-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praxe
MK2-Dependent p38b Signalling Protects Hindgut Enterocytes against JNK-Induced Apoptosis under Chronic Stress
The integrity of the intestinal epithelium is crucial for the barrier function of the gut. Replenishment of the gut epithelium by intestinal stem cells contributes to gut homeostasis, but how the differentiated enterocytes are protected against stressors is less well understood. Here we use the Drosophila larval hindgut as a model system in which damaged enterocytes are not replaced by stem cell descendants. By performing a thorough genetic analysis, we demonstrate that a signalling complex consisting of p38b and MK2 forms a branch of SAPK signalling that is required in the larval hindgut to prevent stress-dependent damage to the enterocytes. Impaired p38b/MK2 signalling leads to apoptosis of the enterocytes and a subsequent loss of hindgut epithelial integrity, as manifested by the deterioration of the overlaying muscle layer. Damaged hindguts show increased JNK activity, and removing upstream activators of JNK suppresses the loss of hindgut homeostasis. Thus, the p38/MK2 complex ensures homeostasis of the hindgut epithelium by counteracting JNK-mediated apoptosis of the enterocytes upon chronic stress.
Published in the journal: . PLoS Genet 7(8): e32767. doi:10.1371/journal.pgen.1002168
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1002168Summary
The integrity of the intestinal epithelium is crucial for the barrier function of the gut. Replenishment of the gut epithelium by intestinal stem cells contributes to gut homeostasis, but how the differentiated enterocytes are protected against stressors is less well understood. Here we use the Drosophila larval hindgut as a model system in which damaged enterocytes are not replaced by stem cell descendants. By performing a thorough genetic analysis, we demonstrate that a signalling complex consisting of p38b and MK2 forms a branch of SAPK signalling that is required in the larval hindgut to prevent stress-dependent damage to the enterocytes. Impaired p38b/MK2 signalling leads to apoptosis of the enterocytes and a subsequent loss of hindgut epithelial integrity, as manifested by the deterioration of the overlaying muscle layer. Damaged hindguts show increased JNK activity, and removing upstream activators of JNK suppresses the loss of hindgut homeostasis. Thus, the p38/MK2 complex ensures homeostasis of the hindgut epithelium by counteracting JNK-mediated apoptosis of the enterocytes upon chronic stress.
Introduction
In its function as a protective barrier, the intestinal epithelium is constantly exposed to stressors from the outside [1]. It acts as a mediator between the bacterial flora and the host's immune system, and the intestinal epithelial cells have to respond to extrinsic and intrinsic factors to ensure their own survival and proper gut homeostasis. Two populations of cells have to orchestrate different aspects of intestinal epithelium survival. While the intestinal stem cells (ISCs) are essential for the proliferative aspects of intestinal homeostasis [2]–[4], the enterocytes (ECs) form the first line of defence against pathogens and stressors. Signalling cascades that are modulated by external signals and by cellular stress are crucial regulators of intestinal epithelial survival. For example, mice with gut-specific knockout of NEMO spontaneously develop intestinal lesions reminiscent of those in inflammatory bowel diseases (IBDs), indicating an essential role for NFκB signalling in EC survival [5], [6]. Recently, ER stress in the ECs has also been found to influence epithelial homeostasis, and mutations in XBP1 are sufficient to trigger an IBD-like phenotype [7]. The p38 stress-activated protein kinase pathway has also been implicated in intestinal disorders [8] but its role in intestinal diseases is still controversially discussed [9], [10].
The p38 SAPK belongs to the MAPK family and is conserved from yeast to humans. In higher eukaryotes, p38 associates with its major target, the MAPK-activated protein kinase MK2. This complex resides in the nucleus in the resting state. Upon stress, p38 is activated by MKK3/MKK6 and phosphorylates MK2, which results in an exposure of the nuclear export signal of MK2 and a subsequent nuclear export of the complex [11]. Another consequence of the p38/MK2 complex formation is the stabilisation of p38 protein. Interestingly, the kinase activity of MK2 is required neither for the nucleo-cytoplasmic shuttling nor for the p38 protein stabilisation [11]–[13]. Conversely, MK2 kinase activity is crucial to phosphorylate small heat-shock proteins, transcription factors (e.g., SRF and HSF-1), and TTP [14]–[16]. The inhibitory phosphorylation of TTP by the p38/MK2 complex has been shown to increase the translation of AU-rich elements (ARE)-containing mRNAs including the mRNA encoding the proinflammatory cytokine TNFα [17]. Furthermore, p38 and MK2 have been shown to act as cytoplasmic checkpoint kinases in parallel to CHK1 [18], [19]. Due to the plethora of p38/MK2 functions, targeting the p38 SAPK branch with inhibitors might lead to harmful side effects. Thus, it is important to understand the roles of p38 signalling in a tissue-specific context. The availability of mouse models has helped to decipher some in vivo roles of p38 SAPK signalling [20], [21] but the complex nature of the intestinal system has hampered a detailed analysis.
Studies in the model organism Drosophila have provided new insights into intestinal maintenance and how different signalling pathways are employed to ensure proper gut homeostasis. The Drosophila gut consists of the fore-, the mid-, and the hindgut. The larval and adult midgut displays a regional specification along the antero-posterior axis and fulfils vital functions such as nutrient absorption [22]. The main part of the embryonic and larval hindgut, the large intestine, is subdivided into a ventral (hv, positive for Delta expression) and a dorsal domain (hd, positive for engrailed expression) [23] that are separated by a single row of boundary cells [24]. The adult hindgut shows a similar yet more complex organisation [25]. The function of the hindgut, however, remains largely unknown. Ultrastructurally, the hd domain is marked by deep infoldings and enriched in elongated mitochondria, resembling the rectal papillae of other insects. Based on these similarities, it has been speculated that the Drosophila large intestine plays a role in ion and/or water resorption [23], [24].
At least four distinct intestinal epithelial cell types are found in Drosophila: intestinal stem cells (ISCs), the hormone-producing enteroendocrine cells (EEs), enterocytes (ECs) and the transient enteroblasts (EBs; progenitors of EEs and ECs). In the adult midgut, ISCs are required for normal gut homeostasis, but in aged and/or stressed individuals the number of midgut ISCs is increased and differentiation is disturbed [26], [27]. The orchestrated activation of Hippo, JNK, JAK/STAT, Notch and EGFR signalling within and between the ISCs, the ECs and the visceral muscles is required to coordinate proliferation, differentiation and cellular turnover in the midgut epithelium [26], [28]–[36]. In contrast to the situation in the midgut, the hindgut stem cells are not required for hindgut homeostasis in the larva and in the adult fly. The hindgut ISCs are rather needed during the shift from larval to adult hindgut, and stress induces proliferation and cell migration in the pylorus region [37]. Thus, especially the adult midgut serves as a good experimental model for the analysis of EC replenishment upon damage, but the mechanisms governing proper stress response in the ECs have remained elusive.
p38 SAPK signalling also plays a role in gut homeostasis in Drosophila. In aged adult midguts, an increase in p38b expression has been observed in Delta-positive stem cells, which appears to be partly due to DREF-mediated transcriptional activation [38], [39]. Knockdown of p38b in the ISCs prevents age - and stress-induced ISC overproliferation and accumulation of aberrant Delta-positive cells, implying a role for p38b in regulating intestinal regeneration [38]. p38 signalling has been shown to be required for DUOX expression in differentiated ECs and for normal differentiation in the ISCs [38], [40]. Consistently, a recent study showed that the larval intestinal epithelium is more susceptible to damage by pathogens in the absence of p38 function [41]. However, the mechanism of p38 action within the ECs remains unclear. In Drosophila, p38 signalling can antagonize the closely related JNK SAPK branch [42]. JNK signalling has also been shown to regulate several aspects of intestinal function. It is required in the midgut ECs to induce autophagy and thereby ensure their survival during oxidative stress [43]. In the ISCs, JNK is needed for proper stress response but strong activation of JNK leads to differentiation defects and loss of gut homeostasis [26]. Whether p38 and JNK influence each other in their intestinal function has not been addressed so far.
In this study, we investigate how the Drosophila larval hindgut is enabled to maintain homeostasis under stress conditions. Using deletion mutants for MK2, p38a and p38b, we show that MK2 and p38b form a complex that is specifically required to protect the dorsal hindgut ECs against chronic stress. In the absence of this p38b/MK2 complex, JNK is activated in patches of hindgut ECs, resulting in JNK-dependent apoptosis, loss of epithelial organisation, and melanisation of hindgut regions. This melanisation of the ECs does not require the recruitment of hemocytes, indicating an epithelial response that might also precede immune activation in mammalian intestinal diseases. Thus, we identify a specific SAPK signalling module required to maintain hindgut epithelial integrity upon stress.
Results
Drosophila MK2 Is Dispensable for Normal Development and Survival at Non-Stress Conditions
The mammalian MAPKAP-K2 is known to be a downstream kinase of the p38 branch of SAPKs [44]. To generate deletion mutants for Drosophila MK2, we mobilised a P-element insertion located in the MK2 locus (Figure 1A). Whereas Δ43 is a null allele as judged by the absence of MK2 protein and by the failure of Δ43 larval lysates to phosphorylate mammalian small heat shock protein 25 in a kinase assay (Figure S1A), the alleles Δ41 and Δ12 are likely to represent hypomorphic alleles. Δ38 is probably also a null allele although the generation of a truncated protein (initiated from alternative Methionine codons) cannot be excluded (Figure 1A). A precise excision allele of the same P-element (Δ1A) was used as control throughout this study. All of the generated alleles show no obvious phenotypic alteration and can be kept as homozygous lines at normal conditions. Since several mutants of the SAPK pathway are sensitive to stresses including high osmolarity [45], [46], we tested the MK2 mutants in various stress assays including oxidative stress (paraquat feeding), UV exposure during late embryonic/early larval development, heavy metals (0.5 mM copper sulphate), high osmolarity (0.2 M NaCl) and SDS (0.2%) (Figure S1B and data not shown). Interestingly, only salt and SDS feeding resulted in a melanotic phenotype in 35% to 45% of the mutant larvae, characterized by the appearance of a “black dot” (BD) in the posterior part of the body (Figure 1B and Figure S1C–S1E). Closer examination revealed that the BD localised to the posterior hindgut, and the affected hindgut epithelium appeared pathologically altered (Figure 1C). All MK2 alleles were analysed for the appearance of BDs and survival at three conditions: normal food, weak salt stress (0.1 M NaCl) and strong salt stress (0.2 M NaCl) (Figure 1D). The behaviour of MK2 hypomorphic larvae indicated that the levels of MK2 get more important with increasing osmolarity. Introducing a genomic rescue construct completely rescued both the BD phenotype and the lethality of Δ43 mutants at 0.2 M NaCl (Figure 1D). Thus, MK2 is not essential at normal conditions but is required when larvae are reared on a high sodium chloride diet.
Fig. 1. MK2 deficient larvae are sensitive to salt stress. 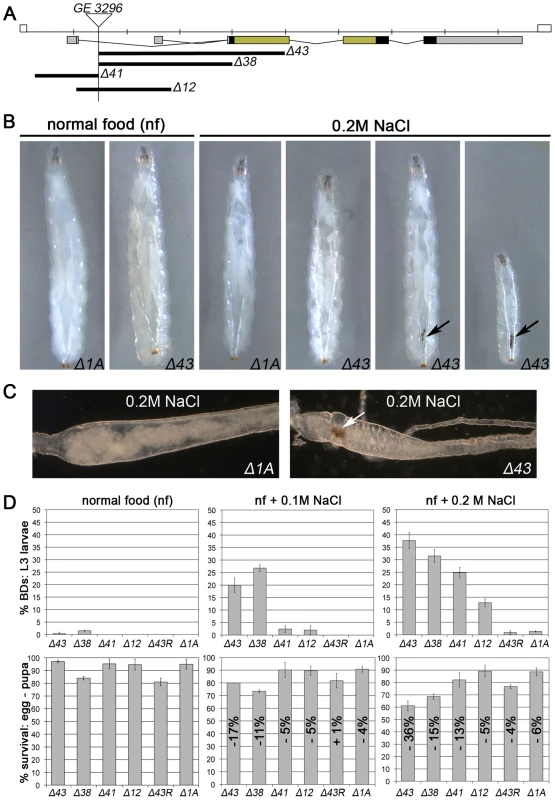
(A) Deletion alleles of MK2 generated by imprecise excision of the P-element GE3296. The untranslated regions (UTRs; grey), protein coding sequence (black), and the sequences encoding the kinase domain (green) are indicated. (B) MK2 null mutants (Δ43) but not wild-type larvae (Δ1A) develop melanisations in the posterior part of the body (black dots/BDs, indicated by arrows) when reared on 0.2 M NaCl food. (C) Bright field pictures of wild-type (Δ1A) and MK2 mutant (Δ43) hindguts of larvae reared on 0.2 M NaCl food. Rectum points to the left; BD indicated by arrow. (D) The different MK2 deletion alleles reared on normal food, 0.1 M and 0.2 M NaCl food, respectively, reveal an increase in BDs with the severity of the stress (upper panel) and a reduction in survival (lower panel) as compared to the control (Δ1A). The decrease in the survival rate is indicated relative to the survival on normal food of the respective genotype. A genomic MK2 rescue construct fully rescues both the BD phenotype and the decreased survival of MK2 mutants (Δ43R). Drosophila MK2 Is Required to Protect the Hindgut from Stress-Induced Apoptosis
We speculated that MK2 is specifically required to protect the hindgut epithelium. In sections of the hindgut, dorsal ECs with BDs were apically ruptured (Figure 2Aii). In wild-type hindguts, ECs of the dorsal domain were not damaged under salt stress conditions (Figure 2Ai and Figure S2C). Similarly, most MK2 mutant hindguts without BDs did not display morphological alterations (Figure S2C). However, a blistering of the apical surface without damaging the apical membrane was occasionally observed (Figure 2Aiii and Figure S2C). Blistering of the apical surface was also observed in MK2 mutant hindguts with BDs in regions away from the BD (Figure S2C). The melanisations occurred at the apical surface within the ECs, probably preceded by the blistering (Figure 2Aiii and Figure S2C). Next, we tested the hindgut tissue for the presence of dying cells. TUNEL staining revealed local clusters of apoptotic hindgut ECs in MK2 mutant larvae (Figure 2Bi and 2Bii) but not in wild-type larvae reared on 0.2 M NaCl (Figure 2Biii). The visceral muscles surrounding the hindgut ECs appeared unaffected in MK2 mutant larvae. A disturbed hindgut musculature was only observed in hindguts with large BDs (Figure 2C). Furthermore, loss of epithelial integrity was evident from the mislocalisation of Neuroglian (Nrg) (Figure 2D). Nrg-GFP was localised laterally in cell-cell junctions in hindguts of wild-type and MK2 mutant larvae (Figure 2Di). In contrast, in MK2 mutant larvae with BDs, Nrg-GFP was normally localised in unaffected regions but displayed a more diffuse pattern close to the BD (Figure 2Dii). This mislocalisation was more pronounced at 0.2 M NaCl (Figure 2Dii and 2Diii). Interestingly, BDs were only found in the dorsal hindgut (hd) compartment, as judged by cell and nuclear size (hd is composed of smaller cells) (Figure 2Dii and 2Diii).
Fig. 2. Increased apoptosis and tissue damage in hindguts of MK2 mutants. 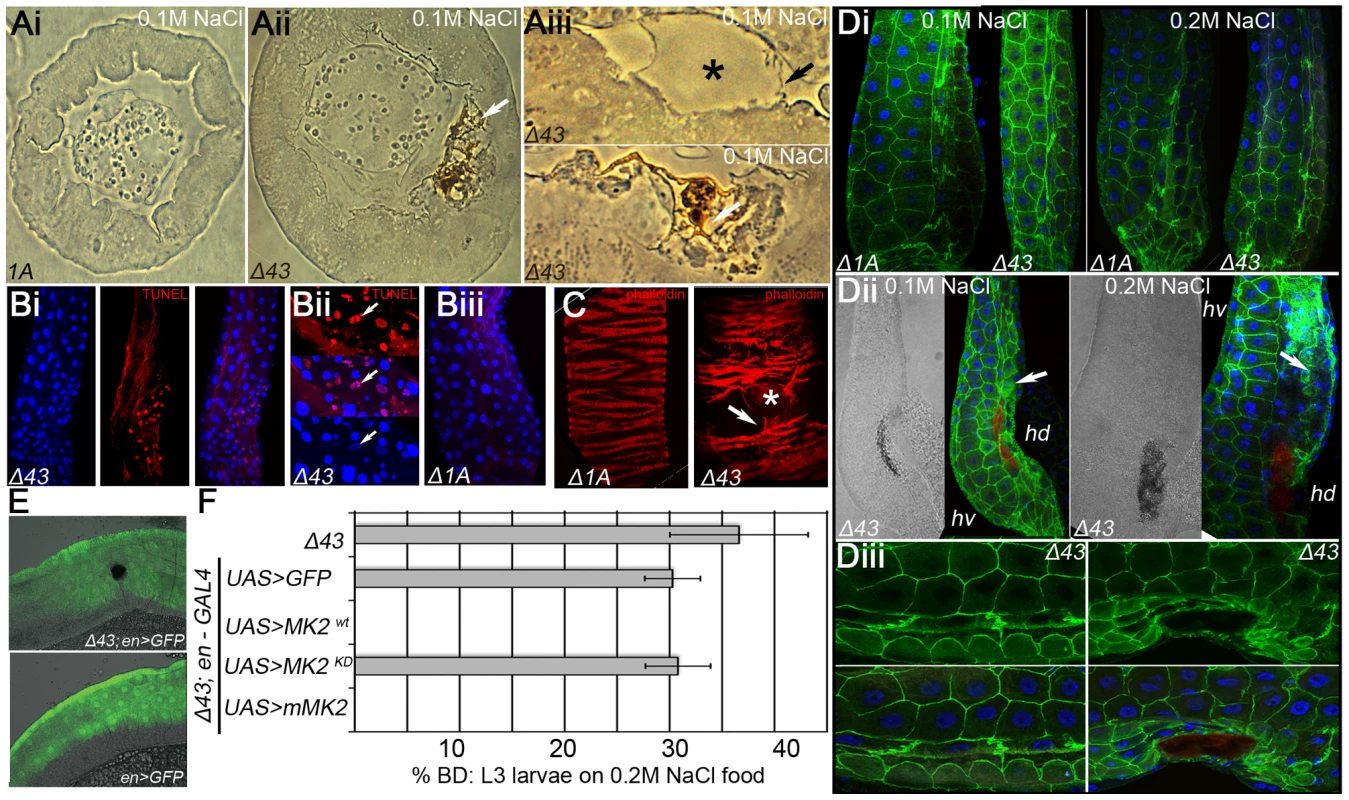
(A) MK2 mutants show ruptures of ECs when reared on high salt diet. Bright field pictures of sections of hindgut tissue of larvae reared on 0.1 M NaCl food are shown. (Ai) wild-type (Δ1A), (Aii) MK2 mutant (Δ43) with BD (white arrow). (Aiii) Higher magnifications of ECs in the dorsal hd domain of MK2 mutants. Blistering of the apical part (upper panel, black asterisk) and melanisation of the apical region (lower panel, white arrow) are frequently observed. The apical surface of the ECs appears to be intact (black arrow). (B) Apoptosis is increased in the hindguts of MK2 mutant larvae reared on high salt conditions. (Bi) TUNEL staining revealed local apoptosis in MK2 mutant hindguts. (Bii) Magnification of apoptotic foci shows the condensed nature of the TUNEL positive nuclei (white arrows). (Biii) No apoptotic cells are present in control hindguts. (C) MK2 mutant larvae reared on high salt food (0.2 M) with large BDs show ruptures of the visceral muscles (right panel, white arrow) in comparison to the regular organisation of wild-type larvae (left panel). The location of the BD is marked by an asterisk. (D) The cell-cell junction marker Nrg-GFP indicates morphological defects in ECs in the hd domain. (Di) Wild-type and MK2 mutant larvae reared on 0.1 M or 0.2 M NaCl food show normal localisation of Nrg-GFP in the ECs. The hd domain points to the right; the rectum points to the bottom. (Dii) In MK2 mutants with BDs, Nrg-GFP is more diffusely localised at the sites of BDs (white arrow). The BD is discernible in the bright field picture and by its autofluorescence in the far-red channel (red) (Diii) Higher magnification of Nrg-GFP (green) signal in MK2 mutants (reared on 0.2 M NaCl food) with (right) or without BD (left). (E) BDs are always found in the engrailed-positive hd domain (green) of the hindgut. (F) Rescue of the MK2 mutant phenotype by en-GAL4 mediated expression of the indicated cDNA constructs. We performed a series of rescue experiments to determine where MK2 function is required. The BD phenotype was rescued by ubiquitous and hindgut-specific but not by midgut-specific expression of MK2 (Figure S2D). Moreover, only wild-type MK2 but not a kinase-dead version of MK2 rescued the BD phenotype. The largest domains of the larval hindgut are the dorsal engrailed-positive (hd) and the ventral Delta-positive (hv) domain. BDs were only observed in the hd domain (Figure 2E). Consistently, engrailed-GAL4 driven expression of a wild-type Drosophila MK2 or a wild-type murine MK2, but not of a kinase-dead Drosophila MK2, rescued the BD phenotype (Figure 2F).
Melanisation in insects can be regarded as a wound healing or defence response and has been shown to be either hemocyte-dependent or hemocyte-independent [47]. We thus attempted to clarify whether BD formation in MK2 mutant larvae was dependent on the recruitment of hemocytes. Staining for the blood cell marker Hemese - either direct (using antibodies) or indirect (using He-GAL4 UAS-GFP) - revealed the absence of blood cells at or within the hindgut epithelium in both wild-type and MK2 mutant hindguts (Figure S2A and data not shown). Consistently, the formation of BDs was still observed in hemocyte-ablated MK2 mutants (Figure S2B).
Together, those results show that Drosophila MK2 kinase function is required in the dorsal hindgut compartment to protect larval hindgut ECs from stress-induced apoptosis upon salt stress.
MK2 Genetically Interacts with p38a and p38b
Mutations in MEKK1 and p38b but not in MKK3/lic and p38a resulted in a strong BD phenotype even at normal food conditions (Figure 3A). To define the roles of p38a and p38b with respect to MK2, we next tested these kinases genetically for the behaviour at normal conditions and under salt stress (Figure 3E). At unstressed conditions, MK2 was required neither for hindgut homeostasis nor for survival. Similarly, p38a mutants did not display BDs or elevated mortality rates. In contrast, MK2; p38a double mutants developed a weak BD phenotype, indicating that p38a and MK2 act in two parallel stress-signalling pathways. p38b mutants had a decreased survival rate, consistent with published findings [46]. p38b is required for hindgut homeostasis even under normal conditions, because larvae lacking p38b function developed BDs on normal food. Interestingly, MK2; p38b double mutants displayed a slight increase in BDs but no increase in lethality rate compared to p38b single mutants. Thus, p38b and MK2 are likely to function in the same pathway but both kinases may have additional independent functions in the hindgut or in other tissues.
Fig. 3. Interactions of MK2 with the p38 SAPK pathway. 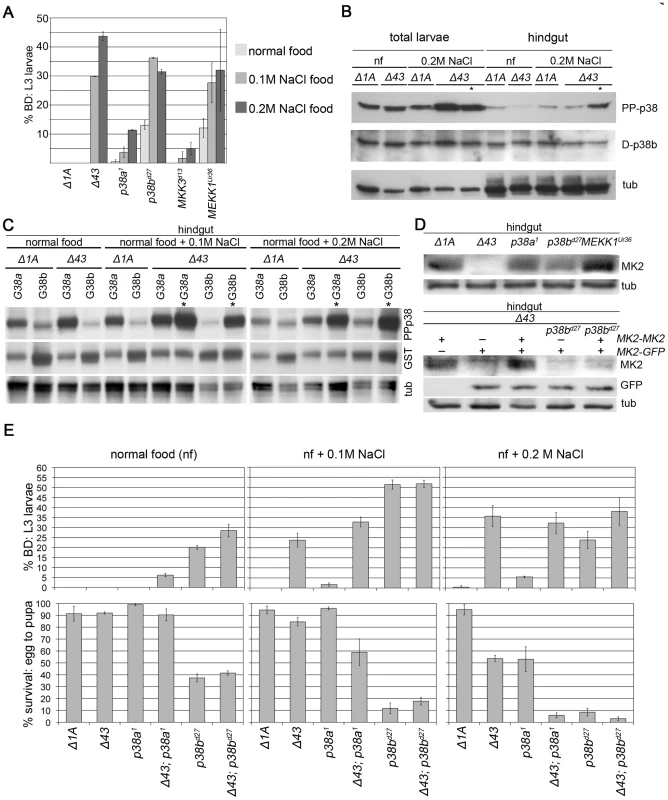
(A) Mutations in genes encoding p38 SAPK pathway members were tested for a BD phenotype (normal food, 0.1 M NaCl, 0.2 M NaCl). MEKK1 and p38b but not MKK3 or p38a show a BD phenotype similar to MK2 mutants. The wild-type control (Δ1A) does not display any BDs. (B) The activating pTGpY phosphorylation (PP-p38) of p38 is increased in MK2 mutants under stress, especially in larvae with BDs (lanes marked by asterisks). (C) GST-tagged p38a (G38a) and p38b (G38b) were overexpressed in wild-type or MK2 mutant larvae to distinguish between the activation of the two p38 kinases. The lanes with lysates of MK2 mutant larvae with BDs are marked by asterisks. (D) Stabilisation of MK2 happens at the level of protein stability or translation and not at the level of transcription. In p38b mutant hindguts MK2 protein levels are reduced (upper blot). GFP expressed under the control of the genomic MK2 locus (MK2-GFP, MK2 coding sequence replaced by sequence encoding GFP) is unaltered in a p38b mutant background. In contrast, MK2 expressed from the same genomic region (MK2-MK2, MK2 coding sequence not replaced) does not result in restoration of MK2 protein levels (lower blot). (E) p38a and p38b deletion alleles (in wild-type and MK2 mutant background, respectively) were assessed for the appearance of BDs (upper panel) and survival (lower panel) on normal food, 0.1 M NaCl and 0.2 M NaCl, respectively. Consistent with a common MK2/p38b pathway, p38b single mutants and MK2; p38b double mutants displayed the same phenotype at both weak (0.1 M NaCl) and strong (0.2 M NaCl) stress conditions. At weak stress conditions, p38a was not crucial for survival and hindgut homeostasis. In contrast, MK2 mutants displayed hindgut defects but no increase in mortality. In agreement with MK2 and p38a acting in two parallel stress-signalling pathways, BD formation was only slightly increased in the MK2; p38a double mutants but the absence of MK2 significantly enhanced the mortality of p38a mutants. This lethality increase of MK2; p38a double mutants was also observed at strong salt stress. At this condition, both MK2 and p38a mutants resulted in increased mortality rates, indicating that both branches of stress signalling are required for survival. Whereas MK2 is specifically required in the hindgut, p38a might be needed in other organs since p38a mutants hardly developed BDs.
Taken together, our genetic data suggest the existence of two major p38 branches in Drosophila that are required to varying extents during normal and different salt stress conditions. The p38a branch is not essential at normal or weak salt stress conditions but required at strong salt stress conditions. In contrast, the p38b branch is required at both normal and salt stress conditions to protect the hindgut. Moreover, the lethality of p38a; p38b double mutants [46] suggests that, besides their specific functions, both p38 kinases engage in a common essential function. MK2 is involved in a sub-branch of the p38b branch and appears to be a key effector of p38b in the hindgut. MK2 becomes more important with increasing stress conditions specifically in this tissue.
p38b Phosphorylation Is Increased in Melanised Larval Hindguts
We next checked the activation status of p38 by Western analysis (Figure 3B). Chronic exposure (from L1 to L3) to 0.2 M NaCl did not increase p38 phosphorylation in total and hindgut lysates of wild-type larvae. In contrast, stronger p38 activation was observed in total larval lysates of MK2 mutants under stress conditions. This strong activation was also apparent in the hindgut but only in larvae with BDs. Since we were not able to distinguish the endogenous p38a and p38b, we overexpressed GST-tagged versions of p38 and analysed their activation status in the hindgut (Figure 3C). GST-p38a was strongly activated even under normal conditions, and the activation was slightly increased under stress conditions. In MK2 mutants, GST-p38a was more strongly activated under all conditions and especially in hindguts with BDs. In contrast, GST-p38b was only weakly phosphorylated in wild-type and MK2 mutant hindguts at all conditions, except in hindguts of MK2 mutant larvae with BDs where a boost in GST-p38b phosphorylation was seen. Thus, the increase in endogenous p38 phosphorylation observed in MK2 mutant larval hindguts is probably due to p38b phosphorylation, suggesting that a negative feedback loop operates from MK2 to the upstream signalling components. Alternatively, the lack of a functional stress-protective MK2/p38b function may increase the stress in the ECs and lead to a vicious cycle that boosts p38b activation.
MK2 Protein Abundance Depends on p38b
In mammalian cells, MK2 is needed to stabilise p38 protein levels [13], which does not appear to be the case in Drosophila (Figure 3B and 3C). We wondered whether the protein levels of MK2 would depend on the presence of the upstream components. No change in MK2 expression was observed in p38a mutants, and a slight increase was detected in MEKK1 mutants. In sharp contrast, MK2 protein levels were reduced in p38b mutants (Figure 3D). Using a genomic MK2 rescue construct and a genomic MK2-GFP reporter in an MK2 null mutant background, we found that transcription from the MK2 locus was unchanged but protein levels were reduced, probably due to destabilisation of MK2 in the absence of p38b (Figure 3D).
MK2 Physically Interacts with p38b
The facts that MK2 and p38b genetically interact and that MK2 protein levels depend on the presence of p38b suggest a close physical interaction of the two kinases. Therefore, we expressed tagged versions of p38a, p38b and MK2 in S2 cells and checked for co-localization of these kinases. In mammalian cells, MK2 is nuclear under normal conditions and translocates to the cytoplasm upon stress [48]. Similarly, in Drosophila S2 cells, GFP-MK2 was mainly found in the nucleus, whereas overexpressed p38a and p38b were largely cytoplasmic (Figure 4A). Co-overexpression of p38b and MK2 (but not of p38a and MK2) resulted in a nuclear-to-cytoplasmic translocation of GFP-tagged MK2 (Figure 4A). Consistently, MK2 was found to bind p38b but not p38a in pull down experiments (Figure 4B). This interaction as well as the nuclear-to-cytoplasmic shuttling was dependent on a four amino acid motif (DPTD) in p38b resembling the common docking motif (CD domain) that is critical for docking interactions in MAPKs [49]. In p38a, the respective four amino acids are EPSV. Exchanging these motifs revealed that the DPTD motif is necessary and sufficient to dock MK2 to p38 proteins (Figure 4A and 4B).
Fig. 4. A p38b/MK2 complex is required for hindgut EC stress protection. 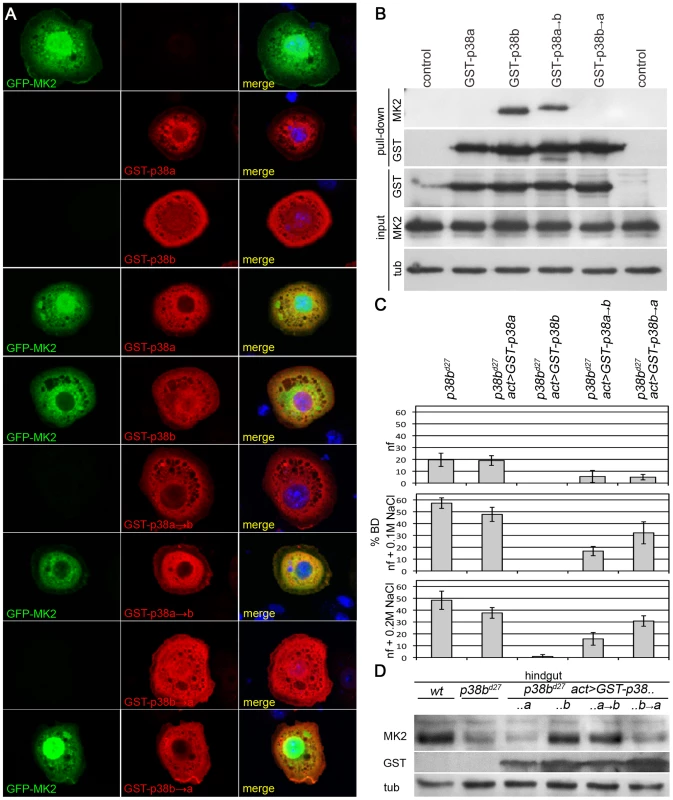
(A) GFP-MK2 (green) localises to the nucleus. GST-p38a and GST-p38b show a broad, more cytoplasmic distribution (red). GST-p38b co-expression shuttles GFP-MK2 to the cytoplasm in a DPTD-motif dependent manner. GST-p38a or GST-p38b→a (DPTD changed to EPSV) do not change the localisation of MK2, but a GST-p38a→b protein mutant can export GFP-MK2 from the nucleus. (B) In pull down experiments, GST-p38b co-precipitates MK2, whereas p38a and the docking mutant p38b→a do not bind MK2. The DPTD motif introduced into p38 (p38a→b) is sufficient for binding to MK2. (C) Whereas overexpression of GST-p38b rescues completely, GST-p38a fails to rescue the BD phenotype of p38b mutants. GST-p38a→b and GST-p38b→a partially rescue the BD phenotype with p38a→b performing better. (D) Docking of MK2 to p38 that harbours a DPTD motif is sufficient to restore wild-type levels of MK2 protein in a p38b mutant background. The expression of GST-p38b completely rescued the BD phenotype of p38b deficient flies, whereas expression of GST-p38a had no influence on the BD phenotype (Figure 4C and Figure S3). Expressing either GST-p38a→b (p38a with docking motif of p38b) or GST-p38b→a (p38b with respective sequences of p38a) resulted in a partial rescue of the BD phenotype. On normal food, both versions rescued partially, indicating that although not essential, binding to MK2 is required for a complete rescue under normal conditions. On 0.1 M and 0.2 M NaCl, GST-p38a→b resulted in substantial but incomplete rescue, suggesting that other aspects of p38b function not mediated by binding to MK2 are required for a complete rescue. Consistently, a GST-p38b→a protein that is not able to bind MK2 also partially rescued the p38b phenotype but to a lesser extent than p38a→b (Figure 4C and Figure S3). MK2 protein levels in the hindgut were restored by expressing p38b or p38a→b but not by p38a or p38b→a (Figure 4D). Thus, binding of p38 to MK2 is required to localise and stabilise MK2 and is important for the stress-protective function in the hindgut.
The Catalytic Activity of p38b Is Required to Localise MK2
The catalytic activity of MK2 and MK2 binding to p38b are required to protect the hindgut epithelial cells upon salt stress. To address whether the catalytic activity of p38b is also necessary, we used GST-tagged non-activatable p38bAGF and kinase-dead p38bKR protein mutants. Whereas co-expression of wild-type GST-p38b and GFP-MK2 led to a nuclear export (>70%) of MK2, the localisation of GFP-MK2 was random when GST-p38bAGF or GST-p38bKR were co-expressed (Figure 5A and 5B). Consistently, the BD phenotype of p38b null mutants was not rescued by re-expression of the kinase-dead or of the non-activatable p38b protein version (Figure 5C). Moreover, at high NaCl stress (0.2 M NaCl), the p38bAGF and p38bKR expressing larvae died. This could be explained by the titration of an upstream kinase of p38b, which might impinge on the activation of other downstream effectors. Thus, the catalytic activity of p38b is required to impact on the subcellular localisation and thereby the proper function of MK2.
Fig. 5. p38b activation and kinase activity are required to protect the hindgut. 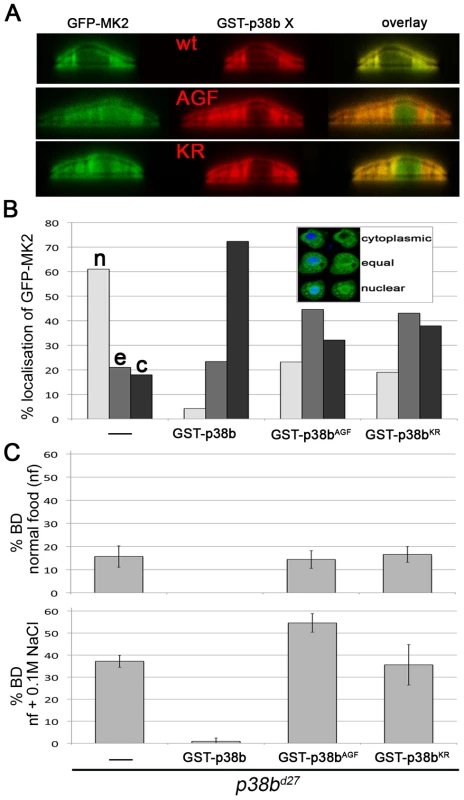
(A) Co-expression of GST-p38b and MK2 results in cytoplasmic localisation of GFP-MK2. When a kinase-dead (KR) or non-activatable (AGF) version of p38b was expressed, the nuclear export was not efficient. (B) Quantification of GFP-MK2 localisation upon GST-p38b, GST-p38bAGF or GST-p38bKR co-expression. Localisation was assigned to three classes: cytoplasmic (c), equal distribution between nucleus and cytoplasm (e), and nuclear localisation (n). (C) Activation and kinase function are required for proper function of p38b in the hindgut. The rate of BDs of p38b mutants upon re-expression of p38b protein variants by means of Act-GAL4 was assessed. JNK Signalling Is Induced in MK2 Mutants and Is Linked to Apoptosis
JNK signalling has been implicated in triggering apoptosis [50]. Furthermore, a JNK antagonizing activity of p38 signalling has been observed in developmental processes [42] and at the systemic level during infection [41]. Thus, we tested whether the cell death observed in MK2 mutant larvae reared on high salt correlated with JNK activation. Indeed, elevated levels of phosphorylated JNK were detected in hindgut lysates of MK2 mutant larvae with BDs (Figure 6A). As in vivo readouts for JNK signalling activity, we used reporters for misshapen and puckered [51], [52]. An induction of both msn>lacZ and puc>lacZ was observed in MK2 mutant larval hindguts, with highest levels adjacent to the BDs (Figure 6B). To exclude that the induction of JNK signalling is a secondary consequence of wound healing or the melanisation process, we checked for puc-GFP induction in stress-challenged MK2 mutant larvae before BD formation. Interestingly, patches with puc-GFP signal were readily detected upon stress in larvae devoid of BDs, and the area of those patches correlated with the strength of the stress (Figure 6C). We noted that puc-GFP was activated in a graded fashion, with highest activity where ECs undergo apoptosis and a BD will ultimately form (Figure 6D). Strong puc-GFP reporter activity co-localised with TUNEL positive cells close to the BDs (Figure 6E). Removing the JNK upstream components TAK1 and MKK4, respectively, in an MK2 mutant background partially suppressed the BD phenotype, indicating that the hindgut epithelial cells are dying due to JNK-induced apoptosis (Figure 6F). Expressing a dominant-negative version of JNK (BskDN) in the dorsal domain using engrailed-GAL4 resulted in a suppression of the BD phenotype (Figure 6G). Furthermore, expression of BskDN specifically in cells with high JNK activity (using puc-GAL4) substantially suppressed BD formation. In contrast, re-expression of MK2 at this stage reduced the number of larvae with BDs only mildly (Figure 6G). Thus, deregulated JNK activation in the hindgut of MK2 mutants precedes and probably causes cell death and BD formation.
Fig. 6. JNK activation leads to EC apoptosis in MK2 deficient hindguts. 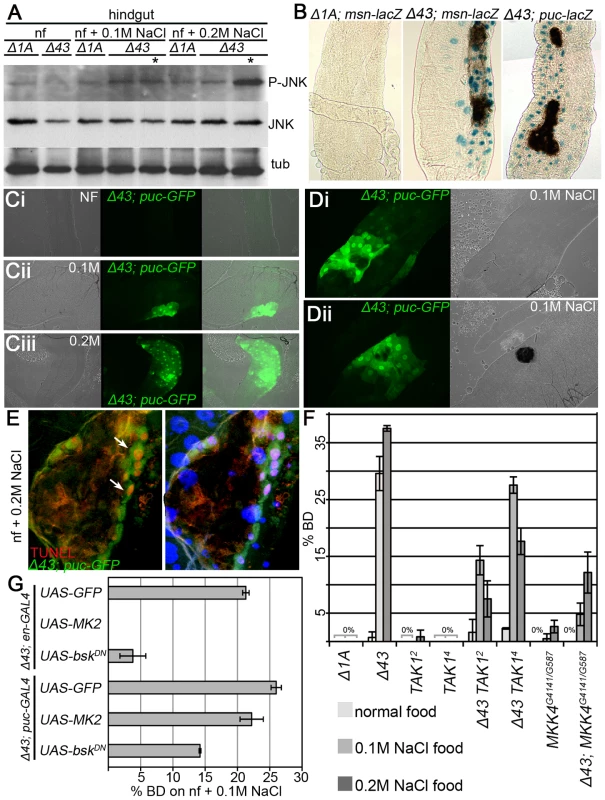
(A) JNK phosphorylation is strongly increased in hindguts of MK2 mutant larvae with BDs reared on 0.2 M NaCl food. Lanes containing lysates of larvae with BDs are marked by asterisks. (B) Reporters of JNK signalling activity (msn-lacZ and puc-lacZ) in wild-type (Δ1A) and MK2 mutant (Δ43) larvae reared on 0.2 M NaCl. In MK2 mutants, the JNK activity reporters get induced, especially close to the BDs. (C) Activation of puc-GFP in Δ43 mutants prior to BD appearance. Whereas no activation of puc-GFP occurs on normal food (Ci), fields of increased puc-GFP appear in the hindgut epithelium on 0.1 M NaCl (Cii) and 0.2 M NaCl (Ciii). The area of the patches and the number of affected ECs correlate with the strength of the stress. (D) puc-GFP induction occurs prior to the melanisation process. Hindgut of an MK2 mutant larva at the initiation of a BD (Di) and of an MK2 mutant larva after BD appearance (Dii), both reared on 0.1 M NaCl food. (E) In Δ43 mutants on 0.2 M NaCl, highest puc-GFP (green) and TUNEL staining (red) co-localise in the BD border region (white arrows). (F) Removing two copies of TAK1 or of MKK4 partially suppresses the BD phenotype of MK2 mutants. (G) Overexpression of MK2 and of BskDN in the dorsal hindgut (by means of en-GAL4) rescues the BD phenotype of MK2 mutants on 0.1 M NaCl food. Whereas re-expression of MK2 using puc-GAL4 does not rescue the BD phenotype on 0.1 M NaCl food, downregulation of JNK signalling using UAS-bskDN in combination with the puc-GAL4 driver results in a substantial suppression of the BD phenotype. Discussion
Gut homeostasis—under normal and stress conditions—is ensured by complex interactions between the intestinal epithelium, the immune system and the gut flora. Drosophila has been used as a simple model organism to address different aspects of intestinal homeostasis. Replenishment of the gut epithelium by ISCs clearly contributes to epithelial homeostasis but how the differentiated ECs are protected against stressors has remained largely unknown. We used the larval hindgut of Drosophila as a simple intestinal model organ to address how stress signalling in the hindgut ECs ensures intestinal homeostasis in the absence of proliferative cells. Our analysis identifies the p38b/MK2 signalling module as a critical component in the protection of hindgut ECs against salt stress.
We propose a model that puts a p38b/MK2 complex in the centre of stress-protection of the hindgut ECs (Figure 7). In the absence of this signalling module, cells are undergoing JNK-dependent apoptosis upon stress. The lesion in the EC monolayer results in the damage of the overlying hindgut musculature. This regional loss of the barrier function leads to systemic defects in the larvae (Figure S4), further weakening the larvae and impairing growth under stress conditions. As a consequence, pathogens and toxic substances might enter the body cavity, eventually resulting in the melanisation of pericardial cells and the induction of cecropin in the midgut (Figure S4).
Fig. 7. Protection of hindgut ECs by p38b/MK2. 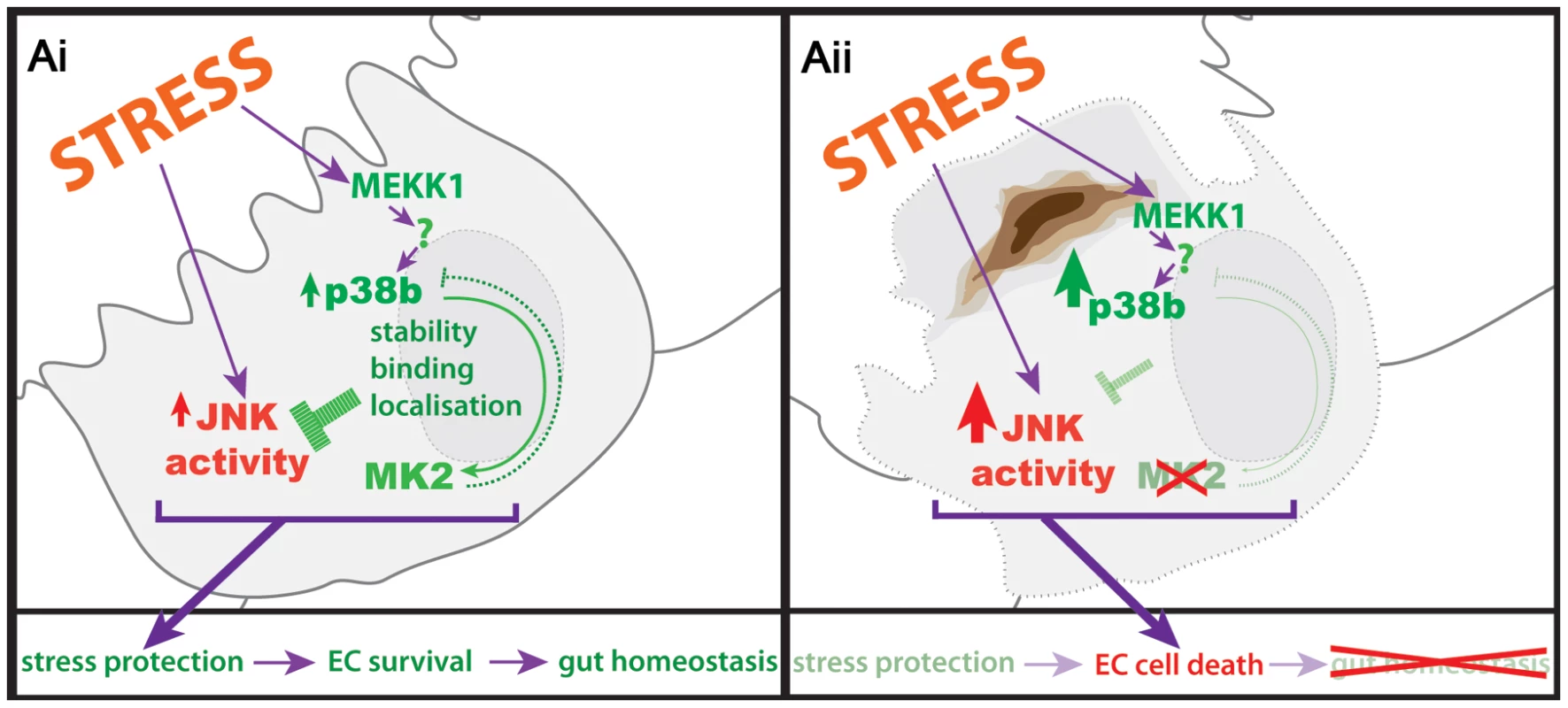
(Ai) Stressors in the gut lumen act on the hindgut ECs and induce p38 and JNK activation. The interaction of p38b with MK2 influences the localisation and protein stability of MK2, ensuring proper stress response by keeping JNK activity low. (Aii) In the absence of a functional p38b/MK2 complex, stress protection of the ECs is reduced and JNK activity is no longer kept in check, resulting in EC cell death and loss of hindgut homeostasis. Interestingly, JNK activation in MK2 mutant hindguts precedes the melanisation, and it consistently occurs in patches. Within these areas, some cells acquire highest amounts of JNK activity and eventually undergo apoptosis. The surrounding cells maintain JNK activity, forming a rim around the scar in the tissue. The number of affected ECs remains roughly constant for a given stress. Presently we do not know what determines the patches with high JNK activity within the tissue. Although the hd domain ECs of the hindgut form a homogeneous epithelium and are facing the same stressor, JNK signalling is only induced in clusters of a certain size but not in surrounding cells. Increased JNK activation was also observed in the p38α deleted intestinal epithelium of a mouse model for IBDs [53]. Furthermore, ulcerations occur in similar patchy patterns in IBDs [54]. Thus, the MK2 mutant phenotype may be useful to decipher how a group of cells within a tissue of genetically identical cells transforms into the weakest link in the chain upon stressful conditions.
Several lines of evidence support the notion that the p38b/MK2 signalling complex is key to EC protection against chronic salt stress. (1) p38b and MK2 mutant larvae both develop BDs upon stress conditions. (2) The severity of the p38b; MK2 double mutant phenotype upon stress is similar to the p38b single mutant phenotype, suggesting that they act in the same signalling pathway. (3) p38b but not p38a physically associates with MK2 via its C-terminal CD domain (DPTD motif). (4) The binding of p38b to MK2 stabilises MK2. (5) Upon co-expression, p38b but not p38a redirects MK2 to the cytoplasm. (6) Both the activation and the catalytic activity of p38b are required to efficiently relocalise MK2. (7) The binding of MK2 to p38 and the catalytic activities of both kinases are essential to protect the ECs of the larval hindgut upon salt stress. Taken together, stabilisation, localisation and activation of MK2 by p38b are required for a proper stress response.
Our genetic analysis also revealed that p38 SAPK signalling is contributing to stress protection in different ways in addition to the pivotal role of the p38b/MK2 complex. First, p38b impacts on hindgut homeostasis in an MK2-independent manner. This is apparent from the p38b mutant larvae that, in contrast to MK2 mutant larvae, develop BDs even at normal conditions. Consistently, a p38b protein version that no longer binds MK2 partially rescues the p38b mutant phenotype. Second, p38a is also required for full stress protection. The strong phenotype of MK2; p38a double mutants underscores the importance of the p38a SAPK pathway upon severe salt stress. However, the double mutants do not display an increase in BD formation but rather a decrease in viability. Thus, p38a may be involved in general stress protection that is not specific to the hindgut tissue. Third, a common p38 SAPK branch, encompassing p38a, p38b and potentially also p38c, is essential as the p38a; p38b double mutants die. Since the p38a1 allele affects the coding sequences of p38a and p38c, the p38a1; p38bd27 double mutants may even represent p38a; p38b; p38c triple mutants. However, it is unclear to date whether p38c is a pseudogene. Recent studies have suggested an involvement of p38c function in immune gene regulation, early larval survival, and fertility [41], [55]. Thus, further studies will be required to clarify whether p38c contributes to p38 signalling subbranches. Finally, the slight increase in BDs seen in MK2; p38b double mutants under normal conditions suggests that MK2 also performs a p38b-independent function in the hindgut. p38b mutants always impact on MK2 signalling since the MK2 protein is not stable and probably not correctly localised if not bound to p38b. Negative feedback regulation acting from MK2 on the activation of p38b further complicates the SAPK signalling network.
What are the upstream components regulating the p38b/MK2 complex? To our surprise, MKK3/Lic does not appear to play a role in the hindgut function of the p38b/MK2 branch. MKK3 but not MKK4 can activate p38 proteins in cell culture [56]. On the other hand, siRNA mediated knockdown of both MKK4 and MKK3 is required to fully block the activation of p38 under certain stresses in S2 cells [57]. Both p38b and MKK3/lic mutants show a strong reduction in p38 activation but no BDs are observed in MKK3/lic mutants. In contrast, mutants for MEKK1, which acts upstream of MKK3, do develop BDs similar to p38b and MK2 mutants. In mammalian cells, it has been shown that p38 can be activated independently of a MAP2 kinase [58]–[60]. However, no activation of p38 occurred in fibroblasts of MKK3 MKK6 double mutant mice [61]. Since MKK4 is a suppressor of the BD phenotype and MKK7 most likely does not activate p38 in Drosophila, a scenario of MAP2K-independent p38 activation could also apply for the p38b/MK2 signalling branch in the larval hindgut (Figure 7).
Interestingly, overexpression of the kinase-dead or of the non-activatable p38b protein version results in an even stronger phenotype than the deletion of p38b, probably by titrating upstream partners that would have additional functions besides activating p38b. A common p38a/p38b activator would be a strong candidate for such an upstream component. Saturating this common p38 activator with p38bKR or p38bAGF would essentially result in a p38a/p38b double mutant situation and therefore would explain the strong phenotype, especially at high salt conditions.
Our analysis of the p38b/MK2 signalling module in hindgut ECs reveals how deletion of SAPK members results in increased sensitivity towards a particular stressor from the molecular level to the level of the whole organism. These findings provide a new model of how hindgut homeostasis is maintained and how different SAPK branches act together in vivo to ensure cellular survival upon stress. The p38 SAPK pathway efficiently protects hindgut ECs over a wide range of stress conditions and for an astonishingly long time period of at least five days without cell replenishment. In this light, a comparative analysis of the larval and the adult hindgut (which has to be maintained for a time period of up to forty days) will be of great interest.
How does p38 interfere with JNK activation in the hindgut epithelium? In mammalian cells, p38α binds to and phosphorylates TAB1 (and potentially TAB2). As a consequence, the activities of TAK1 and thereby JNK are reduced [62]. TAB1 is not conserved in Drosophila but TAB2 might be involved in a similar negative feedback loop. TAB2 has been implicated in JNK activation in response to peptidoglycans and lipopolysaccharides. However, in the absence of TAB2, no change in JNK activation in response to Sorbitol or NaCl has been observed [63], [64]. Alternatively, p38 might induce a JNK phosphatase. p38α has been shown to impact on JNK activation by inducing DUSP1/MKP-1 in mammalian cells. Sustained JNK activation due to a loss of DUSP1/MKP-1 resulted in increased cell death in response to UV stress [65]. Interestingly, p38 activation was also increased in DUSP1/MKP-1 mutant cells but p38 activity was not linked to cell death induction. Similarly, MK2 mutant larvae bearing BDs display an overactivation of both p38 and JNK, and blocking JNK only is sufficient to prevent the BD phenotype.
Despite the more complex nature of mammalian guts, the strong conservation of stress-signalling pathways and the similar demands of ECs make it likely that our results will also be important in the context of various diseases of the intestinal system. A variety of different signalling pathways have been implicated in IBDs, underscoring the complex nature of these diseases. p38 and MK2 are critical regulators of TNFα production and are thereby associated with IBDs [8] but the role of p38 SAPK in IBDs has remained controversial [9], [10]. Our study identifies a crucial role of p38b/MK2 signalling in the first line of defence against a particular stressor in a model system devoid of an adaptive immune system. The consequences of lacking this immune system-independent protective function of a SAPK branch might parallel early steps of IBD development in intestinal epithelial cells. Indeed, a recent study has revealed tissue-specific effects of p38α in a DSS-induced mouse model for IBDs [53]. Deleting p38α in the myeloid lineage had beneficial effects, consistent with the inflammatory nature of IBDs. In contrast, p38α deletion in the intestinal epithelium resulted in a loss of gut homeostasis, marked by increased proliferation and by a reduction in goblet cells. Thus, our studies on Drosophila p38 signalling and its role in the larval hindgut provide a basis to specifically address the role of ECs in the maintenance of an intestinal epithelium in the absence of proliferation and immune response.
Materials and Methods
Fly Media and Stock Keeping
1 litre Drosophila media contains 100 g fresh yeast, 55 g cornmeal, 10 g wheat flour, 75 g sugar, and 8 g bacto-agar. For stress medium, Drosophila media were boiled and sodium chloride was added from a 5 M stock solution. 15 ml/l of a stock solution containing 33 g/L nipagin and 66 g/L nipasol in 96% EtOH was added to prevent growth of mould and bacteria. All crosses and experiments were performed at 25°C.
Fly Stocks
GE3296 was remobilised to generate the MK2 deletion mutants. y w; MK2 (genomic rescue), y w; MK2-GFP (genomic GFP reporter); y w; 86Fb pattB [GST-X] (X…p38a, p38b, p38a>b, p38b>a, p38bKR, p38bAGF), y w; 51D [MK2], y w; 51D [MK2KD], y w; 51D [mouseMK2], y w; nbyn2-GAL4 were generated in this study. For overexpression analyses, the following lines were used: y w; cad-GAL4/CyO y+, y w; byn-GAL4/TM6B, y w; en-GAL4/CyO y+, y w; NP1-GAL4/CyO y+, y w; da-GAL4 and y w; Act-GAL4. For genetic interactions, the following lines were used: y w; FRT82B D-p38a1/TM6B [66], y w; p38bd27/CyO y+ [46], y w licorned13/Binsn [46], y w; MKK4414/TM6B, y w; MKK4589/TM6B [67]; w dTAK12, w dTAK14 [63], y w; MEKK1Ur36/TM6B [45]. For hemocyte ablation, y w; He-GAL4, UAS-GFP flies were crossed to y w; UAS-Bax flies [68]. The following reporter lines have been used: nrgG00305 [69], y w; cecA1-lacZ [70]; y w; pucE69, y w; UAS-EGFP; puc-GAL4/TM6B (gift from K. Basler), and y w; msn-lacZ [52].
Stress Experiments
Females were allowed to lay eggs overnight on apple agar plates. Eggs were collected and 80–120 eggs were transferred to the different food vials. For BD quantification, larvae were analysed before reaching L3 wandering stage. For survival quantification, dead embryos were counted 24 h after seeding to the food and survival to pupae was recorded, respectively.
Plasmid Constructs and Transgenic Animals
For the MK2 genomic rescue construct, the genomic region between CG15771 and CG15770 was PCR-amplified and cloned into pCasper3. For the genomic MK2-GFP reporter, the same region was used but the MK2 coding sequence was replaced by the GFP coding sequence.
For overexpression constructs, the MK2 coding sequence was amplified and cloned into pENTR/D-TOPO (Invitrogen). For the kinase-dead MK2 protein, the mutation leading to the K49A substitution was introduced by PCR mutagenesis. The inserts were shuttled into the destination vector pTGW (http://www.ciwemb.edu/labs/murphy/Gateway%20vectors.html) for N-terminal GFP tagging. To express untagged MK2, the MK2 coding sequence was cut from the pENTR/D-TOPO and ligated into a pUAST-attB vector.
GST-tagged p38a and p38b overexpression constructs were generated by ligating the GST coding sequence in frame to the p38a or p38b coding sequence, and the resulting fusion sequences were cloned into a pUAST-attB vector. The constructs encoding the p38a or p38b protein mutants were generated by PCR mutagenesis. In the p38a→b and the p38b→a protein mutants, the EPSV motif was changed to DPTD and vice versa. The kinase-dead or non-activatable p38b protein mutants were generated by introducing mutations in the coding sequence that result in the K53R substitution and in the replacement of the TGY dual phosphorylation motif by AGF, respectively.
pUAST-attB based constructs were injected into embryos carrying a landing site (vas-φC31-zh2A; ZH-attP-51D for chromosome II and vas-φC31-zh2A; ZH-attP-86Fb for chromosome III, [71]). The MK2 genomic rescue and reporter lines were generated by co-injecting the respective plasmid with Δ2–3 helper plasmid into y w embryos.
Cell Transfection and Pull Downs
Transfection of S2 cells was done using the Effectene Transfection Reagent (Qiagen) according to the manufacturer's protocol. After four days of protein expression, cells were harvested and lysed. Pull down of GST-p38 proteins was performed using glutathione sepharose beads (Pharmacia Biotech AB). 10% of the lysates was loaded as input and the complete pull down sample was loaded onto SDS PAGE.
Dissection and Fixation of Larval Tissues
Larvae were collected in PBS on ice. Three to five larvae were transferred to new vials containing 400 µl ice-cold PBS. Larvae were dissected and the desired organs were transferred into a microfuge tube containing 500 µl of ice-cold 4% paraformaldehyde in PBS. Hindguts and midguts were fixed for 40–50′ at 4°C. Subsequently, the fixative was removed by three washing steps with cold PBS. Fixed preparations were stored in PBS at 4°C until furthre use.
Preparation of Cells for IHC
Cover slips were washed by dipping into 100% EtOH and air-dried. They were then incubated in 0.15% ConA solution (in ddH2O) for 1–2 h, washed with ddH2O and air-dried overnight.
ConA slides were placed into a small Petri dish and covered with Schneider's medium (approx. 1 ml). 200 µl S2 cells were added and allowed to settle onto the discs for 45′ to max. 90′. Cover slips were then washed once with ice-cold PBS, and 1 ml 4%PFA was added for 5′ fixation on ice followed by 10′ fixation at room temperature. Cover slips were washed three times with PBS. Cells were then permeabilised with PBT for 10′ and stored in PBS until further processing.
Antibody Stainings
Specimens were blocked by incubating in 2% NDS in PBS with 0.2% Triton X-100 (or 0.3% Triton X-100 for hindguts) for 1 h (larval tissues) or 30′ (S2 cells), respectively. Primary antibodies were added in PBS with 2% NDS and 0.2% Triton X-100 for 1 h (S2 cells) or overnight (larval tissues), respectively. Before secondary antibodies were added, samples were washed three times in PBS with 0.2% Triton X-100. Secondary antibodies were added in PBS with 2% NDS and 0.2% Triton X-100 for 1 h (S2 cells and larval tissues).
Western blot (WB) membranes were blocked in 3% membrane blocking agent (GE Healthcare) for one hour. Membranes were incubated with the primary antibodies overnight and one hour with the secondary antibodies in 3% membrane blocking agent.
Primary antibodies: rabbit anti-GST (1∶5,000 WB or 1∶500 IHC, Sigma G7781); rabbit anti-activated JNK (1∶1000 WB, Promega V793A); rabbit anti-pTGpY-p38 (1∶1000 WB, Cell Signaling 4631); mouse anti-Tubulin (1∶10,000 WB, Sigma T9026); rabbit anti-D-p38b (1∶1000 WB, [72]), and mouse anti-Hemese (1∶50, [73], [74]). The anti-Drosophila MK2 antibody was generated by Eurogentec by immunising a rabbit with the peptide H20-QPKTTPLTDDYVTSN-COOH, and the final bleed was used 1∶500 in WB.
Secondary antibodies: HRP-coupled anti-mouse IgG (Jackson ImmunoResearch; 1∶10,000; WB), HRP-coupled anti-rabbit IgG (Jackson ImmunoResearch; 1∶10,000; WB), and Cy3-coupled anti-rabbit IgG (1∶500; IHC).
Other Histological Stainings
Larvae were dissected and fixed by standard procedures. After washing with PBS, 500 µl X-gal staining solution was added, and the samples were incubated at 37°C in the dark. The staining progress was observed every 10′, and the staining reaction was stopped by two washes with PBT.
Alexa Fluor 594-conjugated phalloidin (Molecular Probes) was used to stain muscles. For apoptosis detection, the TUNEL assay kit ApopTag RED In Situ Detection Kit (Millipore S7165) was used.
Hindgut Sections
Larvae were dissected on ice and hindguts were immediately fixed in 2.5% glutaraldehyde, 1% paraformaldehyde, 1% potassium ferrocyanide, 0.1 M cacodylate buffer for 80′. After washing three times in 0.1 M cacodylate buffer, hindguts were postfixed in 1% osmium tetroxide, 1% potassium ferrocyanide, 0.1 M cacodylate buffer (pH 7.4) for 60′. Hindguts were then dehydrated in an ascending acetone series (30%>50%>70%>90%>100% 3′ each and 5′ 100%). The samples were incubated overnight in a 1∶1 acetone∶Spurr solution. After equilibration in Spurr solution for 4 h, samples were embedded in Spurr solution and hardened at 65°C overnight. 2 µm sections were made with a Supercut Reichert-Jung 2050 microtome, and sections were mounted in DPX Mountant for histology (Fluka).
Supporting Information
Zdroje
1. DanemanRRescignoM 2009 The gut immune barrier and the blood-brain barrier: are they so different? Immunity 31 722 735
2. BarkerNRidgwayRAvan EsJHvan de WeteringMBegthelH 2009 Crypt stem cells as the cells-of-origin of intestinal cancer. Nature 457 608 611
3. BarkerNvan EsJHJaksVKasperMSnippertH 2008 Very long-term self-renewal of small intestine, colon, and hair follicles from cycling Lgr5+ve stem cells. Cold Spring Harb Symp Quant Biol 73 351 356
4. BarkerNvan EsJHKuipersJKujalaPvan den BornM 2007 Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449 1003 1007
5. NenciABeckerCWullaertAGareusRvan LooG 2007 Epithelial NEMO links innate immunity to chronic intestinal inflammation. Nature 446 557 561
6. PasparakisM 2008 IKK/NF-kappaB signaling in intestinal epithelial cells controls immune homeostasis in the gut. Mucosal Immunol 1 Suppl 1 S54 57
7. KaserALeeAHFrankeAGlickmanJNZeissigS 2008 XBP1 links ER stress to intestinal inflammation and confers genetic risk for human inflammatory bowel disease. Cell 134 743 756
8. WaetzigGHSeegertDRosenstielPNikolausSSchreiberS 2002 p38 mitogen-activated protein kinase is activated and linked to TNF-alpha signaling in inflammatory bowel disease. J Immunol 168 5342 5351
9. MalamutGCabaneCDubuquoyLMalapelMDerijardB 2006 No evidence for an involvement of the p38 and JNK mitogen-activated protein in inflammatory bowel diseases. Dig Dis Sci 51 1443 1453
10. ArulampalamVPetterssonS 2002 Uncoupling the p38 MAPK kinase in IBD: a double edged sword? Gut 50 446 447
11. ter HaarEPrabhakarPLiuXLepreC 2007 Crystal structure of the p38 alpha-MAPKAP kinase 2 heterodimer. J Biol Chem 282 9733 9739
12. Ben-LevyRHooperSWilsonRPatersonHFMarshallCJ 1998 Nuclear export of the stress-activated protein kinase p38 mediated by its substrate MAPKAP kinase-2. Curr Biol 8 1049 1057
13. KotlyarovAYannoniYFritzSLaassKTelliezJB 2002 Distinct cellular functions of MK2. Mol Cell Biol 22 4827 4835
14. HittiEIakovlevaTBrookMDeppenmeierSGruberAD 2006 Mitogen-activated protein kinase-activated protein kinase 2 regulates tumor necrosis factor mRNA stability and translation mainly by altering tristetraprolin expression, stability, and binding to adenine/uridine-rich element. Mol Cell Biol 26 2399 2407
15. WangXKhalequeMAZhaoMJZhongRGaestelM 2006 Phosphorylation of HSF1 by MAPK-activated protein kinase 2 on serine 121, inhibits transcriptional activity and promotes HSP90 binding. J Biol Chem 281 782 791
16. HeidenreichONeiningerASchrattGZinckRCahillMA 1999 MAPKAP kinase 2 phosphorylates serum response factor in vitro and in vivo. J Biol Chem 274 14434 14443
17. BrookMTchenCRSantaluciaTMcIlrathJArthurJS 2006 Posttranslational regulation of tristetraprolin subcellular localization and protein stability by p38 mitogen-activated protein kinase and extracellular signal-regulated kinase pathways. Mol Cell Biol 26 2408 2418
18. ReinhardtHCAslanianASLeesJAYaffeMB 2007 p53-deficient cells rely on ATM - and ATR-mediated checkpoint signaling through the p38MAPK/MK2 pathway for survival after DNA damage. Cancer Cell 11 175 189
19. ReinhardtHCHasskampPSchmeddingIMorandellSvan VugtMA 2010 DNA damage activates a spatially distinct late cytoplasmic cell-cycle checkpoint network controlled by MK2-mediated RNA stabilization. Mol Cell 40 34 49
20. SumaraGFormentiniICollinsSSumaraIWindakR 2009 Regulation of PKD by the MAPK p38delta in insulin secretion and glucose homeostasis. Cell 136 235 248
21. SchwermannJRathinamCSchubertMSchumacherSNoyanF 2009 MAPKAP kinase MK2 maintains self-renewal capacity of haematopoietic stem cells. EMBO J 28 1392 1406
22. MurakamiSAMatsumotoAYamaokaITanimuraT 1993 Novel tissue units of regional differentiation in the gut epithelium of Drosophila, as revealed by P-element-mediated detection of enhancer. Development Genes and Evolution 203 243 249
23. MurakamiRShiotsukiY 2001 Ultrastructure of the hindgut of Drosophila larvae, with special reference to the domains identified by specific gene expression patterns. J Morphol 248 144 150
24. IwakiDDLengyelJA 2002 A Delta-Notch signaling border regulated by Engrailed/Invected repression specifies boundary cells in the Drosophila hindgut. Mech Dev 114 71 84
25. TakashimaSMkrtchyanMYounossi-HartensteinAMerriamJRHartensteinV 2008 The behaviour of Drosophila adult hindgut stem cells is controlled by Wnt and Hh signalling. Nature 454 651 655
26. BiteauBHochmuthCEJasperH 2008 JNK activity in somatic stem cells causes loss of tissue homeostasis in the aging Drosophila gut. Cell Stem Cell 3 442 455
27. ChoiYJHwangMSParkJSBaeSKKimYS 2008 Age-related upregulation of Drosophila caudal gene via NF-kappaB in the adult posterior midgut. Biochim Biophys Acta 1780 1093 1100
28. BuchonNBroderickNAKuraishiTLemaitreB 2010 Drosophila EGFR pathway coordinates stem cell proliferation and gut remodeling following infection. BMC Biol 8 152
29. BiteauBJasperH 2011 EGF signaling regulates the proliferation of intestinal stem cells in Drosophila. Development 138 1045 1055
30. StaleyBKIrvineKD 2010 Warts and Yorkie mediate intestinal regeneration by influencing stem cell proliferation. Curr Biol 20 1580 1587
31. ShawRLKohlmaierAPoleselloCVeelkenCEdgarBA 2010 The Hippo pathway regulates intestinal stem cell proliferation during Drosophila adult midgut regeneration. Development 137 4147 4158
32. KarpowiczPPerezJPerrimonN 2010 The Hippo tumor suppressor pathway regulates intestinal stem cell regeneration. Development 137 4135 4145
33. BeebeKLeeWCMicchelliCA 2010 JAK/STAT signaling coordinates stem cell proliferation and multilineage differentiation in the Drosophila intestinal stem cell lineage. Dev Biol 338 28 37
34. MathurDBostADriverIOhlsteinB 2010 A transient niche regulates the specification of Drosophila intestinal stem cells. Science 327 210 213
35. JiangHPatelPHKohlmaierAGrenleyMOMcEwenDG 2009 Cytokine/Jak/Stat signaling mediates regeneration and homeostasis in the Drosophila midgut. Cell 137 1343 1355
36. FreSPallaviSKHuygheMLaeMJanssenKP 2009 Notch and Wnt signals cooperatively control cell proliferation and tumorigenesis in the intestine. Proc Natl Acad Sci U S A 106 6309 6314
37. FoxDTSpradlingAC 2009 The Drosophila hindgut lacks constitutively active adult stem cells but proliferates in response to tissue damage. Cell Stem Cell 5 290 297
38. ParkJSKimYSYooMA 2009 The role of p38b MAPK in age-related modulation of intestinal stem cell proliferation and differentiation in Drosophila. Aging (Albany NY) 1 637 651
39. ParkJSKimYSKimJGLeeSHParkSY 2010 Regulation of the Drosophila p38b gene by transcription factor DREF in the adult midgut. Biochim Biophys Acta 1799 510 519
40. HaEMLeeKASeoYYKimSHLimJH 2009 Coordination of multiple dual oxidase-regulatory pathways in responses to commensal and infectious microbes in drosophila gut. Nat Immunol 10 949 957
41. ChenJXieCTianLHongLWuX 2010 Participation of the p38 pathway in Drosophila host defense against pathogenic bacteria and fungi. Proceedings of the National Academy of Sciences of the United States of America 107 20774 20779
42. BalakirevaMRosseCLangevinJChienYCGhoM 2006 The Ral/exocyst effector complex counters c-Jun N-terminal kinase-dependent apoptosis in Drosophila melanogaster. Mol Cell Biol 26 8953 8963
43. WuHWangMCBohmannD 2009 JNK protects Drosophila from oxidative stress by trancriptionally activating autophagy. Mech Dev
44. GaestelM 2006 MAPKAP kinases - MKs - two's company, three's a crowd. Nat Rev Mol Cell Biol 7 120 130
45. InoueHTatenoMFujimura-KamadaKTakaesuGAdachi-YamadaT 2001 A Drosophila MAPKKK, D-MEKK1, mediates stress responses through activation of p38 MAPK. EMBO J 20 5421 5430
46. CullyMGenevetAWarnePTreinsCLiuT 2010 A role for p38 stress-activated protein kinase in regulation of cell growth via TORC1. Mol Cell Biol 30 481 495
47. MinakhinaSStewardR 2006 Melanotic mutants in Drosophila: pathways and phenotypes. Genetics 174 253 263
48. EngelKKotlyarovAGaestelM 1998 Leptomycin B-sensitive nuclear export of MAPKAP kinase 2 is regulated by phosphorylation. EMBO J 17 3363 3371
49. TanoueTAdachiMMoriguchiTNishidaE 2000 A conserved docking motif in MAP kinases common to substrates, activators and regulators. Nature cell biology 2 110 116
50. IgakiT 2009 Correcting developmental errors by apoptosis: lessons from Drosophila JNK signaling. Apoptosis
51. GliseBBourbonHNoselliS 1995 hemipterous encodes a novel Drosophila MAP kinase kinase, required for epithelial cell sheet movement. Cell 83 451 461
52. BraunALemaitreBLanotRZacharyDMeisterM 1997 Drosophila immunity: analysis of larval hemocytes by P-element-mediated enhancer trap. Genetics 147 623 634
53. OtsukaMKangYJRenJJiangHWangY 2010 Distinct effects of p38alpha deletion in myeloid lineage and gut epithelia in mouse models of inflammatory bowel disease. Gastroenterology 138 1255 1265, 1265 e1251–1259
54. LeightonJAShenBBaronTHAdlerDGDavilaR 2006 ASGE guideline: endoscopy in the diagnosis and treatment of inflammatory bowel disease. Gastrointest Endosc 63 558 565
55. DavisMMPrimroseDAHodgettsRB 2008 A member of the p38 mitogen-activated protein kinase family is responsible for transcriptional induction of Dopa decarboxylase in the epidermis of Drosophila melanogaster during the innate immune response. Mol Cell Biol 28 4883 4895
56. HanZSEnslenHHuXMengXWuIH 1998 A conserved p38 mitogen-activated protein kinase pathway regulates Drosophila immunity gene expression. Mol Cell Biol 18 3527 3539
57. ZhuangZHZhouYYuMCSilvermanNGeBX 2006 Regulation of Drosophila p38 activation by specific MAP2 kinase and MAP3 kinase in response to different stimuli. Cell Signal 18 441 448
58. TannoMBassiRGorogDASaurinATJiangJ 2003 Diverse mechanisms of myocardial p38 mitogen-activated protein kinase activation: evidence for MKK-independent activation by a TAB1-associated mechanism contributing to injury during myocardial ischemia. Circ Res 93 254 261
59. LuGKangYJHanJHerschmanHRStefaniE 2006 TAB-1 modulates intracellular localization of p38 MAP kinase and downstream signaling. J Biol Chem 281 6087 6095
60. RuddCE 2005 MAPK p38: alternative and nonstressful in T cells. Nat Immunol 6 368 370
61. BranchoDTanakaNJaeschkeAVenturaJJKelkarN 2003 Mechanism of p38 MAP kinase activation in vivo. Genes & development 17 1969 1978
62. CheungPCCampbellDGNebredaARCohenP 2003 Feedback control of the protein kinase TAK1 by SAPK2a/p38alpha. The EMBO journal 22 5793 5805
63. GeukingPNarasimamurthyRBaslerK 2005 A genetic screen targeting the tumor necrosis factor/Eiger signaling pathway: identification of Drosophila TAB2 as a functionally conserved component. Genetics 171 1683 1694
64. ZhuangZHSunLKongLHuJHYuMC 2006 Drosophila TAB2 is required for the immune activation of JNK and NF-kappaB. Cellular signalling 18 964 970
65. StaplesCJOwensDMMaierJVCatoACKeyseSM 2010 Cross-talk between the p38alpha and JNK MAPK pathways mediated by MAP kinase phosphatase-1 determines cellular sensitivity to UV radiation. The Journal of biological chemistry 285 25928 25940
66. CraigCRFinkJLYagiYIpYTCaganRL 2004 A Drosophila p38 orthologue is required for environmental stress responses. EMBO Rep 5 1058 1063
67. GeukingPNarasimamurthyRLemaitreBBaslerKLeulierF 2009 A non-redundant role for Drosophila Mkk4 and hemipterous/Mkk7 in TAK1-mediated activation of JNK. PLoS ONE 4 e7709 doi:10.1371/journal.pone.0007709
68. DefayeAEvansICrozatierMWoodWLemaitreB 2009 Genetic ablation of Drosophila phagocytes reveals their contribution to both development and resistance to bacterial infection. J Innate Immun 1 322 334
69. MorinXDanemanRZavortinkMChiaW 2001 A protein trap strategy to detect GFP-tagged proteins expressed from their endogenous loci in Drosophila. Proc Natl Acad Sci U S A 98 15050 15055
70. RoosEBjorklundGEngstromY 1998 In vivo regulation of tissue-specific and LPS-inducible expression of the Drosophila Cecropin genes. Insect Mol Biol 7 51 62
71. BischofJMaedaRKHedigerMKarchFBaslerK 2007 An optimized transgenesis system for Drosophila using germ-line-specific phiC31 integrases. Proc Natl Acad Sci U S A 104 3312 3317
72. Adachi-YamadaTNakamuraMIrieKTomoyasuYSanoY 1999 p38 mitogen-activated protein kinase can be involved in transforming growth factor beta superfamily signal transduction in Drosophila wing morphogenesis. Mol Cell Biol 19 2322 2329
73. KuruczEZettervallCJSinkaRVilmosPPivarcsiA 2003 Hemese, a hemocyte-specific transmembrane protein, affects the cellular immune response in Drosophila. Proc Natl Acad Sci U S A 100 2622 2627
74. KuruczEVacziBMarkusRLaurinyeczBVilmosP 2007 Definition of Drosophila hemocyte subsets by cell-type specific antigens. Acta Biol Hung 58 Suppl 95 111
Štítky
Genetika Reprodukční medicína
Článek The T-Box Factor MLS-1 Requires Groucho Co-Repressor Interaction for Uterine Muscle SpecificationČlánek B Chromosomes Have a Functional Effect on Female Sex Determination in Lake Victoria Cichlid FishesČlánek Distinct Cdk1 Requirements during Single-Strand Annealing, Noncrossover, and Crossover RecombinationČlánek Specification of Corpora Cardiaca Neuroendocrine Cells from Mesoderm Is Regulated by Notch SignalingČlánek Ongoing Phenotypic and Genomic Changes in Experimental Coevolution of RNA Bacteriophage Qβ and
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2011 Číslo 8- Je „freeze-all“ pro všechny? Odborníci na fertilitu diskutovali na virtuálním summitu
- Kazuistika – Perspektivy využití precizované medicíny v rámci personalizované specifické terapie onkologických pacientů
- Nobelova cena za chemii pro genetické nůžky: Objev, který změní naši budoucnost?
- Technologie na bázi RNA v klinické praxi: od přebarvených petúnií k terapii vzácných a dosud jen obtížně léčitelných chorob u lidí
- „Nepředstavovali jsme si, že náš výzkum povede přímo ke vzniku nových léků, dokonce ještě za našeho života“
-
Všechny články tohoto čísla
- Polo, Greatwall, and Protein Phosphatase PP2A Jostle for Pole Position
- Genome-Wide Association Analysis of Incident Coronary Heart Disease (CHD) in African Americans: A Short Report
- The T-Box Factor MLS-1 Requires Groucho Co-Repressor Interaction for Uterine Muscle Specification
- B Chromosomes Have a Functional Effect on Female Sex Determination in Lake Victoria Cichlid Fishes
- Analysis of DNA Methylation in a Three-Generation Family Reveals Widespread Genetic Influence on Epigenetic Regulation
- PP2A-Twins Is Antagonized by Greatwall and Collaborates with Polo for Cell Cycle Progression and Centrosome Attachment to Nuclei in Drosophila Embryos
- Discovery of Sexual Dimorphisms in Metabolic and Genetic Biomarkers
- Pervasive Sharing of Genetic Effects in Autoimmune Disease
- DNA Methylation and Histone Modifications Regulate Shoot Regeneration in by Modulating Expression and Auxin Signaling
- Mutations in and Reveal That Cartilage Matrix Controls Timing of Endochondral Ossification by Inhibiting Chondrocyte Maturation
- Variance of Gene Expression Identifies Altered Network Constraints in Neurological Disease
- Frequent Beneficial Mutations during Single-Colony Serial Transfer of
- Increased Gene Dosage Affects Genomic Stability Potentially Contributing to 17p13.3 Duplication Syndrome
- Distinct Cdk1 Requirements during Single-Strand Annealing, Noncrossover, and Crossover Recombination
- Hunger Artists: Yeast Adapted to Carbon Limitation Show Trade-Offs under Carbon Sufficiency
- Suppression of Scant Identifies Endos as a Substrate of Greatwall Kinase and a Negative Regulator of Protein Phosphatase 2A in Mitosis
- Temporal Dynamics of Host Molecular Responses Differentiate Symptomatic and Asymptomatic Influenza A Infection
- MK2-Dependent p38b Signalling Protects Hindgut Enterocytes against JNK-Induced Apoptosis under Chronic Stress
- Specification of Corpora Cardiaca Neuroendocrine Cells from Mesoderm Is Regulated by Notch Signaling
- Genome-Wide Gene-Environment Study Identifies Glutamate Receptor Gene as a Parkinson's Disease Modifier Gene via Interaction with Coffee
- Identification of Functional Toxin/Immunity Genes Linked to Contact-Dependent Growth Inhibition (CDI) and Rearrangement Hotspot (Rhs) Systems
- Genomic Analysis of the Necrotrophic Fungal Pathogens and
- Celsr3 Is Required for Normal Development of GABA Circuits in the Inner Retina
- Genetic Architecture of Aluminum Tolerance in Rice () Determined through Genome-Wide Association Analysis and QTL Mapping
- Predisposition to Cancer Caused by Genetic and Functional Defects of Mammalian
- Regulation of p53/CEP-1–Dependent Germ Cell Apoptosis by Ras/MAPK Signaling
- and but Not Interact in Genetic Models of Amyotrophic Lateral Sclerosis
- Gamma-Tubulin Is Required for Bipolar Spindle Assembly and for Proper Kinetochore Microtubule Attachments during Prometaphase I in Oocytes
- Ongoing Phenotypic and Genomic Changes in Experimental Coevolution of RNA Bacteriophage Qβ and
- Genetic Architecture of a Reinforced, Postmating, Reproductive Isolation Barrier between Species Indicates Evolution via Natural Selection
- -eQTLs Reveal That Independent Genetic Variants Associated with a Complex Phenotype Converge on Intermediate Genes, with a Major Role for the HLA
- The GATA Factor ELT-1 Works through the Cell Proliferation Regulator BRO-1 and the Fusogen EFF-1 to Maintain the Seam Stem-Like Fate
- and Control Optic Cup Regeneration in a Prototypic Eye
- A Comprehensive Map of Mobile Element Insertion Polymorphisms in Humans
- An EMT–Driven Alternative Splicing Program Occurs in Human Breast Cancer and Modulates Cellular Phenotype
- Evidence for Hitchhiking of Deleterious Mutations within the Human Genome
- A Broad Brush, Global Overview of Bacterial Sexuality
- Global Chromosomal Structural Instability in a Subpopulation of Starving Cells
- A Pre-mRNA–Associating Factor Links Endogenous siRNAs to Chromatin Regulation
- Glutamine Synthetase Is a Genetic Determinant of Cell Type–Specific Glutamine Independence in Breast Epithelia
- The Repertoire of ICE in Prokaryotes Underscores the Unity, Diversity, and Ubiquity of Conjugation
- Genome-Wide Association Analysis of Autoantibody Positivity in Type 1 Diabetes Cases
- Natural Polymorphism in BUL2 Links Cellular Amino Acid Availability with Chronological Aging and Telomere Maintenance in Yeast
- Chromosome Painting Reveals Asynaptic Full Alignment of Homologs and HIM-8–Dependent Remodeling of Chromosome Territories during Meiosis
- Ku Must Load Directly onto the Chromosome End in Order to Mediate Its Telomeric Functions
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- An EMT–Driven Alternative Splicing Program Occurs in Human Breast Cancer and Modulates Cellular Phenotype
- Chromosome Painting Reveals Asynaptic Full Alignment of Homologs and HIM-8–Dependent Remodeling of Chromosome Territories during Meiosis
- A Pre-mRNA–Associating Factor Links Endogenous siRNAs to Chromatin Regulation
- Discovery of Sexual Dimorphisms in Metabolic and Genetic Biomarkers
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



