans. We also assessed the involvement of the RecA, RecO, RecF, UvrD and DdrB proteins in the deletion process. The role of these proteins in the progression of replication forks was also discussed.
Results
Efficient RecA-independent recombination between direct repeats in D. radiodurans
To investigate the role of recombination proteins in the occurrence of repeat-mediated deletion events in D. radiodurans, we constructed a mutated tetA allele bearing an internal duplication and a spc cassette inserted between the duplicated regions. The engineered tetA allele was inserted into the dispensable amyE locus of chromosome 1 and provided a recombination substate in which the direct repeats (438 bp long) were separated by a 1,479 bp spacer (Fig 1A). Precise deletion of one of the direct repeats and the intervening sequence restores the wild type tetA allele. The presence of a functional tetA gene on one copy of chromosome 1 suffices to confer tetracycline resistance to the cells, although D. radiodurans bacteria contain 4 to 10 genome equivalents per cell. In contrast, when the deletion of a gene generates a loss-of-function mutant, all the copies of the gene must be eliminated to detect the mutant phenotype, and failure to obtain a homozygote provides a first indication that the gene might encode a function essential for cell viability [3,17].
Fig. 1. Deletion frequency between repeated sequences depends on the genetic context. 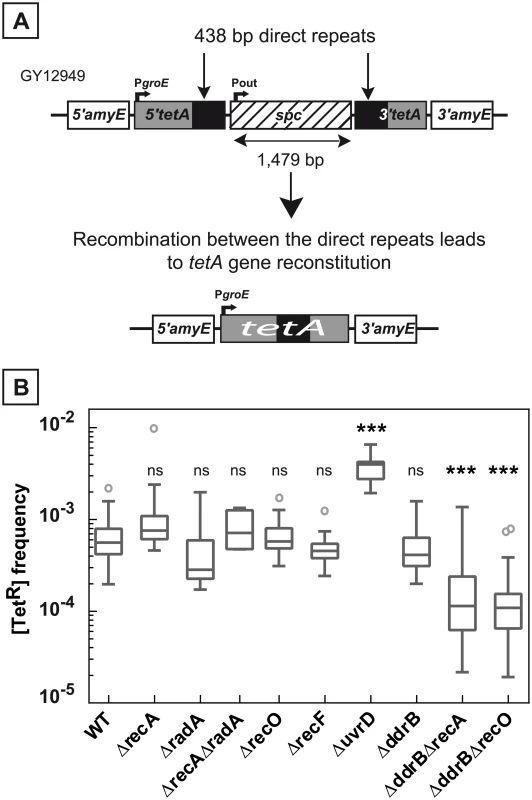
A. Schematic representation of the genetic recombination assay. The 5’ and 3’ parts of the tetA gene, containing 438 bp repeats (black boxes) separated by an intervening sequence of 1,479 bp encoding a spc gene conferring resistance to spectinomycin, were introduced into the dispensable amyE gene locus. Recombination between the repeats leads to the tetA gene reconstitution conferring tetracycline resistance to bacteria. B. Medians of [TetR] frequencies in WT (GY12949), ΔrecA (GY15184), ΔradA (GY12956), ΔrecAΔradA (GY12971), ΔrecO (GY12963), ΔrecF (GY12955), ΔuvrD (GY12953), ΔddrB (GY16016), ΔddrB ΔrecA (GY16628), and ΔddrB ΔrecO (GY16638) mutant strains are represented by Tukey box plots. Outliers are represented by open circles. Medians were calculated from 12 to 53 independent values (except for medians calculated from at least 4 independent values in ΔradA and ΔrecA ΔradA bacteria). Statistically significant differences in the medians of recombination frequencies of the mutants, compared to those observed in strain GY12949, were calculated using the non-parametric Dunn's multiple comparison test: * P < 0.05; ** P < 0.01; *** P < 0.001; ns if P > 0.05. The frequency of spontaneous deletion events in a population was estimated by measuring the frequency of [TetR] bacteria. As shown in Fig 1B, the median of the frequencies of [TetR] in the wild type bacteria was 6.5x10-4 and did not decrease in ΔrecA bacteria (median value: 8x10-4). Does the apparent RecA-independent high frequency of deletion events result from a functional redondancy of RecA activities in the cells? We tested the involvement of the RadA protein, a RecA-related protein, and showed that wild type frequencies of deletions were found in ΔradA and in ΔrecA ΔradA bacteria (Fig 1B), suggesting that the RadA protein was not required in RecA-independent recombination to compensate for the absence of the RecA protein. We also observed that the frequency of [TetR] bacteria was not reduced in cells devoid of RecF or RecO proteins required for loading RecA onto its single-stranded DNA substrate (Fig 1B). Altogether, our results suggest an important contribution of RecA-independent mechanisms in the generation of deletions between repeated DNA sequences in D. radiodurans.
To test whether the same 438 bp homologous fragments can undergo efficient RecA-dependent recombination, we constructed a plasmid-by-chromosome recombination assay in which the recombining tetA fragments were placed in a different configuration, one being located at the chromosomal amyE locus and the other on a resident plasmid (Fig 2A). In this assay, the reconstitution of a functional tetA gene resulted from the integration of the plasmid into the chromosomal DNA as verified by PCR analysis of few [TetR] colonies (S1 Fig). The integration event is well tolerated by the cell, since we used a low copy number plasmid p15002, a derivative of plasmid pI8 maintained at 4 to 10 copies per cell in D. radiodurans [18]. As can be seen in Fig 2B, the frequency of [TetR] bacteria dropped from a median value of 2x10-5 in the wild type to less than 5 x 10−7 in cells devoid of the RecA or the RecF proteins, indicating that the 438 bp homologous fragments recombine through a classical RecA-promoted strand exchange mechanism. In contrast, loss of the RadA protein did not impair recombination efficiency. In E. coli, the RadA protein has been involved in processing of branched recombination intermediates. However single radA mutants have a modest effect on recombination and DNA survival while they show a strong synergistic effect in combination with mutations in the recG or the ruvAB Holliday junction proteins [19,20].
Fig. 2. Recombination between chromosomal and plasmid DNA is RecA- and RecF-dependent. 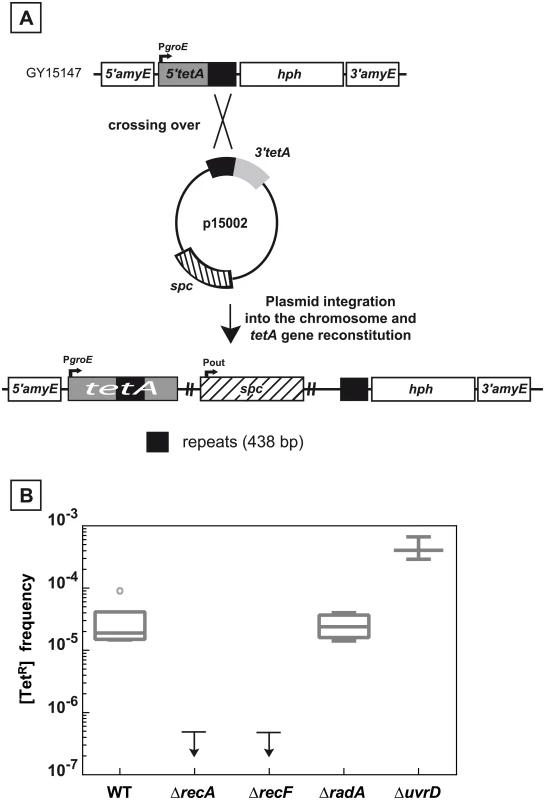
A. Schematic representation of the recombination assay between chromosomal and plasmid DNA. The 5’tetA and the 3’tetA regions of the tetA gene, containing 438 bp repeats, were introduced into the chromosomal dispensable amyE gene and into the p11554 plasmid giving rise to plasmid p15002, respectively. One crossing over between the two 438 bp repeated sequences (black boxes) leads to the reconstitution of a functional tetA gene and the integration of the plasmid into chromosomal DNA. B. Medians of [TetR] frequencies in WT (GY15147), ΔrecA (GY15158), ΔrecF (GY15160), ΔradA (GY15149), and ΔuvrD (GY15156) bacteria, all containing the p15002 plasmid, are calculated from at least 3 independent values and represented by Tukey boxplots. Outliers are represented by open circles. The small arrows attached to the horizontal line representing the upper limit of detectable [TetR] frequencies indicate that [TetR] frequencies were < 5 10−7 for ΔrecA and ΔrecF bacteria. Differences between the DNA repeats reduces the frequency of the deletion events
The presence of mutations in one of the 438 bp repeats (Fig 3A) reduced the frequency of the deletion events between the chromosomal repeats. A single mutation sufficed to significantly decrease the frequency of the deletion events as shown by median values that decreased by a factor of 4.8 in recA+ bacteria and 4.6 in ΔrecA bacteria, when compared to fully homologous repeats. The decrease was greater when 3 mutations were present in one of the repeats, yielding reduction factors of 10.0 and 19.0 in recA+ and ΔrecA bacteria, respectively (Fig 3B). A plot of the frequency of [TetR] bacteria as a function of the number of differences shows a linear decrease in the deletion frequency with similar regression slopes in recA+ and ΔrecA bacteria (S2 Fig).
Fig. 3. Effect of base pair changes in the repeats on their recombination frequencies. 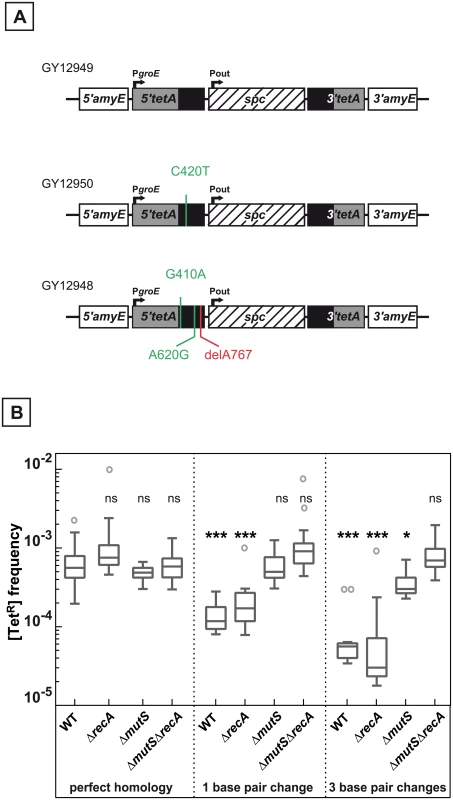
A. Schematic representation of the recombination substrates containing 1 or 3 bp changes. The positions of the base pair changes calculated from the initiation codon in the 5’tetA region are indicated. B. Medians of [TetR] frequencies calculated from 14 to 43 independent values in WT (GY12949), ΔrecA (GY15184), ΔmutS (GY12978), ΔmutS ΔrecA (GY16620) bacteria containing identical repeated sequences, WT (GY12950), ΔrecA (GY16624), ΔmutS (GY12980), ΔmutS ΔrecA (GY16618) containing one base difference in the repeated sequences, and WT (GY12948), ΔrecA (GY16622), ΔmutS (GY12979), ΔmutS ΔrecA (GY16616) containing 3 base differences in the repeated sequences, are represented by Tukey boxplots. Outliers are represented by open circles. Statistically significant differences in the medians of recombination frequencies of the mutants and WT containing sequence differences in the repeated sequence compared to GY12949 were calculated using the non-parametric Dunn's multiple comparison test: * P < 0.05; ** P < 0.01; *** P < 0.001; ns if P > 0.05. A similar analysis was performed in recA+ ΔmutS and ΔrecA ΔmutS bacteria devoid of the MutS protein, the key enzyme involved in mismatch recognition. The data (Fig 3B and S2 Fig) show that in this case the differences between the repeats did not significantly affect the deletion frequency. These results suggest that, as in homologous recombination intermediates, a heteroduplex DNA is formed during RecA-independent processes leading to the reconstitution of a functional tet gene and that an efficient mismatch repair aborts recombination between the DNA repeats in ΔrecA as well as in recA+ bacteria.
Absence of UvrD increases the frequency of deletions between the repeats
It was previously shown that uvrD mutations stimulate RecA-dependent recombination [21–23] and enhance tandem repeat deletions in E. coli [23]. Here, we show that the absence of UvrD enhanced the efficiency of RecA-dependent recombination between chromosomal and plasmid DNA by a factor of 21.2 (Fig 2B), suggesting that deinococcal UvrD protein possesses an anti-RecA activity as previously shown for the E. coli UvrD protein [24,25]. The absence of UvrD also enhanced the frequency of deletions between the chromosomal direct repeats by a factor of 7.1 (Fig 1B). This increase may be due to the anti-RecA activity of UvrD protein that can possibly inhibit RecA-dependent recombination between the repeated sequences in a recA+ uvrD+ background. However, the absence of UvrD might also disturb DNA replication, and thus increase genome instability. A clue to understand how the absence of the UvrD protein might be involved, independently of its anti-RecA activity, in the stimulation of deletion events requires an analysis of its effects in a recombination-deficient background. Unfortunately, we were unable to obtain homozygotes for recA, recF or recO deletion in combination with a uvrD deletion, even after extensive purification steps (S3 Fig), suggesting that uvrD deletion is colethal with a recA, recF or recO deletion. We propose that the UvrD protein, by displacing obstacles downstream of the replisome, plays an important role in the progression of replication forks (see discussion).
Loss of viability of ΔddrB ΔrecA and ΔddrB ΔrecO double mutants
Previous in vitro and in vivo results suggest that the DdrB protein plays a major role in a single strand annealing process (SSA) that operates early in genome reconstitution after DNA damage [4,6]. Single strand annealing is the only activity of DdrB known besides binding to single strand DNA [5,6]. Thus, to analyse the involvement of SSA in generating deletions via a RecA-independent pathway, we decided to construct double mutants devoid of DdrB and RecA or RecO proteins. Homogenotization of ΔddrB ΔrecA and ΔddrB ΔrecO double mutants was difficult, requiring 7 steps of purification, suggesting growth inhibition of the mutated cells (S3 Fig). Thus, we compared the growth rate and the plating efficiency of the double mutants with those of the single ΔddrB, ΔrecA and ΔrecO mutants and of the parental wild type strain. Wild type and ΔddrB bacteria exhibited a generation time of 105 min. The recombination deficient bacteria grew more slowly, as ΔrecA and ΔrecO bacteria during exponential growth showed a generation time of 285 min whereas ΔddrB ΔrecO and ΔddrB ΔrecA exhibited a generation time of 370 min. Moreover, during exponential growth phase, the single ΔrecA and ΔrecO mutants had a 15 fold decreased plating efficiency as compared with the wild type, whereas the ΔddrB ΔrecA and ΔddrB ΔrecO double mutants had a 35 fold decreased plating efficiency (Fig 4). These results suggest that the DdrB protein may be involved in management of blocked replication forks in the absence of the RecA or RecO proteins. Another striking result was the increased lethality of the recombination deficient mutants in late stationary phase. Indeed, after reaching a plateau after 6 hours of incubation for the wild type and ΔddrB bacteria, the number of CFU did not decrease during 70 additional hours of incubation. In contrast, the single ΔrecA and ΔrecO recombination deficient mutants and the double ΔddrB ΔrecA and ΔddrB ΔrecO mutants reached a plateau after 18 to 20 hours of incubation and the number of CFU decreased 2–3 orders of magnitude after 30 hours of incubation (Fig 4) suggesting that DNA lesions are generated during prolonged stationary phase and require recombination functions to be repaired.
Fig. 4. Impaired growth and stationary-phase lethality of recombination-deficient mutant cells. 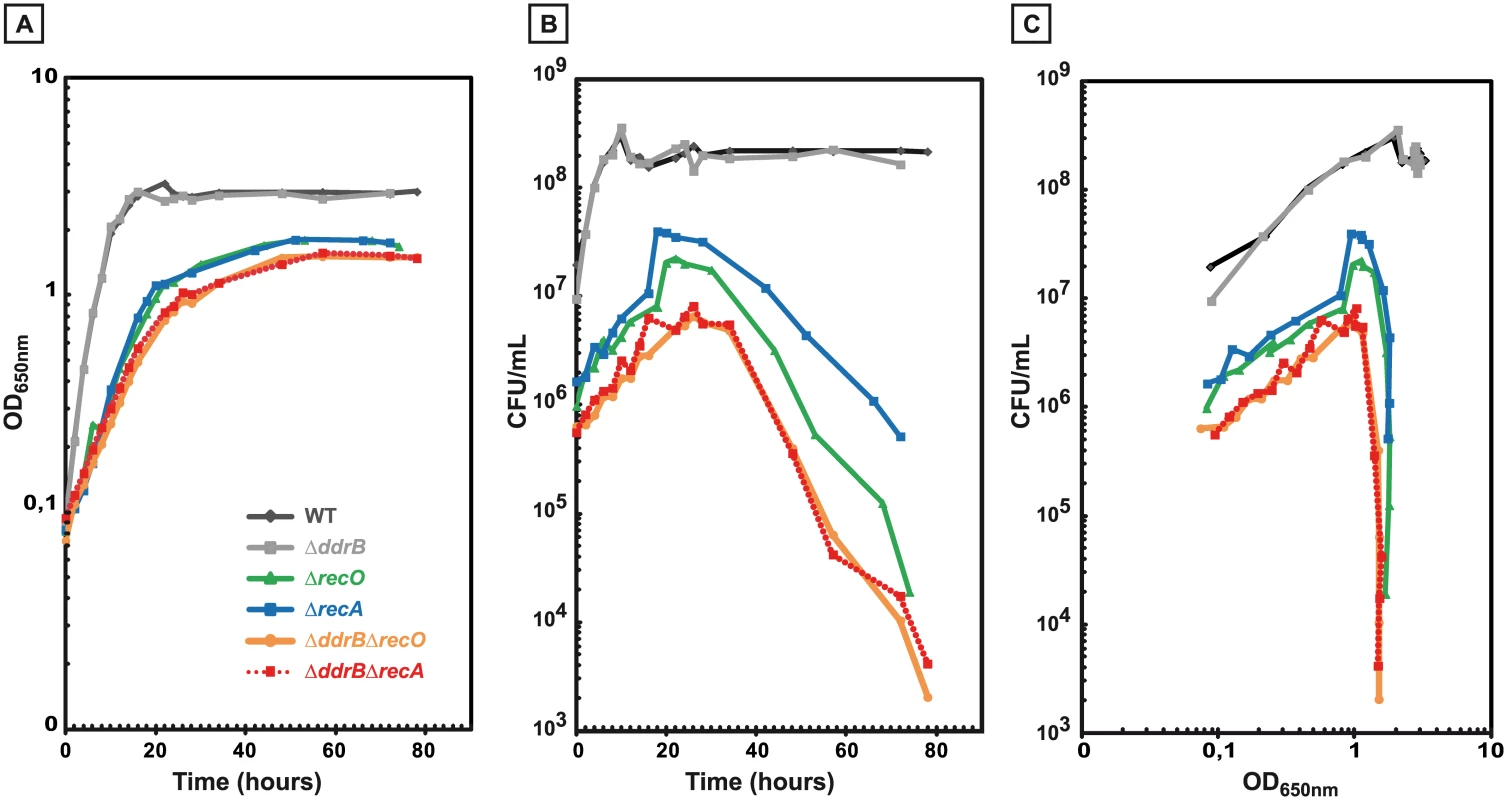
GY9613 (WT) (black diamonds), GY13915 (ΔddrB) (grey squares), GY15125 (ΔrecO) (green triangles), GY12968 (ΔrecA) (blue squares), GY16626 (ΔddrB ΔrecA) (red squares and interrupted lines), GY16636 (ΔddrB ΔrecO) (orange circles) were grown from independent colonies at 30°C to an OD650nm = 0.1 (time 0 of the growth curves). A. OD650nm as a function of time. B. Colony forming units as a function of time. C. Colony forming units as a function of OD650nm. An important role of DdrB in the recombination process between repeated sequences in the absence of RecA or RecO proteins
We found that the absence of DdrB had a strong negative effect on the frequencies of deletions events between the chromosomal repeats generated via a RecA-independent pathway. Indeed, when the repeats are separated by 1,479 bp, the median values of the deletion frequencies in ΔddrB ΔrecA and ΔddrB ΔrecO bacteria decreased by a factor of 4.9 and 5.1 respectively, as compared to their ΔrecA and ΔrecO counterparts (Fig 1B). These results indicate that 80% of the [TetR] bacteria generated in the absence of RecA or RecO proteins were formed in a DdrB-dependent manner, suggesting a major role of single strand annealing in RecA-independent recombination between the direct repeats. We can also hypothesize that the DdrB protein might be involved in the stabilization of DNA polymerase template switching intermediates.
The frequency of deletion events does not decrease when the length of the spacers between the repeated sequences was as large as 10,500 bp
In E. coli, in which both RecA-dependent and RecA-independent mechanisms can contribute to recombination between direct repeats, deletion events become increasingly RecA-dependent as the distance between the repeated sequences increases [9]. To verify if this also applies in D. radiodurans, we modified the test deletion construct shown in Fig 1 by replacing the 1,479 bp spacer with sequences of increasing length to analyse the impact of the distance between the repeats on the incidence of deletions and their genetic control (Fig 5A).
Fig. 5. Deletion frequencies between repeated sequences separated by 3,500, 6,500, and 10,500 bp. 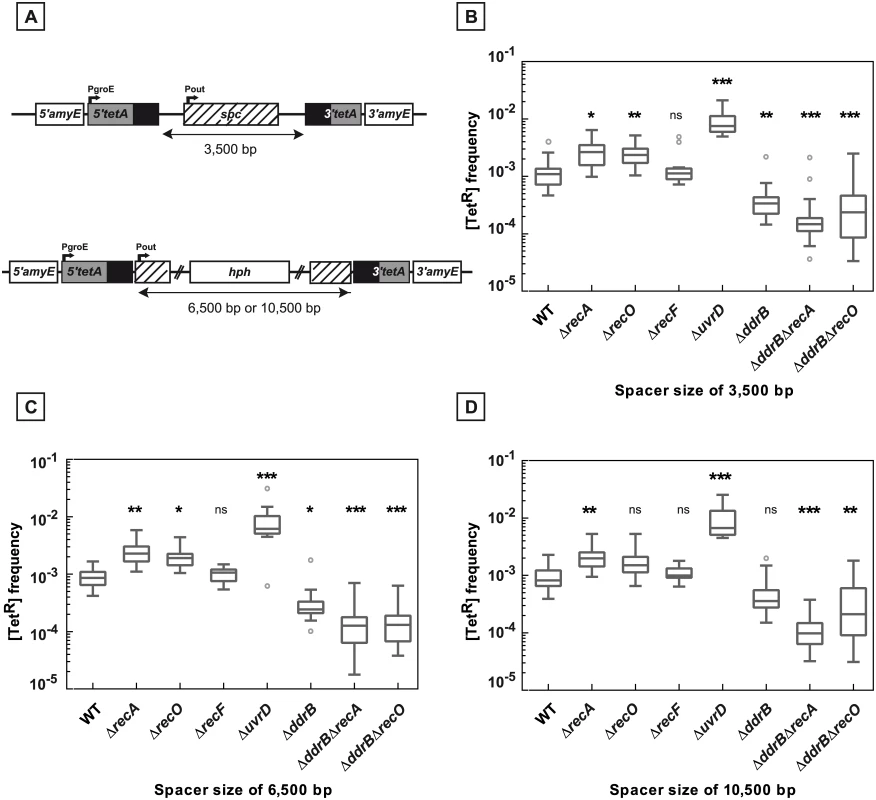
A. Schematic representation of the constructions used. B., C., D. Bacteria contain 3,500 bp (panel B), 6,500 bp (panel C), and 10,500 bp (panel D) intervening sequences between the tetA repeats. The medians of [TetR] frequencies calculated from 10 to 35 independent values in the tested strains are represented by Tukey boxplots. Outliers are represented by open circles. Statistically significant differences in the medians of recombination frequencies of the mutants compared to the WT GY16209, GY16227, and GY16235 in panel B, C, and D, respectively, were calculated using the non-parametric Dunn's multiple comparison test: * P < 0.05; ** P < 0.01; *** P < 0.001; (ns) if P > 0.05. B. WT (GY16209), ΔrecA (GY16238), ΔrecO (GY16262), ΔrecF (GY16264), ΔuvrD (GY16608), ΔddrB (GY16268), ΔddrB ΔrecA (GY16630), ΔddrB ΔrecO (GY16640) C. WT (GY16227), ΔrecA (GY16244), ΔrecO (GY16278), ΔrecF (GY16276), ΔuvrD (GY16612), ΔddrB (GY16282), ΔddrB ΔrecA (GY16632), ΔddrB ΔrecO (GY16642) D. WT (GY16235), ΔrecA (GY16252), ΔrecO (GY16290), ΔrecF (GY16292), ΔuvrD (GY16614), ΔddrB (GY16296), ΔddrB ΔrecA (GY16634), ΔddrB ΔrecO (GY16644). We found that the increase of the distance between the repeats from 1,479 bp (Fig 1A) up to 10,500 bp (Fig 5A) had no effect on the deletion frequency in recA+ as well as in ΔrecA or ΔrecF hosts (compare the [TetR] frequencies in Figs 1B, 5B, 5C and 5D). Likewise, the distance between the repeats had no effect on the stimulation of deletion events by the absence of the UvrD protein (Figs 1B, 5B, 5C and 5D).
In contrast, the involvement of DdrB in the deletion events became more apparent when the distance between the repeats increased. Indeed, while a DdrB deficiency had no effect on the frequency of deletions in a recA+ background when the spacer between the repeats was 1,479 bp long, it produced a 2 to 3-fold decrease in the deletion frequency when the spacer length increased (Figs 1B, 5B, 5C and 5D). When the ddrB deletion was associated with a recA deletion, the reduction factors were found to be between 18 - and 20-fold if the length of the intervening sequences was ≥ 3,500bp as compared to their single ΔrecA counterparts (Fig 5B, 5C and 5D). A similar effect of a ddrB deletion was also observed in cells devoid of the RecO protein (Fig 5B, 5C and 5D). These results suggest that, in the absence of the RecA-promoted homologous recombination, approximately 95% of the recombination events were dependent on the DdrB protein and may be related to an SSA pathway.
Induction of recombination between direct repeats by ionizing radiation
We used our deletion assay to analyze the impact of the presence of repeated sequence on the stability of the genome in γ-irradiated cells during the process of genome reconstitution. We showed that the frequency of repeat-induced deletions restoring a functional tetA gene increased as a function of the dose of γ-irradiation used (Fig 6A). Thus, we further exposed the cells to 5 kGy γ-irradiation, a dose producing hundreds of DNA double strand breaks [26]. The repeats in the tested cells were separated either by 1,479 bp (“short” spacer), 3,500 bp, 6,500 bp, or by 10,500 bp (“long” spacer) sequences. The deletion analysis was performed only in a recA+ background, since the extreme radio-sensitivity of ΔrecA or ΔrecF bacteria (cell survival was less than 10−5 after exposure to 5 kGy) precluded the inclusion of these cells in the genetic assay. The deletions were induced by exposure to γ-irradiation independently of the length of the spacers (Fig 6B).
Fig. 6. γ-irradiation induced recombination between repeated sequences. 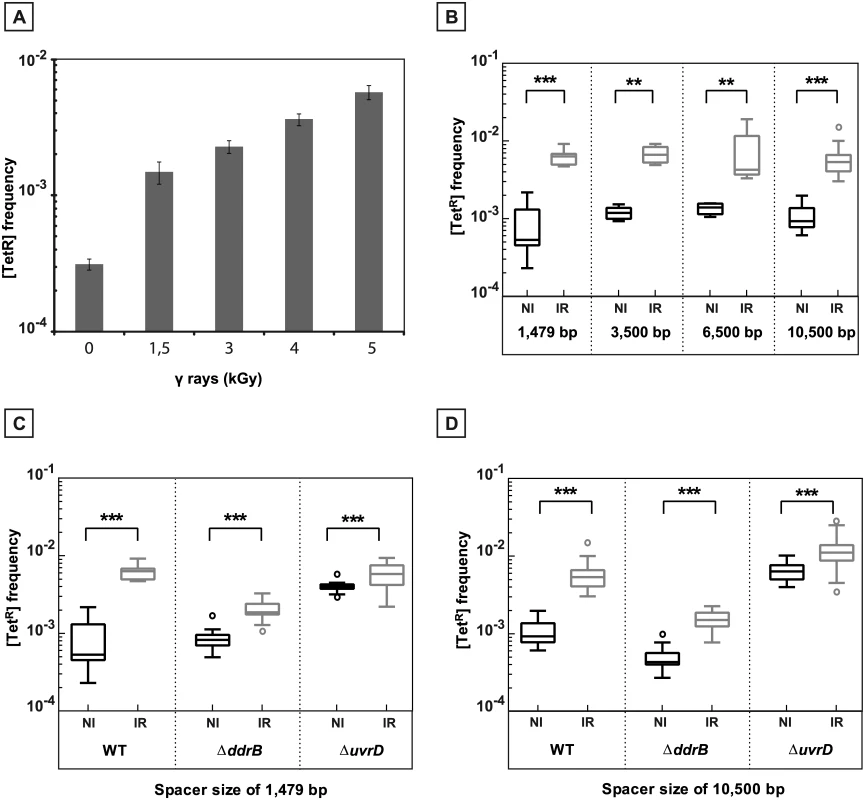
A. Induction of recombination between repeated sequences separated by 1,479 bp as a function of the radiation dose. [TetR] frequencies were measured in at least 5 independent cultures after 20 hours of post irradiation incubation of GY12949 in TGY medium after exposure to different γ-irradiation doses. B. Induction by exposure to γ-irradiation of recombination between repeats separated by intervening sequences of increasing length: 1,479 bp (GY12949), 3,500 bp (GY16209), 6,500 bp (GY16227) and 10,500 bp (GY16235). C. γ-promoted induction of recombination between overlapping sequences separated by 1,479 bp in WT (GY12949), ΔddrB (GY16016), and ΔuvrD (GY12953) bacteria. D. γ-promoted induction of recombination between overlapping sequences separated by 10,500 bp in WT (GY16235), ΔddrB(GY16296), ΔuvrD(GY16614). B., C., D. Medians of the [TetR] frequencies calculated from 5 to 30 independent values are represented by Tukey boxplots. Outliers were represented by open circles. Statistically significant differences in the medians of recombination frequencies between irradiated and the corresponding non-irradiated bacteria were calculated using the non-parametric Mann-Withney test: * P < 0.05; ** P < 0.01; *** P < 0.001; (ns) if P> 0.05. NI: non-irradiated bacteria. IR: irradiated bacteria. As shown in Fig 6, in the wild type bacteria, the deletion frequency increased by 11.9 fold and 5.9 fold after irradiation when the repeats are separated by the”short” spacer and the “long” spacer, respectively (compare left panels in Fig 6C and 6D). In cells devoid of the UvrD protein, the frequency of deletions moderately increased after irradiation (induction factors of 1.45 and 1.7 for the “short” and “long” spacer, respectively), likely because these cells already have an elevated spontaneous level of recombination in the absence of irradiation (compare right panels in Fig 6C and 6D). In contrast, cells devoid of the DdrB protein showed a marked reduction in the induced levels of deletion events (compare middle panels in Fig 6C and 6D), suggesting that single strand annealing might play an important role in the generation of the [TetR] recombinants during genome reconstitution after irradiation.
Discussion
Repeated sequences are targeted by recombination processes leading to amplifications, deletions, and other rearrangements of the genetic material. These events play an important role in genome plasticity and rapid adaptation to environmental challenges, but are also potential source of genome instability and can be deleterious to an organism (see for review [27]). D. radiodurans contains an enhanced number of repetitive sequences as compared to other bacteria, including insertion sequences, small non-coding repeats, and intragenic repeats (see for review [28]). This bacterium is also known for its capacity to reconstitute an entire genome from a myriad of fragments after exposure to elevated γ-irradiation doses. Genome reconstitution occurs through extended synthesis dependent single strand annealing (ESDSA) followed by classical homologous recombination. In heavily irradiated cells, a RecA-independent single strand annealing (SSA) process takes place before ESDSA allowing the assembly of small fragments into substrates that can be further processed through ESDSA. The single strand annealing activity of the DdrB protein plays a major role in this early step of DNA double strand break repair [4].
Here, we show that DdrB also plays a key role in RecA-independent recombination between direct repeats, leading to the occurrence of deletions, and substantially contributes to the induction of deletions in γ-irradiated recA+ bacteria. Our results points to an important contribution of a DdrB-dependent pathway in D. radiodurans genome plasticity.
Efficient mechanisms ensure recombination between chromosomal direct repeats in D. radiodurans cells devoid of the RecA protein
Here, we determined the frequencies of spontaneous and radiation-induced recombination between chromosomal direct repeats and investigated the role of RecA and of other key recombination and repair proteins in the occurrence of these events in D. radiodurans. We found that recombination events restoring a functional tetA gene occurred at a very high frequency. In a recA+ background, the median frequency of [TetR] cells was equal to 6.5 x 10−4 (Figs 1, 5B, 5C and 5D), values more than 10-fold higher than those measured in a study that used a similar substrate (chromosomal 358 bp direct repeats separated by an intervening sequence of 850 bp) to measure recombination in Helicobacter pylori, a bacterium known for its high recombination proficiency [29]. Moreover, inactivation of recA resulted in a 10-fold decrease in recombination between the direct repeats in H. pylori [29]. In contrast, introduction of a ΔrecA, ΔrecF or ΔrecO mutation in the D. radiodurans tester strains did not change the elevated recombination frequencies between the repeats (Figs 1, 5B, 5C and 5D). In E. coli and in B. subtilis, the distance between the repeats plays a key role in determining the mechanisms involved in the recombination processes, the efficiency of RecA-independent recombination decreasing sharply when the distance between the repeats increases [9, 10, 30]. No such proximity effect was observed in D. radiodurans. Indeed, the frequency of appearance of the recombinants in our assay remained elevated in ΔrecA (and ΔrecF or ΔrecO) bacteria as in the parental rec+ bacteria when the spacer between the repeats increased from 1,479 up to 10,500 bp (Fig 5).
Our results suggest that, in the absence of RecA (or in the absence of “facilitator” proteins required for loading RecA onto its single-stranded DNA substrate), alternate pathways ensure recombination between repeated sequences in D. radiodurans. These RecA-independent pathways do not necessarily predominate in recA+ bacteria, although we observed a similar frequency of recombinants in recA+ and ΔrecA bacteria. Indeed, ΔrecA (or ΔrecF or ΔrecO) bacteria had a low plating efficiency with less than 10% of cells able to form colonies.
Does replication slippage account for RecA-independent recombination between chromosomal direct repeats?
In E. coli, mutations in components of the DNA Pol III holoenzyme result in elevated levels of tandem repeat rearrangements, supporting the idea that RecA-independent recombination occurs during the process of chromosome replication [14]. A replication slipped misalignment model [14] proposed that a pause in DNA synthesis and dissociation of the polymerase from its template allows the nascent strand to translocate to a new pairing position. Slipped misalignment is thought to occur on single-stranded DNA and thus more frequently during lagging-strand synthesis [14]. The availability of single-stranded DNA on the lagging-strand template, and thus, the length of the Okazaki fragments, might constitute parameters that govern the efficiency of the deletion events and might explain the strong dependence of the deletion frequencies on the proximity of the repeated sequences [14]. Our findings that the distance between the repeats in the 1–11 kb range has no influence on the deletion frequency raise the question as to whether these events were generated through a slipped misalignment mechanism, thus implying the presence of very large single stranded DNA regions on the lagging strand template in D. radiodurans. A response to this question awaits better knowledge of the replication machinery in D. radiodurans and a determination of the average size of Okazaki fragments in this bacterium.
Involvement of single strand annealing in RecA-independent recombination between chromosomal direct repeats
We found that the frequencies of the deletion events in ΔrecA ΔddrB (or ΔrecO ΔddrB) bacteria were reduced 5-fold and 18 - to 20-fold as compared with those measured in the single ΔrecA (or ΔrecO) mutant counterparts (Figs 1B and 5) in strains containing repeats separated by 1479 bp and 3,500 to 10,500 bp, respectively. These results strongly suggest that the DdrB protein strongly stimulates RecA-independent recombination in D. radiodurans. The DdrB protein was shown to bind single stranded DNA [5] and to mediate in vitro fast annealing of complementary oligomers [6]. In vivo, the single strand annealing activity of DdrB is supported by its involvement in plasmid establishment during natural transformation [4]. Thus, we propose that RecA-independent recombination between direct repeats occurs mainly through a DdrB-dependent single strand annealing (SSA) pathway. SSA was first proposed to explain circularization of linear duplex phage DNA containing terminal repetitions by annealing complementary terminal single overhangs [31]. The SSA model was postulated later to take place in eukaryotic cells [32,33] where it is facilitated by RPA and RAD52 in a RAD51-independent manner [34,35]. SSA involves an initial DNA double strand break in the sequence between the duplications followed by the action of a 5’ to 3’ exonuclease to expose single stranded regions in both repeats that are subsequently aligned and annealed by the RAD52-RPA-ssDNA ternary complex. Annealed intermediates are then processed by digestion of the displaced single stranded DNA, polymerase filling-in and ligation to generate the final recombination product (For review, see [36]).
Although DdrB does not share sequence similarity with the eukaryotic RAD52 protein, it might act as its functional equivalent [37,38]. The activity of the RAD52 protein is strongly stimulated by the presence of RPA [39,40]. In contrast, the single strand annealing activity of DdrB is not stimulated but rather inhibited by inclusion of the SSB protein in the in vitro annealing reaction [4,6]. The deinococcal SSB protein is an essential protein and the DdrB protein is unable, even when overexpressed, to replace SSB for cell viability [41]. SSB is crucial for all aspects of DNA metabolism [42] while DdrB seems to have a more specialized role in DNA repair and plasmid transformation by stimulating the SSA pathway. Both DdrB and SSB bind to 3’ single stranded tails of resecting ends [5]. The ends can be engaged in two alternative pathways: annealing to complementary ssDNA in the SSA pathway, or, depending on the formation of a RecA nucleofilament, invasion of homologous dsDNA to promote strand exchange in homologous recombination or to prime DNA synthesis in ESDSA. Polymerization of RecA on ssDNA requires the displacement of SSB or DdrB from ssDNA. The SSB protein can be efficiently displaced through the action of RecO and RecR proteins [43,44]. DdrB protein binds more tightly than SSB to ssDNA [5] and might be displaced with more difficulty from ssDNA. We propose that, in D. radiodurans, homologous recombination and SSA might also compete for substrate in making deletions between direct repeats. Reams et al. [45] proposed that, in Salmonella enterica, a single-strand annealing pathway might also be activated to generate duplication between tandem copies of the ribosomal RNA genes (rrn) when two single-stranded DNA ends are provided and neither strand is coated with inhibitory RecA protein. Under these conditions, the activation of single-strand annealing might compensate the loss of homologous recombination [45].
A single strand annealing mechanism is involved in the rescue of stalled replication fork in ΔrecA bacteria
We found that ΔrecA (or ΔrecO) bacteria had a 15 fold decreased plating efficiency as compared with the wild type during exponential growth phase (Fig 4). This important lethal sectoring suggests that problems resulting in the arrest of replication fork (see for review [46]) occur at high frequency in D. radiodurans, and that RecA-mediated recombination plays a key role in the recovery of stalled replication forks in this bacterium. A further (2-fold) decreased plating efficiency was observed when a DdrB deficiency was combined with a RecA (or RecO) deficiency (Fig 4). These results are consistent with a single strand annealing model but also with any model that envisions annealing of complementary DNA strands, for example misannealing of the direct repeats during recovery from replication fork collapse in cells devoid of the RecA protein. D. radiodurans also seems to be very sensitive to prolonged stationary phase, with a rapid loss of cell viability when proteins involved in homologous recombination were absent, suggesting that DNA double strand breaks were generated in “old” cells and could not be repaired in the absence of RecA or RecO proteins.
Absence of UvrD increases the frequency of recombination between the repeats
Another important feature was the increased recombination frequency between repeated sequences measured in a D. radiodurans mutant devoid of the UvrD protein at a level almost equivalent to those measured after irradiation of wild type bacteria (Fig 6). It was previously shown that uvrD mutations enhance tandem repeat deletion in the E. coli chromosome [23,47] and stimulate RecA-dependent recombination [21,48,49]. In E. coli, mutations in uvrD induce the SOS response, a common phenotype in cells with replication defects [50]. The obstacles possibly encountered by replication forks during their progression are multiple, such as tightly bound proteins, nicks or DNA lesions. The Rep helicase acts by dislodging proteins in front of replication forks [51–53] and its absence results in a marked slowing down of replication progression [54], suggesting increased fork arrest. Simultaneous inactivation of Rep and UvrD helicases is lethal in E. coli [23] suggesting that UvrD might partially substitute for the Rep protein in ensuring replication progression [55]. In favour of this hypothesis, it was recently shown that UvrD displaces the obstacles downstream of the replisome in vitro [52] and plays a major role to displace transcription complexes [56]. Moreover, it was proposed that UvrD acts at blocked replication forks by clearing RecA, facilitating replication fork reversal [57, 58], a hypothesis supported by the ability of UvrD to directly remove RecA nucleoprotein filaments in vitro [25].
In D. radiodurans, we previously showed that inactivation of uvrD results in a marked slowing down of replication progression in un-irradiated cells [3]. During post-irradiation recovery through ESDSA, the absence of UvrD results in a delayed kinetics of DNA double strand break repair that coincided with delayed and less extensive DNA synthesis than that observed in the wild type cells [3]. D. radiodurans bacteria are naturally devoid of the RecB and RecC proteins, and it was suggested that UvrD, in association with the RecJ exonuclease, might play an important role in the processing of DNA double strand ends required for priming of DNA synthesis, but also may act in the DNA synthesis elongation step of ESDSA and more generally may play an important role for the progression of replication forks [3]. It is important to notice that we were unable to obtain mutants devoid of the RecJ protein [3], and recJ mutants constructed by Hua and his collaborators were shown to grow very slowly and to be thermosensitive [59].
In D. radiodurans, we were unable to delete the recA gene when bacteria were devoid of the UvrD protein, suggesting colethality of uvrD and recA deficiencies. These results are reminiscent of phenotypes observed in particular rad3 mutants of Saccharomyces cerevisiae. The RAD3 gene, a homolog of the human gene XPD, encodes a helicase which is a component of the NER apparatus as part of the transcription factor TFIIH. Interestingly rad3-101 and rad3-102 mutants accumulate DNA double strand breaks and are lethal when in combination with mutations in recombinational repair genes, strongly suggesting that Rad3 protein influences either the generation of DNA double strand breaks or their processing by homologous recombination [60].
We propose that the absence of UvrD in D. radiodurans may disturb the progression of the replication fork, and thus might, as RAD3 in S. cerevisiae, influence the generation of DNA double strand break, favouring recombination and also single strand annealing between DNA repeats.
Induction of recombination between direct repeats by ionizing radiation
We used our assay to analyze the impact of the presence of repeated sequence on the stability of the genome in γ-irradiated cells. We found that exposure to a dose of 5 kGy γ-irradiation increased the recombination level about 10-fold in the wild type but to a lesser extent in cells devoid of the DdrB protein, suggesting that SSA might play an important role in recombination between the duplicated sequences during the process of genome reconstitution.
In D. radiodurans, interplasmidic recombination between homologous regions was previously shown to be induced by exposure to γ-radiation [61]. Moreover, when two TetS alleles were inserted on the same chromosome into two randomly distant sites, 2% of TetR bacteria were found among the surviving cells exposed to 17.5 kGy, whereas TetR isolates were only very rarely found without irradiation [62]. Interestingly, when two slightly different E. coli plasmids were inserted in the D.radiodurans genome generating adjacent duplication insertions, circular derivatives of the tandemly integrated plasmids were formed in the first 1.5 h postirradiation before the onset of recA-dependent repair in cells exposed to 17.5 kGy γ-irradiation. These circular derivatives had structures consistent with the hypothesis that DNA repair occurred immediately postirradiation by a recA-independent single strand annealing process [63]. These authors proposed that SSA may be a preparatory step for further DNA repair in wild-type D. radiodurans, a hypothesis in accordance with our recent results, suggesting that DdrB-dependent single-strand annealing might facilitate the assembly of the myriad of small fragments generated by extreme radiation exposure to generate suitable substrates for subsequent ESDSA-promoted genome reconstitution [4]. Genome reassembly in irradiated D. radiodurans cells was considered for a long time as an error-free process since no genome rearrangements were detected after post-irradiation DNA repair. Gross chromosomal rearrangements were detected for the first time in recA+ D. radiodurans cells exposed to extremely high γ-doses (25 kGy) and in recA mutant cells that survived 5 kGy γ-radiation [64]. The recA mutants were also shown to be prone to spontaneous DNA rearrangements during normal exponential growth [64]. These authors presumed that SSA, by pairing ectopic repetitive sequences, may be the main source of these chromosomal rearrangements, a hypothesis reinforced by our results suggesting an important role of SSA in recombination between repeated sequences (this work), in DNA double strand break repair in cells devoid of the RecA protein [4,6], and in early reassembly of small DNA fragments when cells were exposed to high γ-doses [4].
Conclusion
Altogether, these results suggest that SSA plays a major role in RecA-independent recombination between repeated sequences in the radioresistant D. radiodurans bacterium. In un-irradiated wild type bacteria, the deletion events might result, as proposed by Susan Lovett in E. coli, from RecA-dependent intermolecular unequal crossing over or intramolecular recombination between the overlapping 5’ and 3’ regions of the tetA gene, and from RecA-independent processes such as replication slippage or template switching [10] or single strand annealing [10,14]. Difficulties in replication can lead to breakage of the fork when replication forks are halted by obstacles or DNA damage in virtually every cell and every cell generation [65,66]. If this occurs in the context of repeated DNA sequences, single-stranded DNA substrates might be generated by resection of the DNA ends, and genetic rearrangements can result through strand-invasion of the broken chromosome with its sister or through SSA at the repeats. Replication of damaged DNA templates can further elevate the probability of fork breakage [67,68]. Moreover, when D. radiodurans cells were exposed to a dose of 5 kGy γ-irradiation, generating hundreds DNA double strand breaks, DdrB-dependent SSA and RecA-dependent ESDSA processes involved in DNA double strand break repair increased the opportunities to generate deletion events when DNA repeats are present in the DNA fragments.
Materials and Methods
Culture, media
D. radiodurans strains were grown at 30°C, 150 rpm in TGY2X (1% tryptone, 0.2% dextrose, 0.6% yeast extract) or plated on TGY1X containing 1.5% agar. E. coli strains were grown at 37°C, 150 rpm in Lysogeny Broth (LB). When necessary, media were supplemented with the appropriate antibiotics used at the following final concentrations: kanamycin, 6 μg/mL; chloramphenicol, 3.5 μg/mL; hygromycin, 50 μg/mL; spectinomycin, 75 μg/mL; tetracycline 2.5 μg/mL for D. radiodurans and kanamycin, 25 μg/mL or spectinomycin 40 μg/mL for E. coli.
Bacterial strains and plasmids
The bacterial strains and plasmids used in this study are listed in Table 1. The E. coli strains used were DH5α as the general cloning host, and SCS110, a dam dcm mutant strain, to propagate plasmids prior introduction into D. radiodurans via transformation [69]. Transformation of D. radiodurans with genomic DNA, PCR products, or plasmid DNA was performed as described [70]. All D. radiodurans strains were derivatives of strain R1 ATCC 13939. The genetic structure and the purity of the mutant strains were checked by PCR. Oligonucleotides used for strain constructions and diagnostic PCR will be provided on request.
Tab. 1. Bacterial strains and plasmids. 
(a) 5’tetA and the 3’tetA contain the 1–779 and the 342–1190 part of the tetA coding region, respectively. They contain a 342–779 overlapping region. The positions are numbered relatively to the first base of the initiation codon of tetA. Construction of tester strains for recombination assays
The strain GY12947 was constructed by the integration of the tetA gene into the dispensable amyE (DR_1472) gene by transformation of GY9613 (ATCC 13939 R1 strain) by the tripartite ligation product containing the 5’ and 3’parts of amyE and the tetA gene amplified by PCR using GY9613 genomic DNA and plasmid p11615 as template, respectively.
GY12947 was then used to amplify the 778 bp of the 5’ part (called 5’tetA) and the 850 bp of the 3’ part (called 3’tetA) of the coding region of the 1,190 bp tetA gene. The 5’tetA and 3’tetA fragments contain 438 bp overlapping sequences. Tripartite ligation products containing the 5’tetA and 3’tetA fragments separated by spacers of 1,479, 3,500 or 4,500 bp containing a spec gene amplified by PCR from plasmid p11554 were used to transform GY12947 giving rise to GY12949, GY16209, GY16211, respectively. Independent clones were saved for each strain and constructions were verified by DNA sequencing. Mutations generated during PCR amplification of 5‘tetA using GY12947 as template were detected in the 438 bp overlapping sequence of the 5‘tetA in two clones of GY12949 and the strains containing mutations in the 5’tetA overlapping region were renamed GY12948 (3 mutations) and GY12950 (1 mutation).
Strains with larger spacers between the tetA overlapping regions were obtained by transformation of GY16209 and GY16211 with a tripartite ligation product of a 3,000 or 6,000 bp PCR fragment containing the hph gene amplified from plasmid p15070, and 5’ and 3’ regions of the spec gene amplified by PCR from GY12949 yielding GY16227 containing a 6,500 bp spacer and GY16235 containing a 10,500 bp spacer, respectively. The right integration of spacers, and the purity of the strains were checked by PCR. The 5’tetA, the 3’tetA and the intervening sequences were checked by DNA sequencing.
For the recombination assays between chromosomal and plasmid DNA, strain GY15102, in which the 5’ region of the tetA gene (5’tetA) and an hph cassette were inserted in the amyE gene, was transformed by the plasmid p15002 obtained by cloning in plasmid p11554 of the 3’tetA region to give rise to strain GY15147. Plasmid and chromosomal DNA contained the same 438 bp homologous sequences as those present in the genomic DNA of strains used for measurement of inter - or intra-chromosomal recombination.
Deletion mutant derivatives of all the recombination tester strains were obtained by transformation of the tester strains with genomic DNA of ΔrecA, ΔmutS, ΔuvrD, ΔrecF, ΔrecO, ΔddrB, ΔradA derivatives of R1 strain, available in the laboratory or constructed by the tripartite ligation method [71]. The ΔrecAΔradA, ΔrecAΔmutS, ΔrecAΔddrB, ΔrecOΔddrB double mutants were obtained by transformation of the single ΔradA, ΔmutS or ΔddrB mutants by genomic DNA of ΔrecA or ΔrecO bacteria. ΔuvrD bacteria were also transformed by genomic DNA of ΔrecA, ΔrecF and ΔrecR bacteria. All strains were extensively purified on selective medium to obtain homogenotization of the deletions before to be checked by PCR for their purity. Attempts to obtain homogenotes containing the ΔuvrDΔrecA allele, the ΔuvrDΔrecF allele and the ΔuvrDΔrecR allele on each of the multiple genomic copies present in D. radiodurans were unsuccesful, even after extensive purification on selective plates (see Results section).
Inter - or intra-chromosomal recombination
Cells were plated on TGY agar and incubated at 30°C for 3 days, or 5 days for ΔrecA, ΔrecO and ΔrecF mutant bacteria and 7 days for double mutant ΔddrB ΔrecA and ΔddrB ΔrecO bacteria. Three to six colonies per strain were inoculated in 3 mL of TGY2X and incubated at 30°C, 150 rpm. Appropriate dilutions of the bacterial cultures, grown to OD650nm = 1.5 were plated on TGY and TGY + tetracycline 2.5 μg/mL. Colonies were counted after 4 to 7 days of incubation at 30°C. The experiments were repeated at least three times, using, when possible, strain isolates obtained independently during the strain constructions.
To measure recombination between repeated sequences after γ-irradiation, bacterial strains were treated as previously described upstream, except that bacterial cultures were grown to an OD650nm = 0.5 before being concentrated by centrifugation in TGY2X to an OD650nm = 10 and irradiated on ice at a dose of 5 kGy with a 60Co irradiation system (LABRA, CEA, Saclay) at a dose rate of 100 Gy/min. Following irradiation, samples of 100 μL were inoculated in 4.9 mL of TGY2X and incubated at 30°C, 150 rpm. After 20 hours of post-irradiation incubation, appropriate dilutions of bacterial culture were plated on TGY and TGY + tetracycline 2.5 μg/mL. Colonies were counted after 4-7days of incubation at 30°C. Unirradiated controls were treated as irradiated cells, except that they were maintained on ice without irradiation during the period when the irradiated cells were exposed to γ-rays.
Recombination between chromosomal and plasmid DNA
Cells containing plasmid p15002 were plated on TGY agar + spectinomycin (75 μg/mL) and incubated at 30°C during 3 days (or 5 days for ΔrecA and ΔrecF bacteria). Three colonies per strain were inoculated in 3 mL of TGY2X + spectinomycin (75 μg/mL) and incubated at 30°C, 150 rpm. Appropriate dilutions of the bacterial cultures grown to OD650nm = 1.5 were plated on TGY and TGY+ tetracycline 2.5 μg/mL. Colonies were counted after 4 to 7 days of incubation at 30°C.
Growth rate and lethal sectoring of ΔddrB ΔrecA and ΔddrB ΔrecO double mutants
Strains were streaked on TGY plates supplemented with the appropriate antibiotics. Independent colonies were inoculated in 3 mL TGY2X supplemented with the appropriate antibiotics (only kanamycin for double mutants) and grown at 30°C to an OD650nm = 1.5. Cultures were then diluted 200 to 5,000 fold and grown overnight at 30°C to an OD650nm = 0.1 (time 0 of the growth curves). Then, the OD650nm was measured and appropriate dilutions were plated on TGY plates at different times during 80 h of incubation at 30°C with agitation (150 rpm). Colonies were counted after 3 (WT or ΔddrB bacteria), 5 (ΔrecA or ΔrecO bacteria) or 7 days (ΔddrB ΔrecA or ΔddrB ΔrecO bacteria) of incubation at 30°C.
Statistical analysis
In Figs 1 and 3 and 5, in order to establish the statistical differences between the [TetR] frequencies measured in mutant and WT strains, non parametric Dunn’s multiple comparison test [75] were used, taking into account the p-value correction and performed with the GraphPad Prism6 software. All the comparisons were bi-sided.
Linear regressions and the slope significances observed in S2 Fig were estimated using the GraphPad Prism6 software.
In Fig 6, statistically significant differences between the irradiated and the non-irradiated conditions were calculated by non-parametric Mann-Withney tests performed in GraphPad Prism 6 software.
Supporting Information
Zdroje
1. Zahradka K, Slade D, Bailone A, Sommer S, Averbeck D, et al. (2006) Reassembly of shattered chromosomes in Deinococcus radiodurans. Nature 443 : 569–573. 17006450
2. Slade D, Lindner AB, Paul G, Radman M (2009) Recombination and replication in DNA repair of heavily irradiated Deinococcus radiodurans. Cell 136 : 1044–1055. doi: 10.1016/j.cell.2009.01.018 19303848
3. Bentchikou E, Servant P, Coste G, Sommer S (2010) A major role of the RecFOR pathway in DNA double-strand-break repair through ESDSA in Deinococcus radiodurans. PLoS Genet 6: e1000774. doi: 10.1371/journal.pgen.1000774 20090937
4. Bouthier de la Tour C, Boisnard S, Norais C, Toueille M, Bentchikou E, et al. (2011) The deinococcal DdrB protein is involved in an early step of DNA double strand break repair and in plasmid transformation through its single-strand annealing activity. DNA Repair (Amst) 10 : 1223–1231.
5. Norais CA, Chitteni-Pattu S, Wood EA, Inman RB, Cox MM (2009) An alternative Deinococcus radiodurans SSB induced by ionizing radiation: The DdrB protein. J Biol Chem.
6. Xu G, Lu H, Wang L, Chen H, Xu Z, et al. (2010) DdrB stimulates single-stranded DNA annealing and facilitates RecA-independent DNA repair in Deinococcus radiodurans. DNA Repair (Amst) 9 : 805–812.
7. Mazin AV, Kuzminov AV, Dianov GL, Salganik RI (1991) Mechanisms of deletion formation in Escherichia coli plasmids. II. Deletions mediated by short direct repeats. Mol Gen Genet 228 : 209–214. 1679526
8. Dianov GL, Kuzminov AV, Mazin AV, Salganik RI (1991) Molecular mechanisms of deletion formation in Escherichia coli plasmids. I. Deletion formation mediated by long direct repeats. Mol Gen Genet 228 : 153–159. 1679524
9. Bi X, Liu LF (1994) recA-independent and recA-dependent intramolecular plasmid recombination. Differential homology requirement and distance effect. J Mol Biol 235 : 414–423. 8289271
10. Lovett ST, Gluckman TJ, Simon PJ, Sutera VA Jr., Drapkin PT (1994) Recombination between repeats in Escherichia coli by a recA-independent, proximity-sensitive mechanism. Mol Gen Genet 245 : 294–300. 7816039
11. Bierne H, Vilette D, Ehrlich SD, Michel B (1997) Isolation of a dnaE mutation which enhances RecA-independent homologous recombination in the Escherichia coli chromosome. Mol Microbiol 24 : 1225–1234. 9218771
12. Saveson CJ, Lovett ST (1997) Enhanced deletion formation by aberrant DNA replication in Escherichia coli. Genetics 146 : 457–470. 9177997
13. Saveson CJ, Lovett ST (1999) Tandem repeat recombination induced by replication fork defects in Escherichia coli requires a novel factor, RadC. Genetics 152 : 5–13. 10224240
14. Michel B (2000) Replication fork arrest and DNA recombination. Trends Biochem Sci 25 : 173–178. 10754549
15. Lovett ST (2004) Encoded errors: mutations and rearrangements mediated by misalignment at repetitive DNA sequences. Mol Microbiol 52 : 1243–1253. 15165229
16. Bzymek M, Lovett ST (2001) Evidence for two mechanisms of palindrome-stimulated deletion in Escherichia coli: single-strand annealing and replication slipped mispairing. Genetics 158 : 527–540. 11404319
17. Nguyen HH, de la Tour CB, Toueille M, Vannier F, Sommer S, et al. (2009) The essential histone-like protein HU plays a major role in Deinococcus radiodurans nucleoid compaction. Mol Microbiol 73 : 240–252. doi: 10.1111/j.1365-2958.2009.06766.x 19570109
18. Meima R, Lidstrom ME (2000) Characterization of the minimal replicon of a cryptic Deinococcus radiodurans SARK plasmid and development of versatile Escherichia coli-D. radiodurans shuttle vectors. Appl Environ Microbiol 66 : 3856–3867. 10966401
19. Beam CE, Saveson CJ, Lovett ST (2002) Role for radA/sms in recombination intermediate processing in Escherichia coli. J Bacteriol 184 : 6836–6844. 12446634
20. Cooper DL, Boyle DC, Lovett ST (2015) Genetic analysis of Escherichia coli RadA: functional motifs and genetic interactions. Mol Microbiol 95 : 769–779. doi: 10.1111/mmi.12899 25484163
21. Konrad EB (1977) Method for the isolation of Escherichia coli mutants with enhanced recombination between chromosomal duplications. J Bacteriol 130 : 167–172. 323226
22. Feinstein SI, Low KB (1986) Hyper-recombining recipient strains in bacterial conjugation. Genetics 113 : 13–33. 3519362
23. Washburn BK, Kushner SR (1991) Construction and analysis of deletions in the structural gene (uvrD) for DNA helicase II of Escherichia coli. J Bacteriol 173 : 2569–2575. 1849510
24. Morel P, Hejna JA, Ehrlich SD, Cassuto E (1993) Antipairing and strand transferase activities of E. coli helicase II (UvrD). Nucleic Acids Res 21 : 3205–3209. 8341594
25. Veaute X, Delmas S, Selva M, Jeusset J, Le Cam E, et al. (2005) UvrD helicase, unlike Rep helicase, dismantles RecA nucleoprotein filaments in Escherichia coli. EMBO J 24 : 180–189. 15565170
26. Cox MM, Battista JR (2005) Deinococcus radiodurans—the consummate survivor. Nat Rev Microbiol 3 : 882–892. 16261171
27. Treangen TJ, Abraham AL, Touchon M, Rocha EP (2009) Genesis, effects and fates of repeats in prokaryotic genomes. FEMS Microbiol Rev 33 : 539–571. 19396957
28. Makarova KS, Aravind L, Wolf YI, Tatusov RL, Minton KW, et al. (2001) Genome of the extremely radiation-resistant bacterium Deinococcus radiodurans viewed from the perspective of comparative genomics. Microbiol Mol Biol Rev 65 : 44–79. 11238985
29. Marsin S, Mathieu A, Kortulewski T, Guerois R, Radicella JP (2008) Unveiling novel RecO distant orthologues involved in homologous recombination. PLoS Genet 4: e1000146. doi: 10.1371/journal.pgen.1000146 18670631
30. Bruand C, Bidnenko V, Ehrlich SD (2001) Replication mutations differentially enhance RecA-dependent and RecA-independent recombination between tandem repeats in Bacillus subtilis. Mol Microbiol 39 : 1248–1258. 11251841
31. Thomas CA (1967) The recombination of DNA molecules. In: press TRU, editor. The Neurosciences A study program. New York. pp. 162–182.
32. Lin FL, Sperle K, Sternberg N (1984) Model for homologous recombination during transfer of DNA into mouse L cells: role for DNA ends in the recombination process. Mol Cell Biol 4 : 1020–1034. 6330525
33. Mezard C, Nicolas A (1994) Homologous, homeologous, and illegitimate repair of double-strand breaks during transformation of a wild-type strain and a rad52 mutant strain of Saccharomyces cerevisiae. Mol Cell Biol 14 : 1278–1292. 8289807
34. Grimme JM, Honda M, Wright R, Okuno Y, Rothenberg E, et al. (2010) Human Rad52 binds and wraps single-stranded DNA and mediates annealing via two hRad52-ssDNA complexes. Nucleic Acids Res 38 : 2917–2930. doi: 10.1093/nar/gkp1249 20081207
35. Rothenberg E, Grimme JM, Spies M, Ha T (2008) Human Rad52-mediated homology search and annealing occurs by continuous interactions between overlapping nucleoprotein complexes. Proc Natl Acad Sci U S A 105 : 20274–20279. doi: 10.1073/pnas.0810317106 19074292
36. Paques F, Haber JE (1999) Multiple pathways of recombination induced by double-strand breaks in Saccharomyces cerevisiae. Microbiol Mol Biol Rev 63 : 349–404. 10357855
37. Sugiman-Marangos S, Junop MS (2010) The structure of DdrB from Deinococcus: a new fold for single-stranded DNA binding proteins. Nucleic Acids Res 38 : 3432–3440. doi: 10.1093/nar/gkq036 20129942
38. Sugiman-Marangos SN, Peel JK, Weiss YM, Ghirlando R, Junop MS (2013) Crystal structure of the DdrB/ssDNA complex from Deinococcus radiodurans reveals a DNA binding surface involving higher-order oligomeric states. Nucleic Acids Res 41 : 9934–9944. doi: 10.1093/nar/gkt759 23975200
39. Shinohara A, Shinohara M, Ohta T, Matsuda S, Ogawa T (1998) Rad52 forms ring structures and co-operates with RPA in single-strand DNA annealing. Genes Cells 3 : 145–156. 9619627
40. Sugiyama T, New JH, Kowalczykowski SC (1998) DNA annealing by RAD52 protein is stimulated by specific interaction with the complex of replication protein A and single-stranded DNA. Proc Natl Acad Sci U S A 95 : 6049–6054. 9600915
41. Lockhart JS, DeVeaux LC (2013) The essential role of the Deinococcus radiodurans ssb gene in cell survival and radiation tolerance. PLoS One 8: e71651. doi: 10.1371/journal.pone.0071651 23951213
42. Shereda RD, Kozlov AG, Lohman TM, Cox MM, Keck JL (2008) SSB as an organizer/mobilizer of genome maintenance complexes. Crit Rev Biochem Mol Biol 43 : 289–318. doi: 10.1080/10409230802341296 18937104
43. Inoue J, Honda M, Ikawa S, Shibata T, Mikawa T (2008) The process of displacing the single-stranded DNA-binding protein from single-stranded DNA by RecO and RecR proteins. Nucleic Acids Res 36 : 94–109. 18000001
44. Hobbs MD, Sakai A, Cox MM (2007) SSB protein limits RecOR binding onto single-stranded DNA. J Biol Chem 282 : 11058–11067. 17272275
45. Reams AB, Kofoid E, Duleba N, Roth JR (2014) Recombination and annealing pathways compete for substrates in making rrn duplications in Salmonella enterica. Genetics 196 : 119–135. doi: 10.1534/genetics.113.158519 24214339
46. Atkinson J, McGlynn P (2009) Replication fork reversal and the maintenance of genome stability. Nucleic Acids Res 37 : 3475–3492. doi: 10.1093/nar/gkp244 19406929
47. Bierne H, Seigneur M, Ehrlich SD, Michel B (1997) uvrD mutations enhance tandem repeat deletion in the Escherichia coli chromosome via SOS induction of the RecF recombination pathway. Mol Microbiol 26 : 557–567. 9402025
48. Zieg J, Maples VF, Kushner SR (1978) Recombinant levels of Escherichia coli K-12 mutants deficient in various replication, recombination, or repair genes. J Bacteriol 134 : 958–966. 350859
49. Arthur HM, Lloyd RG (1980) Hyper-recombination in uvrD mutants of Escherichia coli K-12. Mol Gen Genet 180 : 185–191. 7003307
50. Ossanna N, Mount DW (1989) Mutations in uvrD induce the SOS response in Escherichia coli. J Bacteriol 171 : 303–307. 2536658
51. Yancey-Wrona JE, Wood ER, George JW, Smith KR, Matson SW (1992) Escherichia coli Rep protein and helicase IV. Distributive single-stranded DNA-dependent ATPases that catalyze a limited unwinding reaction in vitro. Eur J Biochem 207 : 479–485. 1321715
52. Guy CP, Atkinson J, Gupta MK, Mahdi AA, Gwynn EJ, et al. (2009) Rep provides a second motor at the replisome to promote duplication of protein-bound DNA. Mol Cell 36 : 654–666. doi: 10.1016/j.molcel.2009.11.009 19941825
53. Atkinson J, Gupta MK, McGlynn P (2011) Interaction of Rep and DnaB on DNA. Nucleic Acids Res 39 : 1351–1359. doi: 10.1093/nar/gkq975 20959294
54. Lane HE, Denhardt DT (1975) The rep mutation. IV. Slower movement of replication forks in Escherichia coli rep strains. J Mol Biol 97 : 99–112. 1100854
55. Taucher-Sholz G, Abdel-Monem M, Hoffman-Berling H (1983) Function of DNA helicases in E. coli. In: Cozarelli NR, editor. Mechanisms of DNA replication and recombination: Alan, R. Liss, Inc., New York. pp. 65–76.
56. Boubakri H, de Septenville AL, Viguera E, Michel B (2010) The helicases DinG, Rep and UvrD cooperate to promote replication across transcription units in vivo. EMBO J 29 : 145–157. doi: 10.1038/emboj.2009.308 19851282
57. Flores MJ, Bidnenko V, Michel B (2004) The DNA repair helicase UvrD is essential for replication fork reversal in replication mutants. EMBO Rep 5 : 983–988. 15375374
58. Flores MJ, Sanchez N, Michel B (2005) A fork-clearing role for UvrD. Mol Microbiol 57 : 1664–1675. 16135232
59. Jiao J, Wang L, Xia W, Li M, Sun H, et al. (2012) Function and biochemical characterization of RecJ in Deinococcus radiodurans. DNA Repair (Amst) 11 : 349–356.
60. Montelone BA, Malone RE (1994) Analysis of the rad3-101 and rad3-102 mutations of Saccharomyces cerevisiae: implications for structure/function of Rad3 protein. Yeast 10 : 13–27. 8203147
61. Daly MJ, Ling O, Minton KW (1994) Interplasmidic recombination following irradiation of the radioresistant bacterium Deinococcus radiodurans. J Bacteriol 176 : 7506–7515. 8002574
62. Daly MJ, Minton KW (1995) Interchromosomal recombination in the extremely radioresistant bacterium Deinococcus radiodurans. J Bacteriol 177 : 5495–5505. 7559335
63. Daly MJ, Minton KW (1996) An alternative pathway of recombination of chromosomal fragments precedes recA-dependent recombination in the radioresistant bacterium Deinococcus radiodurans. J Bacteriol 178 : 4461–4471. 8755873
64. Repar J, Cvjetan S, Slade D, Radman M, Zahradka D, et al. (2010) RecA protein assures fidelity of DNA repair and genome stability in Deinococcus radiodurans. DNA Repair (Amst) 9 : 1151–1161.
65. Cox MM, Goodman MF, Kreuzer KN, Sherratt DJ, Sandler SJ, et al. (2000) The importance of repairing stalled replication forks. Nature 404 : 37–41. 10716434
66. Kuzminov A (1999) Recombinational repair of DNA damage in Escherichia coli and bacteriophage lambda. Microbiol Mol Biol Rev 63 : 751–813, table of contents. 10585965
67. Kuzminov A (1995) Instability of inhibited replication forks in E. coli. Bioessays 17 : 733–741. 7661854
68. Michel B, Ehrlich SD, Uzest M (1997) DNA double-strand breaks caused by replication arrest. EMBO J 16 : 430–438. 9029161
69. Meima R, Rothfuss HM, Gewin L, Lidstrom ME (2001) Promoter cloning in the radioresistant bacterium Deinococcus radiodurans. J Bacteriol 183 : 3169–3175. 11325946
70. Bonacossa de Almeida C, Coste G, Sommer S, Bailone A (2002) Quantification of RecA protein in Deinococcus radiodurans reveals involvement of RecA, but not LexA, in its regulation. Mol Genet Genomics 268 : 28–41. 12242496
71. Mennecier S, Coste G, Servant P, Bailone A, Sommer S (2004) Mismatch repair ensures fidelity of replication and recombination in the radioresistant organism Deinococcus radiodurans. Mol Genet Genomics 272 : 460–469. 15503140
72. Bentchikou E, Servant P, Coste G, Sommer S (2007) Additive effects of SbcCD and PolX deficiencies in the in vivo repair of DNA double strand breaks in Deinococcus radiodurans. J Bacteriol.
73. Devigne A, Ithurbide S, Bouthier de la Tour C, Passot F, Mathieu M, et al. (2015) DdrO is an essential protein that regulates the radiation desiccation response and the apoptotic-like cell death in the radioresistant Deinococcus radiodurans bacterium. Mol Microbiol.
74. Satoh K, Kikuchi M, Ishaque AM, Ohba H, Yamada M, et al. (2012) The role of Deinococcus radiodurans RecFOR proteins in homologous recombination. DNA Repair (Amst) 11 : 410–418.
75. Dunn OJ (1964) Multiple Comparisons Using Rank Sums. Technometrics 6 : 241–252.
Štítky
Genetika Reprodukční medicína
Článek Evidence of Selection against Complex Mitotic-Origin Aneuploidy during Preimplantation DevelopmentČlánek A Novel Route Controlling Begomovirus Resistance by the Messenger RNA Surveillance Factor PelotaČlánek A Follicle Rupture Assay Reveals an Essential Role for Follicular Adrenergic Signaling in OvulationČlánek Canonical Poly(A) Polymerase Activity Promotes the Decay of a Wide Variety of Mammalian Nuclear RNAsČlánek FANCI Regulates Recruitment of the FA Core Complex at Sites of DNA Damage Independently of FANCD2Článek Hsp90-Associated Immunophilin Homolog Cpr7 Is Required for the Mitotic Stability of [URE3] Prion inČlánek The Dedicated Chaperone Acl4 Escorts Ribosomal Protein Rpl4 to Its Nuclear Pre-60S Assembly SiteČlánek Chromatin-Remodelling Complex NURF Is Essential for Differentiation of Adult Melanocyte Stem CellsČlánek A Systems Approach Identifies Essential FOXO3 Functions at Key Steps of Terminal ErythropoiesisČlánek Integration of Posttranscriptional Gene Networks into Metabolic Adaptation and Biofilm Maturation inČlánek Lateral and End-On Kinetochore Attachments Are Coordinated to Achieve Bi-orientation in OocytesČlánek MET18 Connects the Cytosolic Iron-Sulfur Cluster Assembly Pathway to Active DNA Demethylation in
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2015 Číslo 10- Kazuistika – Perspektivy využití precizované medicíny v rámci personalizované specifické terapie onkologických pacientů
- Nobelova cena za chemii pro genetické nůžky: Objev, který změní naši budoucnost?
- Technologie na bázi RNA v klinické praxi: od přebarvených petúnií k terapii vzácných a dosud jen obtížně léčitelných chorob u lidí
- „Nepředstavovali jsme si, že náš výzkum povede přímo ke vzniku nových léků, dokonce ještě za našeho života“
- Bezplatné služby pro diagnostiku ATTRv amyloidózy pro kardiology
-
Všechny články tohoto čísla
- Gene-Regulatory Logic to Induce and Maintain a Developmental Compartment
- A Decad(e) of Reasons to Contribute to a PLOS Community-Run Journal
- DNA Methylation Landscapes of Human Fetal Development
- Single Strand Annealing Plays a Major Role in RecA-Independent Recombination between Repeated Sequences in the Radioresistant Bacterium
- Evidence of Selection against Complex Mitotic-Origin Aneuploidy during Preimplantation Development
- Transcriptional Derepression Uncovers Cryptic Higher-Order Genetic Interactions
- Silencing of X-Linked MicroRNAs by Meiotic Sex Chromosome Inactivation
- Virus Satellites Drive Viral Evolution and Ecology
- A Novel Route Controlling Begomovirus Resistance by the Messenger RNA Surveillance Factor Pelota
- Sequence to Medical Phenotypes: A Framework for Interpretation of Human Whole Genome DNA Sequence Data
- Your Data to Explore: An Interview with Anne Wojcicki
- Modulation of Ambient Temperature-Dependent Flowering in by Natural Variation of
- The Ciliopathy Protein CC2D2A Associates with NINL and Functions in RAB8-MICAL3-Regulated Vesicle Trafficking
- PPP2R5C Couples Hepatic Glucose and Lipid Homeostasis
- DCA1 Acts as a Transcriptional Co-activator of DST and Contributes to Drought and Salt Tolerance in Rice
- Intermediate Levels of CodY Activity Are Required for Derepression of the Branched-Chain Amino Acid Permease, BraB
- "Missing" G x E Variation Controls Flowering Time in
- The Rise and Fall of an Evolutionary Innovation: Contrasting Strategies of Venom Evolution in Ancient and Young Animals
- Type IV Collagen Controls the Axogenesis of Cerebellar Granule Cells by Regulating Basement Membrane Integrity in Zebrafish
- Loss of a Conserved tRNA Anticodon Modification Perturbs Plant Immunity
- Genome-Wide Association Analysis of Adaptation Using Environmentally Predicted Traits
- Oriented Cell Division in the . Embryo Is Coordinated by G-Protein Signaling Dependent on the Adhesion GPCR LAT-1
- Disproportionate Contributions of Select Genomic Compartments and Cell Types to Genetic Risk for Coronary Artery Disease
- A Follicle Rupture Assay Reveals an Essential Role for Follicular Adrenergic Signaling in Ovulation
- The RNAPII-CTD Maintains Genome Integrity through Inhibition of Retrotransposon Gene Expression and Transposition
- Canonical Poly(A) Polymerase Activity Promotes the Decay of a Wide Variety of Mammalian Nuclear RNAs
- Allelic Variation of Cytochrome P450s Drives Resistance to Bednet Insecticides in a Major Malaria Vector
- SCARN a Novel Class of SCAR Protein That Is Required for Root-Hair Infection during Legume Nodulation
- IBR5 Modulates Temperature-Dependent, R Protein CHS3-Mediated Defense Responses in
- NINL and DZANK1 Co-function in Vesicle Transport and Are Essential for Photoreceptor Development in Zebrafish
- Decay-Initiating Endoribonucleolytic Cleavage by RNase Y Is Kept under Tight Control via Sequence Preference and Sub-cellular Localisation
- Large-Scale Analysis of Kinase Signaling in Yeast Pseudohyphal Development Identifies Regulation of Ribonucleoprotein Granules
- FANCI Regulates Recruitment of the FA Core Complex at Sites of DNA Damage Independently of FANCD2
- LINE-1 Mediated Insertion into (Protein of Centriole 1 A) Causes Growth Insufficiency and Male Infertility in Mice
- Hsp90-Associated Immunophilin Homolog Cpr7 Is Required for the Mitotic Stability of [URE3] Prion in
- Genome-Scale Mapping of σ Reveals Widespread, Conserved Intragenic Binding
- Uncovering Hidden Layers of Cell Cycle Regulation through Integrative Multi-omic Analysis
- Functional Diversification of Motor Neuron-specific Enhancers during Evolution
- The GTP- and Phospholipid-Binding Protein TTD14 Regulates Trafficking of the TRPL Ion Channel in Photoreceptor Cells
- The Gyc76C Receptor Guanylyl Cyclase and the Foraging cGMP-Dependent Kinase Regulate Extracellular Matrix Organization and BMP Signaling in the Developing Wing of
- The Ty1 Retrotransposon Restriction Factor p22 Targets Gag
- Functional Impact and Evolution of a Novel Human Polymorphic Inversion That Disrupts a Gene and Creates a Fusion Transcript
- The Dedicated Chaperone Acl4 Escorts Ribosomal Protein Rpl4 to Its Nuclear Pre-60S Assembly Site
- The Influence of Age and Sex on Genetic Associations with Adult Body Size and Shape: A Large-Scale Genome-Wide Interaction Study
- Parent-of-Origin Effects of the Gene on Adiposity in Young Adults
- Chromatin-Remodelling Complex NURF Is Essential for Differentiation of Adult Melanocyte Stem Cells
- Retinoic Acid Receptors Control Spermatogonia Cell-Fate and Induce Expression of the SALL4A Transcription Factor
- A Systems Approach Identifies Essential FOXO3 Functions at Key Steps of Terminal Erythropoiesis
- Protein O-Glucosyltransferase 1 (POGLUT1) Promotes Mouse Gastrulation through Modification of the Apical Polarity Protein CRUMBS2
- KIF7 Controls the Proliferation of Cells of the Respiratory Airway through Distinct Microtubule Dependent Mechanisms
- Integration of Posttranscriptional Gene Networks into Metabolic Adaptation and Biofilm Maturation in
- Lateral and End-On Kinetochore Attachments Are Coordinated to Achieve Bi-orientation in Oocytes
- Protein Homeostasis Imposes a Barrier on Functional Integration of Horizontally Transferred Genes in Bacteria
- A New Method for Detecting Associations with Rare Copy-Number Variants
- Histone H2AFX Links Meiotic Chromosome Asynapsis to Prophase I Oocyte Loss in Mammals
- The Genomic Aftermath of Hybridization in the Opportunistic Pathogen
- A Role for the Chaperone Complex BAG3-HSPB8 in Actin Dynamics, Spindle Orientation and Proper Chromosome Segregation during Mitosis
- Establishment of a Developmental Compartment Requires Interactions between Three Synergistic -regulatory Modules
- Regulation of Spore Formation by the SpoIIQ and SpoIIIA Proteins
- Association of the Long Non-coding RNA Steroid Receptor RNA Activator (SRA) with TrxG and PRC2 Complexes
- Alkaline Ceramidase 3 Deficiency Results in Purkinje Cell Degeneration and Cerebellar Ataxia Due to Dyshomeostasis of Sphingolipids in the Brain
- ACLY and ACC1 Regulate Hypoxia-Induced Apoptosis by Modulating ETV4 via α-ketoglutarate
- Quantitative Differences in Nuclear β-catenin and TCF Pattern Embryonic Cells in .
- HENMT1 and piRNA Stability Are Required for Adult Male Germ Cell Transposon Repression and to Define the Spermatogenic Program in the Mouse
- Axon Regeneration Is Regulated by Ets–C/EBP Transcription Complexes Generated by Activation of the cAMP/Ca Signaling Pathways
- A Phenomic Scan of the Norfolk Island Genetic Isolate Identifies a Major Pleiotropic Effect Locus Associated with Metabolic and Renal Disorder Markers
- The Roles of CDF2 in Transcriptional and Posttranscriptional Regulation of Primary MicroRNAs
- A Genetic Cascade of Modulates Nucleolar Size and rRNA Pool in
- Inter-population Differences in Retrogene Loss and Expression in Humans
- Cationic Peptides Facilitate Iron-induced Mutagenesis in Bacteria
- EP4 Receptor–Associated Protein in Macrophages Ameliorates Colitis and Colitis-Associated Tumorigenesis
- Fungal Infection Induces Sex-Specific Transcriptional Changes and Alters Sexual Dimorphism in the Dioecious Plant
- FLCN and AMPK Confer Resistance to Hyperosmotic Stress via Remodeling of Glycogen Stores
- MET18 Connects the Cytosolic Iron-Sulfur Cluster Assembly Pathway to Active DNA Demethylation in
- Sex Bias and Maternal Contribution to Gene Expression Divergence in Blastoderm Embryos
- Transcriptional and Linkage Analyses Identify Loci that Mediate the Differential Macrophage Response to Inflammatory Stimuli and Infection
- Mre11 and Blm-Dependent Formation of ALT-Like Telomeres in Ku-Deficient
- Genome Wide Identification of SARS-CoV Susceptibility Loci Using the Collaborative Cross
- Identification of a Single Strand Origin of Replication in the Integrative and Conjugative Element ICE of
- The Type VI Secretion TssEFGK-VgrG Phage-Like Baseplate Is Recruited to the TssJLM Membrane Complex via Multiple Contacts and Serves As Assembly Platform for Tail Tube/Sheath Polymerization
- The Dynamic Genome and Transcriptome of the Human Fungal Pathogen and Close Relative
- Secondary Structure across the Bacterial Transcriptome Reveals Versatile Roles in mRNA Regulation and Function
- ROS-Induced JNK and p38 Signaling Is Required for Unpaired Cytokine Activation during Regeneration
- Pelle Modulates dFoxO-Mediated Cell Death in
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Single Strand Annealing Plays a Major Role in RecA-Independent Recombination between Repeated Sequences in the Radioresistant Bacterium
- The Rise and Fall of an Evolutionary Innovation: Contrasting Strategies of Venom Evolution in Ancient and Young Animals
- Genome Wide Identification of SARS-CoV Susceptibility Loci Using the Collaborative Cross
- DCA1 Acts as a Transcriptional Co-activator of DST and Contributes to Drought and Salt Tolerance in Rice
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Autoři: prof. MUDr. Pavel Horák, CSc., doc. MUDr. Ludmila Brunerová, Ph.D., doc. MUDr. Václav Vyskočil, Ph.D., prim. MUDr. Richard Pikner, Ph.D., MUDr. Olga Růžičková, MUDr. Jan Rosa, prof. MUDr. Vladimír Palička, CSc., Dr.h.c.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



