-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
Top novinky
ReklamaSynthetic Lethality of Cohesins with PARPs and Replication Fork Mediators
Synthetic lethality has been proposed as a way to leverage the genetic differences found in tumor cells to affect their selective killing. Cohesins, which tether sister chromatids together until anaphase onset, are mutated in a variety of tumor types. The elucidation of synthetic lethal interactions with cohesin mutants therefore identifies potential therapeutic targets. We used a cross-species approach to identify robust negative genetic interactions with cohesin mutants. Utilizing essential and non-essential mutant synthetic genetic arrays in Saccharomyces cerevisiae, we screened genome-wide for genetic interactions with hypomorphic mutations in cohesin genes. A somatic cell proliferation assay in Caenorhabditis elegans demonstrated that the majority of interactions were conserved. Analysis of the interactions found that cohesin mutants require the function of genes that mediate replication fork progression. Conservation of these interactions between replication fork mediators and cohesin in both yeast and C. elegans prompted us to test whether other replication fork mediators not found in the yeast were required for viability in cohesin mutants. PARP1 has roles in the DNA damage response but also in the restart of stalled replication forks. We found that a hypomorphic allele of the C. elegans SMC1 orthologue, him-1(e879), genetically interacted with mutations in the orthologues of PAR metabolism genes resulting in a reduced brood size and somatic cell defects. We then demonstrated that this interaction is conserved in human cells by showing that PARP inhibitors reduce the viability of cultured human cells depleted for cohesin components. This work demonstrates that large-scale genetic interaction screening in yeast can identify clinically relevant genetic interactions and suggests that PARP inhibitors, which are currently undergoing clinical trials as a treatment of homologous recombination-deficient cancers, may be effective in treating cancers that harbor cohesin mutations.
Published in the journal: . PLoS Genet 8(3): e32767. doi:10.1371/journal.pgen.1002574
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1002574Summary
Synthetic lethality has been proposed as a way to leverage the genetic differences found in tumor cells to affect their selective killing. Cohesins, which tether sister chromatids together until anaphase onset, are mutated in a variety of tumor types. The elucidation of synthetic lethal interactions with cohesin mutants therefore identifies potential therapeutic targets. We used a cross-species approach to identify robust negative genetic interactions with cohesin mutants. Utilizing essential and non-essential mutant synthetic genetic arrays in Saccharomyces cerevisiae, we screened genome-wide for genetic interactions with hypomorphic mutations in cohesin genes. A somatic cell proliferation assay in Caenorhabditis elegans demonstrated that the majority of interactions were conserved. Analysis of the interactions found that cohesin mutants require the function of genes that mediate replication fork progression. Conservation of these interactions between replication fork mediators and cohesin in both yeast and C. elegans prompted us to test whether other replication fork mediators not found in the yeast were required for viability in cohesin mutants. PARP1 has roles in the DNA damage response but also in the restart of stalled replication forks. We found that a hypomorphic allele of the C. elegans SMC1 orthologue, him-1(e879), genetically interacted with mutations in the orthologues of PAR metabolism genes resulting in a reduced brood size and somatic cell defects. We then demonstrated that this interaction is conserved in human cells by showing that PARP inhibitors reduce the viability of cultured human cells depleted for cohesin components. This work demonstrates that large-scale genetic interaction screening in yeast can identify clinically relevant genetic interactions and suggests that PARP inhibitors, which are currently undergoing clinical trials as a treatment of homologous recombination-deficient cancers, may be effective in treating cancers that harbor cohesin mutations.
Introduction
Defects in cohesin-associated genes are emerging as potential drivers of tumor genomic instability and progression. Mutations in cohesin genes have been identified in several tumor types (Reviewed in [1]). Sequencing of over 200 human orthologs of yeast chromosome instability (CIN) genes from 130 colon tumors found that cohesin genes are mutated in 8% of tumor samples [2]. In a recent study, Solomon et al. found the cohesin gene STAG2 mutated in 21% of Ewing's sarcomas and in 19% of both melanoma and glioblastoma tumors [3]. Furthermore, altered cohesin gene expression, either overexpression or underexpression is characteristic of many tumors [4]–[7]. It has been shown that loss of cohesin subunits induces genomic instability in human cancers and the associated aneuploidy, as is observed in many cell lines with mutations in cohesin, can itself lead to further genomic instability [2], [3], [8]. These observations and the elevated mutational frequency of cohesin in diverse tumor types suggest that cohesin dysfunction may contribute to tumor development and progression.
Cohesins maintain sister chromatid cohesion and screens for defects in sister chromatid cohesion have identified the core cohesin complex, composed of Smc1, Smc3, Scc1 and Scc3, and additional accessory and regulatory proteins [9]. Cohesins form a ring structure that is thought to encircle sister chromatids and physically tether them together until it is cleaved by separase during anaphase [10], [11]. Cohesin proteins contribute to DNA repair and the regulation of gene expression in addition to chromosome segregation (Reviewed in [12]). Although much is known about the function of cohesin in regulating sister chromatid cohesion and DNA damage repair, it is not as yet clear which aspects of cohesin biology might contribute to tumor progression.
One approach to understanding the functional spectrum associated with a gene of interest relies on the identification of genetic interactions with other gene mutations. Negative genetic interactions occur when the double mutant shows a synthetic growth defect manifested as severe slow growth or lethality (synthetic sickness/lethality) when compared to both single mutants. Synthetic sick or lethal interactions with genes mutated in cancer can identify potential therapeutic targets [13], [14]. A clinically relevant example of a synthetic lethal (SL) genetic interaction is the SL interaction between mutations in breast cancer susceptibility genes BRCA1 or BRCA2 and loss of function of the Poly-ADP Ribose Polymerases (PARP). Two groups found that BRCA1 - and BRCA2-defective cells are sensitive to knockdown of PARP or chemical inhibition of PARP activity [15], [16]. This has lead to the development of PARP inhibitors as chemotherapeutics. PARP inhibitors are being evaluated in Phase II clinical trials for use in homologous recombination (HR) deficient breast and ovarian tumors [17]–[20].
In addition to identifying synthetic lethal interactions that could represent potential drug targets, comprehensive genetic interaction networks can also lead to new functional insights [21]. Mapping global genetic interactions in human cells is feasible but techniques lag behind those currently available in budding yeast. Synthetic genetic array (SGA) is a large-scale genetic interaction screening approach in yeast that facilitates the collection and analysis of positive and negative genetic interaction data [22]–[24]. The use of yeast as a model organism to identify conserved genetic interactions with potential cancer therapeutic value has proven effective [14]. The inclusion of the metazoan animal model, C. elegans, in the genetic interaction testing pipeline can also contribute new insights as nematodes have a gene complement more akin to humans and contain several cancer-relevant genes not found in yeast, such as BRCA1, BRCA2, TP53 and the family of poly(ADP)-ribose polymerases (PARPs) [25]–[28]. Furthermore, C. elegans mutants and double mutants also present informative phenotypes, such as apoptotic defects, cell cycle checkpoint dysfunction, and chromosome loss, in the context of a multicellular animal model, which can lead to a better understanding of the biological processes affected by specific genetic interactions [29]. The large number of genetic interactions that can be identified by comprehensive genetic interaction screens such as SGA can identify key processes or pathways that when disrupted result in synthetic lethality. These pathways could be targeted for SL therapeutic intervention even if the specific genes are not well conserved from the yeast to humans.
In this study, we performed digenic SGA screens in S. cerevisiae using three hypomorphic cohesin mutations to identify common processes required for survival when cohesin is mutated. Interactions were tested for conservation in C. elegans using an assay for defects in somatic cell proliferation [30]. We found that proteins mediating replication fork progression and stability are required in cohesin mutants of both S. cerevisiae and C. elegans. Based on these findings we predicted that other mediators of replication fork stability not conserved in yeast, such as PARP, would be required for viability in higher eukaryotic cells with mutations in cohesin. To test this prediction, we expanded the screen in C. elegans to include PAR metabolism (pme) mutations and found that pme mutants genetically interact with him-1/SMC1 in C. elegans. We found that this genetic interaction was conserved in cultured human cells and small molecule PARP inhibitors, currently being evaluated in clinical trials, were effective in inhibiting growth in cohesin depleted cultured human cells. Beginning with systematic screens in a simple model eukaryote this study identifies conserved and clinically relevant genetic interactions between cohesin and replication fork modulators including the chemotherapeutic target PARP.
Results
Systematic quantitative analysis of cohesin genetic interactions
Synthetic genetic array (SGA) technology was used to screen temperature sensitive (ts) alleles of two cohesin components (smc1-259, scc1-73) and one cohesin loader (scc2-4) against ∼95% of genes in S. cerevisiae as represented by non-essential gene deletions [31], ts [32] or decreased abundance by mRNA perturbation (DAmP) [33] alleles. All three cohesin alleles have mutations in similar regions as those identified in colon tumors (SMC1, SCC2) or in the Catalogue of Somatic Mutations in Cancer (COSMIC) (SCC1) (Table 1, Figure S1).
Tab. 1. Mutations in cohesin genes seen in tumors. 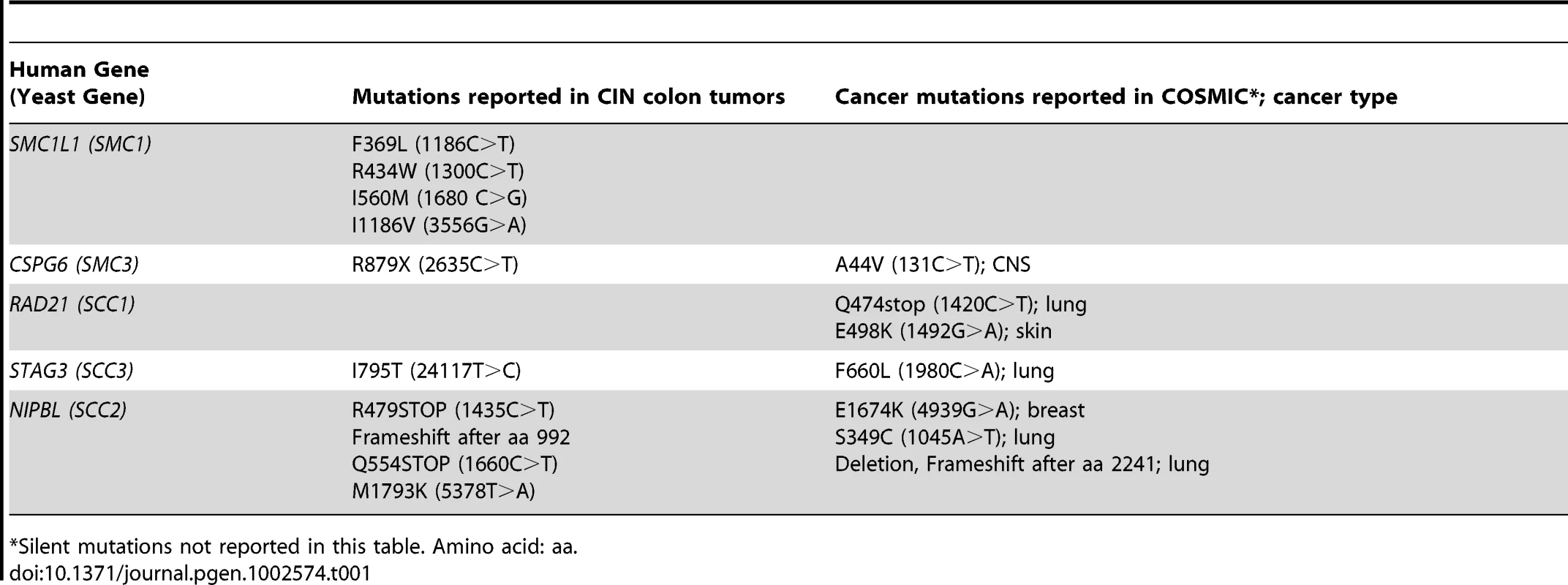
Silent mutations not reported in this table. Amino acid: aa. The interaction data was sequentially filtered using several criteria to increase quality and focus. Only negative genetic interactions with a p-value less than 0.05 and a large interaction magnitude (E-C value less than −0.3, see Methods) were considered. Filtering based on magnitude enriched for interactions that cause a severe fitness defect when compared to the single mutant. To reduce false positives and increase the potential of identifying drug targets that interact with mutations in multiple cohesin subunits, we eliminated genes that interacted with only a single cohesin query gene, leaving 55 genes (Figure S2). Finally, to focus on interactions that may have relevance to the biology of cohesins and cancer, we eliminated genes that did not have an obvious human homolog (see Figure 1A, Table S1). Using these criteria, 39 of 55 genes (71%), defining 90 putative negative genetic interactions, had an identifiable homolog in humans (Table S1). Six genes were removed from further analysis for technical reasons, leaving 33 genes comprising 78 genetic interactions.
Fig. 1. S. cerevisiae cohesin genetic interaction network. 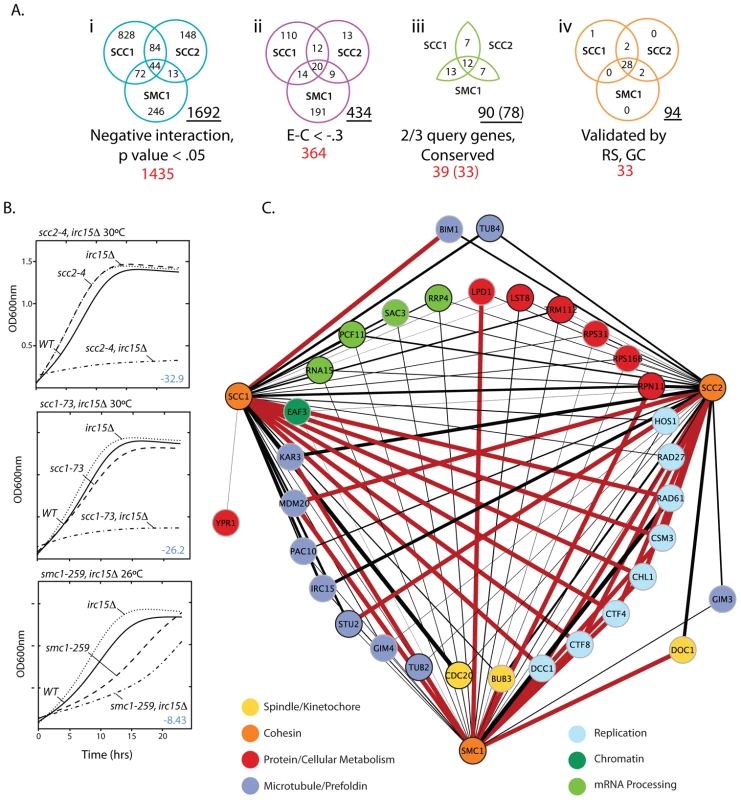
A) Venn diagrams depicting how SGA data was filtered. i. Interactions that had a negative interaction value and were statistically significant (p-value<0.05). ii. Interactions that had a relatively weak interaction score (Experimental value – Control value>−.3) were eliminated to enrich for biologically significant interactions. iii. Genes were eliminated if they failed to interact with ≥2 of the cohesin query genes and if they did not have a human homolog (Table S3). iv. Summary of the final network after random spore and growth curve retesting and validation. The total number of interactions in a given Venn diagram is underlined. The total number of genes is shown in red. The numbers in brackets indicate interacting genes remaining after removal of 6 genes for technical reasons. B) Representative subset of growth curve data. The T-stat, which takes into account the interaction magnitude and statistical significance, is shown in blue. All growth curve data can be found in Figures S3, S4, S5, S6 and Tables S1, S2, S3. C) Expanded view of the final network summarized in A iv. Red lines indicate SL interactions and black lines represent SS interactions. The black line thickness represents interaction strength. To validate this subset of interactions identified in the primary screen, 99 double heterozygous diploids (33 genetic interaction genes by 3 cohesin query genes) were reconstructed, the specific gene deletions confirmed by DNA analysis, and double mutant phenotypes retested by random spore analysis. To assess whether growth defects were greater than additive, growth curve analysis was performed on all viable double mutants (Figure 1B; Figures S3, S4, S5, S6, S7; Tables S2, S3, S4). Random spore and growth curve analyses achieved several goals; 1) Reduced the false positive rate by removing genes with an incorrect well address in the high throughput arrays (8% of hits), 2) Eliminated condition artifacts by ensuring that genetic interactions were reproducible under the same drug selective conditions as SGA (random spore) and in rich medium (growth curve), 3) Yielded an additional quantitative measure of each synthetic sick interaction, and 4) Identified additional true positives not identified under SGA conditions.
During subsequent testing 4 out of 78 (5%) interactions identified by SGA did not result in a negative genetic interaction and an additional 20 interactions not observed by SGA were identified for a total of 94 negative genetic interactions with the three cohesin query genes (Figure 1C). 29 (31%) of these interactions involve essential genes, highlighting the importance of screening against essential gene collections.
Negative genetic interactions with cohesin are conserved in C. elegans
One of our goals is to identify interactions that are conserved in mammalian cells and thus relevant to the development of therapeutics. We hypothesized that interactions that are conserved among eukaryotes are more likely to be conserved in higher animals and therefore we tested the validated S. cerevisiae interactions in the model metazoan, C. elegans. We used a visual screen that monitors defects in development of the C. elegans vulva to identify synthetic genetic interactions that resulted in defects in somatic cell proliferation [30]. These vulval cell divisions occur late in nematode development [34], so perturbations do not affect organismal viability, which allows us to screen for interactions using gene mutations or RNA interference (RNAi) knockdowns that are potentially embryonic lethal. We tested the C. elegans homolog of all genes that had a verified genetic interaction with one of the cohesin genes in S. cerevisiae and for which there was an RNAi construct available (Table S1). Cohesins are essential genes, so we used a viable hypomorphic mutation in the C. elegans SMC1 ortholog, him-1(e879), and treated these worms with RNAi by feeding. Adult worms were bleached to obtain embryos that hatched into onto plates seeded with bacteria expressing different dsRNA constructs. When the embryos hatch into L1 larva they eat the dsRNA-expressing bacteria, which initiates systemic RNAi knockdown. An increased frequency of defects in the mutant treated with RNAi, compared to the predicted additive effect for the mutant and RNAi separately, is indicative of a genetic interaction. We observed a clear increase over the predicted additive frequency of Pvl in 23/28 (82%) interactions tested (Figure 2) demonstrating that most interactions are conserved between S. cerevisiae and C. elegans.
Fig. 2. C. elegans genetic interactions. 
Graph depicting the frequency of worms with a protruding vulva (Pvl) when VC2010 (WT) and SMC1/him-1(e879) strains are treated with various RNAi constructs. The RNAis tested targeted the C. elegans homologs of genes that interact with one of the cohesin query genes in the S. cerevisiae validated network (Figure 1C). Homologs with associated e-value BLAST scores can be found in Table S1. Interactions are ranked by the difference between the frequency of Pvl in VC2010 and him-1 strains. The predicted value is the sum of both the him-1 and N2 background Pvl frequencies and the effect of the RNAi on WT. unc-22 and 6 randomly chosen RNAi clones from chromosome 1 are included as negative controls. Error bars represent SEM. Analysis of cohesin network sub-groups
Each gene in the network was assigned to one of several broad functional groups based on gene descriptions reported in the Saccharomyces Genome Database (SGD). These groups were the spindle, microtubules and kinetochore (39%), mRNA processing (12%), replication factors (24%), and a group that contains genes involved in general cell metabolism (24%).
One hypothesis for a genetic interaction is that two mutations that cause the same phenotype, when combined cause a cumulative phenotype that breaches the tolerance level of the cell. One phenotype of cohesin mutants is increased CIN and we therefore investigated whether the interactors also cause CIN when mutated. For this analysis we used information collected from SGD and a recently performed screen for CIN in essential genes [35] (Table S5). We found most of the genes, except those in the metabolism group, cause CIN as measured by a variety of assays such as chromosome transmission fidelity (CTF) and gross chromosomal rearrangements (GCR) in yeast [35].
Cohesin mutations are synthetic lethal with mutations in replication fork mediators
To probe the identified interactions further, we profiled the interactions based on strength by filtering the network to include only SL interactions. We hypothesized that these interactions would indicate the most critical processes when cohesin genes are disrupted (Figure 3A). This analysis revealed five genes specifically involved in replication fork progression and stability that were SL with all three cohesin alleles tested. Ctf8p and Dcc1p are components of the alternative replication factor C Ctf18 clamp loader (altRFCCTF18) that controls the speed and restart activity of the replication fork [36]. Rad61p acts with Pds5p to bind cohesin and regulates its association with chromatin [37], [38]. Chl1p is a DNA helicase that interacts with CTF18 and physically with Eco1p [39]. Ctf4p (human AND1) has a role in coupling the Mmc2-7p helicase replication progression complex to DNA polymerase α [40]. Csm3p functions in a complex with Tof1p and Mrc1p to control stable pausing of the replication fork [41], [42]. We collectively call these genes replication fork mediators. The SL interaction of replication fork mediators with multiple cohesin mutants suggests that cohesin mutations sensitize cells to perturbations of the replication fork.
Fig. 3. Sub-optimal cohesin requires replication fork mediators. 
A) The validated S. cerevisiae cohesion network was filtered to include only SL interactions. Gene nodes are colored according to the legend in Figure 1C. B) Brood size (total number of eggs laid), embryonic lethality, and percentage of male progeny and C) Frequency of Pvl in the indicated single and double mutants. Error bars represent SEM. To investigate the biology underlying the interaction between cohesin and replication fork mediator mutations the frequency of apoptosis in the C. elegans germline was analyzed. In the C. elegans germ line, nuclei with DNA damage are removed by apoptosis and increased germline apoptosis can be indicative of increased DNA damage [43]. Apoptosis levels were quantified in the germline of the him-1(e879) mutant and were found to be elevated above that of wild type (Figure S8). When him-1 mutant animals were treated with RNAi against csm-3, rad-61, ctf-8, ctf-4 and dcc-1 an increase in apoptosis was detected. Apoptotic bodies in these cases were typically found in large clusters, rather than being distributed throughout the pachytene region, reminiscent of irradiated animals (Figure S8). These results suggested that the defects in both HIM-1 and replication fork mediator function leads to DNA damage that results in increased apoptosis.
We have shown that cohesin mutations are synthetic lethal with mutations in genes that mediate replication fork progression (Figure 1, Figure 2, Figure S8) but are not synthetic lethal with any DNA repair mutants (Table S6). These results suggest that the cohesin mutations lead to replication fork progression defects but not directly to DNA damage. This is further supported by the observation that the yeast cohesin mutations do not result in Rad52p foci accumulation, which are indicative of HR repair intermediates [44]. Additionally, elimination of the HR repair pathway by deletion of RAD51 in replication fork mediator-cohesin double mutants did not rescue lethality, suggesting that toxic recombination intermediates are not the cause for lethality in a cohesin mutant background (Figure S9). These data suggest that the interaction between cohesin mutants and fork mediators is intimately tied to the regulation of replication progression and led us to further investigate this relationship.
him-1 genetically interacts with the PARP pathway in C. elegans
The finding that replication fork mediator mutations are synthetic lethal with hypomorphic mutations in cohesin led us to hypothesize that inhibitors of replication fork stability could result in specific killing of tumors containing cohesin mutations. At present there are no small molecule inhibitors of the replication fork mediators identified in our SGA screen. However, in higher eukaryotes there are additional factors that protect and regulate the replication fork. An early mediator of replication fork stability is the family of Poly (ADP-ribose) polymerases (PARPs). PARPs have been shown to localize to stalled forks and mediate restart [45]. Furthermore, there are effective small molecule inhibitors of PARP and although PARPs are not present in yeast they are present in C. elegans. To assess whether PARP metabolism plays a role in maintaining viability in a cohesin mutant, we made double mutants with him-1(e879) and the five PAR metabolism enzyme (pme) genes in C. elegans. pme-1 and pme-2 are the C. elegans orthologs of PARP1 and PARP2, respectively [28]. pme-3 and pme-4 are homologs of Poly (ADP-ribose) glycohydrolase (PARG), which depolymerizes ADP-ribose polymers into monomeric ADP-ribose units [46]. pme-5 is the C. elegans ortholog of PARP5, which is also known as TANKYRASE [47]. All him-1; pme double mutants exhibit decreased brood sizes and an increase in the frequency of arrested embryos, suggesting that him-1 interacts with all identified members of the PAR metabolism family in C. elegans (Figure 3B). Strikingly, him-1; pme-2(ok344) double mutants had a very high frequency of protruding vulva phenotype (Figure 3C) indicative of somatic cell proliferation defects.
PARP inhibition reduces the viability of SMC1 depleted HCT116 cells
The strong negative genetic interactions in C. elegans between a hypomorphic cohesin mutation and the pme mutations prompted us to test whether this interaction is conserved in human cells using the colon cancer-derived, near diploid cell line, HCT116 [48]. We used an early generation PARP inhibitor, benzamide [49], to inhibit PARP function in a panel of HCT116 cells treated individually with siRNA against several cohesin genes (SMC1, SMC3, SCC2/NIPBL, SCC1/RAD21, SCC3/STAG1 and SCC3/STAG3). High content digital imaging microscopy (HC-DIM) was used to count Hoescht stained nuclei. Although HC-DIM is not necessary to count nuclei it allows more nuclei to be counted and more technical replicates to be performed in a timely manner. This preliminary assay suggested that HCT116 cells depleted of cohesin were sensitive to PARP inhibition (Figure S10). We further investigated this interaction using a more specific, third generation PARP inhibitor, olaparib, which has been evaluated in phase II clinical trials in the treatment of HR deficient breast and ovarian cancers [17], [19], [20]. BRCA1 siRNA treated cells were used as a positive control for PARP inhibition as BRCA1-deficient cells are highly sensitive to PARP inhibitors [15], [16]. Accordingly, we found BRCA1 siRNA treated cells were very sensitive to olaparib (Figure 4). We also found by visual inspection that SMC1 siRNA treated cells exhibited reduced cell proliferation in response to a range of olaparib concentrations using a 24 well plate survival assay whereas untreated and GAPDH siRNA treated cells appeared only mildly affected (Figure 4A). We quantified the specific sensitivity using an expanded 10 cm dish survival assay where cells treated with siRNAs were continuously exposed to 0.6 µM olaparib and colonies were stained and counted after a 10 day period (Figure 4B, 4C, 4E). We also saw evidence that HCT116 SMC1 knockdown cells exposed to olaparib exhibited proliferation defects using HC-DIM. Overall, we observed a significant dose dependent decrease in cell number in the BRCA1 and SMC1 treated cells as compared to GAPDH (Figure 4D).
Fig. 4. SMC1 siRNA treated human cells are sensitive to the PARP inhibitor olaparib. 
All experiments were performed with HCT116, a near diploid, colon cancer derived cell line that is MMR deficient. In all panels HCT116 cells are treated with siRNA targeting GAPDH, BRCA1 or SMC1 or untreated (in indicated). A) 24 well plate clonagenic survival assay with HCT116 siRNA treated cells exposed to olaparib concentrations up to 1.5 µM. B) 10 cm dish clonagenic survival assay looking at the number of colonies after 10 days in the presence of 0.6 µM olaparib. C) Normalized colony numbers from the clonagenic assay. D) HC-DIM counting Hoescht positive nuclei of siRNA treated cells after 3 days of olaparib exposure. E) Western blot of samples collected from the 10 cm survival assay (B). Error bars represent SEM. PARP inhibition reduces viability in HTB-38, a human cell line with low parylation levels
We were concerned that the effect of PARP inhibition was complicated by the fact that HCT116 cells have a high level of endogenous PARP activity due to deficiencies in mismatch repair (MMR) [50], [51]. Consistent with previous reports, we observed relatively high PAR levels, which are indicative of PARP activity, in untreated HCT116 cells (Figure 5A). We also found that HCT116 cells treated with 0.6 µM olaparib showed a 54% decrease in viability as measured by a colony forming assay. To rule out the confounding effects of elevated PAR in the HCT116 cell line, we repeated the colony forming assay, with and without olaparib treatment, on a second colon cancer-derived cell line that was MMR-proficient, HTB-38. First, we confirmed that, as previously reported, HTB-38 cell lines did not exhibit increased PAR levels (Figure 5A). We found that GAPDH and untransfected HTB-38 cells were insensitive to olaparib (Figure 5B, 5C) unlike the HCT116 cells that showed mild sensitivity to olaparib (Figure 4B). However, olaparib treatment decreased viability in the HTB-38 cells treated with siRNA targeting either BRCA1 or SMC1, confirming the sensitivity to PARP inhibitors of SMC1-depleted cells. To determine whether the sensitivity to PARP inhibition was limited to SMC1 or was more general, extending to defects in other cohesin components, we treated cells with siRNA targeting the cohesin components SMC3 or RAD21/SCC1. We observed growth defects of similar strengths among all cohesin subunits tested. These experiments demonstrate that cells with cohesin defects are sensitive to PARP inhibition.
Fig. 5. SMC1 and PARP genetically interact in HTB-38 human cells. 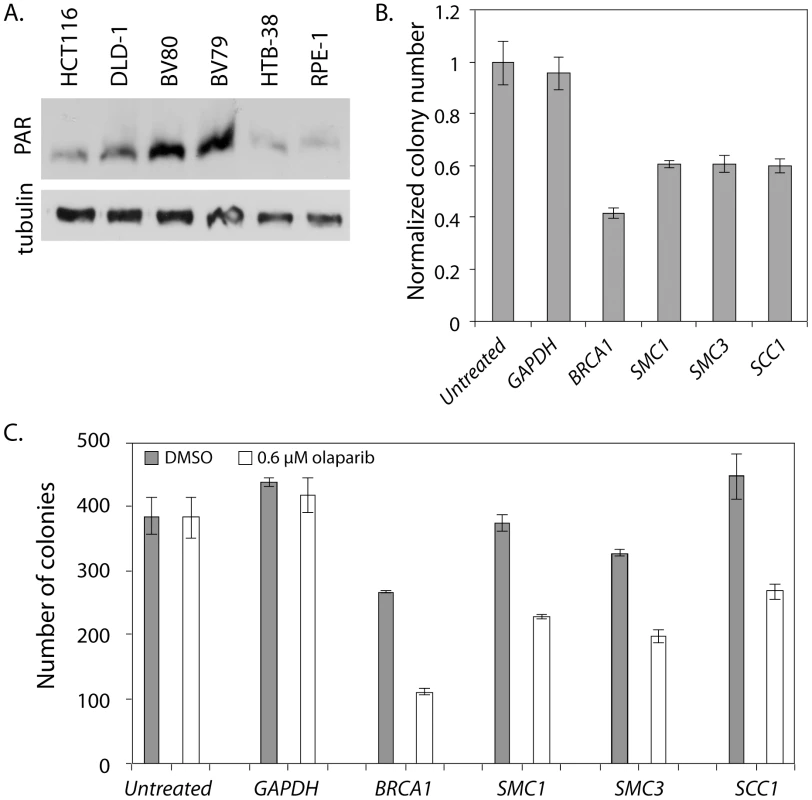
All experiments were performed with HTB-38, a near triploid colon cancer derived cell line that is MMR proficient. A) Western blot stained with anti-PAR. BV80 is MLH1−/− and is the matched line to BV79 which is MLH1+/−. RPE-1 was used as a control cell line for measuring PAR levels. RPE-1 is an hTERT immortalized retina epithelial cell line. B) Normalized colony numbers for HTB-38 cells treated with the indicated siRNA and exposed to 0.6 µM olaparib. C) Raw colony numbers for the clonagenic assay shown in B. Error bars represent SEM. Discussion
Cohesin dysfunction appears to have a significant role in the formation and progression of tumors (Reviewed in [1]). Here we show that replication fork stability genes are required for viability in cohesin mutants across species. Using data from yeast SGA analyses we were able to predict synthetic lethal interactions in human cells between cohesin mutations and PARP inhibitors, even though PARP is not present in yeast, demonstrating the power of large-scale genetic interaction screens for synthetic lethality with genes mutated in tumors.
The spectrum of cohesin genetic interactions
SL interactions in model organisms can identify candidate genes or pathways that can be targeted for inhibition leading to specific killing of tumor cells with specific mutations [13]. We used SGA technology in yeast to generate a network of negative genetic interactions, using hypomorphic mutations in cohesin as query genes, with the aim of elucidating the genetic pathways needed for survival when cohesin function is compromised. By overlaying the results from three separate SGA screens, using two cohesin core components (SMC1 and SCC1) and one cohesin loader (SCC2) as queries, we identified common interactions with compromised cohesin rather than those that were allele or component-specific. While the screens found scores of single negative interactions with each of the three query mutants (Figure 1A), the goal of this study was to identify interactions that are more likely to be SL with the wide-range of cohesin-associated mutations observed in human tumors. Negative genetic interactions specific to only one of the three cohesin mutants may reveal specific aspects of cohesin components and these interactions warrant further investigation.
The filtered S. cerevisiae interaction set identifies many processes that would be predicted to interact with cohesin dysfunction. For example, mutations affecting the mitotic spindle, kinetochore and microtubules, including prefoldin, were found to result in synthetic growth defects with at least two of the three query mutants. Given the role of cohesin in regulating chromosome segregation leading to the anaphase transition [9], mutations affecting the spindle or kinetochore would be predicted to interact with cohesin mutations. Other interactions that do not appear to be related to cohesin function may represent more general effects on cell viability; for example ten interactions were identified with components of mRNA processing and the translation machinery. In these cases, further work will be needed to ascertain whether this is a general synergistic effect on viability or whether the interaction results in a specific defect.
When interactions were filtered based on strength, keeping only those interactions that were SL, the predominant genes were those involved in mediating replication fork progression. These included two of the three genes in the alternative RFCCTF18, the replisome components CSM3 and CTF4, the replication and cohesion-associated helicase CHL1, and the cohesin regulator RAD61. All of these interactors are known to mediate replication fork stability and progression [36]–[42], [52]–[54]. Furthermore, mutations affecting these genes also result in chromosome cohesion defects [55], thereby linking replication fork stability and sister chromatid cohesion. The SL interactions are not specific to mutations that accelerate or impair replication fork progression. In fact, the Alt-RFCCTF18, which promotes replication fork progression, does so by regulating the acetylation of Smc3p [36], which in turn inhibits the association of Smc3p and Rad61p [52], [53]. In contrast to the Alt-RFCCTF18, Rad61p binding to Smc3p slows replication fork progression [36]. Rad61p is known to destabilize the association between cohesin and chromosomes [38]. The requirement for RAD61 in the cohesin mutants is particularly interesting given the correlation of elevated expression of the human RAD61 ortholog, WAPL, with certain cancers. Its importance in maintaining the viability of tumor cells was demonstrated by the fact that knockdown of WAPL in cervical cancer cell lines resulted in cell death [4]. Similarly, we observe synthetic lethality when we knockout RAD61 in the cohesin mutants, thereby demonstrating that Rad61p function is needed when cohesin function is compromised.
The synthetic lethality of the cohesin mutations with replication fork mediator mutations suggested that the replisome must be stabilized when cohesin is mutated to ensure proper progression. Interestingly, cohesin mutants did not show negative genetic interactions with DNA repair genes such as the RAD52-complementation group of HR genes, the RecQ helicase SGS1, or the structure-specific endonuclease MUS81. In addition, the cohesin mutants do not accumulate Rad52p foci [44], which is indicative of increased DNA damage or DNA repair intermediates. These data suggested that cohesin mutations on their own do not lead to DNA double strand breaks or fork collapse, both of which require HR for resolution. Cohesin dysfunction may impact replication fork dynamics, but these events are tolerable in the presence of fork stabilizing proteins. Cohesin may also have a role in modulating fork progression. As vertebrate replicons span between approximately 60 and 140 kb [56], [57] and cohesin is associated, on average, with DNA every 10–20 kb [58]–[61], each fork must theoretically pass several cohesin complexes [36]. One mechanism to prevent fork collapse is to minimize the distance between the leading strand helicase and the polymerase. If the polymerase stalls and the helicase continues to advance, single stranded DNA is exposed and the fork becomes more fragile. Rad61p association with cohesin is thought to induce a closed cohesin conformation that limits fork progression [36]. When the cohesin ring changes to a more restrictive conformation it may prevent helicase and polymerase separation. Support for cohesin moderating fork progression comes from the finding that human SMC1 is phosphorylated by ATR in response to S phase stress [62] and that this modification is required for activation of the replication checkpoint.
PARP inhibition reduces the viability of cohesin-depleted cells
In higher eukaryotes, additional factors regulate replication fork stability and progression, one example being the family of poly(ADP-ribose) polymerases (PARPs). PARPs play a major role in DNA metabolism including aspects of repair and replication (reviewed in [63]). PARP was recently found to be activated by stalled replication forks and promoted replication fork restart [45]. We found that loss of PARP and PARG orthologs synergized with a mutation in him-1/SMC1 in C. elegans resulting in a decrease in cell and organismal viability. In human cells, depletion of SMC1, SMC3 or SCC1 by siRNA caused sensitivity to the PARP-inhibitor olaparib. Interestingly, this sensitivity was comparable to that observed when we depleted BRCA1 by siRNA and then treated cells with olaparib. The interaction between multiple cohesin subunits and PARP was found to be cell line independent, suggesting a robust interaction. Recent emerging hypotheses have proposed alternate fork restart pathways where one branch is HR mediated and the other requires PARP [63]. It has been suggested that BRCA1/2 are synthetic lethal with PARP1/2 because of an inability to restart stalled or collapsed forks. This hypothesis is interesting given the connection we observe between cohesin and fork progression mediators.
Given our findings regarding the importance of replication fork mediators in the presence of cohesin mutations, it is possible that PARP activity is needed for replication fork stability or lesion bypass in the cohesin mutants or knockdown cell lines. However, while synthetic lethal interactions between cohesin mutations and replication fork mediators led to testing PARP inhibitors, the mechanism of the PARP-cohesin interaction is not known because PARP and cohesin each have multiple functions that could be co-dependent. For example, it is possible that the synthetic lethality of cohesin knockdown with PARP inhibition is related to the role of cohesin in HR [64] PARP inhibition is known to be effective in killing cells with defective HR, such as BRCA1, BRCA2 and ATM-deficient cells [15], [16]. It is possible that the synthetic lethality of cohesin knockdown and PARP inhibition is due to either or both mechanisms. Although further investigation is needed to identify the specific mechanism of lethality, our data indicate PARP inhibition is a potential treatment for tumors with cohesin or cohesin-related mutations, which represent a significant proportion of colorectal, ovarian, breast and other tumors [1]–[3].
Materials and Methods
S. cerevisiae strain construction and SGA screens
Temperature sensitive (ts) cohesin alleles were marked with URA3 as described previously [65], [66] and were used as query genes in SGA screens. Query genes were screened against the non-essential deletion collection [31] and a collection of DAmP [33] and ts [32] alleles representing essential genes. SGA screens were performed in biological triplicate, each with three technical replicates using a Singer RoToR essentially as described [67]. SCC1 and SCC2 were screened at 30°C while SMC1 was screened at 25°C due to slow growth at higher temperatures. See Tables S7 and S8 for a full list of S. cerevisiae strains used in this study.
Double mutant reconstruction and random spore analysis
Double heterozygous mutants were recreated by mating each of the single mutants and selecting on −ura, +G418 plates. The cohesin mutant parent strains were the same query genes used for SGA screens and the other parent was pulled directly from the array plates. Heterozygotes were confirmed by PCR analysis to confirm the identity of the array strain. External primers, unique to the upstream and downstream sequences of each gene locus assayed, were used in PCR reactions in most cases. Random spore was performed at 25°C as previously described [30]. Tetrad analysis would have been a true independent measure of a genetic interaction but the cohesin mutants, singly, exhibit relatively high rates of random spore death, making tetrad analysis infeasible on this scale. Briefly, spores were plated onto haploid selection plates containing canavanine and thialysine. Single selection plates were additionally either −ura or +G418. Double mutants that did not grow on the double selection plates (−ura, +G418) were considered SL. Several isolates of viable double mutants were isolated from random spore analysis for growth curve analysis.
S. cerevisiae growth curves
All viable double mutants were analyzed by growth curve analysis as previously described [30] with slight modifications. Briefly, strains were grown overnight in YPD, diluted to an optical density600 nm (OD) of 0.2 and incubated for 4 hours at 25°C. Strains were then diluted to an OD of 0.3 and 100 ul was added to each well of a 96 well plate. 100 ul of fresh YPD was added to each well for a final OD of 0.15. For each plate, fifteen replicates were performed for wild type (WT), while three replicates were performed for each of the other strains analyzed. OD readings were made every 30 min, after 3 min of shaking, over a period of 24 hrs at 26°C (5 plates) or 30°C (6 plates) on a Tecan M1000. Growth curves for each strain can be found in Figure S3.
Growth curve analysis
Strain fitness was defined as the logarithm of the area under the curve (AUC) and was calculated using Simpson's rule in R [68]. Separate, parallel analyses were performed for plates grown at 26°C and 30°C. Measurements were normalized for plate effects such that the average estimated strain fitness for the wild type curves was constant across each set. Interaction effects were assessed with a linear model of the form: is the fitness for a double-knockout of query gene and non-essential gene , is the fitness for the wild type growth curves, is the single-knockout effect for each of the three query genes (scc1-73, smc1-259, scc2-4), is the single-knockout effect for each of the other non-essential genes assessed, and is the double-knockout interaction effect associated with genes and . Under additive neutrality, non-interacting gene pairs will have an interaction term of zero:Values of indicate SS interactions, while values of indicate alleviating interactions. After fitting this model, the significance of the estimated interaction effects was assessed. See Text S1 and Tables S2, S3, S4 and Figures S4, S5, S6, S7 for additional details and statistical analysis.
Homolog identification
Homology was determined using protein BLAST to query the S. cerevisiae protein sequence against the non-redundant protein sequences database using default parameters. S. cerevisiae ORF translation sequences were obtained from the Saccharomyces Genome Database (SGD) and Homo sapiens (taxid:9606) and Caenorhabditis elegans (taxid:6239) organisms were queried for sequence matches. The algorithm used was blastp (protein-protein BLAST) and all other parameters were set as default settings. Homology was considered to exist if an identified match had an expect value less than 1e-05. In cases where no homologous match was identified in either C. elegans or H. sapiens, ‘No human, C. elegans match’ was entered into Table S1. In some cases, such as with the S. cerevisiae gene DCC1, no direct C. elegans sequence match could be found. However, if the identified H. sapiens protein sequence match was used as a BLAST query against the C. elegans database a match, K09H9.2, could be obtained. In analogous situations, such as with the S. cerevisiae gene MDM20, the C. elegans sequence obtained through BLASTp was used to query the human database for homologous sequences. In these cases if the identified match had an expect value greater than 1e-05 the literature was consulted to determine whether the identified protein was considered homologous by other independent groups. An example where the literature was consulted was to identify the human homolog of S. cerevisiae RAD61, WPL-1.
C. elegans genetic interactions and SYTO12 staining
The Pvl somatic cell proliferation assay was performed as previously described [30]. SYTO12 staining was performed on young adult worms that had been pre-treated with RNAi by feeding by bleaching gravid adults onto RNAi plates. Worms were incubated in the dark with 33 µM SYTO12 in M9 buffer for 3 hrs and then destained for 1 hour on fresh RNAi plates. Images were captured immediately after destaining on a Zeiss Axioplan 2 microscope using a 40× lens.
C. elegans double mutant strain construction and brood analysis
See Table S9 for strains used. him-1(e879) males were mated to either pme-1(ok988), pme-2(ok344), pme-2(tm3401), pme-3(gk120), pme-4(ok980) or pme-5(ok446) hermaphrodites. Homozygous him-1 mutants were followed by the high frequency of males (∼10%) and all other mutations were followed by PCR. pme-1 him-1 animals were balanced by hT2 and homozygous him-1; pme-2(ok344) animals were balanced by mIn1.
Cell culture and siRNA
Cells were cultured as previously described [14]. siRNAs and transfection reagents were purchased from Dharmacon. For siRNA transfection experiments 300,000 and 550,000 cells were seeded into 6-well dishes for HCT116 and HTB-38 cell lines, respectively. Transfection occurred 24 hours after seeding using ON-TARGETplus 25 nM siRNA pools. Transfection reagent was removed 12 hours post-transfection and following an additional 6–8 hours of recovery cells were transferred to fresh culture dishes and allowed to expand for 3–10 days, depending on the assay. For the clonogenic survival assays 1000 and 500 cells were seeded per well in 24 well dishes and 10 cm plates, respectively. Cells were allowed to attach for 10 hours and then olaparib was added at the indicated concentration. Cells were fixed with 4% paraformaldehyde (PFA) in phosphate buffered saline (PBS) and stained using 0.1% crystal violet in 95% ethanol after 7 and 10 days for the 24 well plate and 10 cm dishes, respectively for HCT116. HTB-38 cells are slower growing and were stained with crystal violet at day 12. Drug exposure was continuous during this time and the 10 cm dishes were supplemented with fresh media containing 0.6 µM olaparib at day 5. High content digital imaging microscopy (HC-DIM) assays were performed by plating 1000 cells per well in 96 well plates. Cells were allowed to settle for 10 hours and then either benzamide or olaparib was added at the indicated concentration. Cells were fixed with PFA after 3 days of drug exposure and stained with Hoescht dye #33258 (Molecular Probes). Plates were subjected to HC-DIM using a Cellomics ArrayScan with a 20× (81 images/well) or 10× (16 images/well) dry lens. The total number of Hoescht-positive nuclei was determined using a Cellomics Target Activation algorithm and normalized to each siRNA treatment (GAPDH, BRCA1 or SMC1). Western blots were performed on protein extracts collected from asynchronous, sub-confluent cells harvested 3 days post-transfection as detailed previously [2]. Olaparib was purchased from Selleck and Benzamide was purchased from Aldrich. Antibodies were purchased from Millipore (BRCA1 07-434), Abcam (SMC1 ab9262, alpha Tubulin ab56476) and Trevigen (PAR 4336-BPC-100). HTB-38 and RPE-1 cells were purchased from ATCC and BV79 and BV80 were kindly provided by B. Vogelstein.
Supporting Information
Zdroje
1. XuHYanMPatraJNatrajanRYanY 2011 Enhanced RAD21 cohesin expression confers poor prognosis and resistance to chemotherapy in high grade luminal, basal and HER2 breast cancers. Breast Cancer Res 13 1 R9
2. BarberTDMcManusKYuenKWReisMParmigianiG 2008 Chromatid cohesion defects may underlie chromosome instability in human colorectal cancers. Proc Natl Acad Sci U S A 105 9 3443 3448
3. SolomonDAKimTDiaz-MartinezLAFairJElkahlounAG 2011 Mutational inactivation of STAG2 causes aneuploidy in human cancer. Science 333 6045 1039 1043
4. OikawaKOhbayashiTKiyonoTNishiHIsakaK 2004 Expression of a novel human gene, human wings apart-like (hWAPL), is associated with cervical carcinogenesis and tumor progression. Cancer Res 64 10 3545 3549
5. GhiselliGIozzoRV 2000 Overexpression of bamacan/SMC3 causes transformation. J Biol Chem 275 27 20235 20238
6. ZhangNGeGMeyerRSethiSBasuD 2008 Overexpression of separase induces aneuploidy and mammary tumorigenesis. Proc Natl Acad Sci U S A 105 35 13033 13038
7. HagemannCWeigelinBSchommerSSchulzeMAl-JomahN 2011 The cohesin-interacting protein, precocious dissociation of sisters 5A/sister chromatid cohesion protein 112, is up-regulated in human astrocytic tumors. Int J Mol Med 27 1 39 51
8. SheltzerJMBlankHMPfauSJTangeYGeorgeBM 2011 Aneuploidy drives genomic instability in yeast. Science 333 6045 1026 1030
9. MichaelisCCioskRNasmythK 1997 Cohesins: Chromosomal proteins that prevent premature separation of sister chromatids. Cell 91 1 35 45
10. UhlmannFLottspeichFNasmythK 1999 Sister-chromatid separation at anaphase onset is promoted by cleavage of the cohesin subunit Scc1. Nature 400 6739 37 42
11. UhlmannFWernicDPoupartMAKooninEVNasmythK 2000 Cleavage of cohesin by the CD clan protease separin triggers anaphase in yeast. Cell 103 3 375 386
12. NasmythKHaeringCH 2009 Cohesin: Its roles and mechanisms. Ann Rev Genet 43 525 558
13. HartwellLHSzankasiPRobertsCJMurrayAWFriendSH 1997 Integrating genetic approaches into the discovery of anticancer drugs. Science 278 1064 8
14. McManusKBarrettINouhiYHieterP 2009 Specific synthetic lethal killing of RAD54B-deficient human colorectal cancer cells by FEN1 silencing. PNAS 106 3276 81
15. BryantHESchultzNThomasHDParkerKMFlowerD 2005 Specific killing of BRCA2-deficient tumors with inhibitors of poly(ADP-ribose) polymerase. Nature 434 7035 913 917
16. FarmerHMcCabeNLordCJTuttANJohnsonDA 2005 Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature 434 7035 917 21
17. AudehMWCarmichaelJPensonRTFriedlanderMPowellB 2010 Oral poly(ADP-ribose) polymerase inhibitor olaparib in patients with BRCA1 or BRCA2 mutations and recurrent ovarian cancer: A proof-of-concept trial. Lancet 376 9737 245 251
18. O'ShaughnessyJOsborneCPippenJEYoffeMPattD 2011 Iniparib plus chemotherapy in metastatic triple-negative breast cancer. N Engl J Med 364 3 205 214
19. KhanOAGoreMLoriganPStoneJGreystokeA 2011 A phase I study of the safety and tolerability of olaparib (AZD2281, KU0059436) and dacarbazine in patients with advanced solid tumors. Br J Cancer 104 5 750 755
20. TuttARobsonMGarberJEDomchekSMAudehMW 2010 Oral poly(ADP-ribose) polymerase inhibitor olaparib in patients with BRCA1 or BRCA2 mutations and advanced breast cancer: A proof-of-concept trial. Lancet 376 9737 235 244
21. CostanzoMBaryshnikovaABellayJKimYSpearED 2010 The genetic landscape of a cell. Science 327 5964 425 431
22. TongAHEvangelistaMParsonsABXuHBaderGD 2001 Systematic genetic analysis with ordered arrays of yeast deletion mutants. Science 294 2364 8
23. VogelsteinBKinzlerK 2004 Cancer genes and the pathways they control. Nat Med 10 789 799
24. CollinsSRMillerKMMaasNLRoguevAFillinghamJ 2007 Functional dissection of protein complexes involved in yeast chromosome biology using a genetic interaction map. Nature 446 7137 806 10
25. BoultonSJMartinJSPolanowskaJHillDEGartnerA 2004 BRCA1/BARD1 orthologs required for DNA repair in Caenorhabditis elegans. Curr Biol 14 1 33 39
26. MartinJSWinkelmannNPetalcorinMIMcIlwraithMJBoultonSJ 2005 RAD-51-dependent and -independent roles of a Caenorhabditis elegans BRCA2-related protein during DNA double-strand break repair. Mol Cell Biol 25 8 3127 3139
27. DerryWBPutzkeAPRothmanJH 2001 Caenorhabditis elegans p53: Role in apoptosis, meiosis, and stress resistance. Science 294 5542 591 595
28. GagnonSNHengartnerMODesnoyersS 2002 The genes pme-1 and pme-2 encode two poly(ADP-ribose) polymerases in Caenorhabditis elegans. Biochem J 368 Pt 1 263 271
29. KirienkoNVManiKFayDS 2010 Cancer models in Caenorhabditis elegans. Dev Dyn 239 5 1413 1448
30. McLellanJO'NeilNTarailoSStoepelJBryanJ 2009 Synthetic lethal genetic interactions that decrease somatic cell proliferation in Caenorhabditis elegans identify the alternative RFC CTF18 as a candidate cancer drug target. Mol Biol Cell 20 24 5306 5313
31. WinzelerEAShoemakerDDAstromoffALiangHAndersonK 1999 Functional characterization of the S. cerevisiae genome by gene deletion and parallel analysis. Science 285 5429 901 906
32. LiZVizeacoumarFJBahrSLiJWarringerJ 2011 Systematic exploration of essential yeast gene function with temperature-sensitive mutants. Nat Biotechnol 29 4 361 367
33. BreslowDKCameronDMCollinsSRSchuldinerMStewart-OrnsteinJ 2008 A comprehensive strategy enabling high-resolution functional analysis of the yeast genome. Nat Methods 5 8 711 718
34. SulstonJEHorvitzHR 1977 Post-embryonic cell lineages of the nematode, Caenorhabditis elegans. Developmental Biology 56 1 110 56
35. StirlingPCBloomMSSolanki-PatilTSmithSSipahimalaniP 2011 The complete spectrum of yeast chromosome instability genes identifies candidate CIN cancer genes and functional roles for ASTRA complex components. PLoS Genet 7 e1002057 doi:10.1371/journal.pgen.1002057
36. TerretMESherwoodRRahmanSQinJJallepalliPV 2009 Cohesin acetylation speeds the replication fork. Nature 462 7270 231 234
37. KuengSHegemannBPetersBHLippJJSchleifferA 2006 Wapl controls the dynamic association of cohesin with chromatin. Cell 127 5 955 967
38. SutaniTKawaguchiTKannoRItohTShirahigeK 2009 Budding yeast Wpl1(Rad61)-Pds5 complex counteracts sister chromatid cohesion-establishing reaction. Curr Biol 19 6 492 497
39. SkibbensRV 2004 Chl1p, a DNA helicase-like protein in budding yeast, functions in sister-chromatid cohesion. Genetics 166 1 33 42
40. TanakaHKatouYYaguraMSaitohKItohT 2009 Ctf4 coordinates the progression of helicase and DNA polymerase alpha. Genes Cells 14 7 807 820
41. BandoMKatouYKomataMTanakaHItohT 2009 Csm3, Tof1, and Mrc1 form a heterotrimeric mediator complex that associates with DNA replication forks. J Biol Chem 284 49 34355 34365
42. KomataMBandoMArakiHShirahigeK 2009 The direct binding of Mrc1, a checkpoint mediator, to Mcm6, a replication helicase, is essential for the replication checkpoint against methyl methanesulfonate-induced stress. Mol Cell Biol 29 18 5008 5019
43. GartnerAMacQueenAJVilleneuveAM 2004 Methods for analyzing checkpoint responses in Caenorhabditis elegans. Methods Mol Biol 280 257 274
44. StirlingPCChanYAMinakerSWAristizabalMJBarrettI 2012 R-loop mediated genome instability in mRNA cleavage and polyadenylation mutants. Genes Dev., Epub Jan. 15, 2012
45. BryantHEPetermannESchultzNJemthASLosevaO 2009 PARP is activated at stalled forks to mediate Mre11-dependent replication restart and recombination. EMBO J 28 17 2601 2615
46. St-LaurentJFGagnonSNDequenFHardyIDesnoyersS 2007 Altered DNA damage response in Caenorhabditis elegans with impaired poly(ADP-ribose) glycohydrolases genes expression. DNA Repair (Amst) 6 3 329 343
47. GravelCStergiouLGagnonSNDesnoyersS 2004 The C. elegans gene pme-5: Molecular cloning and role in the DNA-damage response of a tankyrase orthologue. DNA Repair (Amst) 3 2 171 182
48. BrattainMGFineWDKhaledFMThompson BrattainDE 1981 Heterogeneity of malignant cells from a human colonic carcinoma. Cancer Res 41 1751 1756
49. LiJHZhangJ 2001 PARP inhibitors. IDrugs 4 7 804 812
50. TentoriLLeonettiCScarsellaMMuziAMazzonE 2006 Inhibition of poly(ADP-ribose) polymerase prevents irinotecan-induced intestinal damage and enhances irinotecan/temozolomide efficacy against colon carcinoma. FASEB J 20 10 1709 1711
51. TakahashiMKoiMBalaguerFBolandCRGoelA 2011 MSH3 mediates sensitization of colorectal cancer cells to cisplatin, oxaliplatin, and a poly(ADP-ribose) polymerase inhibitor. J Biol Chem 286 14 12157 12165
52. BeckouetFHuBRoigMBSutaniTKomataM 2010 An Smc3 acetylation cycle is essential for establishment of sister chromatid cohesion. Mol Cell 39 5 689 699
53. RowlandBDRoigMBNishinoTKurzeAUluocakP 2009 Building sister chromatid cohesion: Smc3 acetylation counteracts an antiestablishment activity. Mol Cell 33 6 763 74
54. Rolef Ben-ShaharTHeegerSLehaneCEastPFlynnH 2008 Eco1-dependent cohesin acetylation during establishment of sister chromatid cohesion. Science 321 5888 563 566
55. MayerMLPotIChangMXuHAneliunasV 2004 Identification of protein complexes required for efficient sister chromatid cohesion. Mol Biol Cell 15 4 1736 1745
56. WalterJNewportJW 1997 Regulation of replicon size in xenopus egg extracts. Science 275 5302 993 995
57. BlumenthalABKriegsteinHJHognessDS 1974 The units of DNA replication in drosophila melanogaster chromosomes. Cold Spring Harb Symp Quant Biol 38 205 223
58. LalorayaSGuacciVKoshlandD 2000 Chromosomal addresses of the cohesin component Mcd1p. J Cell Biol 151 5 1047 1056
59. ParelhoVHadjurSSpivakovMLeleuMSauerS 2008 Cohesins functionally associate with CTCF on mammalian chromosome arms. Cell 132 3 422 433
60. WendtKSPetersJM 2009 How cohesin and CTCF cooperate in regulating gene expression. Chromosome Res 17 2 201 214
61. WendtKSYoshidaKItohTBandoMKochB 2008 Cohesin mediates transcriptional insulation by CCCTC-binding factor. Nature 451 7180 796 801
62. KimSTXuBKastanMB 2002 Involvement of the cohesin protein, Smc1, in atm-dependent and independent responses to DNA damage. Genes Dev 16 5 560 570
63. HelledayT 2011 The underlying mechanism for the PARP and BRCA synthetic lethality: Clearing up the misunderstandings. Mol Oncol 5 4 387 393
64. SjogrenCNasmythK 2001 Sister chromatid cohesion is required for postreplicative double-strand break repair in Saccharomyces cerevisiae. Curr Biol 11 12 991 995
65. Ben-AroyaSCoombesCKwokTO'DonnellKABoekeJD 2008 Toward a comprehensive temperature-sensitive mutant repository of the essential genes of Saccharomyces cerevisiae. Mol Cell 30 248 58
66. Ben-AroyaSPanXBoekeJDHieterP 2010 Making temperature-sensitive mutants. Methods Enzymol 470 181 204
67. TongAHLesageGBaderGDDingHXuH 2004 Global mapping of the yeast genetic interaction network. Science 303 5659 808 13
68. R Development Core Team: R Foundation for Statistical Computing. 2008 R: A language and environment for statistical computing.
Štítky
Genetika Reprodukční medicína
Článek Physiological Notch Signaling Maintains Bone Homeostasis via RBPjk and Hey Upstream of NFATc1Článek Intronic -Regulatory Modules Mediate Tissue-Specific and Microbial Control of / TranscriptionČlánek Probing the Informational and Regulatory Plasticity of a Transcription Factor DNA–Binding DomainČlánek Repression of Germline RNAi Pathways in Somatic Cells by Retinoblastoma Pathway Chromatin ComplexesČlánek An Alu Element–Associated Hypermethylation Variant of the Gene Is Associated with Childhood ObesityČlánek Three Essential Ribonucleases—RNase Y, J1, and III—Control the Abundance of a Majority of mRNAsČlánek Genomic Tools for Evolution and Conservation in the Chimpanzee: Is a Genetically Distinct Population
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2012 Číslo 3- Kazuistika – Perspektivy využití precizované medicíny v rámci personalizované specifické terapie onkologických pacientů
- Nobelova cena za chemii pro genetické nůžky: Objev, který změní naši budoucnost?
- Technologie na bázi RNA v klinické praxi: od přebarvených petúnií k terapii vzácných a dosud jen obtížně léčitelných chorob u lidí
- „Nepředstavovali jsme si, že náš výzkum povede přímo ke vzniku nových léků, dokonce ještě za našeho života“
- Bezplatné služby pro diagnostiku ATTRv amyloidózy pro kardiology
-
Všechny články tohoto čísla
- Comprehensive Research Synopsis and Systematic Meta-Analyses in Parkinson's Disease Genetics: The PDGene Database
- Genomic Analysis of the Hydrocarbon-Producing, Cellulolytic, Endophytic Fungus
- Networks of Neuronal Genes Affected by Common and Rare Variants in Autism Spectrum Disorders
- Akirin Links Twist-Regulated Transcription with the Brahma Chromatin Remodeling Complex during Embryogenesis
- Too Much Cleavage of Cyclin E Promotes Breast Tumorigenesis
- Imprinted Genes … and the Number Is?
- Genetic Architecture of Highly Complex Chemical Resistance Traits across Four Yeast Strains
- Exploring the Complexity of the HIV-1 Fitness Landscape
- MNS1 Is Essential for Spermiogenesis and Motile Ciliary Functions in Mice
- A Fundamental Regulatory Mechanism Operating through OmpR and DNA Topology Controls Expression of Pathogenicity Islands SPI-1 and SPI-2
- Evidence for Positive Selection on a Number of MicroRNA Regulatory Interactions during Recent Human Evolution
- Variation in Modifies Risk of Neonatal Intestinal Obstruction in Cystic Fibrosis
- PIF4–Mediated Activation of Expression Integrates Temperature into the Auxin Pathway in Regulating Hypocotyl Growth
- Critical Evaluation of Imprinted Gene Expression by RNA–Seq: A New Perspective
- A Meta-Analysis and Genome-Wide Association Study of Platelet Count and Mean Platelet Volume in African Americans
- Mouse Genetics Suggests Cell-Context Dependency for Myc-Regulated Metabolic Enzymes during Tumorigenesis
- Transcriptional Control in Cardiac Progenitors: Tbx1 Interacts with the BAF Chromatin Remodeling Complex and Regulates
- Synthetic Lethality of Cohesins with PARPs and Replication Fork Mediators
- APOBEC3G-Induced Hypermutation of Human Immunodeficiency Virus Type-1 Is Typically a Discrete “All or Nothing” Phenomenon
- Interpreting Meta-Analyses of Genome-Wide Association Studies
- Error-Prone ZW Pairing and No Evidence for Meiotic Sex Chromosome Inactivation in the Chicken Germ Line
- -Dependent Chemosensory Functions Contribute to Courtship Behavior in
- Diverse Forms of Splicing Are Part of an Evolving Autoregulatory Circuit
- Phenotypic Plasticity of the Drosophila Transcriptome
- Physiological Notch Signaling Maintains Bone Homeostasis via RBPjk and Hey Upstream of NFATc1
- Precocious Metamorphosis in the Juvenile Hormone–Deficient Mutant of the Silkworm,
- Igf1r Signaling Is Indispensable for Preimplantation Development and Is Activated via a Novel Function of E-cadherin
- Accurate Prediction of Inducible Transcription Factor Binding Intensities In Vivo
- Mitochondrial Oxidative Stress Alters a Pathway in Strongly Resembling That of Bile Acid Biosynthesis and Secretion in Vertebrates
- Mammalian Neurogenesis Requires Treacle-Plk1 for Precise Control of Spindle Orientation, Mitotic Progression, and Maintenance of Neural Progenitor Cells
- Tcf7 Is an Important Regulator of the Switch of Self-Renewal and Differentiation in a Multipotential Hematopoietic Cell Line
- REST–Mediated Recruitment of Polycomb Repressor Complexes in Mammalian Cells
- Intronic -Regulatory Modules Mediate Tissue-Specific and Microbial Control of / Transcription
- Age-Dependent Brain Gene Expression and Copy Number Anomalies in Autism Suggest Distinct Pathological Processes at Young Versus Mature Ages
- A Genome-Wide Association Study Identifies Variants Underlying the Shade Avoidance Response
- -by- Regulatory Divergence Causes the Asymmetric Lethal Effects of an Ancestral Hybrid Incompatibility Gene
- Genome-Wide Association and Functional Follow-Up Reveals New Loci for Kidney Function
- A Natural System of Chromosome Transfer in
- Cell Size and the Initiation of DNA Replication in Bacteria
- Probing the Informational and Regulatory Plasticity of a Transcription Factor DNA–Binding Domain
- Repression of Germline RNAi Pathways in Somatic Cells by Retinoblastoma Pathway Chromatin Complexes
- Temporal Transcriptional Profiling of Somatic and Germ Cells Reveals Biased Lineage Priming of Sexual Fate in the Fetal Mouse Gonad
- Rapid Analysis of Genome Rearrangements by Multiplex Ligation–Dependent Probe Amplification
- Metabolic Profiling of a Mapping Population Exposes New Insights in the Regulation of Seed Metabolism and Seed, Fruit, and Plant Relations
- The Atypical Calpains: Evolutionary Analyses and Roles in Cellular Degeneration
- The Silkworm Coming of Age—Early
- Development of a Panel of Genome-Wide Ancestry Informative Markers to Study Admixture Throughout the Americas
- Balanced Codon Usage Optimizes Eukaryotic Translational Efficiency
- The Min System and Nucleoid Occlusion Are Not Required for Identifying the Division Site in but Ensure Its Efficient Utilization
- Neurobeachin, a Regulator of Synaptic Protein Targeting, Is Associated with Body Fat Mass and Feeding Behavior in Mice and Body-Mass Index in Humans
- Statistical Analysis of Readthrough Levels for Nonsense Mutations in Mammalian Cells Reveals a Major Determinant of Response to Gentamicin
- Gene Reactivation by 5-Aza-2′-Deoxycytidine–Induced Demethylation Requires SRCAP–Mediated H2A.Z Insertion to Establish Nucleosome Depleted Regions
- The miR-35-41 Family of MicroRNAs Regulates RNAi Sensitivity in
- Genetic Basis of Hidden Phenotypic Variation Revealed by Increased Translational Readthrough in Yeast
- An Alu Element–Associated Hypermethylation Variant of the Gene Is Associated with Childhood Obesity
- Modelling Human Regulatory Variation in Mouse: Finding the Function in Genome-Wide Association Studies and Whole-Genome Sequencing
- Novel Loci for Adiponectin Levels and Their Influence on Type 2 Diabetes and Metabolic Traits: A Multi-Ethnic Meta-Analysis of 45,891 Individuals
- Polycomb-Like 3 Promotes Polycomb Repressive Complex 2 Binding to CpG Islands and Embryonic Stem Cell Self-Renewal
- Insulin/IGF-1 and Hypoxia Signaling Act in Concert to Regulate Iron Homeostasis in
- EMF1 and PRC2 Cooperate to Repress Key Regulators of Arabidopsis Development
- Three Essential Ribonucleases—RNase Y, J1, and III—Control the Abundance of a Majority of mRNAs
- Contrasted Patterns of Molecular Evolution in Dominant and Recessive Self-Incompatibility Haplotypes in
- A Machine Learning Approach for Identifying Novel Cell Type–Specific Transcriptional Regulators of Myogenesis
- Genomic Tools for Evolution and Conservation in the Chimpanzee: Is a Genetically Distinct Population
- Nos2 Inactivation Promotes the Development of Medulloblastoma in Mice by Deregulation of Gap43–Dependent Granule Cell Precursor Migration
- Intracranial Aneurysm Risk Locus 5q23.2 Is Associated with Elevated Systolic Blood Pressure
- Heritability and Genetic Correlations Explained by Common SNPs for Metabolic Syndrome Traits
- A Genome-Wide Association Study of Nephrolithiasis in the Japanese Population Identifies Novel Susceptible Loci at 5q35.3, 7p14.3, and 13q14.1
- DNA Damage in Nijmegen Breakage Syndrome Cells Leads to PARP Hyperactivation and Increased Oxidative Stress
- DNA Resection at Chromosome Breaks Promotes Genome Stability by Constraining Non-Allelic Homologous Recombination
- Genetic Analysis of Floral Symmetry in Van Gogh's Sunflowers Reveals Independent Recruitment of Genes in the Asteraceae
- A Splice Site Variant in the Bovine Gene Compromises Growth and Regulation of the Inflammatory Response
- Promoter Nucleosome Organization Shapes the Evolution of Gene Expression
- The Nucleoside Diphosphate Kinase Gene Acts as Quantitative Trait Locus Promoting Non-Mendelian Inheritance
- The Ciliogenic Transcription Factor RFX3 Regulates Early Midline Distribution of Guidepost Neurons Required for Corpus Callosum Development
- Phosphorylation of the RNA–Binding Protein HOW by MAPK/ERK Enhances Its Dimerization and Activity
- A Genome-Wide Scan of Ashkenazi Jewish Crohn's Disease Suggests Novel Susceptibility Loci
- Parkinson's Disease–Associated Kinase PINK1 Regulates Miro Protein Level and Axonal Transport of Mitochondria
- LMW-E/CDK2 Deregulates Acinar Morphogenesis, Induces Tumorigenesis, and Associates with the Activated b-Raf-ERK1/2-mTOR Pathway in Breast Cancer Patients
- Mapping the Hsp90 Genetic Interaction Network in Reveals Environmental Contingency and Rewired Circuitry
- Autoregulation of the Noncoding RNA Gene
- The Human Pancreatic Islet Transcriptome: Expression of Candidate Genes for Type 1 Diabetes and the Impact of Pro-Inflammatory Cytokines
- Spo0A∼P Imposes a Temporal Gate for the Bimodal Expression of Competence in
- Antagonistic Regulation of Apoptosis and Differentiation by the Cut Transcription Factor Represents a Tumor-Suppressing Mechanism in
- A Downstream CpG Island Controls Transcript Initiation and Elongation and the Methylation State of the Imprinted Macro ncRNA Promoter
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- PIF4–Mediated Activation of Expression Integrates Temperature into the Auxin Pathway in Regulating Hypocotyl Growth
- Metabolic Profiling of a Mapping Population Exposes New Insights in the Regulation of Seed Metabolism and Seed, Fruit, and Plant Relations
- A Splice Site Variant in the Bovine Gene Compromises Growth and Regulation of the Inflammatory Response
- Comprehensive Research Synopsis and Systematic Meta-Analyses in Parkinson's Disease Genetics: The PDGene Database
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Autoři: prof. MUDr. Pavel Horák, CSc., doc. MUDr. Ludmila Brunerová, Ph.D., doc. MUDr. Václav Vyskočil, Ph.D., prim. MUDr. Richard Pikner, Ph.D., MUDr. Olga Růžičková, MUDr. Jan Rosa, prof. MUDr. Vladimír Palička, CSc., Dr.h.c.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



