-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praxe
Comparative and Functional Genomics of PD630 for Biofuels Development
The Actinomycetales bacteria Rhodococcus opacus PD630 and Rhodococcus jostii RHA1 bioconvert a diverse range of organic substrates through lipid biosynthesis into large quantities of energy-rich triacylglycerols (TAGs). To describe the genetic basis of the Rhodococcus oleaginous metabolism, we sequenced and performed comparative analysis of the 9.27 Mb R. opacus PD630 genome. Metabolic-reconstruction assigned 2017 enzymatic reactions to the 8632 R. opacus PD630 genes we identified. Of these, 261 genes were implicated in the R. opacus PD630 TAGs cycle by metabolic reconstruction and gene family analysis. Rhodococcus synthesizes uncommon straight-chain odd-carbon fatty acids in high abundance and stores them as TAGs. We have identified these to be pentadecanoic, heptadecanoic, and cis-heptadecenoic acids. To identify bioconversion pathways, we screened R. opacus PD630, R. jostii RHA1, Ralstonia eutropha H16, and C. glutamicum 13032 for growth on 190 compounds. The results of the catabolic screen, phylogenetic analysis of the TAGs cycle enzymes, and metabolic product characterizations were integrated into a working model of prokaryotic oleaginy.
Published in the journal: . PLoS Genet 7(9): e32767. doi:10.1371/journal.pgen.1002219
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1002219Summary
The Actinomycetales bacteria Rhodococcus opacus PD630 and Rhodococcus jostii RHA1 bioconvert a diverse range of organic substrates through lipid biosynthesis into large quantities of energy-rich triacylglycerols (TAGs). To describe the genetic basis of the Rhodococcus oleaginous metabolism, we sequenced and performed comparative analysis of the 9.27 Mb R. opacus PD630 genome. Metabolic-reconstruction assigned 2017 enzymatic reactions to the 8632 R. opacus PD630 genes we identified. Of these, 261 genes were implicated in the R. opacus PD630 TAGs cycle by metabolic reconstruction and gene family analysis. Rhodococcus synthesizes uncommon straight-chain odd-carbon fatty acids in high abundance and stores them as TAGs. We have identified these to be pentadecanoic, heptadecanoic, and cis-heptadecenoic acids. To identify bioconversion pathways, we screened R. opacus PD630, R. jostii RHA1, Ralstonia eutropha H16, and C. glutamicum 13032 for growth on 190 compounds. The results of the catabolic screen, phylogenetic analysis of the TAGs cycle enzymes, and metabolic product characterizations were integrated into a working model of prokaryotic oleaginy.
Introduction
Bio-Diesel is an energy-rich portable fuel derived mainly from triacylglycerols (TAGs). Biodiesel and related fuels are extracted from oleaginous organisms, both photosynthetic and non-photosynthetic, that use available energy sources to fix carbon into high levels of stored lipids. In chemoheterotrophic organisms TAGs are synthesized by bioconversion of organic compounds such as the sugars and organic acids derived from globally-abundant cellulosic biomass. A genetic understanding of the oleaginous metabolism of chemoheterotrophic species like Rhodococcus provides critical insight for biofuels development.
The high GC content Gram-positive Actinomycetales bacteria Rhodococcus opacus PD630 and Rhodococcus jostii RHA1, a close relative that has a completely sequenced genome [1], were previously shown to accumulate large amounts of TAGs and wax esters (WEs) [2], [3], [4]. Rhodococcus species present an attractive target for industrial processes due to high substrate tolerances and high density culturing on a rapid time scale as compared to many photosynthetic organisms [5], [6]. The oleaginous metabolism of Rhodococcus goes beyond abundant lipid biosynthesis to include diverse hydrocarbon catabolism. R. jostii RHA1 was isolated from soil containing 1,2,3,4,5,6-hexachlorocyclohexane (Lindane) [7], while R. opacus PD630 was enriched on phenyldecane as a sole carbon source after isolation from soil sampled at a gas works plant [2]. Rhodococcus can catabolize and detoxify several aromatic hydrocarbons that contaminate soil from industrial waste products. These toxic substrates include polychlorinated biphenyls (PCBs) [8], [9], [10], [11] and other halogenated compounds such as Lindane that was used in large quantity for agricultural practices.
Limitation of an essential nutrient stimulates enzymatic conversion of the non-limiting essential nutrients into stored polymers such as phosphorous conversion to poly-phosphate [4], acetyl - and other short acyl-CoAs conversion to polyhydroxyalkanoates (PHAs) [12], [13], [14], or the production of TAGs and WEs from these same short chain acyl-CoA primers [2], [15]. Most prokaryotes store carbon as polyhydroxyalkanoic acids (PHAs) when other essential nutrients such as reduced nitrogen are limiting. By contrast bacteria in the order Actinomycetales have uniquely developed a storage lipid cycle that leads to accumulation of TAGs and WEs [16]. Abundant TAGs accumulation in Rhodococcus provides a pool of fatty acids for β-oxidation as cellular fuel, components of the plasma membrane, and substrates for the enzymatic production of the very-long and highly-modified extracellular lipids characteristic of Actinomycetales.
Lipid metabolism in the genus Mycobacterium has been a major focus of scientific research due to the effect pharmacological inhibitors of lipid biosynthesis such as isoniazid [17], [18], [19], thiolactamycin, and pyrazinamide [20] have on killing pathogenic mycobacteria. Whole-genome views of lipid metabolism in mycobacteria reveal these bacteria have developed several lipid biosynthesis systems and a large number of genes to support diverse and abundant lipid biosynthesis. In Mycobacterium an order to the enzyme activity of the lipid synthases has been established through genetic, biochemical, and pharmacological evidence; wherein lipids are biosynthesized de novo by the multifunctional FAS type 1a enzyme followed by further elongation via the FAS II system and the multifunctional MAS-family type 1b synthases. Collectively these 3 fatty acid synthase systems produce 2 classes of fatty acyl-CoAs that differ in chain length. One class contains lipids <20 carbons (C20) that are components of the plasma membrane and the storage lipids TAGs and WEs. Another class contains lipids >C26 that are built by multiple synthases and can reach lengths as long as C90 in some Mycobacterium species [19], [21] but only C60 in Rhodococcus opacus [22] and C54 molecules have been observed in R. equi [23]. The longer chain length lipids are used in Actinomycetales to build a protective extracellular coat that helps these bacteria survive in harsh environments whether it be the phagosome of a macrophage or contaminated soil enriched in toxic organic compounds. The interplay between multiple lipid biosynthesis systems in Actinomycetales [19] requires genetic understanding for engineering the flow of carbon to desired lipid types.
Results
Metabolic Reconstruction of Rhodococcus opacus PD630
To establish a genetic model of Rhodococcus metabolism, we generated a high quality draft sequence of the Rhodococcus opacus PD630 genome. DNA sequencing with 454 shotgun and 3 kb paired-end reads resulted in 16 large scaffolds containing 9.27 Mb of assembled DNA sequence. We stitched the gapped genome scaffolds based on extensive chromosomal-synteny with the complete genome sequences of related Rhodococcus species R. jostii RHA1 and R. opacus B4 (Figure S1).
The R. opacus PD630 genome contained 8632 genes that underwent metabolic reconstruction using pathway tools software [24] resulting in a model containing 1735 metabolic reactions. Enzymes were connected to metabolic reactions based on enzyme commission numbers (EC#) that were assigned by the EFICAz2 algorithm [25] and by gene-name recognition within pathway tools software. This automated EC# assignment allowed for multiple genomes to undergo metabolic reconstruction in parallel. Comparisons between metabolic reconstructions for a set of 8 phylogenetically related and one outlier species Ralstonia eutropha H16 that were assembled in this way can be browsed at (http://tinyurl.com/opacuscyc14-5-comparative). The resulting initial metabolic reconstruction of R. opacus PD630 Opacuscyc14.5_comparative contains 400 metabolic pathways and 135 transport reactions.
A more complete metabolic reconstruction of R. opacus PD630 by pathway hole filling using the pathway tools 14.5 software, additional EC # assignments made with the database at Kyoto Encyclopedia of Gene and Genomes KEGG (http://www.genome.jp/kegg/), and limited manual curation (outlined in Figure S2) resulted in a metabolic reconstruction containing an additional 282 metabolic reactions, 44 metabolic pathways, and eight transport reactions. Opacuscyc_14.5 was improved by refining the metabolic model of TAGs biosynthesis and degradation by metabolic product characterization of uncommon fatty acids that accumulate to high levels in Rhodococcus. The results of a screen for bacterial growth on 190 metabolic compounds were used as a multi-genic test of the reconstruction, described in more detail below. Comparison of the results of the catabolic screen with the metabolic pathway predictions revealed that precision was 65% before refinement and 71% after refinement (Table S1). The current working model of Opacuscyc14.5 contains 2017 metabolic reactions, 444 metabolic pathways, and 143 transport reactions that can be browsed at (http://tinyurl.com/4dv5m32).
Rhodococcus Oleaginy Resulted from Key Genes That Emerged in Actinomycetales
Several eubacteria of the order of Actinobacteria including Rhodococcus, Corynebacterium, Mycobacterium, and Bifidobacterium are distinguished for having both the type 1a fatty acid synthase (FAS) (Figure 1a) and a FAS II lipid biosynthesis system. FAS is a polyketide synthase related protein containing all of the necessary enzymatic activities for de novo lipid biosynthesis (Figure 1b). In mycobacteria FAS has been shown to elongate C2 and C3 carbon acyl-CoAs into C16, C18, C24, and C26 fatty acyl-CoAs [26], [27]. In the suborder of bacteria Corynebacterineae, the FAS II system of lipid biosynthesis elongates FAS product fatty acyl-CoAs through the enzymatic activity of 6 families of enzymes (Figure 1c) for biosynthesis of mycolic acids and related cell wall associated lipids. The FAS II system in these Actinomycetales operates on C16–C26 length fatty acyl-CoAs not the usual short chain lipid biosynthesis substrates characteristic of the related enzymes in bacteria, chloroplasts, and mitochondria. The number of unique FAS genes is shown in Figure 1a in the context of a phylogenetic tree of genera built by AMPHORA [28]. Unlike the widespread taxonomic representation of the FAS II genes, the FAS type 1a gene in Rhodococcus has genus representation in prokaryotes is limited to only within Actinomycetales. The FAS gene is likely to have emerged in Actinobacteria (Figure 1a) and was horizontally transferred to only those eukaryotic branches containing fungi and stramenopiles (Figure 2a).
Fig. 1. Phylogenetic and metabolic pathway features of lipid biosynthesis in Rhodococcus. 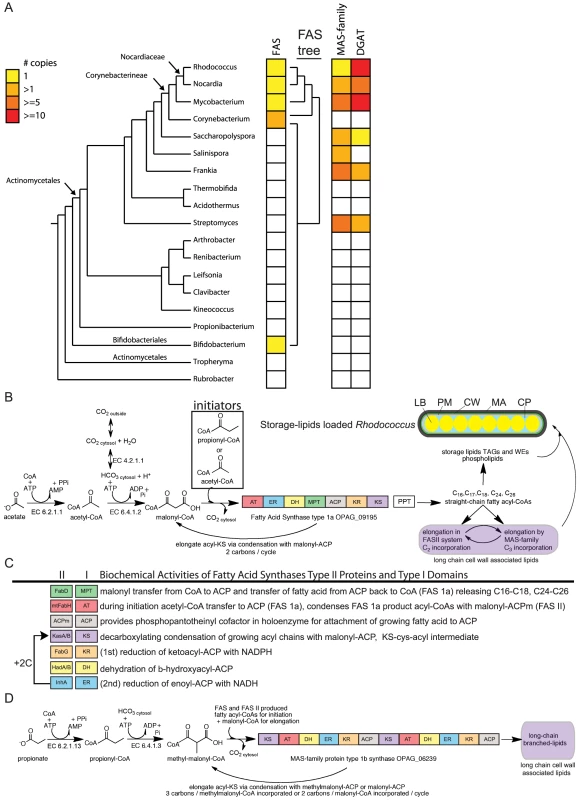
a) A phylogenetic representation of oleaginous genes in Actinomycetales. An AMPHORA-based tree of related genera (left) provides context to the copy number of unique oleaginous genes within each genera presented in three columns to the right. Remarkable oleaginous genes in Actinobacteria are fatty acid synthase (FAS) type 1a, MAS-family type 1b, and diacylglycerol acyl-transferases (DGATs). Oleaginous gene family members in each genus were counted and color-coded from 0 to >10. A phylogenetic tree of the FAS type 1a gene is presented to the right of the FAS column. b) Fatty acids biosynthesis in Rhodococcus. Acetyl-CoA is the product of many biochemical reactions and a limiting substrate in lipid biosynthesis. Acetyl-CoA can also be generated from acetic acid found in the environment by ligation to –CoA. ATP-hydrolysis enables bicarbonate coupling to acetyl-CoA forming malonyl-CoA, the substrate used for elongation. The lipid biosynthesis -CoA substrates are incorporated in Rhodococcus by three fatty acid biosynthesis systems that begins with multifunctional FAS type 1a. This synthase contains the following enzyme activities: malonyl palmityl transferase (MPT), acetyl transferase (AT), keto synthase (KS), keto reductase (KR), dehydratase (DH), enoyl reductase (ER), acyl carrier protein (ACP) catalyzing decarboxylating condensation of malonyl-ACP with acetyl-CoA, propionoyl-CoA, or acyl-CoAs to generate straight-chain fatty acyl-CoAs that range in size (C16–C26) and include odd-carbon fatty acids. Phosphopantotheinyl transferase (PPT) converts the ACP domain of FAS into the active form via attachment of phosphopantotheinate to a serine residue and is encoded by the down stream gene in the Rhodococcus bicistronic FAS operon. During inititiation malonyl-ACP is condensed with either acetyl-CoA or propionoyl-CoA to form a C4 or C5 straight-chain intermediate, respectively. Further elongation cycles consume malonyl-ACP for C2 additions with concomitant release of CO2 for each round of Claisen-type condensation reaction. The fatty acyl-CoA products of FAS <C20 are attached to glycerol catalyzing production of phospholipids and triacylglycerols (TAGs) or processed by number of other routes. Fatty acyl-CoAs can be further elongated by the FAS II system or MAS-family proteins. Wax esters (WEs) are generated by transesterification of fatty acyl-CoAs with fatty alcohols. TAGs, WEs, phospholipds, FAS II and MAS products come together to form compartments within Rhodococcus. Lipid bodies (yellow, LB) house the stored lipids surrounded by cytoplasm (blue, CP). The Plasma Membrane (orange, PM) is surrounded by cell wall (green, CW). The mycolic acid layer (black, MA) on the outside of Rhodococcus cells contains the long chain lipids. 1c) Comparison of biochemical activities of type II proteins and type I protein domains. Colors of FAS II genes correspond to related function in type 1 protein domains. Notable differences between synthase type 1 domains and type 2 proteins include: the MPT domain is dual functional in FAS type 1a thus releasing –CoA products through palmitoyl-CoA activity. The AT domain in type 1a provides acetyl-CoA and propionoyl-CoA substrates for condensation. In type 1b synthase the AT domain provides long chain acyl-CoAs for condensation on the KS domains. 1d) Methyl-branched lipid biosynthesis model for type 1b synthase activity from related mycobacterial protein PKS12. The C3 organic salt propionate can be ligated to CoA generating propionoyl-CoA, a substrate for Rhodococus lipid biosynthesis that is also produced in several metabolic degradation pathways. Propionoyl-CoA can be used directly by FAS during initiation or carboxylated to form methylmalonyl-CoA making it a substrate for a second multifunctional fatty acid synthase with a type 1b protein domain architecture present in Rhodococcus OPAG_06239. The type 1b synthase belongs to the mycocerosic acid synthase (MAS)-family of proteins that in Mycobacterium were shown to incorporate methylmalonyl-CoA into growing fatty acid chains creating methyl-branched lipids. Fig. 2. Phylogenetic analysis of type 1a and type 1b fatty acids synthases. 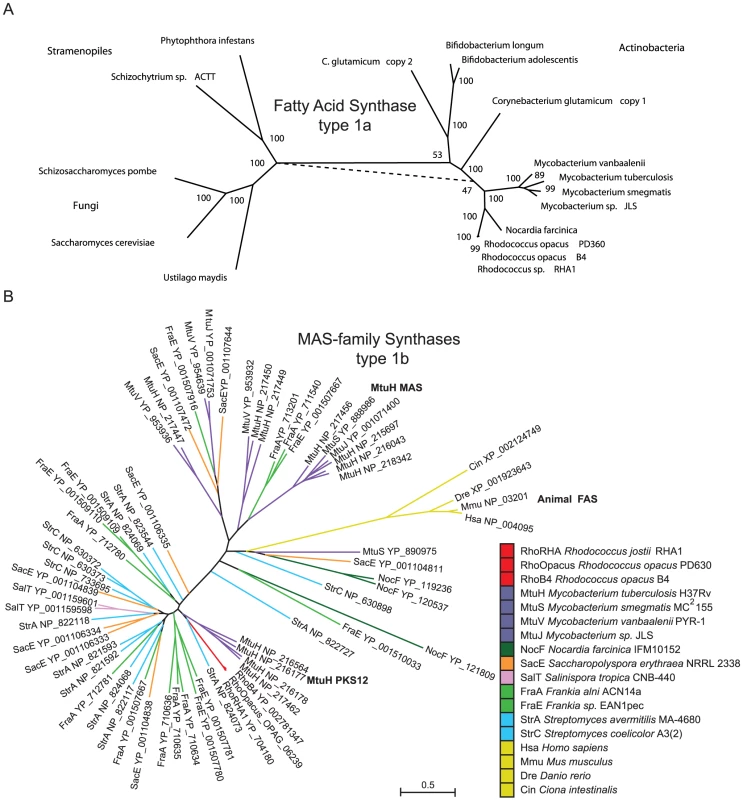
a) A phylogenetic tree that shows FAS type 1a is found in Actinobacteria, Stramenopiles, and Fungi. b) A phylogenetic tree of MAS-family type 1b proteins. This gene family is expanded in the genera Mycobacterium, Frankia, and Streptomyces but present in single copy in Rhodococcus belonging to branch containing the PKS12 from M. tuberculosis NP_216564 that also contains similar protein domain architecture. Our genome-based metabolic reconstruction revealed close metabolic pathway relationships when we compared the lipid metabolism enzymes of the phylogenetically related genera Mycobacterium, Nocardia, and Rhodococcus. These Actinomycetales contain multiple pathways for lipid biosynthesis utilizing a unique combination of multifunctional fatty acid synthases (FAS), a type 1a synthase and a related MAS-family type 1b synthase that generate linear - and branched-fatty acids respectively (Figure 1D). Diacyl glycerol acyl transferase (DGAT) enzymes convert fatty and other carboxylic acids into TAGs [29]. DGAT genes are also of limited genus representation in eubacteria and interestingly are present in six out of seven genera that also contain at least one gene of the MAS-family type 1b synthases (Figure 1a). Closely related genera to Rhodococcus share expanded gene-families of DGATs [29], [30]. A phylogenetic tree based on 701 shared genes present in a single copy and general genomic features of representative species used for comparisons with Rhodococcus are presented in Figure S3.
The phylogenetic distance between the bacterial taxa that contain the FAS type 1a suggest this gene has likely been horizontally transferred within Actinomycetales. The FAS gene was duplicated in Corynebacterium and horizontally transferred between Bifidobacterium and Corynebacterium. The FAS type 1a protein from Rhodococcus is highly related, ranging between 60–65% amino acid identity, to the enzyme in mycobacteria. The FAS protein is comprised of 3128 amino acids with 7 domains (Figure 1b), six of which catalyze distinct biochemical reactions (Figure 1c). Multiple sequence alignment of Actinomycetales FAS type 1a proteins reveals conservation across all domains (Dataset S1 and Dataset S2). The order of type 1a synthase protein domains, high sequence similarity, and presence of all three key lipid biosynthesis systems (FAS type 1a, MAS-family type 1b, and FAS II) suggest that the substrates and products are similar for the shared enzymatic network of lipid synthases within the suborder Nocardiacae and Mycobacterium. A working model for carbon flow in Rhodococcus lipid biosynthesis is presented in Figure 1b.
Enzymes from Expanded Gene Families Generate Lipid Biosynthesis Primers
Acetyl-CoA is the product of many catabolic reactions and a key substrate in lipid biosynthesis (http://tinyurl.com/4fgl6zo). Acetate and longer chain carboxylic acids captured from the environment can be converted to -CoA derivatives by CoA synthetases (EC 6.2.1.1) thus feeding these organic acids into lipid metabolism (Figure 1b). The first committed step in lipid biosynthesis is catalyzed by acetyl-CoA carboxylases that function as α/β complexes in Rhodococcus and related bacteria of the suborder Corynebacterineae to generate malonyl-CoA that is utilized by FAS type 1a, MAS-family type 1b, and FAS II enzymes for fatty acid biosynthesis (Figure 1b and 1d). Malonyl-CoA is generated by ATP-hydrolysis dependent carboxylation of acetyl-CoA in reaction EC 6.4.1.2 by an expanded family of AccA (α) and AccD (β) enzymes in Rhodococcus (Figure 3a). Homologous Rhodococcus AccA and AccD proteins were analyzed phylogenetically with those from related species (Figure S4). Some paralogous enzymes from these gene families were reported to recognize the –CoA derivatives of distinct carbon chain length organic acids [31], [32], [33], [34] suggesting that a diverse pool of organic acids could be carboxylated and incorporated into cellular lipids by Rhodococcus.
Fig. 3. Analysis of genes and gene families implicated in the TAGs cycle. 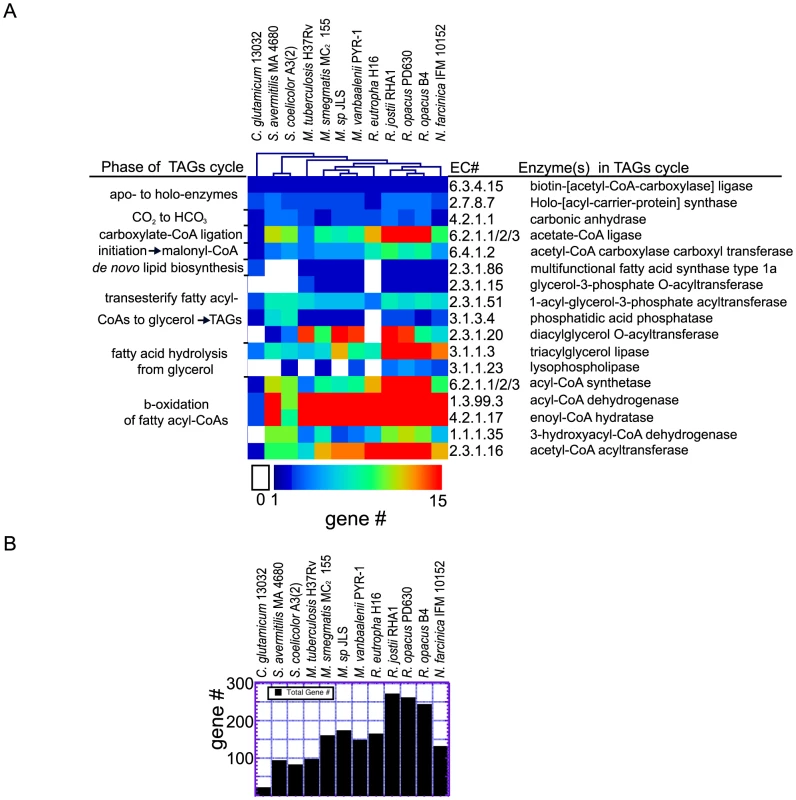
a) The number of genes in the TribeMCL gene-clusters implicated by metabolic reconstruction for each reaction of the TAGs cycle are displayed in a heat map with species clustered according to their TAGs cycle genetic profile. The number of genes corresponding to TribeMCL gene-clusters implicated for each biochemical reaction (EC number) is color coded from 0 to >15. b) The sum of unique genes for each bacterial species implicated for the TAGs cycle by metabolic reconstruction and TribeMCL analysis. The pool of propionoyl-CoA in Rhodococcus can be converted into methylmalonyl-CoA via ATP dependent carboxylation by propionoyl-CoA carboxylase in reaction EC 6.4.1.3 (Figure 1d). In Mycobacterium and Streptomyces methylmalonyl-CoA is a lipid biosynthetic substrate that is incorporated into methyl-branched lipids by the type 1b fatty acid synthases, a member of the mycocerosic acid synthase MAS-family of proteins [35]. OPAG_06239 is homologous (41% identity over 3527 amino acids) to a protein within the MAS-family with all of the same twelve functional domains and common domain architecture as PKS12 (Figure 1d), suggesting a related role in branched-lipid biosynthesis that was shown in M. tuberculosis to be production of C30–34 length methyl-branched phospholipids containing a 4,8,12,16, 20-pentamethylpentacosyl lipid subunit [36]. Only one MAS-family type 1b gene is encoded in R. opacus PD630 and related R. opacus B4, and R. jostii RHA1 genomes; whereas in the genera Mycobacterium, Frankia, and Streptomyces expansion of this gene-family has occurred (Figure 2b). In Mycobacterium the MAS-family synthases generate methyl-branched lipids adding to the diversity observed in their cell wall lipids that have been shown to be key in persistence, pathogenicity, and immune recognition [37], [38], [39]. By contrast, Streptomyces also contain several MAS-family proteins but do not encode for the FAS type 1a protein (Figure 1a). Streptomyces are rich in methyl-branched lipids [40], [41] that are shorter than the methyl-branched lipids observed in Mycobacterium indicating a difference in the biosynthetic workload between MAS-family and FAS II genes that could result from the absence of the type 1a synthase gene in Streptomyces.
Large Expansions of Gene Families Implicated in the Rhodococcus TAGs Cycle of Biosynthesis and Catabolism
The TAGs cycle includes twenty distinct enzymatic reactions starting from acetyl-CoA. The biochemical details of this cycle are presented with the corresponding EC number (Figure S5)(http://tinyurl.com/TAGs-cycle). The large expansions in homologous genes implicated in the TAGs cycle we identified in our initial metabolic reconstruction led us to further analyze the gene-families. We grouped the implicated TAGs cycle genes and families of genes based on protein similarity using the TribeMCL algorithm [42] on a small set of related bacterial species. We found that the genus Rhodococcus was deeply enriched in TAGs cycle genes including gene-families of very different sizes and there were no metabolic deficiencies in the multi-step metabolic-cycle (Figure 3a). 261 candidate R. opacus PD630 TAGs cycle genes were identified for the TAGs cycle reactions (Figure 3b). The largest gene family in the Rhodococcus TAGs cycle corresponds to the FAD dependant acyl-CoA dehydrogenases that operate in the β-oxidation of fatty acyl-CoAs (EC 1.3.99.3). R. opacus PD630 contains 71 of these acyl-CoA dehydrogenase genes whereas Corynebacterium glutamicum 13032 only contains two genes predicted for this reaction (Figure 3a). Large gene families identified in the Rhodococcus TAGs cycle also include: DGATs (EC 2.3.1.20) resolved in a phylogenetic tree (Figure S6), TAG lipases (EC 3.1.1.3), acyl-CoA synthetases (EC 6.2.1.3), enoyl-CoA hydratases (EC 4.2.1.17), and acetyl-CoA C-acyltransferases (EC 2.3.1.16).
Rhodococcus species contained at least 261 genes that could contribute to this metabolic cycle without consideration of membrane transport genes as they have yet to be defined for each catabolic substrate. The TAGs cycle -CoA ligases/synthetases that ligate –CoA with carboxylic acids play a role in initiation as well a β-oxidation of lipids have been grouped into 1 category represented as (EC 6.2.1.1/2/3) because we could not resolve the homologous enzymes that vary in their substrate chain length specificity (acetyl - EC 6.2.1.1, propionoyl - EC 6.2.1.2, acyl - EC 6.2.1.3). The largest number of genes in R. opacus PD630 dedicated to the TAGs cycle were mostly attributed to the acetate/acyl-CoA synthetases (EC 6.2.1.1/2/3)(18 genes), acyl-CoA dehydrogenases (EC 1.3.99.3)(71 genes), and enoyl-CoA hydratases (EC 4.2.1.17) (64 genes). These large gene families are central to the β-oxidation pathway suggesting that Rhodococcus can catabolize and extract energy through aerobic respiration from a diverse range of carboxylic acids as well as biosynthesize from these compounds a diverse array of lipid products.
The related Actinomycetale C. glutamicum 13032 provided stark contrast to the large number of lipid metabolism genes we observed in Rhodococcus. C. glutamicum 13032 had four pathway holes (Figure 3a) with a total of 19 genes implicated in the TAGs cycle (Figure 3b). A substrate-permissive glycerol-3 phosphate acyl transferase could bypass the EC 2.3.1.15 pathway hole allowing for production of essential phospholipids; however the 3-hydroxyacyl-CoA dehydrogenase in the β-oxidation of fatty acids (EC 1.1.1.35) is missing in C. glutamicum 13032, a result that is consistent with the observed deficiency in catabolism of fatty acids described below. C. glutamicum 13032 is also missing the DGAT enzyme responsible for the final step in TAGs and WE biosynthesis. A complete table of genes for the twelve species presented in Figure 3 can be found in Table S4.
Odd-Carbon Straight-Chain Lipid Accumulation in Rhodococcus
During fermentation of glucose, R. opacus PD630 and R. jostii RHA1 produced abundant lipids that were likely odd in carbon number based on their elution profile on gas chromatograph-flame ionizing detector analysis (GC-FID) (Figure 4a and 4b). The odd-carbon lipid species were found in Rhodococcus TAGs that were purified by thin layer chromatography (TLC) prior to conversion into fatty acid methyl esters (FAMEs) for assay by GC-FID (Figure 4c and 4d). During growth on glucose these odd-carbon fatty acids increase in relative abundance accumulating to as much as 30% in R. opacus PD630 and 40% in R. jostii RHA1 of the total lipids detected in the GC-FID assay (Figure 4e). Fermentation analytics of media concentrations of ammonium and glucose as well as cellular total fatty acids, and residual dry weight (Figure S7) indicated glucose depletion at 96 hours further stimulated R. jostii RHA1 to produce higher levels of odd-carbon lipids than R. opacus PD630.
Fig. 4. Identification and purification of odd-carbon straight-chain fatty acids generated by Rhodococcus. 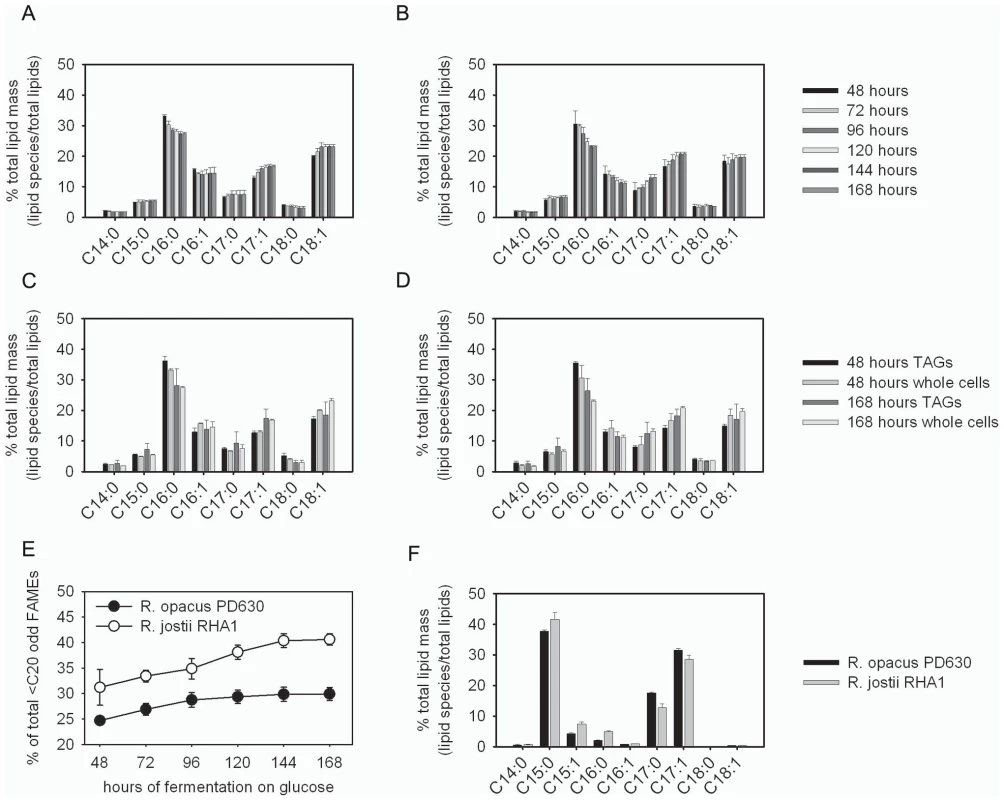
a and b) GC-FID analysis of FAMEs synthesized during fermentation. Freeze-dried whole cells fermented on glucose of R. opacus PD630 (a) and R. jostii RHA1 (b). c and d) GC-FID analysis of FAMEs derived from TLC-purified TAGs of R. opacus PD630 (c) and R. jostii RHA1 (d) grown on glucose e) odd lipids C15∶0, C17∶0, and C17∶1 increase during fermentation of both R. opacus PD630 and R. jostii RHA1. f) Rhodococcus grown on propionate generates mostly C15∶0, C17∶0, and C17∶1 fatty acids that were used for purification and confirmation by mass spectrometry and structural 1H-NMR. These analyses demonstrated that the purified Rhodococcus odd-carbon fatty acids were straight-chain. Chemical Identity and Structure of Straight-Chain Odd-Carbon Lipids Stored in TAGs
The FAS type 1a enzyme purified from Mycobacterium phlei was shown to convert malonyl-CoA and the C3 substrate propionoyl-CoA into undefined fatty acids without propionoyl-CoA carboxylase in vitro [27], suggesting that straight-chain odd-carbon lipids could be made by the type 1a FAS. We analyzed the chemical identity and structure of the Rhodococcus stored odd-carbon fatty acids to evaluate whether these lipids are methyl-branched or straight-alkyl chains in order to provide insight into the enzyme(s) responsible for this uncommon lipid biosynthesis. The known substrate preferences of the FAS type 1a enzymes are straight-chain substrates (acetyl-CoA, propionyl-CoA, and malonyl-CoA [26], [27]) while the MAS-family type 1b synthases incorporate the C3 methyl-branched lipid substrate methylmalonyl-CoA [35], [37], [39], [43], [44].
To determine the identity and chemical structure of the putative Rhodococcus odd-carbon lipids we purified them as FAMEs by taking advantage of their enrichment when R. opacus PD630 and R. jostii RHA1 were grown on propionate as the sole carbon source (Figure 4f). Propionate can be converted intracellularly to propionoyl-CoA through the activity of propionoyl-CoA ligase (EC 6.2.1.2)(Figure 1b) allowing for degradation via the methylcitrate cycle [45] or incorporation into lipids via two routes. In the straight-chain lipid biosynthesis pathway diagramed in Figure 1b, propionoyl-CoA is a substrate in the initial condensation reaction with malonyl-ACP. By contrast, in the branched-lipid biosynthesis pathway diagrammed in Figure 1d, propionoyl-CoA is first converted into methylmalonyl-CoA prior to incorporation into lipids by the MAS-family type 1b synthases.
FAMEs from propionate grown cells were purified via reverse phase HPLC then analyzed on a coupled gas chromatograph/electron ionization-mass spectrophotometer GC-EI-MS. We identifed ions with masses that corresponded to the methyl esters of pentadecanoic acid m/z 256 Da C15∶0 (Figure S8), heptadecanoic acid m/z 284 Da C17∶0 (Figure S9), and heptadecenoic acid m/z 282 Da C17∶1 (Figure S10). Fragmentation of these full length FAMEs resulted in ions that also matched previously reported spectra for electron ionized ions with these odd-carbon chain length FAMEs [46].
To discriminate between structural isomers with the same molecular weight (straight-chain and methyl-branched lipids), we performed 1H-NMR on the HPLC-purified FAMEs from Rhodococcus (Figure S11). We saw evidence of the methyl esters as expected from the transesterification of cellular lipids with methanol at 3.68 ppm; however, the FAMEs purified from Rhodococcus showed no evidence of methyl-branching at 0.86 ppm along the aliphatic chain of the C15∶0, C17∶0, and C17∶1 fatty acids as compared to the branched control 16-methylheptadecanoate. The NMR spectra in addition to the mass spectra demonstrate that these odd-carbon lipids contained straight-chain alkanes and a cis-alkene, not the methyl-branched forms expected from a methylmalonyl-CoA intermediate generated by propionoyl carboxylase in reaction EC 6.4.1.3. The absence of methyl-branched lipids in the shorter odd-carbon storage lipids indicated that de novo biosynthesis of methyl-branched lipids in Rhodococcus if present is restricted to the longer chain length lipids as previously described for the cell wall associated lipids in Mycobacterium. The identification of the stored pentadecanoic acid, heptadecanoic acid, and heptadecenoic acid odd-carbon straight-chain fatty acids could result from the FAS type 1a enzyme that is known to incorporate the three carbon molecule propionoyl-CoA and malonyl-CoA [27]. The odd-carbon fatty acids isolated from Rhodococcus grown on glucose contained predominantly seventeen carbons, that is in the range where most FAS type 1a products are released from the type 1a FAS (C16–18) [26].
Catabolic Phenotyping of Four Industrially Utilized Bacterial Species
Rhodococcus fermentation of low-cost organic substrates into oil requires a more complete understanding of the catabolic capabilities of these species. We tested 190 organic compounds as the sole carbon source in time course growth assays with four soil-derived bacterial species including the Actinomycetales R. opacus PD630, R. jostii RHA1, Corynebacterium glutamicum 13032, and the Gram-negative β-proteobacterium Ralstonia eutropha H16. Four chemical categories of compounds including carboxylic acids, nitrogen containing, carbohydrates and alcohols, and oligosaccharides were tested in these bacterial time course growth assays. Compounds capable of supporting growth yielded growth values that were clustered hierarchically to show catabolic-relationships between growth substrates and the species being compared.
Carbohydrates and alcohols are an important class of compounds to evaluate for converting natural organic streams such as cellulose and cellulose derived sugars, sugarcane, and beet sugars into biofuels. Seventeen oligosaccharides were screened for growth resulting in the identification of 13, 8, 3, and 0 growth substrates for R. opacus PD630, R. jostii RHA1, C. glutamicum 13032, and R. eutropha H16 respectively (Figure 5a). R. eutropha H16 showed no ability to degrade the oligosaccharides tested and relatively few monosaccharides were catabolized indicating that this species has many pathways to catabolize organic streams such as the rich pool of carboxylic acids defined below or fix CO2 by using the energy derived from splitting H2 [47], [48], [49]. Corynebacterium and Rhodococcus catabolized the disaccharides sucrose and maltose as well as the trisaccharide maltotriose. Rhodococcus species grew poorly on disaccaharide maltose but well on the trisaccharide maltotriose suggesting a possible membrane-transport preference. R. opacus PD630 gene OPAG_05551 is a glycogen hydrolase that contains the predicted activity to account for maltotriose growth that are part of an operon containing OPAG_05551 that breaks down glycogen (glucose α1–4) with Pi to yield glucose-1-P. Glucose 1-P isomerase is also utilized in galactose catabolism to generate the glucose 6-P that is common to glycolysis, pentose phosphate, and Entner Duoderoff catabolic pathways that are all complete pathways in our sequence based metabolic reconstructions of Rhodococcus (http://tinyurl.com/GLC-6-P).
Fig. 5. Screens of four bacterial species for growth on carbohydrates and alcohols. 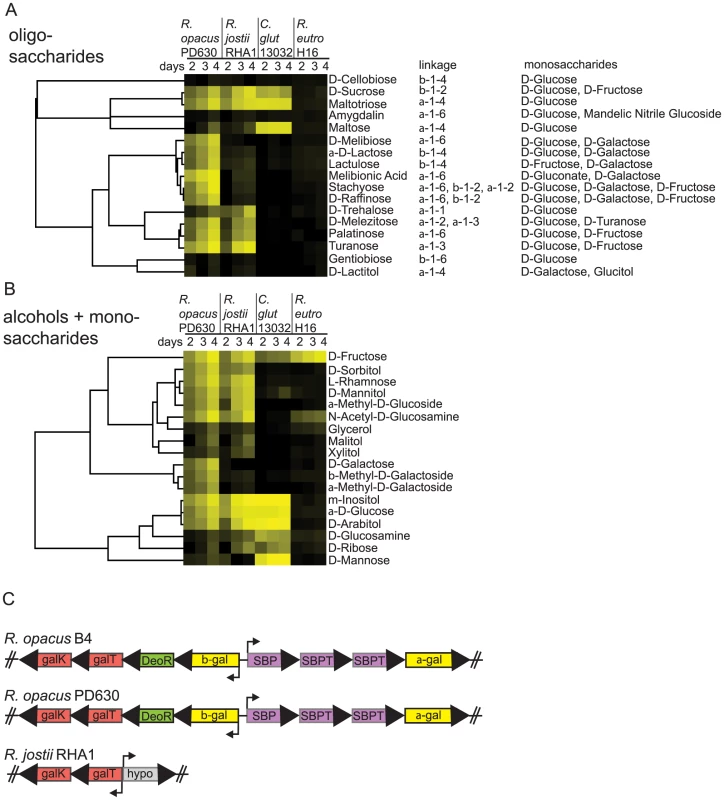
a) Compounds were clustered according to how R. opacus PD630, R. jostii RHA1, C. glutamicum 13032, and R. eutropha H16 were able to grow on oligosaccharides from 2–4 days. Yellow indicates evidence of growth. b) alcohol and monosaccharide compounds were clustered according to bacterial growth as in a. c) Comparison of three Rhodococcus chromosomes revealed that R. opacus B4 and R. opacus PD630 shared two divergent operons dedicated to galactose and oligogalactoside metabolism but R. jostii RHA1 only had a small piece of this chromosomal region containing the GalK and GalT genes omitting α- and β-galactosidases, Solute Binding Protein (SBP), two Solute Binding Protein Transporter (SBPT) proteins, and a DeoR family transcriptional regulatory protein. Galactose Metabolism Distinguishes Rhodococcus opacus PD630
The compound-specific growth assays indicated that galactose and oligogalactoside metabolisms differ between R. jostii RHA1 and R. opacus PD630 (Figure 5b). The ability of R. opacus PD630 to metabolize galactose enables efficient growth on the oligogalactosides lactose, lactitol, melibionic acid, melibiose, lactulose, raffinose, and stachyose (Figure 5a). A search of our metabolic reconstruction for the genetic basis of the galactose phenotypic differences between R. opacus PD630 and R. jostii RHA1 led to identification of a galactose-catabolic region of Rhodococcus genomes that is shared between the two R. opacus species B4 and PD630, but not in the closely related R. jostii RHA1 species. Two divergent polycistrons in the galactose catabolic region are fully syntenic between the R. opacus PD630 and R. opacus B4 species that contain the hydrolytic α - and β-galactosidases as well as solute binding protein (SBP), solute binding protein transporters (SBPT), and transcription regulatory protein of the DeoR family (Figure 5c) that could collectively hydrolyze and transport mono - and oligo-galactosides supporting growth on these compounds.
Rhodococcus Degrades a Diverse Pool of Carboxylic Acids
Carboxylic acids appear to be an important carbon source for Rhodococcus and R. eutropha H16. To determine the utilization of carboxylic acids as carbons source, we tested 64 carboxylic acids for growth using the previously described assay (Figure 6). These analyses led to the identification of 39 growth substrates for R. opacus PD630 and R. jostii RHA1 (61% of carboxylates tested), 15 growth substrates for C. glutamicum 13032 (21% of carboxylates tested), and 34 growth substrates for R. eutropha H16 (53% of carboxylates tested)(Figure 6). Acetic and propionic acids support growth in all species tested as did other common intermediates in central metabolism such as pyruvic, succinic, and citric acids. All of the species tested grew on gluconic acid that is degraded by the pentose phosphate pathway.
Fig. 6. Screens of four bacterial species for growth on carboxylate compounds (organic acids). 
Compounds were clustered as in Figure 5a. Similar to the observed galacto-saccharide phenotypes, R. opacus PD630 uniquely degrades D-galactonic acid-g-lactone (Figure 6). By contrast, growth on the short chain hydroxy acid glycolic acid is specific to R. jostii RHA1 and R. eutropha H16 (Figure 6). Growth was observed for the longer chain α and β hydroxybutyric acids by both Rhodococcus species and R. eutropha H16, whereas weak growth on γ hydroxybutyric acid was again only seen for R. jostii RHA1 (Figure 6) suggesting that this species has a more diverse hydroxy-acid metabolism.
R. opacus PD630, R. jostii RHA1, and R. eutropha H16 grew well on the C10 dicarboxylate sebacic acid suggesting that β-oxidation of these longer chain dicarboxylates provides rapid growth. β-oxidation could also explain the growth on Tween 40 and Tween 80 compounds by Rhodococcus and Ralstonia (Figure 6). The Tween compounds are converted to fatty acids upon ester-hydrolysis by cutinase proteins encoded for in the genomes of Actinomycetales [50]. Tween 20 supported growth of only R. eutropha H16 likely due to toxicity for the other species resulting from the hydrolytic release of a C12 fatty acid. C. glutamicum 13032 is limited in carboxylates catabolism as seen by the inability to catabolize sebacic acid and the longer chain Tween 40 and Tween 80 (Figure 6). A limited carboxylate catabolic-profile observed for C. glutamicum 13032 in our growth assay is consistent with genes missing for 3-hydroxyacyl-CoA dehydrogenase within the fatty acids β-oxidation pathway (Figure 3a).
C. glutamicum Degrades Sialic Acid
The identification of the sialic acid or N-acetylneuraminic acid, an abundant component of extracellular glycoproteins, as a growth substrate for the soil bacterium C. glutamicum 13032 (Figure 6) was unexpected because this pathway has only been described for bacteria that colonise animals [51], [52]. Examination of the C. glutamicum 13032 genome reveals a likely sialic acid catabolic operon containing genes for a secreted sialidase (cg2935), a sialic acid ABC transporter (cg2937–2940) of the satABCD type seen in Haemophilus ducreyi [53] and a full set of catabolic genes (cg2928–9 and cg2931–3) genes that would allow C. glutamicum to degrade sialic acid to fructose-6-phosphate, pyruvate, and ammonia [51]. A related but incomplete catabolic operon is seen in C. diptheriae NCTC 13129. The sialic acid catabolic genes are not conserved in the other species tested nor were related transporters shown to facilitate uptake of sialic acid [54], consistent with these phenotypic results.
Diverse Nitrogen Metabolism in Rhodococcus
Rhodococcus displayed the most diverse catabolism of nitrogenous compounds in our species-comparative time course growth assays. Clustered heats maps relating the time course growth of 38 nitrogenous compounds tested. 26 nitrogenous compounds supported growth in at least one of the species tested (Figure S12). A notable feature of the Rhodococcus species nitrogenous metabolism is the ability to catabolize the branched amino acids that result in the formation of propionoyl-CoA thereby establishing a cycle of propionoyl-CoA in carbon storage and amino acid catabolism (http://tinyurl.com/propionyl-CoA). A ranked order list of growth values on 190 compounds for each species in the screen is presented in Table S2.
Expanded Families of Membrane Transport Proteins in R. opacus PD630
Comparison of the protein domains (Pfam database, Sanger Institute UK) within a set of related Actinomycetales and the Gram-negative outlier R. eutropha H16 revealed that the R. opacus PD630 genome contained 3 membrane transport protein families in the top 9 expanded families of total encoded protein domains (Figure S13). The membrane transport major facilitator super family (MFS) is the most prevalent Pfam domain in R. opacus PD630. There are 229 MFS genes in R. opacus PD630 as compared to 176 in R. jostii RHA1, 104 in R. eutropha H16, and 47 in C. glutamicum 13032 (Figure S14). The high relative number of membrane transporters likely enables the broad catabolism we observed in R. opacus PD630 and R. jostii RHA1.
Cholesterol Degradation by a Large Genomic Region
The ability to degrade sterols is shared between Rhodococcus and Mycobacterium. Following intravenous injection, M. tuberculosis has been observed to colonize lung tissue that is rich in lipid bodies and cholesterol crystals [55]. Genetic analysis of M. tuberculosis cholesterol catabolic pathways showed this sterol to be an essential carbon source during M. tuberculosis infections in mouse lung models [56], [57], [58]. Complete sterols degradation requires catabolism of both of the aliphatic branched side chain as well as the terpene polycyclic rings; however catabolism studies of M. tuberculosis showed that 14C labeling at the fourth position of the steroid A ring was released as CO2; whereas the label at position 26 within the sterol branched side chain was converted into phthiocerol dimycocerosate (PDIM) [57]. This study indicated that part of the sterol was being degraded for energy and the other was being used for assembly of cell wall associated branched-lipids. Sterol A and B ring degradation results in propionoyl-CoA and pyruvate while side chain degradation results in a 2∶1 formation of propionoyl-CoA: acetyl-CoA. In M. tuberculosis the the propionoyl-CoA from cholesterol degradation is converted into methylmalonyl-CoA then incorporated into branched-fatty acids such as PDIM by the PKS12 MAS-family type 1b synthase [35], [37], [43], [44], [59].
A large region (∼0.28 Mb) of Rhodococcus chromosomal DNA has been identified through transcriptomic and genetic analysis of R. jostii RHA1 grown on cholesterol as a sole source of carbon [60]. Within this large chromosomal region, there are six clusters of genes that encode for the multiple enzymes dedicated to sterol degradation including: membrane transport, side chain degradation, sterol A and B ring degradation, and sterol C and D ring degradation, for recent review [61]. We performed whole genome alignments between R. opacus PD630, R. jostii RHA1, and R. opacus B4, M. sp JLS, M. vanbaalenii PYR-1, M. smegmatis MC2 155, M. tuberculosis H37Rv, C. glutamicum 13032, and S. avermitilis MA 4680 to evaluate gene conservation within the six gene clusters implicated in cholesterol degradation (Figure S14). We found extensive conservation in the cholesterol degradation genes between Mycobacterium and Rhodococcus as has been reported previously [60]. Our analysis indicates Streptomyces contains many of the key genes for cholesterol degradation but is lacking homologues to the Mce sterol-transport genes [57]. C. glutamicum 13022 did not contain the genes implicated for cholesterol degradation. The only major difference found between the previously described sterol degradation chromosomal region in R. jostii RHA1 and R. opacus PD630 is the presence of a transposase gene in gene cluster 2 (OPAG_09155).
Discussion
The Rhodococcus genomes encode multiple biosynthetic pathways for making lipids and expanded gene families within those pathways that contribute to the diversity and abundance of lipid products seen with some Actinomycetales. We demonstrated that Rhodococcus TAGs contain the uncommon straight-chain odd-carbon lipids pentadecanoate, heptadecanoate, and heptadecenoate by mass and structural determination. The high abundance of the straight-chain odd-carbon lipids are yield-controllable through feeding of the three carbon organic salt propionate. Propionate is converted to propionoyl-CoA, a metabolite that is generated during the catabolism of the branched amino acids isoleucine, valine, and threonine as well as sterols. Propionoyl-CoA levels likely increase intracellularly during nitrogen and glucose starvation resulting from elevated protein and amino acid degradation as the physiology of these cells adapt to these nutrient limitations. Elevated propionoyl-CoA has been proposed to explain increased production of the branched-lipid PDIM when mycobacteria is grown on cholesterol as a sole source of carbon [59]. The FAS type 1a protein in the genus Rhodococcus is highly related to the mycobacterial protein that has been demonstrated to incorporate propionoyl-CoA during de novo lipid biosynthesis and produce lipids with the same lengths observed in the stored lipids TAGs and WEs. Our genomic analysis indicates conservation of all three interconnected lipid biosynthesis systems in Nocardiacae and Mycobacterium namely FAS type 1a, MAS-family type 1b, and FAS II. This group of bacteria also shares the ability to store lipids in high abundance. This conserved lipid biosynthetic network provides means for extrapolating functional information derived from decades of lipid metabolism research in Mycobacterium to the elaborate lipid metabolism in Rhodococcus.
Pharmacological studies in combination with enzyme activities performed with purified systems demonstrated that Mycobacterium uses FAS type 1a to convert two and three carbon organic acids into straight-chain fatty acids containing 16, 18, 24, and 26 carbons [20], [26], [27]. FAS type 1a products are released as the –CoA derivative of a fatty acid due to the dual activity of the FAS type 1a malonyl palmitoyl transferase (MPT) domain [21]. The FAS acyl-CoA products can then proceed through multiple lipid biosynthetic pathways. Studies of mycobacterial lipid metabolism have established a genetic model for this related oleaginous bacterium that connects multiple biosynthetic systems for the building of storage, plasma membrane, as well as very long chain cell wall-associated lipids. The close phylogenetic relationship between the genus Mycobacterium and Rhodococcus resulted from common ancestry wherein these genera share many features of their elaborate lipid metabolisms.
Much of what is known about Actinomycetales odd-carbon lipid metabolism comes from work that described longer chain extracellular lipids that are methyl-branched. Prior to this study, it was unclear whether the <C20 odd-carbon lipids in Rhodococcus are methyl-branched as a result of incorporating methylmalonyl-CoA [16]. Using 1H-NMR to analyze purified lipids we could distinguish between the structural isomers of branched and straight-chain fatty acids demonstrating that the stored odd-carbon lipids in Rhodococcus are straight-chain. The odd-carbon storage lipids in Rhodococcus could result from propionoyl-CoA and malonyl-ACP condensing in the initiation phase of FAS type 1a biosynthesis (Figure 1b). The substrate requirements for production of straight-chain odd-carbon lipids matches the reported substrate specificity of the multifunctional FAS type 1a enzyme and its product chain lengths. The combination of metabolic product identification and metabolic pathway mapping through genomic sequence supports a serial order to the elaborate network of lipid biosynthesis in Rhodococcus, similar to Mycobacterium, wherein FAS type 1a initiates and elongates fatty acids releasing –CoA products that are in the range of C16–C18 and C24–C26. FAS produced fatty acyl-CoAs can be further elongated in the type II system or by the MAS-family type 1b synthase.
The Rhodococcus type 1b synthase OPAG_06239, like the homologous PKS12 gene from Mycobacterium, contains two modules consisting of 6 protein domains in a direct repeat (Figure 1d). The PKS12 enzyme displays dual specificity for malonyl-CoA and methylmalonyl-CoA within the same polypeptide and oligomerizes in a tail-to-head fashion to perform multiple elongation cycles on C16 - and C18-CoA molecules resulting in C30–34 multiply methyl-branched fatty acids. Enzyme assays with single modules (6 domains) of PKS12 and site-specific mutations within the PKS12 acetyl transferase (AT) domains demonstrated that the N-terminal module incorporates methylmalonyl-CoA whereas the C-terminal module incorporates malonyl-CoA [43]. This mode of biosynthesis results in branched fatty acids that contain a methyl-branch at every fourth carbon from the point of initial condensation by the type 1b enzyme. This alternating of elgonation substrates mechanism explains the curious methylation at every fourth carbon in the mannosyl-β-1-phosphomycoketide (MPM) molecules isolated from Mycobacterium [36]. In Mycobacterium, the enzymes of the type 1b MAS-family and FAS II system share a common preference for longer chain length fatty acyl-CoAs resulting in the observed order in biosynthesis enzyme activity. The order of conserved domains and sequence homology of the Rhodococcus type 1b synthase OPAG_06239 suggests that this enzyme will function similarly in some aspects of branched lipid biosynthesis that is characteristic of the MAS-family enzymes in the related Mycobacterium species; however no methyl-branched lipids have been identified in Rhodococcus to date.
In species such as Mycobacterium that have both FAS type 1a and 1b synthases, there is a distribution in labor amongst the synthases that is dictated by chain length; wherein FAS type 1a functions as the initiating synthase and the type 1b synthase incorporates methyl branch substrate methylmalonyl-CoA during lipid biosynthesis of longer chain length lipids. In more distantly related Actinomycetales, the genera Saccharopolyspora, Salinspora, Frankia, and Streptomyces we observed a type 1b but no type 1a synthase (Figure 1a); thus preventing the distribution of labor observed in Nocardiacae and Mycobacterium. Streptomyces have been reported to store abundant methyl-branched lipids [41] that are shorter in chain length than those isolated from Mycobacterium. The observation of odd-carbon lipids in the TAGs from Rhodococcus intrigued us as a possibility that type 1b synthase activity was contributing to the accumulated TAGs. The chemical analysis we performed indicated the stored TAGs were straight-chain lipids of the appropriate length to have been built by type 1a FAS through iterative biosynthesis with two of this enzymes known substrates (propionyl-CoA and malonyl-CoA). How is the strict distribution of labor observed between type 1a and the other type II and 1b synthases maintained in Nocadiacae and Mycobacterium? The crystal structure of the related FAS from S. cerevisae [62] revealed this synthase is a hexamer complex of apoenzymes. A hexamer complex also explains the behavior in ultracentrifugation studies of the FAS type 1a complex from mycobacteria [26]. The structural studies of fungal FAS provided a structure-based model wherein fatty acids are biosynthesized within the cavity of a 2.6 MDa β barrel structure. The enzyme FAS-ACP domain with growing acyl chain accesses individual catalytic domains shuttling biochemical intermediates from one active site to the next within the FAS type 1a hexamer complex [62]. Fatty acid products of the appropriate length are released as acyl-CoAs by the MPT domain of FAS type 1a. The FAS hexamer in Mycobacterium display a bimodal distribution of product chain lengths that are (C16–18 and C24–26) [26]. We propose the FAS 1a acyl-CoA products become accessible to the other synthases once released from within the β barrel structure of the FAS hexamer by the palmitoyl transferase activity of the MPT domain.
Acetyl/acyl transferase (AT) domains in type 1 fatty acid synthases display remarkable substrate diversity. In the type 1a synthase, the AT domain loads acetyl–CoA on the FAS-ACP domain then is subsequently transferred to a cysteine in the keto synthase (KS) domain. The second substrate for condensation is the elongation substrate malonyl-CoA that is transfered by the FAS-MPT domain to the FAS-ACP domain then delivered to the KS domain for decarboxylating condensation with acetyl-CoA resulting in acetoacetyl-ACP. The type 1b synthases that inititiate fatty acid biosynthesis, like the mammalian FAS, lack the MPT domain thus use their AT domain to load both acetyl-CoA and malonyl-CoA. MAS-family type 1b enzymes display significant substrate diversity by varying both the acyl substrates (acetyl-CoA, acyl-CoAs) as well as elongation substrates (malonyl-CoA, methylmalonyl-CoA). We found seven Actinomycetales genera that contained the MAS-family type 1b synthases compared to 4 genera that encode the type 1a synthase. Six out of seven Actinomycetales genera that encode type 1b synthases also encode at least one storage enzyme of the DGAT/WE family. We conclude the relatively widespread taxonomic representation of the type 1b synthases and the diversity of substrates these enzymes react on suggests that there are lipid variants yet to be identified.
The large number of genes dedicated to lipid metabolism in Actinomycetales is the result of gene duplications, multifunctional FAS type 1a and MAS-family type 1b gene emergence, horizontal gene transfer, and emergence of the DGAT/WE enzymes that catalyze the transesterification of fatty acyl-CoAs with diacylglycerol. We performed phylogenetic analysis on genes identified by metabolic reconstruction in an attempt to predict the most likely activities for each enzyme encoded in the R. opacus PD630 genome. We used the TribeMCL algorithm to describe the size of the gene family and all of the family members for a set of related Actinomycetales. Phylogenetic tree formation with these related species was used to resolve the orthologous from paralogous enzymes, thus facilitating the extrapolation of functional data for related proteins to our in silico model of Rhodococcus Opacuscyc14.5. The broad catabolism and elaborate lipid biosynthesis described in Rhodococcus indicates that there is substantial enzymatic activity on chemically-related compounds that could be explained by expansion in gene-family sizes and genetic drift to encode enzymes with slightly variant substrate recognitions from ancestrally-related enzymes but still catalyze similar chemical reactions. The 8632 genes in R. opacus PD630 provide a large arsenal of metabolic enzymes for an oleaginous lifestyle within soil. We found protein domains contained in transport proteins to be the most abundant class of domains encoded in Rhodococcus as well as a distinctive profile of transporter types that are distinguishing for Rhodococcus (Figure S13). Identification of structural genes and operons that play a role in the lipid body assembly process have already begun to benefit from genomic sequence [63].
Chemoheterotrophic organisms capable of fermenting sugars and a broad spectrum of organic compounds derived from cellulosic and other natural resource biomasses through bioconversion provides an industrial process to convert agricultural side-, natural resource-, and industrial waste-streams into fungible fuels. R. opacus PD630 differs from R. jostii RHA1 in its ability to catabolize the cellulosic sugar galactose and oligogalactosides. Both Rhodococcus species degraded the cellulosic sugars glucose and rhamnose; however, none of the species tested were able to degrade the disaccharide cellobiose containing β1–4 linked glucose. Cellulose catabolism requires hydrolysis of β1–4 linked glucose, indicating that these species do not encode for these hydrolases. The most abundant component of hemicellulose is xylan that is broken down to xylose. Consistent with the cellobiose deficiency these species were unable to catabolize xylose. Genetic complementation of cellulosic degradation pathways in Rhodococcus provides a streamlined approach for cellulosic biomass conversion into oil-based fuels. Rhodococcus species catabolized a diverse array of carboxylic acids that corresponded to an expansion in the acyl-CoA ligase gene family that link organic acids with biosynthesis and catabolism. Organic acids are produced in the mixed-acid fermentations of cellulose degrading organisms thus indicating a potential next-generation strategy for 2-phase fermentations of cellulosic biomass to TAGs that could be done without genetic modification.
Our working model of the R. opacus PD630 metabolism began with genome sequence that allowed phylogenetic comparisons to be made with related species that have been studied in far greater molecular detail. Phenotypic information about catabolism in Rhodococcus provided a powerful multigenic test that guides the metabolic reconstruction towards completion through phenotype-directed pathway curation. Literature-based pathway curation united the reported biochemical reactions of M. phylei FAS type 1a protein with metabolic products that we purified and characterized during metabolic model refinement of R. opacus PD630. The improvements to our genetic model of R. opacus PD630 metabolism provides a template for further refinement with the integration of data from genetics, biochemistry, metabolomics, lipidomics, and transcriptomics that will be the focus of future work.
Materials and Methods
Bacterial Species Used in This Study
R. opacus PD630 was obtained from the DSMZ strain 44193. R. eutropha H16 ATCC17699 and C. glutamicum ATCC13032 were from ATCC. R. jostii RHA1 was a gift from Lindsay Eltis at the University of British Columbia.
Rhodococcus opacus PD630 DNA Sequencing and Genome Annotation
Genomic DNA was extracted from R. opacus PD630 as in [64] without the addition of mutanolysin. Genomic DNA was sequenced at 454 Life Sciences (A Roche Company) according to manufacturer protocol for shotgun (Rapid Library) and 3 kb paired-end reads using GS FLX Titanium sequencing chemistry. The resulting DNA sequence was assembled using the GS De Novo Assembler software version 2.0. Open Reading Frames (ORFs) were predicted from assembled genome sequence using a combination of in silico ORF predictions and gene mapping from the annotated R. jostii RHA1 genome. In silico ORFs were predicted using GeneMark [65] and Glimmer3 [66]. For synteny based gene-mapping Nucmer [67] was used to find local alignments between every contiguous sequence read (contig) in the R. jostii RHA1 and every contig in the R. opacus PD630. A whole-genome synteny map was then built from these local alignments by chaining together collinear hits, allowing up to 10,000 bases of undetectable similarity between anchors, then filtering out chains that overlap a larger chain on either sequence by more than 90% of their length. The remaining chains, corresponding to syntenic regions of DNA, were globally aligned using LAGAN [68]. Each transcript in the R. jostii RHA1 genome was mapped onto the R. opacus PD630 by attempting two mapping techniques and then selecting the transcript with the higher-scoring global alignment to the reference transcript's coding sequence. The first method projects the boundaries of each gene onto the target genome using the coordinates in the raw whole-genome alignment. The second method uses the whole-genome alignment to define a target region containing the reference transcript, and then uses GeneWise to build a gene model by aligning the reference protein to the target region. Final ORFs were defined by comparison of in silico ORFs and mapped ORFs with hits to Pfam [69] and the top blast hits against the non-redundant protein database. ORFs with overlap to non-coding RNA features (see below) were reviewed and removed when appropriate. Discrepancies in the final ORFs were resolved via manual review. Ribosomal RNAs (rRNAs) were identified with RNAmmer [70]. The tRNA features were identified using tRNAScan [71]. Other non-coding features were identified with RFAM [72]. Every annotated gene in the Rhodococcus opacus PD630 genome is assigned a locus number of the form OPAG_##### both at the Broad Institute web site and in GenBank with accession ABRH01000000.
Stitching the Genome Assembly Based on Synteny
The R. opacus PD630 V3 genome assembly contained 293 (contigs), containing 16 scaffolds longer than 2,000 residues. The 16 large scaffolds of the R. opacus PD630 genome, 10 of which were largely syntenic with R. jostii RHA1 and R. opacus B4 linear chromosomes, were ordered and oriented to reflect the observed synteny followed by the smaller contigs. Nucmer and mummerplot, components of the mummer 3.0 software package [67], were used to align the genomes and create graphical representations of those alignments. The mummerplot graph files were annotated to show boundaries between each of the scaffolds in the mummerplot graphs. Through manual inspection of these graphs, the syntenic order and orientation of these contigs were assembled into a stitched FASTA file with the addition of 500 N nucleotides at scaffold termini.
Metabolic Reconstruction
Enzymes for nine comparative species in Opacuscyc14.5_compare were computationally predicted using the EFICAz2 algorithm, enzyme prediction using gene-name matching, pathway prediction [24], transcription unit prediction [73], transporter prediction [74], and pathway hole filling [75] was performed with the Pathway Tools 14.5 software [24]. These databases can be accessed at (http://rhodocyc.broadinstitute.org). In addition, Opacuscyc14.5_working also predicted enzymes using homology to proteins with an EC# assignment in the database at Kyoto Encyclopedia of Gene and Genomes KEGG (http://www.genome.jp/kegg/). Subsequent literature-based manual curation was used to refine Opacuscyc14.5 TAGs biosynthesis and degradation cycle.
Phylogenetic Analysis of FAS, MAS, PKS12, and DGAT Gene Families
A set of directed gene pairs was generated by performing an all-against-all BLASTP search (min % aligned = 10 and e-value<1e−5) between a comparative set of genomes (all accession numbers available in Table S3). Genes were clustered using OrthoMCL [76] with a Markov inflation index of 1.5 and a maximum e-value of 1e−5. Gene clusters were identified to which the M. tuberculosis FAS, MAS, PKS12, and DGAT genes belonged to. These clusters had all members plotted on the AMPHORA phylogenetic tree of the Actinobacteria [28]. In the phylogenetic analysis of FAS genes, we included three fungal FAS genes and two stramenopile FAS genes, while in the phylogenetic analysis of MAS-family genes we added four animal FAS genes. Amino acid sequences were aligned using MAFFT [77] using the E-INS-i method. A maximum likelihood phylogeny was estimated using the PROTGAMMABLOSUM62 model in RAxML [78] with 1000 bootstrap replicates. FAS type 1a protein alignments were visualized using Jalview [79] and domains were annotated based on predictions from the Conserved Domain Database [80].
Phylogenetic Resolution of Acc and DGAT Proteins
A custom blast database containing seven Rhodococcus & Mycobacterium genomes, including R. opacus B4, R. opacus PD630, R. jostii RHA1, M. tuberculosis H37Rv, M. vanbaalenii PYR-1, M. sp JLS & M. leprae Br4923 was generated. Protein sequences annotated as either AccA or AccD within Mycobacterium tuberculosis H37Rv were identified and accessions extracted into separate lists, whereas the WS/DGAT protein accessions were identified from the R. jostii RHA1 genome annotation. Each set of amino acid sequences were blasted against the database using BLASTP with an expect threshold of 1E−30. Unique matches were identified and whole sequences extracted for alignment with MUSCLE v3.7 [81], using a maximum of 24 iterations. Alignments were manually checked with ClustalX v2.0 [82], at which point WS/DGAT sequences without any residues aligning to the proposed active site motif (H[L/S/P]xxxDG) [4] were rejected. The multiple sequence alignments were converted to phylip format for passing to ProtTest v2.4 [83], which determines the best-fit substitution model and produces a phylogenetic tree with maximum likelihood estimation, using PhyML v3.0 [84]. Newick formatted trees were represented with iTOL [85].
Mass and Structural Analysis of Odd-Carbon Lipids
GC-MS analyses were conducted on a TraceGC Ultra DSQ mass spectrometer (Thermo Scientific) equipped with an AT-5 ms column from Alltech (60 m×0.25 mm i.d. ×0.25 mm df). The injector and transfer line were maintained at 280°C while the ion source was set at 180°C in electron ionization (EI) mode. High purity helium was used as carrier gas at a flow rate of 1 mL/minute. The sample was injected onto the column in split mode and heated at 40°C for a minute. The GC oven temperature was increased to 280°C at 20°C per minute.
The NMR spectra were recorded on a Varian Inova instrument, operating at 500 MHz for 1H and 125 MHz for 13C, equipped with a three channel, 5 mm, indirect detection probe, with z-axis gradients. The solvent was chloroform-d, and the temperature was 25°C. The chemical shifts for 1H were referenced to the residual solvent signal, 7.27 ppm on the tetramethylsilane scale. Proton spectra were acquired in 4 transients, with a 30° pulse, an acquisition time of 5 s and no relaxation delay. The intensity of the signals was referenced to the signal of the terminal methyl in the alkyl chain, at 0.88 ppm, 3H.
Purifications of Odd-Carbon Lipids
100 ml cultures of R. opacus PD630 and R. jostii RHA1 were grown in defined media [5] with substitution of 1% propionate/0.056% NH4SO4 for 1 week at 30°C with agitation followed by addition of 1 gram of propionate from sterile 20% stock solution. 50 mls of culture was collected after 2 weeks by pelleting and freeze dried prior to lipid extraction. 50 mg of dried cells were transesterified in a 2 ml volume of 50% CHCl3/42.5% methanol/7.5% H2SO4 for 2.5 hours at 100°C in sealed 16×125 ml glass tubes (Kimble Glass Co.). Rhodococcus fatty acid methyl esters (FAMEs) preps were then concentrated under a stream of N2, resuspended in dichloromethane (DCM), and analyzed (10-mL injections) using an Agilent 1050 High Performance Liquid Chromatography (HPLC; Agilent Technologies, Inc, Wilmington, DE USA) coupled to a Wakosil II RS-Prep C18 column (5 mm, 20 mm×250 mm; Wako Chemicals USA, Inc., Richmond, VA USA) and a Sedere Sedex 75 Evaporative Light Scattering Detector (ELSD; SEDERE, Alfortville Cedex, France) for determination of relevant fractionation range. The ELSD drift tube was set at 50°C, and the air nebulization pressure at 3.5 bar. The dual solvent system used is a linear gradient program based on the method published by Mansour [86], with an extended run time, and substitutes (DCM) for chloroform. Starting with 98% acetonitrile (MeCN) and 2% DCM, the program ramps to 60% MeCN linearly by 100 minutes, holds at that ratio until 105 minutes, then ramps to 100% DCM by 110 minutes holding until 115 min, and returns to the starting conditions by 120 minutes. Methyl 12-methyltetradecanoate (C15∶0) and Methyl 16-methylheptadecanoate (C18∶0) branched FAMEs standards (Sigma) were used to determine the HPLC retention times of analytes of interest. Fractions were collected for each FAME species: 17∶1 eluted at 12.8 minutes, C 15∶0 eluted at 13.5, C17∶0 eluted at 16.6 minutes for subsequent GC-EI-MS and 1H-NMR analysis.
TLC purification of TAGs was performed as previously described for single solvent system [5] followed by scraping of the TAG species that were detected by water staining. Scraped TAGs were extracted with 1∶1 (vol/vol) chloroform methanol for 1 Hr prior to filtration with 0.2 µM PTFE membrane (VWR International). Extracts were dried under nitrogen then subjected to transesterification and analyzed by (GC-FID) [5].
TAGs Cycle Comparative Analysis
R. opacus PD630 TAGs cycle genes were identified through metabolic reconstruction and literature-based curation. The genes assigned to biochemical reactions within the TAGs cycle were analyzed to identify all significant gene pairs (BLASTP, E< = 1e-5) followed by Markov clustering as implemented by TribeMCL [42]. Candidate gene families were then aligned using MAFFT [77] and manually curated in Jalview 2.6.1 [79] using neighbor joining trees and curation of alignments. Because we were classifying by EC number which is a broad classification of function and not necessarily homology, a strict cutoff for percent overlap or percent similarity was not used to retain as many members of particular EC category as annotated in KEGG (http://www.genome.jp/kegg/). A full list of curated EC # related Gene families implicated at each biochemical step of the TAGs cycle are represented with 11 other bacterial species in Table S4. Multiexperiment viewer [87] was used to hierarchically cluster the species in the TAGs cycle and visualize in a heat map. Clustering was pearson correlated with average linkage. The threshold for color-saturation was set to 15 genes per reaction category.
Phenotype Growth Assays on 190 Compounds
Chemical compounds capable of supporting growth as a sole source of carbon were identified using a tetrazolium-based growth assay developed by Biolog Incorporated; wherein growth of cells and aerobic respiration were measured by Dye D (Biolog, Inc) reduction resulting in purple color and turbidity read at 590 nm absorbance in Fluorostar plate reader. Innoculation cultures were grown for 1 day on LB agar plates at 30°C from which 3.8E−3 ODu/well were transferred from 0.5 ml water resuspensions after measurement in nanodrop (Thermo Scientific) at 600 nm. During growth, plates were wrapped in aluminum foil and measured at 44, 72, and 96 hours. The measured A590 values were normalized by first subtracting values from uninoculated plates. The average of normalized values were background subtracted with average values of inoculated wells that lacked a carbon source (negative control). The background subtracted growth values were separated into chemical categories by filtering compound classifications in Excel. The normalized growth values for each compound were clustered using Cluster 3.0 [88] with centered correlation. Heat maps were generated in Java TreeView 1.1.3 [88]. Growth substrates were identified at 96 hours incubation for compounds that had A590 values >0.2.
Supporting Information
Zdroje
1. McLeodMPWarrenRLHsiaoWWArakiNMyhreM 2006 The complete genome of Rhodococcus sp. RHA1 provides insights into a catabolic powerhouse. Proc Natl Acad Sci U S A 103 15582 15587
2. AlvarezHMMayerFFabritiusDSteinbuchelA 1996 Formation of intracytoplasmic lipid inclusions by Rhodococcus opacus strain PD630. Arch Microbiol 165 377 386
3. AlvarezHMSteinbuchelA 2002 Triacylglycerols in prokaryotic microorganisms. Appl Microbiol Biotechnol 60 367 376
4. HernandezMMohnWMartinezERostEAlvarezA 2008 Biosynthesis of storage compounds by Rhodococcus jostii RHA1 and global identification of genes involved in their metabolism. BMC genomics 9 600
5. KurosawaKBoccazziPde AlmeidaNMSinskeyAJ 2010 High-cell-density batch fermentation of Rhodococcus opacus PD630 using a high glucose concentration for triacylglycerol production. Journal of Biotechnology 147 212 218
6. HughesJArmitageYCSymesKC 1998 Application of whole cell rhodococcal biocatalysts in acrylic polymer manufacture. Antonie van Leeuwenhoek 74 107 118
7. SetoMKimbaraKShimuraMHattaTFukudaM 1995 A Novel Transformation of Polychlorinated Biphenyls by Rhodococcus sp. Strain RHA1. Appl Environ Microbiol 61 3353 3358
8. RobrockKRMohnWWEltisLDAlvarez-CohenL 2010 Biphenyl and ethylbenzene dioxygenases of Rhodococcus jostii RHA1 transform PBDEs. Biotechnol Bioeng
9. GoncalvesERHaraHMiyazawaDDaviesJEEltisLD 2006 Transcriptomic assessment of isozymes in the biphenyl pathway of Rhodococcus sp. strain RHA1. Appl Environ Microbiol 72 6183 6193
10. MathieuJMMohnWWEltisLDLeBlancJCStewartGR 2010 7-ketocholesterol catabolism by Rhodococcus jostii RHA1. Appl Environ Microbiol 76 352 355
11. PuglisiECahillMJLessardPACapriESinskeyAJ 2010 Transcriptional response of Rhodococcus aetherivorans I24 to polychlorinated biphenyl-contaminated sediments. Microb Ecol 60 505 515
12. PeoplesOPSinskeyAJ 1989 Poly-beta-hydroxybutyrate (PHB) biosynthesis in Alcaligenes eutrophus H16. Identification and characterization of the PHB polymerase gene (phbC). Journal of Biological Chemistry 264 15298 15303
13. SlaterSCVoigeWHDennisDE 1988 Cloning and expression in Escherichia coli of the Alcaligenes eutrophus H16 poly-beta-hydroxybutyrate biosynthetic pathway. Journal of Bacteriology 170 4431 4436
14. JendrossekD 2009 Polyhydroxyalkanoate Granules Are Complex Subcellular Organelles (Carbonosomes). J Bacteriol 191 3195 3202
15. RajakumariSGrillitschKDaumG 2008 Synthesis and turnover of non-polar lipids in yeast. Progress in Lipid Research 47 157 171
16. AlvarezHMAS 2010 Biology of Rhodococcus. Microbiology Monographs 16 263 289
17. ArgyrouAVettingMBlanchardJ 2007 New insight into the mechanism of action of and resistance to isoniazid: interaction of Mycobacterium tuberculosis enoyl-ACP reductase with INH-NADP. Journal of the American Chemical Society 129 9582 9583
18. TimminsGDereticV 2006 Mechanisms of action of isoniazid. Molecular Microbiology 62 1220 1227
19. RamanKRajagopalanPChandraN 2005 Flux Balance Analysis of Mycolic Acid Pathway: Targets for Anti-Tubercular Drugs. PLoS Comput Biol 1 e46 doi:10.1371/journal.pcbi.0010046
20. ZimhonyOCoxJWelchJVilchezeCJacobsW 2000 Pyrazinamide inhibits the eukaryotic-like fatty acid synthetase I (FASI) of Mycobacterium tuberculosis. Nature Medicine 6 1043 1047
21. SchweizerEHofmannJ 2004 Microbial type I fatty acid synthases (FAS): major players in a network of cellular FAS systems. Microbiology and molecular biology reviews: MMBR 68 501 517
22. SutcliffeIC 1998 Cell envelope composition and organisation in the genus Rhodococcus. Antonie Van Leeuwenhoek 74 49 58
23. HsuF-FSoehlKTurkJHaasA 2011 Characterization of mycolic acids from the pathogen Rhodococcus equi by tandem mass spectrometry with electrospray ionization. Analytical Biochemistry 409 112 122
24. KarpPDPaleySMKrummenackerMLatendresseMDaleJM 2010 Pathway Tools version 13.0: integrated software for pathway/genome informatics and systems biology. Brief Bioinform 11 40 79
25. ArakakiAHuangYSkolnickJ 2009 EFICAz2: enzyme function inference by a combined approach enhanced by machine learning. BMC Bioinformatics 10 107
26. KikuchiSRainwaterDLKolattukudyPE 1992 Purification and characterization of an unusually large fatty acid synthase from Mycobacterium tuberculosis var. bovis BCG. Archives of Biochemistry and Biophysics 295 318 326
27. VanceDEMitsuhashiOBlochK 1973 Purification and properties of the fatty acid synthetase from Mycobacterium phlei. J Biol Chem 248 2303 2309
28. WuMEisenJA 2008 A simple, fast, and accurate method of phylogenomic inference. Genome Biol 9 R151
29. KalscheuerRSteinbuchelA 2003 A novel bifunctional wax ester synthase/acyl-CoA:diacylglycerol acyltransferase mediates wax ester and triacylglycerol biosynthesis in Acinetobacter calcoaceticus ADP1. J Biol Chem 278 8075 8082
30. AlvarezAFAlvarezHMKalscheuerRWaltermannMSteinbuchelA 2008 Cloning and characterization of a gene involved in triacylglycerol biosynthesis and identification of additional homologous genes in the oleaginous bacterium Rhodococcus opacus PD630. Microbiology 154 2327 2335
31. GandeRDoverLGKrumbachKBesraGSSahmH 2007 The two carboxylases of Corynebacterium glutamicum essential for fatty acid and mycolic acid synthesis. J Bacteriol 189 5257 5264
32. PortevinDde Sousa-D'AuriaCMontrozierHHoussinCStellaA 2005 The acyl-AMP ligase FadD32 and AccD4-containing acyl-CoA carboxylase are required for the synthesis of mycolic acids and essential for mycobacterial growth: identification of the carboxylation product and determination of the acyl-CoA carboxylase components. J Biol Chem 280 8862 8874
33. GandeRGibsonKJBrownAKKrumbachKDoverLG 2004 Acyl-CoA carboxylases (accD2 and accD3), together with a unique polyketide synthase (Cg-pks), are key to mycolic acid biosynthesis in Corynebacterianeae such as Corynebacterium glutamicum and Mycobacterium tuberculosis. J Biol Chem 279 44847 44857
34. DiacovichLPeiruSKurthDRodriguezEPodestaF 2002 Kinetic and Structural Analysis of a New Group of Acyl-CoA Carboxylases Found in Streptomyces coelicolor A3(2). J Biol Chem 277 31228 31236
35. RainwaterDLKolattukudyPE 1985 Fatty acid biosynthesis in Mycobacterium tuberculosis var. bovis Bacillus Calmette-Guerin. Purification and characterization of a novel fatty acid synthase, mycocerosic acid synthase, which elongates n-fatty acyl-CoA with methylmalonyl-CoA. Journal of Biological Chemistry 260 616 623
36. MatsunagaIBhattAYoungDCChengTYEylesSJ 2004 Mycobacterium tuberculosis pks12 produces a novel polyketide presented by CD1c to T cells. J Exp Med 200 1559 1569
37. SirakovaTDubeyVKimH-JCynamonMKolattukudyP 2003 The Largest Open Reading Frame (pks12) in the Mycobacterium tuberculosis Genome Is Involved in Pathogenesis and Dimycocerosyl Phthiocerol Synthesis. Infect Immun 71 3794 3801
38. SirakovaTDDubeyVSCynamonMHKolattukudyPE 2003 Attenuation of Mycobacterium tuberculosis by disruption of a mas-like gene or a chalcone synthase-like gene, which causes deficiency in dimycocerosyl phthiocerol synthesis. J Bacteriol 185 2999 3008
39. SirakovaTDThirumalaAKDubeyVSSprecherHKolattukudyPE 2001 The Mycobacterium tuberculosis pks2 gene encodes the synthase for the hepta - and octamethyl-branched fatty acids required for sulfolipid synthesis. The Journal of Biological Chemistry 276 16833 16839
40. GrafeUReinhardtGKrebsDRothMBormannEJ 1982 Biochemical characteristics of non-streptomycin-producing mutants of Streptomyces griseus. II. Lipids and fatty acid composition of vegetative mycelia. Z Allg Mikrobiol 22 97 106
41. KaddorCBiermannKKalscheuerRSteinbuchelA 2009 Analysis of neutral lipid biosynthesis in Streptomyces avermitilis MA-4680 and characterization of an acyltransferase involved herein. Appl Microbiol Biotechnol 84 143 155
42. EnrightAJKuninVOuzounisCA 2003 Protein families and TRIBES in genome sequence space. Nucleic Acids Res 31 4632 4638
43. ChopraTBanerjeeSGuptaSYadavGAnandS 2008 Novel Intermolecular Iterative Mechanism for Biosynthesis of Mycoketide Catalyzed by a Bimodular Polyketide Synthase. PLoS Biol 6 e163 doi:10.1371/journal.pbio.0060163
44. RainwaterDLKolattukudyPE 1983 Synthesis of mycocerosic acids from methylmalonyl coenzyme A by cell-free extracts of Mycobacterium tuberculosis var. bovis BCG. Journal of Biological Chemistry 258 2979 2985
45. HorswillAREscalante-SemerenaJC 1999 Salmonella typhimurium LT2 catabolizes propionate via the 2-methylcitric acid cycle. J Bacteriol 181 5615 5623
46. DavidFTienpontBSandraP 2008 Chemotaxonomy of bacteria by comprehensive GC and GC-MS in electron impact and chemical ionisation mode. Journal of Separation Science 31 3395 3403
47. LenzOLudwigMSchubertTBurstelIGanskowS 2010 H2 conversion in the presence of O2 as performed by the membrane-bound [NiFe]-hydrogenase of Ralstonia eutropha. Chemphyschem 11 1107 1119
48. PohlmannAFrickeWFReineckeFKusianBLiesegangH 2006 Genome sequence of the bioplastic-producing “Knallgas” bacterium Ralstonia eutropha H16. Nat Biotechnol 24 1257 1262
49. SchwartzEVoigtBZuhlkeDPohlmannALenzO 2009 A proteomic view of the facultatively chemolithoautotrophic lifestyle of Ralstonia eutropha H16. Proteomics 9 5132 5142
50. ParkerSKCurtinKMVasilML 2007 Purification and characterization of mycobacterial phospholipase A: an activity associated with mycobacterial cutinase. J Bacteriol 189 4153 4160
51. VimrEKalivodaKDeszoESteenbergenS 2004 Diversity of microbial sialic acid metabolism. Microbiology and molecular biology reviews: MMBR 68 132 153
52. SeveriEHoodDWThomasGH 2007 Sialic acid utilization by bacterial pathogens. Microbiology 153 2817 2822
53. PostDMungurRGibsonBMunsonR 2005 Identification of a novel sialic acid transporter in Haemophilus ducreyi. Infection and Immunity 73 6727 6735
54. SeveriEHosieAHawkheadJThomasG 2009 Characterization of a novel sialic acid transporter of the sodium solute symporter (SSS) family and in vivo comparison with known bacterial sialic acid transporters. FEMS Microbiology Letters 304 47 54
55. HunterRLOlsenMJagannathCActorJK 2006 Trehalose 6,6′-dimycolate and lipid in the pathogenesis of caseating granulomas of tuberculosis in mice. Am J Pathol 168 1249 1261
56. RengarajanJBloomBRRubinEJ 2005 Genome-wide requirements for Mycobacterium tuberculosis adaptation and survival in macrophages. Proc Natl Acad Sci U S A 102 8327 8332
57. PandeyAKSassettiCM 2008 Mycobacterial persistence requires the utilization of host cholesterol. Proc Natl Acad Sci U S A 105 4376 4380
58. HuYvan der GeizeRBesraGSGurchaSSLiuA 2010 3-Ketosteroid 9alpha-hydroxylase is an essential factor in the pathogenesis of Mycobacterium tuberculosis. Mol Microbiol 75 107 121
59. YangXNesbittNMDubnauESmithISampsonNS 2009 Cholesterol metabolism increases the metabolic pool of propionate in Mycobacterium tuberculosis. Biochemistry 48 3819 3821
60. Van der GeizeRYamKHeuserTWilbrinkMHHaraH 2007 A gene cluster encoding cholesterol catabolism in a soil actinomycete provides insight into Mycobacterium tuberculosis survival in macrophages. Proceedings of the National Academy of Sciences 104 1947 1952
61. YamKCOkamotoSRobertsJNEltisLD 2011 Adventures in Rhodococcus - from steroids to explosives. Can J Microbiol 57 155 168
62. LomakinIBXiongYSteitzTA 2007 The Crystal Structure of Yeast Fatty Acid Synthase, a Cellular Machine with Eight Active Sites Working Together. Cell 129 319 332
63. MacEachranDPPropheteMESinskeyAJ 2010 The Rhodococcus opacus PD630 heparin-binding hemagglutinin homolog TadA mediates lipid body formation. Appl Environ Microbiol 76 7217 7225
64. LessardPAO'BrienXMCurrieDHSinskeyAJ 2004 pB264, a small, mobilizable, temperature sensitive plasmid from Rhodococcus. BMC Microbiol 4 15
65. IsonoKMcIninchJDBorodovskyM 1994 Characteristic features of the nucleotide sequences of yeast mitochondrial ribosomal protein genes as analyzed by computer program GeneMark. DNA Res 1 263 269
66. DelcherALHarmonDKasifSWhiteOSalzbergSL 1999 Improved microbial gene identification with GLIMMER. Nucleic Acids Res 27 4636 4641
67. DelcherALPhillippyACarltonJSalzbergSL 2002 Fast algorithms for large-scale genome alignment and comparison. Nucleic Acids Res 30 2478 2483
68. BrudnoMDoCBCooperGMKimMFDavydovE 2003 LAGAN and Multi-LAGAN: efficient tools for large-scale multiple alignment of genomic DNA. Genome Res 13 721 731
69. FinnRDMistryJSchuster-BocklerBGriffiths-JonesSHollichV 2006 Pfam: clans, web tools and services. Nucleic Acids Res 34 D247 251
70. LagesenKHallinPRodlandEAStaerfeldtHHRognesT 2007 RNAmmer: consistent and rapid annotation of ribosomal RNA genes. Nucleic Acids Res 35 3100 3108
71. LoweTMEddySR 1997 tRNAscan-SE: a program for improved detection of transfer RNA genes in genomic sequence. Nucleic Acids Res 25 955 964
72. Griffiths-JonesSMoxonSMarshallMKhannaAEddySR 2005 Rfam: annotating non-coding RNAs in complete genomes. Nucleic Acids Res 33 D121 124
73. RomeroPRKarpPD 2004 Using functional and organizational information to improve genome-wide computational prediction of transcription units on pathway-genome databases. Bioinformatics 20 709 717
74. LeeTPaulsenIKarpP 2008 Annotation-based inference of transporter function. Bioinformatics 24 i259 i267
75. GreenMKarpP 2004 A Bayesian method for identifying missing enzymes in predicted metabolic pathway databases. BMC Bioinformatics 5 76
76. LiLStoeckertCRoosD 2003 OrthoMCL: Identification of Ortholog Groups for Eukaryotic Genomes. Genome Research 13 2178 2189
77. KatohKKumaKTohHMiyataT 2005 MAFFT version 5: improvement in accuracy of multiple sequence alignment. Nucleic Acids Res 33 511 518
78. StamatakisA 2006 RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 22 2688 2690
79. WaterhouseAMProcterJBMartinDMClampMBartonGJ 2009 Jalview Version 2–a multiple sequence alignment editor and analysis workbench. Bioinformatics 25 1189 1191
80. Marchler-BauerALuSAndersonJBChitsazFDerbyshireMK 2011 CDD: a Conserved Domain Database for the functional annotation of proteins. Nucleic Acids Res 39 D225 229
81. EdgarR 2004 MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic acids research 32 1792 1797
82. LarkinMABlackshieldsGBrownNPChennaRMcGettiganPA 2007 Clustal W and Clustal X version 2.0. Bioinformatics 23 2947 2948
83. AbascalFZardoyaRPosadaD 2005 ProtTest: selection of best-fit models of protein evolution. Bioinformatics 21 2104 2105
84. GuindonSGascuelO 2003 A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Systematic biology 52 696 704
85. LetunicIBorkP 2007 Interactive Tree Of Life (iTOL): an online tool for phylogenetic tree display and annotation. Bioinformatics 23 127 128
86. MansourMP 2005 Reversed-phase high-performance liquid chromatography purification of methyl esters of C(16)–C(28) polyunsaturated fatty acids in microalgae, including octacosaoctaenoic acid [28 : 8(n-3)]. J Chromatogr A 1097 54 58
87. SaeedAIBhagabatiNKBraistedJCLiangWSharovV 2006 TM4 microarray software suite. Methods Enzymol 411 134 193
88. de HoonMJImotoSNolanJMiyanoS 2004 Open source clustering software. Bioinformatics 20 1453 1454
Štítky
Genetika Reprodukční medicína
Článek Produces a Long Noncoding RNA and Has General Functions in the Germline, Stem Cells, and BrainČlánek Inference of Relationships in Population Data Using Identity-by-Descent and Identity-by-StateČlánek A Genome-Wide Meta-Analysis of Six Type 1 Diabetes Cohorts Identifies Multiple Associated LociČlánek Retrotransposon-Induced Heterochromatin Spreading in the Mouse Revealed by Insertional PolymorphismsČlánek Essential Roles of BCCIP in Mouse Embryonic Development and Structural Stability of ChromosomesČlánek The Receptor Tyrosine Kinase Alk Controls Neurofibromin Functions in Drosophila Growth and LearningČlánek PCNA Ubiquitination Is Important, But Not Essential for Translesion DNA Synthesis in Mammalian CellsČlánek Genome-Wide Association Study Identifies Four Loci Associated with Eruption of Permanent Teeth
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2011 Číslo 9- Je „freeze-all“ pro všechny? Odborníci na fertilitu diskutovali na virtuálním summitu
- Kazuistika – Perspektivy využití precizované medicíny v rámci personalizované specifické terapie onkologických pacientů
- Nobelova cena za chemii pro genetické nůžky: Objev, který změní naši budoucnost?
- Technologie na bázi RNA v klinické praxi: od přebarvených petúnií k terapii vzácných a dosud jen obtížně léčitelných chorob u lidí
- „Nepředstavovali jsme si, že náš výzkum povede přímo ke vzniku nových léků, dokonce ještě za našeho života“
-
Všechny články tohoto čísla
- Temporal Trends in Results Availability from Genome-Wide Association Studies
- Association of eGFR-Related Loci Identified by GWAS with Incident CKD and ESRD
- Genetic Variants at Chromosomes 2q35, 5p12, 6q25.1, 10q26.13, and 16q12.1 Influence the Risk of Breast Cancer in Men
- Large-Scale Gene-Centric Analysis Identifies Novel Variants for Coronary Artery Disease
- Genetic Association for Renal Traits among Participants of African Ancestry Reveals New Loci for Renal Function
- Transcriptome Kinetics Is Governed by a Genome-Wide Coupling of mRNA Production and Degradation: A Role for RNA Pol II
- Conserved Regulation of p53 Network Dosage by MicroRNA–125b Occurs through Evolving miRNA–Target Gene Pairs
- Heterozygous Mutations of Are Associated with an Increased Risk of Isolated Metopic Craniosynostosis in Humans and Mice
- Study of FoxA Pioneer Factor at Silent Genes Reveals Rfx-Repressed Enhancer at and a Potential Indicator of Esophageal Adenocarcinoma Development
- Cholesterol Metabolism Is Required for Intracellular Hedgehog Signal Transduction
- Produces a Long Noncoding RNA and Has General Functions in the Germline, Stem Cells, and Brain
- Age-Dependent Recombination Rates in Human Pedigrees
- Sequence Conservation and Functional Constraint on Intergenic Spacers in Reduced Genomes of the Obligate Symbiont
- Sex Chromosome Mosaicism and Hybrid Speciation among Tiger Swallowtail Butterflies
- A Negative Feedback Loop That Limits the Ectopic Activation of a Cell Type–Specific Sporulation Sigma Factor of
- Phased Whole-Genome Genetic Risk in a Family Quartet Using a Major Allele Reference Sequence
- Mutations in or near the Transmembrane Domain Alter PMEL Amyloid Formation from Functional to Pathogenic
- Inactivation of Alters Melanosome Shape But Has Only a Subtle Effect on Visible Pigmentation
- Novel Interactions between Actin and the Proteasome Revealed by Complex Haploinsufficiency
- Germline Genetic Variants Disturbing the /LIN28 Double-Negative Feedback Loop Alter Breast Cancer Susceptibility
- Separation of Recombination and SOS Response in RecA Suggests LexA Interaction Sites
- Inference of Relationships in Population Data Using Identity-by-Descent and Identity-by-State
- Misregulation of Scm3p/HJURP Causes Chromosome Instability in and Human Cells
- A Noncoding Point Mutation of Causes Multiple Developmental Malformations and Obesity in Twirler Mice
- The Evolutionarily Conserved Longevity Determinants HCF-1 and SIR-2.1/SIRT1 Collaborate to Regulate DAF-16/FOXO
- A Genome-Wide Metabolic QTL Analysis in Europeans Implicates Two Loci Shaped by Recent Positive Selection
- Bacterial Communities of Diverse Species: Ecological Context of a Host–Microbe Model System
- A Genome-Wide Meta-Analysis of Six Type 1 Diabetes Cohorts Identifies Multiple Associated Loci
- Elongator Complex Influences Telomeric Gene Silencing and DNA Damage Response by Its Role in Wobble Uridine tRNA Modification
- Elevated Proteasome Capacity Extends Replicative Lifespan in
- Genome-Wide Analysis of Heteroduplex DNA in Mismatch Repair–Deficient Yeast Cells Reveals Novel Properties of Meiotic Recombination Pathways
- MicroRNA Predictors of Longevity in
- An Iterative Genetic and Dynamical Modelling Approach Identifies Novel Features of the Gene Regulatory Network Underlying Melanocyte Development
- Atypical AT Skew in Firmicute Genomes Results from Selection and Not from Mutation
- Retrotransposon-Induced Heterochromatin Spreading in the Mouse Revealed by Insertional Polymorphisms
- Genomic Analysis of QTLs and Genes Altering Natural Variation in Stochastic Noise
- The Abnormal Phenotypes of Cartilage and Bone in Calcium-Sensing Receptor Deficient Mice Are Dependent on the Actions of Calcium, Phosphorus, and PTH
- Cell Type–Specific Transcriptome Analysis Reveals a Major Role for and miR-200b in Mouse Inner Ear Morphogenesis
- Essential Roles of BCCIP in Mouse Embryonic Development and Structural Stability of Chromosomes
- IAP1-Mediated Ubiquitylation Controls Activation of the Initiator Caspase DRONC Independent of Protein Degradation
- VANG-1 and PRKL-1 Cooperate to Negatively Regulate Neurite Formation in
- The Receptor Tyrosine Kinase Alk Controls Neurofibromin Functions in Drosophila Growth and Learning
- Comparative and Functional Genomics of PD630 for Biofuels Development
- Identification of Type 1 Diabetes–Associated DNA Methylation Variable Positions That Precede Disease Diagnosis
- PCNA Ubiquitination Is Important, But Not Essential for Translesion DNA Synthesis in Mammalian Cells
- Genetic Effects at Pleiotropic Loci Are Context-Dependent with Consequences for the Maintenance of Genetic Variation in Populations
- Genome-Wide Association Study Identifies Four Loci Associated with Eruption of Permanent Teeth
- Bmp and Nodal Independently Regulate Expression to Maintain Unilateral Nodal Activity during Left-Right Axis Specification in Zebrafish
- Inter-Allelic Prion Propagation Reveals Conformational Relationships among a Multitude of [] Strains
- Emergence and Modular Evolution of a Novel Motility Machinery in Bacteria
- Histone Methyltransferase MET-2 Shields the Male X Chromosome from Checkpoint Machinery and Mediates Meiotic Sex Chromosome Inactivation
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Retrotransposon-Induced Heterochromatin Spreading in the Mouse Revealed by Insertional Polymorphisms
- The Evolutionarily Conserved Longevity Determinants HCF-1 and SIR-2.1/SIRT1 Collaborate to Regulate DAF-16/FOXO
- Genome-Wide Analysis of Heteroduplex DNA in Mismatch Repair–Deficient Yeast Cells Reveals Novel Properties of Meiotic Recombination Pathways
- Association of eGFR-Related Loci Identified by GWAS with Incident CKD and ESRD
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



