-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
Genome-Wide Association Analysis of Incident Coronary Heart Disease (CHD) in African Americans: A Short Report
African Americans have the highest rate of mortality due to coronary heart disease (CHD). Although multiple loci have been identified influencing CHD risk in European-Americans using a genome-wide association (GWAS) approach, no GWAS of incident CHD has been reported for African Americans. We performed a GWAS for incident CHD events collected during 19 years of follow-up in 2,905 African Americans from the Atherosclerosis Risk in Communities (ARIC) study. We identified a genome-wide significant SNP (rs1859023, MAF = 31%) located at 7q21 near the PFTK1 gene (HR = 0.57, 95% CI 0.46 to 0.69, p = 1.86×10−08), which replicated in an independent sample of over 8,000 African American women from the Women's Health Initiative (WHI) (HR = 0.81, 95% CI 0.70 to 0.93, p = 0.005). PFTK1 encodes a serine/threonine-protein kinase, PFTAIRE-1, that acts as a cyclin-dependent kinase regulating cell cycle progression and cell proliferation. This is the first finding of incident CHD locus identified by GWAS in African Americans.
Published in the journal: . PLoS Genet 7(8): e32767. doi:10.1371/journal.pgen.1002199
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1002199Summary
African Americans have the highest rate of mortality due to coronary heart disease (CHD). Although multiple loci have been identified influencing CHD risk in European-Americans using a genome-wide association (GWAS) approach, no GWAS of incident CHD has been reported for African Americans. We performed a GWAS for incident CHD events collected during 19 years of follow-up in 2,905 African Americans from the Atherosclerosis Risk in Communities (ARIC) study. We identified a genome-wide significant SNP (rs1859023, MAF = 31%) located at 7q21 near the PFTK1 gene (HR = 0.57, 95% CI 0.46 to 0.69, p = 1.86×10−08), which replicated in an independent sample of over 8,000 African American women from the Women's Health Initiative (WHI) (HR = 0.81, 95% CI 0.70 to 0.93, p = 0.005). PFTK1 encodes a serine/threonine-protein kinase, PFTAIRE-1, that acts as a cyclin-dependent kinase regulating cell cycle progression and cell proliferation. This is the first finding of incident CHD locus identified by GWAS in African Americans.
Introduction
Coronary heart disease (CHD) is the leading cause of death worldwide [1]. In the United States, African Americans are the most vulnerable population with regard to CHD risk factors and mortality. A recent American Heart Association report showed that African Americans are twice as likely to die from a heart-related disease compared to other ethnicities [2]. The presence of multiple CHD risk factors is 50% more likely in African Americans than in the population of European ancestry. Hypertension and diabetes are more prevalent and the highest rate of obesity is found in African American women [2]. The factors underlying these disparities are not well understood. Socioeconomic status and health care accessibility play an important role [3]. However, genetic factors are known to influence the risk of CHD [4] and population differences in the frequency and effects of these genetic factors also likely have a role.
Progress in the discovery of susceptibility genes for multiple chronic diseases and their risk factors has been made possible in recent years through genome-wide association studies (GWAS); African Americans were noticeably absent from most of these studies. GWAS have the advantage of discovering new genetic variants underlying a disease without a priori knowledge of gene location or function. More than 20 variants for CHD have been discovered in samples of European-descent so far [5]–[11].
In this study, we took advantage of rich longitudinal data on CHD incident events in the Atherosclerosis Risk in Communities (ARIC) study and performed a GWAS of incident CHD events in African Americans. Results that reached pre-specified genome-wide significance were investigated in African Americans from the Women's Health Initiative (WHI) [12].
Results/Discussion
Genome-wide association analysis for incident CHD was carried out in African Americans from the ARIC study. After 19 years of follow-up, 362 individuals developed CHD and 2,543 individuals were free of CHD. Descriptive statistics for multiple cardiovascular disease risk factors at the baseline examination are provided in Table 1. Three loci reached the pre-specified genome-wide significance threshold (p<5×10−8) (Table 2) and were considered for replication in a sample of African American women from WHI. Results are shown in Table 2. For all variants, the direction of effect was consistent in both studies, but only the variant rs1859023 reached statistical significance in WHI (Table 2). The rs1859023 minor allele A (frequency = 0.31) had a protective effect on CHD risk with a hazard ratio (HR) of 0.57 (95% CI 0.46 to 0.69, p = 1.86×10−08) in ARIC and 0.81 (95% CI 0.70 to 0.93, p = 0.005) in WHI. After adjustment for CHD risk factors (smoking, diabetes, LDL, BMI and hypertension), the result was only slightly less significant (HR = 0.59, 95% CI 0.48 to 0.73, p = 8.53×10−07 in the ARIC study). After exclusions of coronary revascularization procedures from the ARIC case definition, the association became stronger (HR 0.53, 95% CI 0.43 to 0.65, p = 2.95×10−09), possibly due to a more homogenous definition of CHD. The pooled hazard ratio of incident CHD for combined ARIC and WHI data was 0.72 (95% CI 0.64 to 0.81, p = 1.29×10−08).
Tab. 1. Baseline characteristics of the ARIC sample. 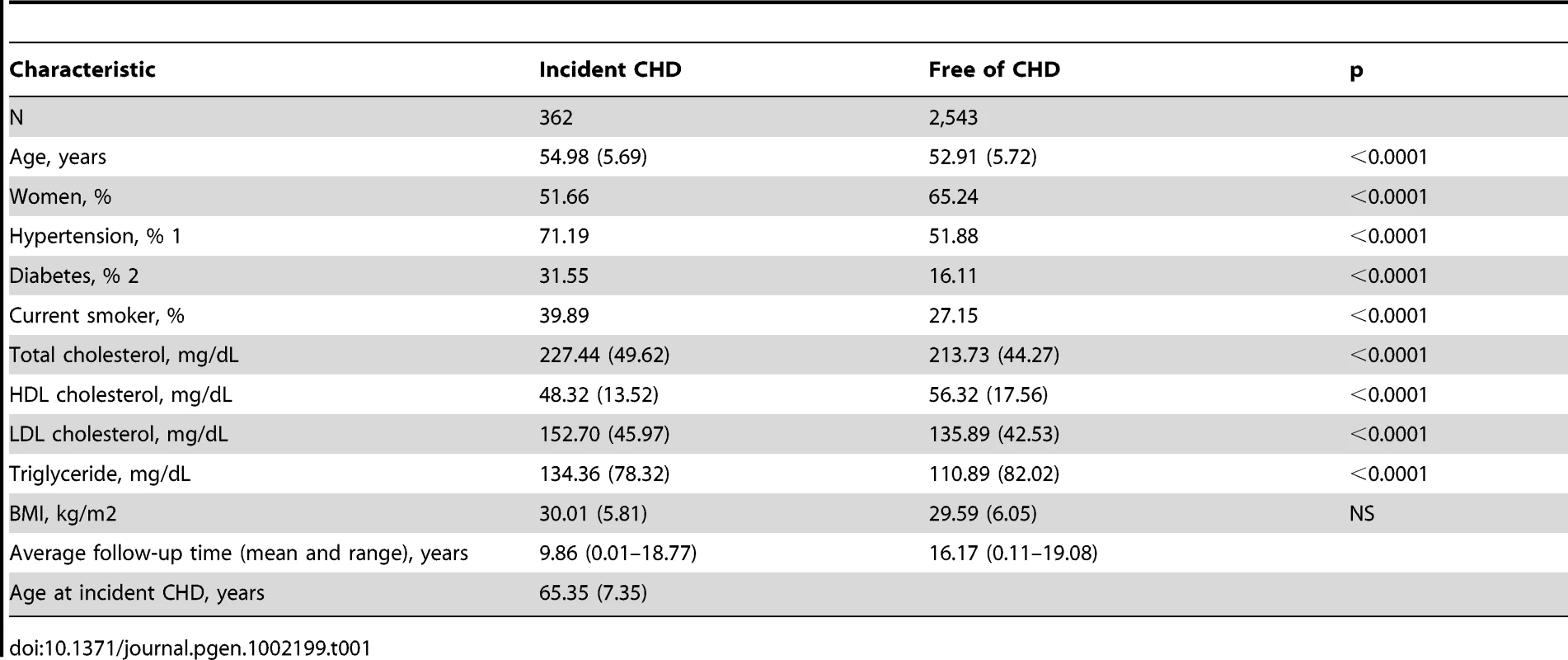
Tab. 2. Association results for genome-wide significant variants associated with incident CHD in ARIC African Americans and replication results in WHI. 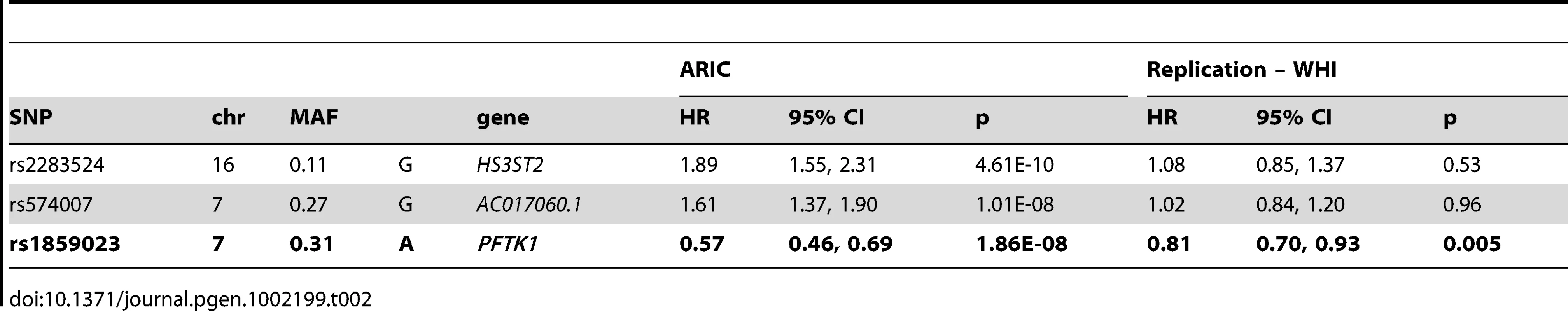
rs1859023 was not significantly associated with any of the traditional CHD risk factors at the baseline ARIC examination (LDL, HDL, SBP, DBP and BMI, data not shown). However, average thickness of the carotid artery, a common measure of subclinical atherosclerosis, was significantly different among rs1859023 genotypes (mean difference in carotid artery thickness per copy of the minor allele β = −0.0168, SE = 0.0047, p = 3.39×10−04) which suggest a role for this variant in the atherosclerotic process. When including carotid artery wall thickness as a covariate in the analysis of incident CHD attenuated but did not abolish the association with rs1859023 (HR = 0.59, 95% CI 0.48 to 0.73, p = 1.58×10−06). We attepted to replicate this observation by testing the assocation of rs1859023 and right coronary fatty streak area (% of intimal surface area) in the PDAY study consisting of 1,452 African Americans and 1,342 European Americans but the results did not reach statistical significance using a one-sided test. In the ARIC European Americans, rs1859023 was not significantly associated with incident CHD. The smallest p-value in the region surrounding rs1859023 (±50 kb) was at rs17869240 (p = 0.01, MAF = 0.07).
rs1859023 is located in an intergenic region in proximity to the PFTK1, CLDN12 and GTPBP10 genes (Figure 1). PFTK1, also known as CDK14, encodes a serine/threonine-protein kinase PFTAIRE-1 that acts as a cyclin-dependent kinase regulating cell cycle progression and cell proliferation [13]. It is highly expressed in heart tissue [14]. In the ARIC study, rs1859023 is in loose linkage disequilibrium (LD) with other SNPs in the region (r2< = 0.4). In the Yoruba HapMap data, there are only 3 SNPs with LD greater than r2 = 0.8, all located within a very short distance of rs1859023 (Figure 1) which implies that the tagging region of rs1859023 is very narrow in African-derived populations. Given the location of the rs1859023 5′ to the PFTK1 gene, these data imply that rs1859023 may affect gene expression. To test this hypothesis, we undertook expression QTL analyses using the resources provided at the SCAN – SNP and CNV Annotation Database (http://www.scandb.org/newinterface/about.html) [15]. rs1859023 predicts the expression of 6 genes with p-values less than 10−4 (Table 3). Interestingly, the evidence of rs1859023 predicting the gene expression is found only in the Yoruba population.
Fig. 1. Regional plot (ARIC) of rs1859023 association with incident CHD and LD in the region arround rs1859023 (YRI) <em class="ref">[<b>22]</b></em>, <em class="ref">[23]</em>. ![Regional plot (ARIC) of rs1859023 association with incident CHD and LD in the region arround rs1859023 (YRI) <em class="ref">[<b>22]</b></em>, <em class="ref">[23]</em>.](https://pl-master.mdcdn.cz/media/cache/media_object_image_small/media/image/563d3dd103e0347a65fcd84e807ddeb8.png)
Tab. 3. Genes whose expression levels are associated with rs1859023. 
A variant close to PFTK1, rs10499903, located ∼60 kb from rs1859023 was associated with ankle brachial index (ABI) in European Americans from the Framingham Heart Study (FHS) [16]. Given the different ethnic backgrounds of the two studies, and expected difference of LD patterns within the region between two samples, the Framingham result strengthens our finding and further suggests a role for the PFTK1 gene region in the atherosclerotic process.
Some of European-discovered CHD genes have been reported to also influence CHD in African Americans [17]. However, this is the first reported GWAS finding of a CHD risk locus in African Americans. Although the sample size is less than contemporary GWAS publications in European-Americans (i.e. often exceeding 100,000 individuals (e.g. [18]), we have combined all of the available well-powered incident CHD data in African Americans with genotype data and are able to present results based on 133,415 person-years of follow-up. In addition, supporting evidence is provided by the association with subclinical atherosclerosis and expression QTL analyses. In conclusion, we have identified a region near the PFTK1 gene as being associated with incident CHD and subclinical atherosclerosis in African Americans. Further studies are needed to examine the cellular or metabolic mechanisms underlying this association, and large population-based studies of minority populations are necessary to more fully understand the impact of genetic factors on multiple phenotypes in those that bear a disproportionate burden of disease.
Materials and Methods
The ARIC (Atherosclerosis Risk in Communities) study is a population-based prospective cohort study of cardiovascular disease and its risk factors [19]. ARIC includes 15,792 persons aged 45–64 years at baseline (1987–89), randomly chosen from four US communities. Of these individuals, 4,266 are self-reported African Americans. Cohort members completed four clinic examinations, conducted approximately three years apart between 1987 and 1998, and followed with annual phone interviews since 1987.
Incident CHD in ARIC was ascertained by contacting participants annually, identifying hospitalizations and deaths during the prior year, and by surveying discharge lists from local hospitals and death certificates from state vital statistics offices for potential cardiovascular events [19]. A CHD event was defined as a validated definite or probable hospitalized MI, a definite CHD death, an unrecognized MI defined by ARIC ECG readings, or coronary revascularization. Participants were excluded from analyses if they had a positive or unknown history of prevalent stroke, transient ischemic attack/stroke symptoms, or CHD at the initial visit and/or being of non–African American ethnicity. Real-time, B-mode ultrasound was used to evaluate the carotid arterial intima-media wall thickness as an indicator of atherosclerosis in the ARIC study and the detailed description of its measurement is described elsewhere [17].
Genotyping was done in 15,020 ARIC participants using the Affymetrix Genome-Wide Human SNP Array 6.0. A total of 3,182 individuals remained after excluding individuals of non African American ethnicity, subjects who did not consent DNA use, unintentional duplicates with higher missing genotype rates, suspected mixed/contaminated samples, scans from one problem plate, samples with a mismatch between called and phenotypic sex, samples with genotype mismatch with 39 previously genotyped SNPs, suspected first-degree relative of an included individual, and genetic outliers based on average IBS statistics and principal components analysis using EIGENSTRAT. SNPs were excluded due to having no chromosome location, being monomorphic, having a call rate <95% and HWE-p<10−5. In this analysis, we considered only variants with a MAF greater than 10%.
Cox proportional hazards models with adjustment for age, gender and the first three principal components derived from EIGENSTRAT were used to estimate CHD hazard rate ratios (HRs) over a 19-year period (362 cases) under an additive genetic model. These analyses were done using PLINK and an R application for survival regression analyses. We define as “genome-wide significant” all associations with p<5×10−8. We define replication to be a significant (p<0.05) and directionally consistent association in an independent sample.
The WHI has two major components: (1) a clinical trial that enrolled and randomized 68,132 women ages 50–79 into at least one of three clinical trials; and (2) an observational study that enrolled 93,676 women ages 50–79 into a parallel prospective cohort study [12]. WHI participants were recruited from 1993–1998 at 40 clinical centers across the U.S. During follow-up, incident CHD events were adjudicated locally and centrally from medical records including hospital discharge summaries, ICD-9 codes, diagnostic, laboratory, surgical, and pathology reports by trained physicians blinded to randomized intervention and exposure status [20]. In the WHI replication sample, CHD was defined as MI, coronary revascularization, hospitalized angina, or CHD death. Definite and probable nonfatal MI required overnight hospitalization and was defined according to an algorithm based on standardized criteria using cardiac pain, cardiac enzymes and troponin levels, and ECG findings. CHD death was defined as death consistent with underlying cause of CHD plus one or more of the following: hospitalization for MI within 28 days prior to death, previous angina or myocardial infarction, death due to a procedure related to CHD, or a death certificate consistent with underlying cause of atherosclerotic CHD.
Of a total of 26,045 (17%) women from minority groups, 8,515 self identified African American women who had consented to genetic research were eligible for the WHI GWAS project. Genotyping was performed on the Affymetrix 6.0 array. After excluding samples due to genotyping failure, cryptic relatedness, and discrepancy between genetic ancestry and self-reported race, there were 8,421 WHI African Americans. Participants were further excluded from analyses if they had a positive or unknown history of prevalent stroke, transient ischemic attack/stroke symptoms, or CHD at the initial visit. A total of 862 incident first CHD events occurred among 8,155 eligible African American women without baseline CHD (Table 4). The mean age at study entry was 61.6+/−7.0 years (range 50–79). The mean baseline age of the cases was 64.2+/−7.2, and the mean baseline age of the non-cases was 61.3+/−6.9. The mean time to CHD event was 5.29+/−3.19 years. The mean age at CHD event was 69.5+/−7.5 years.
Tab. 4. Baseline characteristics of the WHI sample. 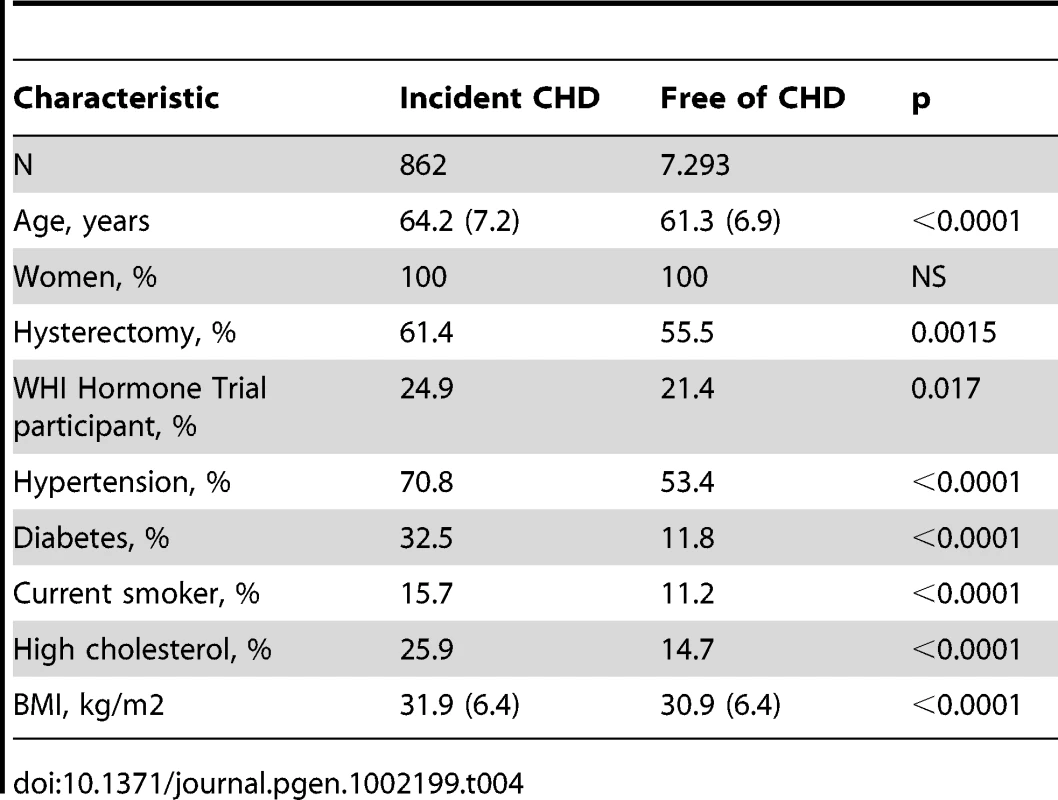
This study was approved by the participating institutional IRBs, and all ARIC and WHI participants provided written informed consent, involving the sharing of data with the scientific community.
The Pathobiolobical Determinants of Atherosclerosis in Youth (PDAY) study is composed of subjects who were 15 to 34 years of age when they died of non-CVD related causes (accidents, homicides or suicides). The purpose of PDAY was to evaluate early development of atherosclerosis [21]. For this replication analysis, we genotyped rs1859023 in 2,794 individuals from PDAY - 1,452 African Americans and 1,342 European Americans and tested the association with fatty streak area in the right coronary artery.
Zdroje
1. WHO 2010 Global burden of coronary heart disease. http://wwwwhoint/cardiovascular_diseases/en/cvd_atlas_13_coronaryHDpdf
2. Lloyd-JonesDAdamsRJBrownTMCarnethonMDaiS Heart disease and stroke statistics–2010 update: a report from the American Heart Association. Circulation 121 e46 e215
3. ClarkLT 2005 Issues in minority health: atherosclerosis and coronary heart disease in African Americans. Med Clin North Am 89 977 1001, 1994
4. LusisAJMarRPajukantaP 2004 Genetics of atherosclerosis. Annu Rev Genomics Hum Genet 5 189 218
5. KathiresanSVoightBFPurcellSMusunuruKArdissinoD 2009 Genome-wide association of early-onset myocardial infarction with single nucleotide polymorphisms and copy number variants. Nat Genet 41 334 341
6. TregouetDAKonigIRErdmannJMunteanuABraundPS 2009 Genome-wide haplotype association study identifies the SLC22A3-LPAL2-LPA gene cluster as a risk locus for coronary artery disease. Nat Genet 41 283 285
7. ErdmannJGrosshennigABraundPSKonigIRHengstenbergC 2009 New susceptibility locus for coronary artery disease on chromosome 3q22.3. Nat Genet 41 280 282
8. OzakiKSatoHInoueKTsunodaTSakataY 2009 SNPs in BRAP associated with risk of myocardial infarction in Asian populations. Nat Genet 41 329 333
9. SamaniNJErdmannJHallASHengstenbergCManginoM 2007 Genomewide association analysis of coronary artery disease. N Engl J Med 357 443 453
10. SamaniNJDeloukasPErdmannJHengstenbergCKuulasmaaK 2009 Large scale association analysis of novel genetic loci for coronary artery disease. Arterioscler Thromb Vasc Biol 29 774 780
11. SchunkertHKonigIRKathiresanSReillyMPAssimesTL Large-scale association analysis identifies 13 new susceptibility loci for coronary artery disease. Nat Genet 43 333 338
12. 1998 Design of the Women's Health Initiative clinical trial and observational study. The Women's Health Initiative Study Group. Control Clin Trials 19 61 109
13. ShuFLvSQinYMaXWangX 2007 Functional characterization of human PFTK1 as a cyclin-dependent kinase. Proc Natl Acad Sci U S A 104 9248 9253
14. YangTChenJY 2001 Identification and cellular localization of human PFTAIRE1. Gene 267 165 172
15. GamazonERZhangWKonkashbaevADuanSKistnerEO SCAN: SNP and copy number annotation. Bioinformatics 26 259 262
16. O'DonnellCJCupplesLAD'AgostinoRBFoxCSHoffmannU 2007 Genome-wide association study for subclinical atherosclerosis in major arterial territories in the NHLBI's Framingham Heart Study. BMC Med Genet 8 Suppl 1 S4
17. KralBGMathiasRASuktitipatBRuczinskiIVaidyaD A common variant in the CDKN2B gene on chromosome 9p21 protects against coronary artery disease in Americans of African ancestry. J Hum Genet 56 224 229
18. TeslovichTMMusunuruKSmithAVEdmondsonACStylianouIM Biological, clinical and population relevance of 95 loci for blood lipids. Nature 466 707 713
19. ARIC 1989 The Atherosclerosis Risk in Communities (ARIC) Study: design and objectives. The ARIC investigators. Am J Epidemiol 129 687 702
20. CurbJDMcTiernanAHeckbertSRKooperbergCStanfordJ 2003 Outcomes ascertainment and adjudication methods in the Women's Health Initiative. Ann Epidemiol 13 S122 128
21. HixsonJE 1991 Apolipoprotein E polymorphisms affect atherosclerosis in young males. Pathobiological Determinants of Atherosclerosis in Youth (PDAY) Research Group. Arterioscler Thromb 11 1237 1244
22. PruimRJWelchRPSannaSTeslovichTMChinesPS LocusZoom: regional visualization of genome-wide association scan results. Bioinformatics 26 2336 2337
23. JohnsonADHandsakerREPulitSLNizzariMMO'DonnellCJ 2008 SNAP: a web-based tool for identification and annotation of proxy SNPs using HapMap. Bioinformatics 24 2938 2939
Štítky
Genetika Reprodukční medicína
Článek The T-Box Factor MLS-1 Requires Groucho Co-Repressor Interaction for Uterine Muscle SpecificationČlánek B Chromosomes Have a Functional Effect on Female Sex Determination in Lake Victoria Cichlid FishesČlánek Distinct Cdk1 Requirements during Single-Strand Annealing, Noncrossover, and Crossover RecombinationČlánek Specification of Corpora Cardiaca Neuroendocrine Cells from Mesoderm Is Regulated by Notch SignalingČlánek Ongoing Phenotypic and Genomic Changes in Experimental Coevolution of RNA Bacteriophage Qβ and
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2011 Číslo 8- Kazuistika – Perspektivy využití precizované medicíny v rámci personalizované specifické terapie onkologických pacientů
- Nobelova cena za chemii pro genetické nůžky: Objev, který změní naši budoucnost?
- Technologie na bázi RNA v klinické praxi: od přebarvených petúnií k terapii vzácných a dosud jen obtížně léčitelných chorob u lidí
- „Nepředstavovali jsme si, že náš výzkum povede přímo ke vzniku nových léků, dokonce ještě za našeho života“
- Bezplatné služby pro diagnostiku ATTRv amyloidózy pro kardiology
-
Všechny články tohoto čísla
- Polo, Greatwall, and Protein Phosphatase PP2A Jostle for Pole Position
- Genome-Wide Association Analysis of Incident Coronary Heart Disease (CHD) in African Americans: A Short Report
- The T-Box Factor MLS-1 Requires Groucho Co-Repressor Interaction for Uterine Muscle Specification
- B Chromosomes Have a Functional Effect on Female Sex Determination in Lake Victoria Cichlid Fishes
- Analysis of DNA Methylation in a Three-Generation Family Reveals Widespread Genetic Influence on Epigenetic Regulation
- PP2A-Twins Is Antagonized by Greatwall and Collaborates with Polo for Cell Cycle Progression and Centrosome Attachment to Nuclei in Drosophila Embryos
- Discovery of Sexual Dimorphisms in Metabolic and Genetic Biomarkers
- Pervasive Sharing of Genetic Effects in Autoimmune Disease
- DNA Methylation and Histone Modifications Regulate Shoot Regeneration in by Modulating Expression and Auxin Signaling
- Mutations in and Reveal That Cartilage Matrix Controls Timing of Endochondral Ossification by Inhibiting Chondrocyte Maturation
- Variance of Gene Expression Identifies Altered Network Constraints in Neurological Disease
- Frequent Beneficial Mutations during Single-Colony Serial Transfer of
- Increased Gene Dosage Affects Genomic Stability Potentially Contributing to 17p13.3 Duplication Syndrome
- Distinct Cdk1 Requirements during Single-Strand Annealing, Noncrossover, and Crossover Recombination
- Hunger Artists: Yeast Adapted to Carbon Limitation Show Trade-Offs under Carbon Sufficiency
- Suppression of Scant Identifies Endos as a Substrate of Greatwall Kinase and a Negative Regulator of Protein Phosphatase 2A in Mitosis
- Temporal Dynamics of Host Molecular Responses Differentiate Symptomatic and Asymptomatic Influenza A Infection
- MK2-Dependent p38b Signalling Protects Hindgut Enterocytes against JNK-Induced Apoptosis under Chronic Stress
- Specification of Corpora Cardiaca Neuroendocrine Cells from Mesoderm Is Regulated by Notch Signaling
- Genome-Wide Gene-Environment Study Identifies Glutamate Receptor Gene as a Parkinson's Disease Modifier Gene via Interaction with Coffee
- Identification of Functional Toxin/Immunity Genes Linked to Contact-Dependent Growth Inhibition (CDI) and Rearrangement Hotspot (Rhs) Systems
- Genomic Analysis of the Necrotrophic Fungal Pathogens and
- Celsr3 Is Required for Normal Development of GABA Circuits in the Inner Retina
- Genetic Architecture of Aluminum Tolerance in Rice () Determined through Genome-Wide Association Analysis and QTL Mapping
- Predisposition to Cancer Caused by Genetic and Functional Defects of Mammalian
- Regulation of p53/CEP-1–Dependent Germ Cell Apoptosis by Ras/MAPK Signaling
- and but Not Interact in Genetic Models of Amyotrophic Lateral Sclerosis
- Gamma-Tubulin Is Required for Bipolar Spindle Assembly and for Proper Kinetochore Microtubule Attachments during Prometaphase I in Oocytes
- Ongoing Phenotypic and Genomic Changes in Experimental Coevolution of RNA Bacteriophage Qβ and
- Genetic Architecture of a Reinforced, Postmating, Reproductive Isolation Barrier between Species Indicates Evolution via Natural Selection
- -eQTLs Reveal That Independent Genetic Variants Associated with a Complex Phenotype Converge on Intermediate Genes, with a Major Role for the HLA
- The GATA Factor ELT-1 Works through the Cell Proliferation Regulator BRO-1 and the Fusogen EFF-1 to Maintain the Seam Stem-Like Fate
- and Control Optic Cup Regeneration in a Prototypic Eye
- A Comprehensive Map of Mobile Element Insertion Polymorphisms in Humans
- An EMT–Driven Alternative Splicing Program Occurs in Human Breast Cancer and Modulates Cellular Phenotype
- Evidence for Hitchhiking of Deleterious Mutations within the Human Genome
- A Broad Brush, Global Overview of Bacterial Sexuality
- Global Chromosomal Structural Instability in a Subpopulation of Starving Cells
- A Pre-mRNA–Associating Factor Links Endogenous siRNAs to Chromatin Regulation
- Glutamine Synthetase Is a Genetic Determinant of Cell Type–Specific Glutamine Independence in Breast Epithelia
- The Repertoire of ICE in Prokaryotes Underscores the Unity, Diversity, and Ubiquity of Conjugation
- Genome-Wide Association Analysis of Autoantibody Positivity in Type 1 Diabetes Cases
- Natural Polymorphism in BUL2 Links Cellular Amino Acid Availability with Chronological Aging and Telomere Maintenance in Yeast
- Chromosome Painting Reveals Asynaptic Full Alignment of Homologs and HIM-8–Dependent Remodeling of Chromosome Territories during Meiosis
- Ku Must Load Directly onto the Chromosome End in Order to Mediate Its Telomeric Functions
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- An EMT–Driven Alternative Splicing Program Occurs in Human Breast Cancer and Modulates Cellular Phenotype
- Chromosome Painting Reveals Asynaptic Full Alignment of Homologs and HIM-8–Dependent Remodeling of Chromosome Territories during Meiosis
- A Pre-mRNA–Associating Factor Links Endogenous siRNAs to Chromatin Regulation
- Discovery of Sexual Dimorphisms in Metabolic and Genetic Biomarkers
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Autoři: prof. MUDr. Pavel Horák, CSc., doc. MUDr. Ludmila Brunerová, Ph.D., doc. MUDr. Václav Vyskočil, Ph.D., prim. MUDr. Richard Pikner, Ph.D., MUDr. Olga Růžičková, MUDr. Jan Rosa, prof. MUDr. Vladimír Palička, CSc., Dr.h.c.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



