-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praxe
CPAF: A Chlamydial Protease in Search of an Authentic Substrate
Bacteria in the genus Chlamydia are major human pathogens that cause an intracellular infection. A chlamydial protease, CPAF, has been proposed as an important virulence factor that cleaves or degrades at least 16 host proteins, thereby altering multiple cellular processes. We examined 11 published CPAF substrates and found that there was no detectable proteolysis when CPAF activity was inhibited during cell processing. We show that the reported proteolysis of these putative CPAF substrates was due to enzymatic activity in cell lysates rather than in intact cells. Nevertheless, Chlamydia-infected cells displayed Chlamydia-host interactions, such as Golgi reorganization, apoptosis resistance, and host cytoskeletal remodeling, that have been attributed to CPAF-dependent proteolysis of host proteins. Our findings suggest that other mechanisms may be responsible for these Chlamydia-host interactions, and raise concerns about all published CPAF substrates and the proposed roles of CPAF in chlamydial pathogenesis.
Published in the journal: . PLoS Pathog 8(8): e32767. doi:10.1371/journal.ppat.1002842
Category: Research Article
doi: https://doi.org/10.1371/journal.ppat.1002842Summary
Bacteria in the genus Chlamydia are major human pathogens that cause an intracellular infection. A chlamydial protease, CPAF, has been proposed as an important virulence factor that cleaves or degrades at least 16 host proteins, thereby altering multiple cellular processes. We examined 11 published CPAF substrates and found that there was no detectable proteolysis when CPAF activity was inhibited during cell processing. We show that the reported proteolysis of these putative CPAF substrates was due to enzymatic activity in cell lysates rather than in intact cells. Nevertheless, Chlamydia-infected cells displayed Chlamydia-host interactions, such as Golgi reorganization, apoptosis resistance, and host cytoskeletal remodeling, that have been attributed to CPAF-dependent proteolysis of host proteins. Our findings suggest that other mechanisms may be responsible for these Chlamydia-host interactions, and raise concerns about all published CPAF substrates and the proposed roles of CPAF in chlamydial pathogenesis.
Introduction
Chlamydia are obligate intracellular bacteria that are responsible for more infections reported to the CDC than all other infectious agents combined [1]. Chlamydia trachomatis causes the most prevalent bacterial sexually transmitted disease in the United States [2] and the most common form of preventable blindness worldwide [3]. Another species, Chlamydia pneumoniae, is a causative agent of community-acquired pneumonia [4]. Despite this wide range of clinical manifestations, Chlamydia spp. display many similarities at the level of the intracellular infection. Chlamydiae replicate within a membrane-bound compartment called the chlamydial inclusion in which the bacterium converts between two specialized forms. During this developmental cycle, chlamydiae usurp or subvert a number of processes within the host cell to support the infection. For instance, Chlamydia alters the host secretory pathway to acquire lipids from post-Golgi vesicles to support growth of the inclusion and bacterial replication [5]–[7]. It also blocks host cell apoptosis, which could otherwise be used as a host defense mechanism against this intracellular pathogen that requires 2–3 days to complete its developmental cycle [8]–[10].
CPAF (chlamydial protease or proteasome-like activity factor) has been proposed to be a major virulence factor in Chlamydia-infected cells [11]. This atypical serine protease [12] is conserved within the Chlamydiales [13], including the distantly related environmental chlamydiae, which include endosymbionts of amoeba [14]. During an infection, CPAF is secreted into the host cytoplasm where it has been reported to cleave or degrade specific host proteins [15]. A rapidly growing number of CPAF substrates has been reported [11], including at least 16 host proteins (Table 1).
Tab. 1. Summary of published CPAF substrates. 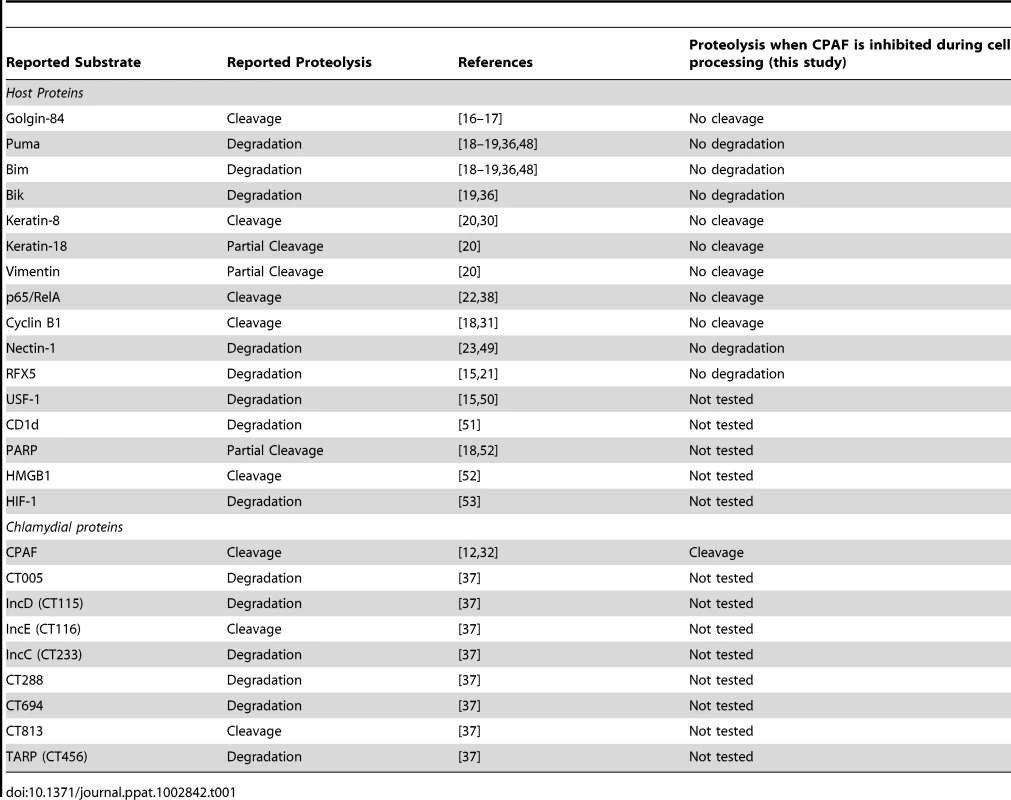
This proteolysis of host proteins by CPAF has been proposed to cause a number of effects on the infected host cell. For example, cleavage of the Golgi matrix protein golgin-84 has been reported to cause fragmentation and reorganization of the Golgi apparatus in Chlamydia-infected cells [16]–[17]. Similarly, degradation of pro-apoptotic BH3-only proteins, such as Puma, Bik, and Bim, has been proposed to mediate resistance to apoptosis [18]–[19]. Cleavage of several intermediate filaments has been linked to the dynamic remodeling of the host cytoskeleton around the growing chlamydial inclusion [20]. In addition, proteolysis of host transcription factors, such as the MHC transcription factor RFX5 [15], [21] and the p65/RelA subunit of NFκB [22], have been implicated as chlamydial strategies for evading the immune response of the host cell. In general, these associations have been inferred from the known functions of reported CPAF substrates, but the direct effects of these proteolytic events in Chlamydia-infected cells have not been examined [11].
The evidence for CPAF as a chlamydial protease that targets host proteins has been largely based on the observed cleavage or degradation of specific host proteins during an infection, as assayed by immunoblotting lysates from Chlamydia-infected cells [15]–[16], [18]–[20], [22]–[23]. In addition, many reports demonstrated that the timing of this proteolysis correlates with the expression of CPAF during the developmental cycle [24] and can be prevented in vitro [15], [19], [23] and in vivo [20], [23] by the CPAF inhibitor lactacystin [15]. Furthermore, the same cleavage or degradation patterns have been reproduced when recombinant CPAF was used in vitro [15], [19]–[20], [23] or overexpressed in uninfected cells [16], [18], [22].
In this study, we demonstrate that the reported cleavage or degradation of 11 published CPAF substrates was abrogated when the enzymatic activity of CPAF was inhibited during cell harvest and lysate preparation. However, we still observed host-pathogen interactions, such as Golgi reorganization and resistance to apoptosis, that are proposed to result from proteolysis of target proteins by CPAF. Our findings indicate that these host-Chlamydia interactions are likely to be mediated by mechanisms other than CPAF-dependent proteolysis of these host proteins. These results invite a reappraisal of previously identified CPAF substrates and re-interpretation of models involving the function of this chlamydial enzyme in the intracellular infection.
Results
Re-examination of Golgin-84 Cleavage during a Chlamydial Infection
The Golgi protein golgin-84 is reported to be cleaved by CPAF in Chlamydia-infected cells [16], but we found that cleavage is dependent on the method of cell harvest and lysis (which we will hereafter collectively refer to as ‘cell processing’). To reproduce the published proteolysis, we harvested infected cells under standard lysis conditions in RIPA buffer [25] and analyzed the cell lysate by SDS-PAGE, followed by immunoblotting with anti-golgin-84 antibodies. Consistent with previous reports, there was progressive conversion of full-length golgin-84 into two cleavage products of ∼78 and ∼65 kDa beginning at 18 hours post infection (hpi) (Figure 1A, top panel), and most of the full-length protein in the extracts was cleaved by 36 hpi [16]–[17]. In striking contrast, there was no golgin-84 cleavage, even as late as 36 hpi, when we treated Chlamydia-infected cells for one hour prior to cell processing with 150 µM of clasto-lactacystin β-lactone, which is the active form of the CPAF inhibitor lactacystin [15], [26] (Figure 1A, compare top and bottom panels). This inhibition of golgin-84 proteolysis is unlikely to be due to the activity of clasto-lactacystin as a proteasome inhibitor [27] because we did not detect inhibition of proteasome function in our infected cells from the one hour treatment (data not shown). These results suggest that the cleavage of golgin-84 occurred during or after cell processing in standard lysis buffers and was prevented by inhibiting CPAF activity prior to these manipulations.
Fig. 1. Golgin-84 cleavage does not occur in Chlamydia-infected cells when CPAF is inhibited during cell processing. 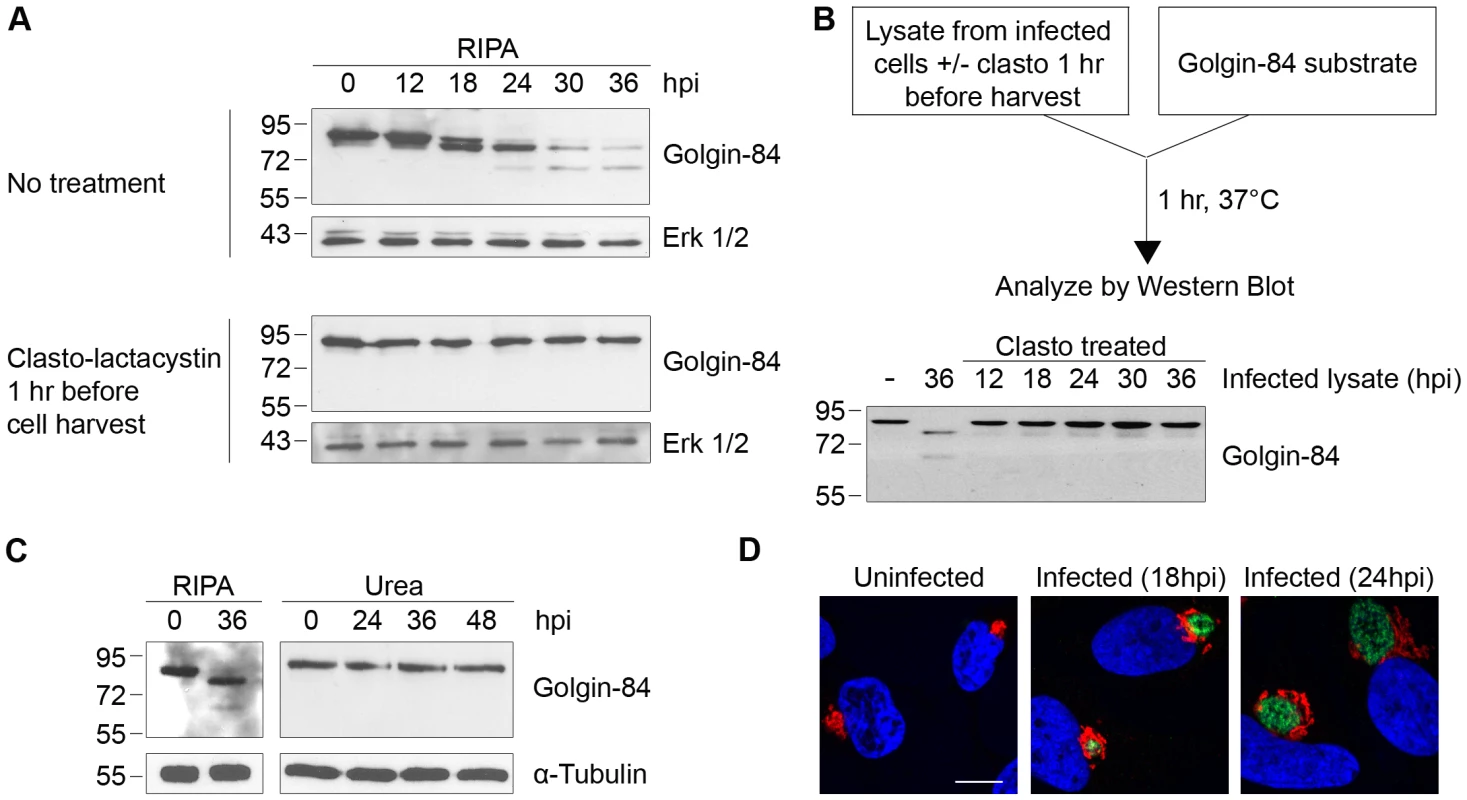
(A) Uninfected (0 hpi) and infected cells at time points between 12 and 36 hpi were treated with methyl acetate as a solvent control (top panel) or 150 µM of the CPAF inhibitor clasto-lactacystin (bottom panel) for 1 hour prior to cell lysis in RIPA buffer. Total cell lysates were separated by SDS-PAGE and probed with antibodies against golgin-84 or Erk 1/2 (loading control). (B) Cell-free degradation assay testing for CPAF activity in lysates prepared from the Chlamydia-infected HeLa cells described in Figure 1A. Each infected cell lysate was incubated with a lysate of uninfected HeLa cells as the source of golgin-84 substrate and reactions were analyzed by immunoblotting with golgin-84 antibodies. (C) Lysates of uninfected (0 hpi) or infected cells from different times in the infection were prepared in RIPA buffer (left panel) or by direct lysis in 8M urea (right panel), separated by SDS-PAGE and analyzed with antibodies to golgin-84 or α-tubulin (loading control). (D) Confocal images of uninfected or Chlamydia-infected HeLa cells examined at 18 and 24 hpi. Cells were stained with antibodies to the Golgi marker α-mannosidase II (red), the chlamydial major outer membrane protein MOMP (green) and the DNA dye Hoechst 33342 (blue) to detect Golgi membranes, the chlamydial inclusion and DNA, respectively. Scale bar, 10 µm. We next developed a cell-free assay to test for CPAF activity in lysates of Chlamydia-infected cells. When we incubated infected cell lysate with a lysate of uninfected HeLa cells as a source of golgin-84 substrate, there was almost complete cleavage of golgin-84 at 37°C (Figure 1B) and at 0°C (Figure S1A). These in vitro experiments demonstrate that CPAF remains active in lysates from Chlamydia-infected cells, even on ice, and thus could cleave a putative substrate during lysate preparation. In contrast, lysates of Chlamydia-infected cells pre-treated with clasto-lactacystin, for one hour before processing at times up to 36 hpi, did not cleave golgin-84 in this in vitro assay (Figure 1B). These results show that CPAF activity during lysate preparation can be abolished by treating the infected cells with clasto-lactacystin for one hour prior to cell processing.
To determine whether clasto-lactacystin was preventing golgin-84 cleavage during the one hour treatment before cell lysis or during cell processing itself, we used an alternative approach to inhibit CPAF activity during cell processing. In these experiments, we lysed cells in urea as a denaturing agent to block enzymatic activity in our lysates [28]. When we lysed Chlamydia-infected cells by adding 8M urea directly to the monolayer, no golgin-84 cleavage was observed even as late as 48 hpi (Figure 1C). These lysates did not contain detectable CPAF activity as verified with the cell-free degradation assay (data not shown). This lack of golgin-84 cleavage was observed for Chlamydia infection of HeLa cells and two other human cell lines when lysed in urea (Figure S1B). Taken together, these results lead us to conclude that the reported CPAF-dependent cleavage of golgin-84 is unlikely to occur in intact cells. Our results are consistent with an explanation that proteolysis occurred during cell processing and is due to CPAF activity in the lysates of Chlamydia-infected cells.
We examined Golgi organization in cells on parallel coverslips because cleavage of golgin-84 has been proposed to induce Golgi fragmentation in Chlamydia-infected cells [17]. In uninfected control cells, Golgi membranes were arranged as an interconnected Golgi ribbon in the pericentriolar region, as detected by immunofluorescence with antibodies to the Golgi marker mannosidase II [29]. In contrast, Golgi membranes of infected cells were reorganized around the growing chlamydial inclusion by 18 and 24 hpi (Figure 1D), which is consistent with previous reports [17]. Thus, Golgi reorganization in our Chlamydia-infected cells occurs in the absence of detectable golgin-84 cleavage and is unlikely to be caused by CPAF-dependent proteolysis of this structural Golgi protein.
Degradation of Pro-apoptotic BH3-only Proteins Is Also Dependent on Cell Processing
Our findings with golgin-84 motivated us to investigate other host proteins reported to be CPAF substrates based on immunoblots of lysates prepared with standard buffers. The resistance of Chlamydia-infected cells to apoptosis [8]–[10] has been proposed to be mediated by CPAF-dependent degradation of BH3-only proteins, including Puma, Bik, and Bim [18]–[19]. We again replicated the complete degradation of each of these proteins by lysing Chlamydia-infected cells in RIPA buffer at 36 hpi (Figure 2A). However, when infected cells were lysed in 8M urea, Puma, Bik, and Bim were unaltered even at 48 hpi (Figure 2A). Thus, as with cleavage of golgin-84, inhibition of CPAF activity during cell processing abolished the published degradation of these pro-apoptotic factors.
Fig. 2. Degradation of BH3-only proteins in Chlamydia-infected cells is prevented by inhibiting CPAF during cell processing. 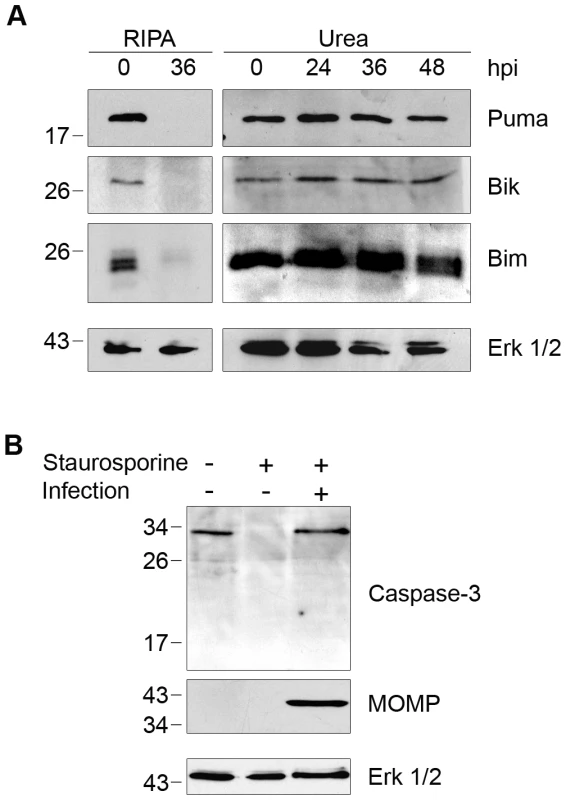
(A) Lysates of uninfected (0 hpi) or infected HeLa cells were prepared in RIPA buffer (left panel) or by direct lysis in 8M urea (right panel) at the indicated times, separated by SDS-PAGE and analyzed by immunoblotting with antibodies to the proapoptotic BH3-only proteins Puma, Bik, or Bim. Equal loading was monitored for each blot with antibodies to Erk 1/2, but only the loading control for the Puma blot is shown as an example. (B) Uninfected or Chlamydia-infected HeLa cells were treated with 1 µM staurosporine to induce apoptosis, which was monitored by the loss of full-length caspase-3. Immunoblots of the lysates were probed with antibodies to caspase-3, MOMP (marker of Chlamydia infection), or Erk 1/2 (loading control). Although Puma, Bik, and Bim were not degraded in Chlamydia-infected cells, these cells were resistant to staurosporine-induced programmed cell death, as previously reported [8]. Staurosporine treatment for 3 hours caused the complete loss of full-length caspase-3 in uninfected cells, but there was no decrease in full-length caspase-3 levels in our C. trachomatis-infected cells (Figure 2B). Based on these results, it is doubtful that the anti-apoptotic effects of a chlamydial infection on the host cell can be attributed to CPAF-dependent degradation of the BH3-only proteins Puma, Bik, and Bim.
Proteolysis of Seven Additional Host Proteins by CPAF Is Dependent on Cell Processing
We next examined the proteolytic effects of CPAF on the three intermediate filaments keratin-8, keratin-18, and vimentin, whose cleavage has been implicated in the growth of the chlamydial inclusion [20], [30]. When we lysed Chlamydia-infected cells at 36 hpi in RIPA buffer (Figure 3A), we observed the conversion of each full-length protein into smaller fragments, as previously reported [20], [30]. In contrast, when we lysed cells directly in 8M urea, we did not detect any proteolytic effects on these proteins over the time course of an infection up to 48 hpi (Figure 3A), indicating that CPAF was unlikely to cleave these proteins in intact cells. CPAF-dependent cleavage of vimentin has been proposed to be important for host cytoskeletal rearrangement into a supportive cage surrounding the chlamydial inclusion [20]. However, we still observed a cage-like vimentin structure around the inclusion in our Chlamydia-infected cells (Figure 3B), which suggests that this reorganization of the host cytoskeleton during an infection does not require vimentin cleavage.
Fig. 3. Cleavage of intermediate filaments in Chlamydia-infected cells is also dependent on cell processing. 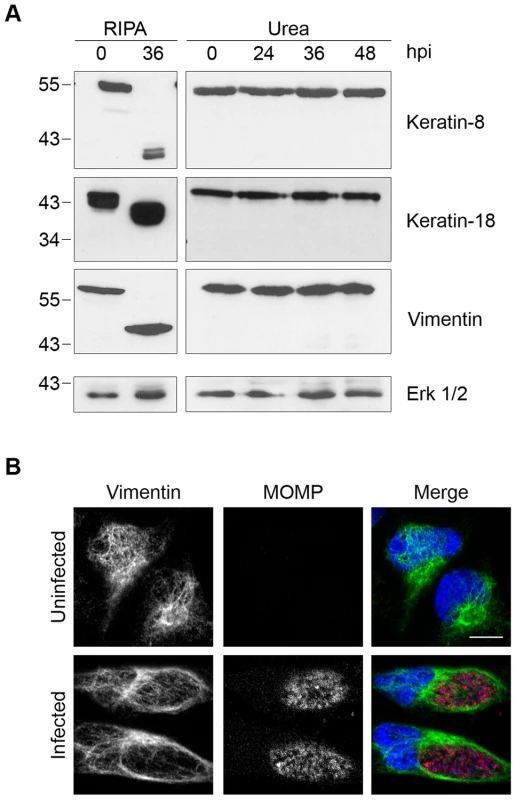
(A) Lysates of uninfected (0 hpi) or infected HeLa cells were prepared in RIPA buffer (left panel) or by direct lysis in 8M urea (right panel) at the indicated times, separated by SDS-PAGE and analyzed by immunoblotting with antibodies to keratin-8, keratin-18, or vimentin. Equal loading for each blot was monitored by blotting for Erk 1/2 (loading control), but only the loading control for keratin-8 is shown. (B) Uninfected and infected HeLa cells at 30 hpi were fixed and stained with antibodies to vimentin (green), the chlamydial major outer membrane protein MOMP (red) and the DNA dye Hoechst 33342 (blue). Representative confocal images are shown. Scale bar, 10 µm. We also examined proteolytic effects of CPAF on four additional host proteins that have been reported to be CPAF substrates. These include the NFκB transcription factor subunit p65/RelA [22], the MHC transcription factor RFX5 [15], [21], the adherens junction protein nectin-1 [23], and the cell cycle protein cyclin B1 [18], [31]. As with the other substrates analyzed, the apparent proteolysis of these four proteins was only observed when Chlamydia-infected cells were lysed in RIPA buffer and not with direct lysis in urea (Figure 4). Taken together, these studies demonstrate that the previously reported proteolysis of 11 published CPAF substrates can be prevented by inhibiting CPAF activity during the processing of Chlamydia-infected cells (Table 1).
Fig. 4. Proteolysis of four additional CPAF substrates is dependent on cell processing. 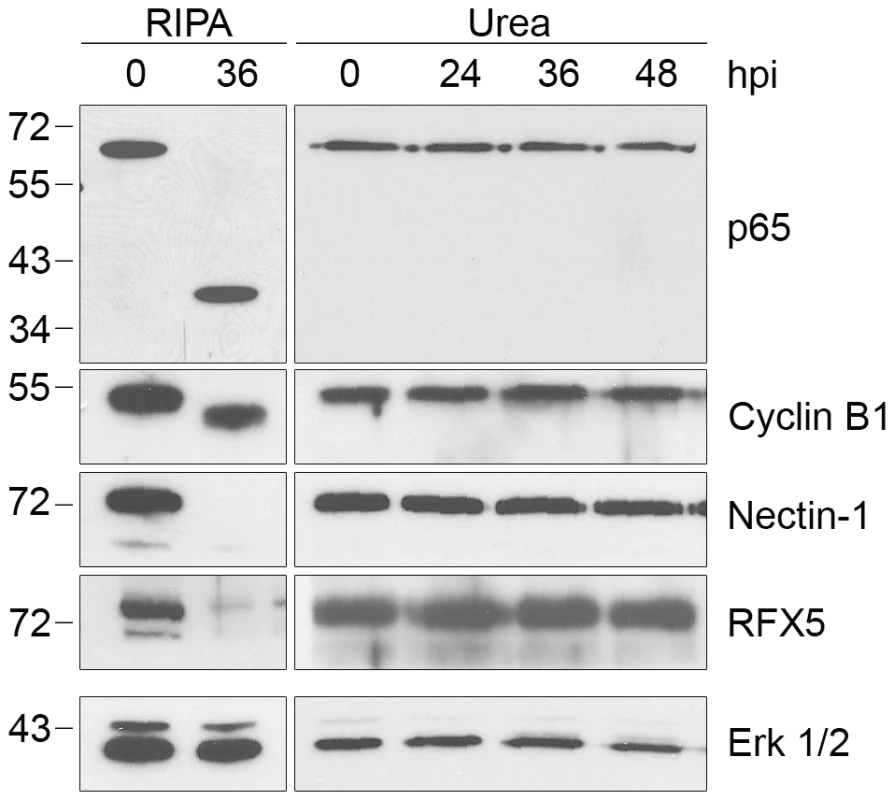
Lysates of uninfected (0 hpi) or infected HeLa cells were prepared in RIPA buffer (left panel) or by direct lysis in 8M urea (right panel) at the indicated times, separated by SDS-PAGE and probed with antibodies to the p65/RelA subunit of NFκB, cyclin B1, nectin-1, or RFX5 as indicated. Equal loading for each blot was monitored by blotting for Erk 1/2, but only the loading control for nectin-1 is shown. Evidence That There Is CPAF Activity in Chlamydia-Infected Cells
Since we did not detect CPAF activity towards reported host substrates when we lysed cells in 8M urea, we examined if CPAF itself was cleaved into its enzymatically-active form in our Chlamydia-infected cells [12], [32]. CPAF is synthesized as a zymogen of 70 kDa that is converted in trans into active N - and C-terminal fragments via an autocatalytic cleavage reaction that requires CPAF proteolytic activity [12], [32]–[35]. Using an antibody that recognizes the C-terminal fragment of CPAF, CPAFc [35], we detected only this cleaved form but not the full-length zymogen in Chlamydia-infected cell lysates prepared with either RIPA buffer or 8M urea (Figure 5). This C-terminal fragment of CPAF accumulated over the course of infection from 24 to 48 hpi (Figure 5). By verifying CPAF as its own substrate, we provide evidence of CPAF catalytic activity in Chlamydia-infected cells even under our most stringent cell processing conditions.
Fig. 5. CPAF is autocatalytically cleaved into its active form in Chlamydia-infected cells. 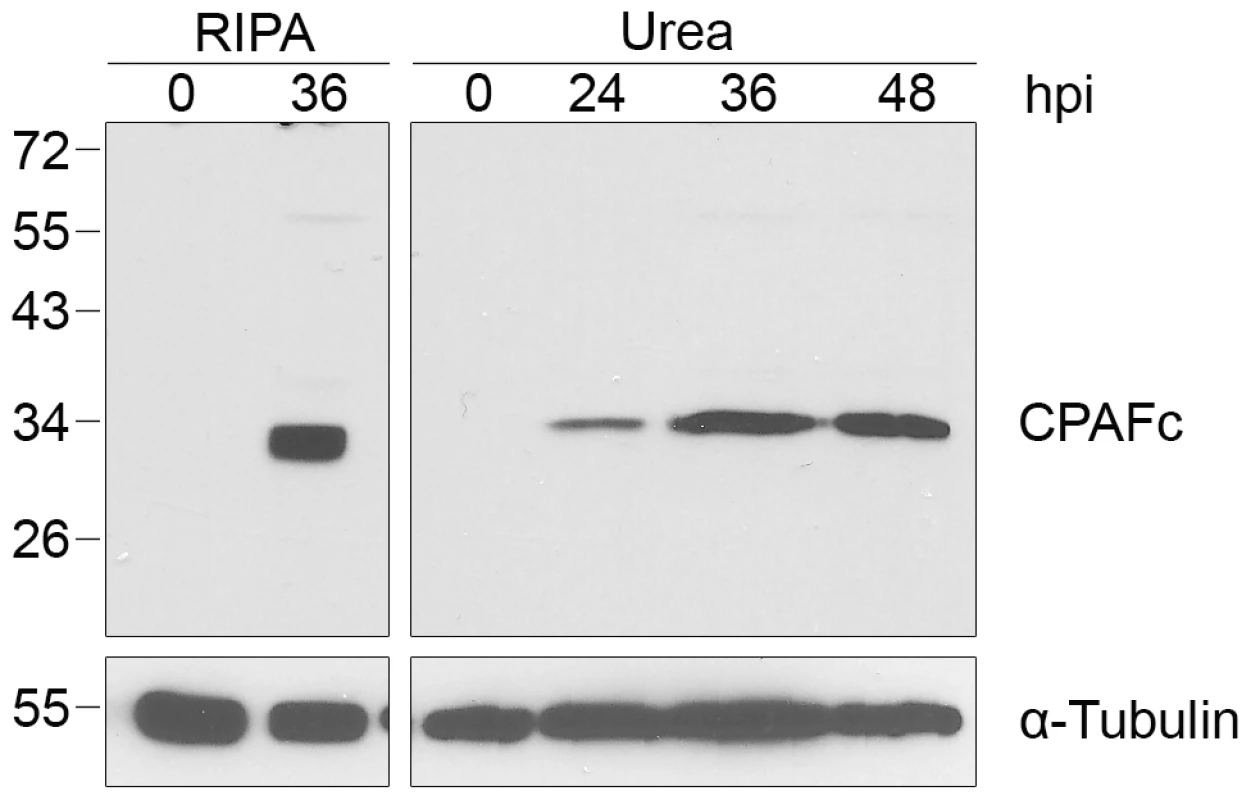
Lysates of uninfected (0 hpi) or infected HeLa cells were prepared in RIPA buffer (left panel) or by direct lysis in 8M urea (right panel) at the indicated times, separated by SDS-PAGE and probed with antibodies to the C-terminal fragment of CPAF (CPAFc) and α-tubulin (loading control). Discussion
We have re-examined the reported proteolysis of specific host proteins by the enzyme CPAF in Chlamydia-infected cells. Specifically, we show that cleavage or degradation of 11 independent host proteins was abrogated by treating cells with the CPAF inhibitor clasto-lactacystin for one hour prior to cell processing or by lysing cells from a monolayer directly in the denaturing agent urea. A major strength of our study is that we can reproduce the reported proteolytic effects of CPAF under standard lysis conditions and then prevent these effects solely by altering the conditions used to process the cells for protein analysis. In addition, there was no detectable CPAF-dependent proteolysis up to 48 hpi, which is many hours after the substrates are reported to be cleaved or degraded [16]–[18], [20], [30], [36]. These surprising results raise questions about the identity of authentic CPAF substrates and the timing and significance of their proteolysis during a Chlamydia infection.
Several lines of evidence have been used to identify putative CPAF substrates. The cleavage or degradation of specific host proteins has been detected by immunoblot analyses of lysates from Chlamydia-infected cells prepared by standard lysis procedures [15]–[16], [18]–[20], [22]–[23]. These proteolysis patterns have been reproduced in vitro by recombinant CPAF [15], [19]–[20], [23] and in vivo by overexpression of CPAF in uninfected cells [16], [18], [22], suggesting that CPAF may be the cognate protease. The involvement of this chlamydial protease in these cleavage reactions has been further supported by data showing that proteolysis of specific substrates can be prevented in vitro by a CPAF inhibitor, such as lactacystin [15], [19], [23]. However, only a few studies have provided evidence of proteolysis in intact infected cells with techniques such as immunofluorescence [23].
How can we reconcile our results with the large body of published data on CPAF and its substrates? Our studies demonstrate that there is CPAF activity in lysates of Chlamydia-infected cells prepared under standard conditions (i.e. lysis of cells in RIPA buffer in the presence of a standard protease inhibitor cocktail on ice). CPAF may remain active in the lysate because it is an atypical serine protease that is not inhibited by a range of protease inhibitors [21], [37]. This enzymatic activity can then cleave or degrade susceptible host proteins in the lysate, even when left on ice, because CPAF is active at 0°C (Figure S1A). When we prevented CPAF activity in lysates obtained from monolayers of Chlamydia-infected cells, we no longer observed proteolysis of these 11 host proteins as late as 48 hpi. These findings lead us to conclude that the published cleavage and degradation of these host proteins in Chlamydia-infected cells are unlikely to have occurred in intact cells but instead are due to in vitro proteolysis by CPAF during cell processing. Our results do not rule out the possibility that there could be a small amount of cleavage or degradation of one or more of these purported substrates below the detection limit of immunoblots. In addition, the specificity with which these proteins are cleaved or degraded by CPAF in vitro suggests that they have the potential to be in vivo CPAF substrates under conditions in a Chlamydia-infected cell that have not yet been elucidated.
These findings emphasize the importance of monitoring CPAF activity during cell processing in order to distinguish between proteolytic activity in vitro and in vivo. Since we did not detect proteolytic effects for any of the 11 published host substrates in this study, the proteolysis of other reported CPAF substrates should be re-examined to verify that they are altered in vivo. However, there is evidence of CPAF enzymatic activity within a Chlamydia-infected cell because CPAF itself was cleaved even though we inhibited CPAF activity during cell harvest and lysis (Figure 5). This cleavage of CPAF is consistent with its autocatalytic processing [34].
The multiple proposed roles of CPAF in mediating host-pathogen interactions during a chlamydial infection merit reappraisal. Our data demonstrate that Chlamydia-induced Golgi reorganization occurs in the absence of detectable golgin-84 cleavage. Also, resistance of Chlamydia-infected cells to apoptosis is unlikely to be caused by the degradation of the pro-apoptotic BH3-only proteins Puma, Bik, and Bim. Similarly, the cage-like vimentin structure surrounding the chlamydial inclusion was present even though vimentin cleavage was not evident. It has been reported that NFκB activation is blocked in Chlamydia-infected cells by cleavage of p65/RelA by either CPAF [22], [38] or another chlamydial protease CT441/Tsp [22], [38]. Our results indicate that this controversy may be moot because we did not detect any appreciable cleavage of p65/RelA. These proposed effects of CPAF have been largely based on the proteolysis of specific host proteins and inferred from the known functions of these proteins in an uninfected cell [18]–[20]. However, there has been little direct evidence showing that CPAF-mediated proteolysis is necessary for the specific host-pathogen interactions during a chlamydial infection. In the absence of detectable CPAF-dependent proteolysis of golgin-84, the three BH3-only proteins, vimentin, and p65/RelA, these host-Chlamydia interactions are probably mediated by alternative mechanisms. For example, it has been proposed that resistance of Chlamydia-infected cells to apoptosis is due to upregulation of the anti-apoptotic factors cIAP-2 and Mcl-1 and not the degradation of BH3-only proteins [39]–[40].
Despite these questions about CPAF, this conserved chlamydial protease is likely to play an important role during the intracellular chlamydial infection. We have shown that CPAF is still cleaved into its active form (Figure 5), suggesting that it can potentially cleave or degrade host targets in an infected cell. Moreover, treatment of infected cells with CPAF inhibitors can prevent normal chlamydial growth and development [16], [37]. However, in light of the current findings, the exact role and timing of CPAF activity in a Chlamydia-infected cell remains to be determined.
Our studies on CPAF have general implications for the analysis of modification enzymes such as kinases or proteases that may retain activity under standard cell processing conditions. This concern is most obvious for proteases that are resistant to standard protease inhibitor cocktails. For example, hundreds of caspase substrates have been identified, but many are considered “bystander” substrates that are not biologically relevant even though they contain specific cleavage sites for these proteases [41]. Our findings underscore the importance of inhibiting the relevant enzymatic activity during cell processing and verifying that this activity is actually blocked.
This study highlights the need to re-evaluate the Chlamydia literature on CPAF. General concerns have been raised about continued CPAF activity during cell lysis [11], [31], but the present study shows that at least 11 of 16 previously identified host CPAF substrates are not observed to be altered during the normal course of infection. Biochemical reports about the activation of CPAF [12], [32]–[35], and studies characterizing antibody responses to CPAF [42]–[45], are likely to be unaffected by our findings. However, the presence of CPAF activity under standard lysis conditions calls for a reassessment of prior studies on CPAF substrates and the role of CPAF in Chlamydia-infected cells.
Materials and Methods
Antibodies
The following antibodies were used in this study: rabbit anti-Bim, rabbit anti-Puma, mouse anti-keratin-8, mouse anti-keratin-18, mouse anti-α-tubulin, and mouse anti-vimentin (all from Sigma-Aldrich); rabbit anti-cyclin B1, mouse anti-nectin-1, mouse anti-p65/RelA and bovine anti-sheep-HRP (all from Santa Cruz Biotechnology); rabbit anti-Bik, rabbit anti-vimentin, and mouse anti-Erk 1/2 (all from Cell Signaling Technology); goat anti-mouse-HRP and goat anti-rabbit-HRP (both from Jackson ImmunoResearch Laboratories); mouse anti-caspase-3 (BD Biosciences); rabbit anti-RFX5 (Rockland Immunochemicals); Alexa Fluor 488 or Alexa Fluor 594-tagged goat anti-mouse or goat anti-rabbit antibodies (Molecular Probes/Invitrogen); mouse anti-CPAFc (generous gift from Dr. Guangming Zhong, University of Texas Health Science Center at San Antonio); sheep anti-golgin-84 (kindly provided by Dr. Martin Lowe, University of Manchester); rabbit anti-α-mannosidase II (kindly provided by Dr. Kelley Moremen, University of Georgia); mouse anti-MOMP (VD4 epitope of the major outer membrane protein from C. trachomatis serovar E [BOUR]) (kind gift from Dr. Ellena Peterson, UC Irvine).
Cell Culture
HeLa cells (ATCC) were grown in 6-well dishes in Advanced DMEM (4.5 g glucose/L) (Invitrogen) supplemented with 2% fetal bovine serum (FBS) (Hyclone/Thermo Fisher) and 2 mM GlutaMAX-I (Invitrogen). HEK 293T cells and retinal pigment epithelial (hTERT RPE-1) cells (both from ATCC) were cultured in DMEM (4.5 g glucose/L) (Invitrogen) supplemented with 10% fetal bovine serum. All cell lines were grown in 5% CO2 at 37°C and screened for Mycoplasma contamination by PCR [46].
Chlamydia Infections
Cell monolayers were infected with C. trachomatis serovar L2 (L2/434/Bu), LGV biovar, at a multiplicity of infection (MOI) of 3 in sucrose-phosphate-glutamic acid (SPG). In parallel, uninfected control experiments were performed as mock infections in SPG alone. Infections were carried out by centrifugation at 700× g in a Sorvall Legend Mach 1.6R centrifuge for 1 hr at room temperature. After centrifugation, the inoculum was replaced by fresh cell culture medium and monolayers were incubated at 37°C and 5% CO2. Chlamydial EBs (elementary bodies) were verified to be free of Mycoplasma contamination by PCR [46].
Cell Processing and Immunoblotting
Lysis in RIPA buffer: Cells were harvested by trypsinization for 3–5 min at 37°C and the trypsinized cells were transferred to a 15 mL conical tube on ice. The dish was washed twice with 1×PBS and the washes were added to the 15 mL conical tube to collect any remaining cells. The cells were pelleted by centrifugation at 1500 rpm for 3 min at 4°C and lysed on ice for 10 min in RIPA buffer (50 mM Tris [pH 7.5], 150 mM NaCl, 0.1% SDS, 0.5% sodium deoxycholate, 1% NP-40) supplemented with protease inhibitors (2 mM pepstatin, 150 mM aprotinin [both from MP Biochemicals], 1 mM leupeptin [Calbiochem], 1 mM PMSF [Acros]). The cells were resuspended by pipetting up and down in approximately 1 mL of ice-cold lysis buffer per 5×106 cells. Lysates were cleared by centrifugation at 13,000× g for 10 min at 4°C and protein concentrations were determined by Bradford assay (BioRad).
Lysis in urea: A solution of 8M urea was supplemented with 325 U/mL of Benzonase Nuclease (Sigma-Aldrich) and added directly to cell monolayers at a volume of 1 mL per 6-well dish for 10 min on ice. Lysates were then pooled and protein concentrations were determined by the DC protein assay (BioRad).
Cell lysates were diluted into Laemmli sample buffer (50 mM Tris-HCl [pH 6.8], 10% glycerol, 2% SDS, 1% 2-mercaptoethanol, 0.1% bromophenol blue). Samples containing equal amounts of protein were loaded and resolved by SDS-PAGE. Proteins were transferred onto nitrocellulose membranes and subjected to immunoblot analysis (Table S1) with enhanced chemiluminescence (90 mM p-Courmaric acid, 250 mM 3-Aminophthalhydrazide, 100 mM Tris-HCl [pH 8.5]).
Clasto-lactacystin treatment: 150 µM of clasto-lactacystin β-lactone (Cayman Chemical), dissolved in methyl acetate, was added to the cell culture medium for 1 hr prior to cell processing. For example, samples of Chlamydia-infected cells at 36 hpi were treated with clasto-lactacystin at 35 hpi for 1 hr and then processed. In parallel control experiments, methyl acetate as the solvent was added to the culture medium.
Immunofluorescence
Cells grown on glass coverslips were fixed in 4% formaldehyde for 10 min at room temperature and blocked in 5% blocking buffer (0.1% Triton X-100, 5% FBS in PBS) or TBS-BSA (0.1% Tween-20, 5% BSA in TBS) for 1 hr. Cells were incubated with primary antibodies for 1 hr at room temperature or overnight at 4°C followed by Alexa-fluorochrome-conjugated secondary antibodies for 30 min at room temperature. Host and chlamydial DNA were stained with Hoechst 33342 (Molecular Probes/Invitrogen). Coverslips were mounted onto glass slides with gelvatol [47] and imaged by confocal microscopy on a Nikon Eclipse Ti-U inverted microscope fitted with a Nikon D-Eclipse confocal laser assembly and a D-Eclipse C1 controller (Nikon). Images were acquired using the Nikon EZ-C1 program and analyzed using Nikon NIS Elements and Adobe Photoshop.
Cell-free Degradation Assays
Chlamydia-infected HeLa cells at various times in the infection were either left untreated, mock-treated with methyl acetate, or treated for 1 hr with 150 µM of clasto-lactacystin, and then lysed in RIPA buffer as described above. 3.5 µg of Chlamydia-infected HeLa cell lysate, as the source of CPAF, was incubated with 27 µg of uninfected HeLa cell lysate, as the source of host protein substrates, at 37°C for 1 hr in CPAF reaction buffer (25 mM Tris [pH 8.0], 150 mM NaCl, 3 mM DTT). For reactions performed at 0°C, 54 µg of Chlamydia-infected HeLa cell lysate from 36 hpi was incubated with 54 µg of uninfected HeLa cell lysate on ice for 10 or 30 min. Reactions were terminated by adding Laemmli sample buffer and boiling for 5 min.
Apoptosis Induction Assay
Uninfected or Chlamydia-infected HeLa cells (MOI of 3, at 24 hpi) were incubated with 1 µM staurosporine in tissue culture medium for 3 hrs. Lysates of the cell monolayers were prepared by direct lysis in 8M urea as previously described, separated by SDS-PAGE and analyzed by immunoblotting with antibodies to caspase-3.
Supporting Information
Zdroje
1. CDC 2011 Summary of notifiable diseases: United States, 2009. MMWR Morb Mortal Wkly Rep 58 1 100
2. SchachterJ 1999 Infection and disease epidemiology. StephensRS Chlamydia: Intracellular Biology, Pathogenesis, and Immunity Washington, D.C. American Society for Microbiology 139 169 editor pp
3. BurtonMJMabeyDC 2009 The global burden of trachoma: a review. PLoS Negl Trop Dis 3 e460
4. BlasiFTarsiaPAlibertiS 2009 Chlamydophila pneumoniae. Clin Microbiol Infect 15 29 35
5. CarabeoRAMeadDJHackstadtT 2003 Golgi-dependent transport of cholesterol to the Chlamydia trachomatis inclusion. Proc Natl Acad Sci U S A 100 6771 6776
6. HackstadtTRockeyDHeinzenRScidmoreM 1996 Chlamydia trachomatis interrupts an exocytic pathway to acquire endogenously synthesized sphingomyelin in transit from the Golgi apparatus to the plasma membrane. EMBO J 15 964 977
7. HackstadtTScidmoreMARockeyDD 1995 Lipid metabolism in Chlamydia trachomatis-infected cells: directed trafficking of Golgi-derived sphingolipids to the chlamydial inclusion. Proc Natl Acad Sci U S A 92 4877 4881
8. FanTLuHHuHShiLMcClartyGA 1998 Inhibition of apoptosis in Chlamydia-infected cells: blockade of mitochondrial cytochrome c release and caspase activation. J Exp Med 187 487 496
9. FischerSFSchwarzCVierJHackerG 2001 Characterization of antiapoptotic activities of Chlamydia pneumoniae in human cells. Infect Immun 69 7121 7129
10. RajalingamKAl-YounesHMullerAMeyerTFSzczepekAJ 2001 Epithelial cells infected with Chlamydophila pneumoniae (Chlamydia pneumoniae) are resistant to apoptosis. Infect Immun 69 7880 7888
11. ZhongG 2009 Killing me softly: chlamydial use of proteolysis for evading host defenses. Trends Microbiol 17 467 474
12. HuangZFengYChenDWuXHuangS 2008 Structural basis for activation and inhibition of the secreted Chlamydia protease CPAF. Cell Host Microbe 4 529 542
13. DongFZhongYArulanandamBZhongG 2005b Production of a proteolytically active protein, chlamydial protease/proteasome-like activity factor, by five different Chlamydia species. Infect Immun 73 1868 1872
14. HornMCollingroASchmitz-EsserSBeierCLPurkholdU 2004 Illuminating the evolutionary history of chlamydiae. Science 304 728 730
15. ZhongGFanPJiHDongFHuangY 2001 Identification of a chlamydial protease-like activity factor responsible for the degradation of host transcription factors. J Exp Med 193 935 942
16. ChristianJGHeymannJPaschenSAVierJSchauenburgL 2011 Targeting of a chlamydial protease impedes intracellular bacterial growth. PLoS Pathog 7 e1002283
17. HeuerDRejman LipinskiAMachuyNKarlasAWehrensA 2009 Chlamydia causes fragmentation of the Golgi compartment to ensure reproduction. Nature 457 731 735
18. PaschenSAChristianJGVierJSchmidtFWalchA 2008 Cytopathicity of Chlamydia is largely reproduced by expression of a single chlamydial protease. J Cell Biol 182 117 127
19. PirbhaiMDongFZhongYPanKZZhongG 2006 The secreted protease factor CPAF is responsible for degrading pro-apoptotic BH3-only proteins in Chlamydia trachomatis-infected cells. J Biol Chem 281 31495 31501
20. KumarYValdiviaRH 2008 Actin and intermediate filaments stabilize the Chlamydia trachomatis vacuole by forming dynamic structural scaffolds. Cell Host Microbe 4 159 169
21. ZhongGLiuLFanTFanPJiH 2000 Degradation of transcription factor RFX5 during the inhibition of both constitutive and interferon gamma-inducible major histocompatibility complex class I expression in Chlamydia-infected cells. J Exp Med 191 1525 1534
22. ChristianJVierJPaschenSAHackerG 2010 Cleavage of the NF-kappaB family protein p65/RelA by the chlamydial protease-like activity factor (CPAF) impairs proinflammatory signaling in cells infected with Chlamydiae. J Biol Chem 285 41320 41327
23. SunJSchoborgRV 2009 The host adherens junction molecule nectin-1 is degraded by chlamydial protease-like activity factor (CPAF) in Chlamydia trachomatis-infected genital epithelial cells. Microbes Infect 11 12 19
24. BellandRJZhongGCraneDDHoganDSturdevantD 2003 Genomic transcriptional profiling of the developmental cycle of Chlamydia trachomatis. Proc Natl Acad Sci U S A 100 8478 8483
25. HoldenPHortonWA 2009 Crude subcellular fractionation of cultured mammalian cell lines. BMC Res Notes 2 243
26. DickLRCruikshankAADestreeATGrenierLMcCormackTA 1997 Mechanistic studies on the inactivation of the proteasome by lactacystin in cultured cells. J Biol Chem 272 182 188
27. FenteanyGStandaertRFLaneWSChoiSCoreyEJ 1995 Inhibition of proteasome activities and subunit-specific amino-terminal threonine modification by lactacystin. Science 268 726 731
28. RajagopalanKVFridovichIHandlerP 1961 Competitive inhibition of enzyme activity by urea. J Biol Chem 236 1059 1065
29. TakizawaPAYucelJKVeitBFaulknerDJDeerinckT 1993 Complete vesiculation of Golgi membranes and inhibition of protein transport by a novel sea sponge metabolite, ilimaquinone. Cell 73 1079 1090
30. DongFSuHHuangYZhongYZhongG 2004c Cleavage of host keratin 8 by a Chlamydia-secreted protease. Infect Immun 72 3863 3868
31. BalsaraZRMisaghiSLafaveJNStarnbachMN 2006 Chlamydia trachomatis infection induces cleavage of the mitotic cyclin B1. Infect Immun 74 5602 5608
32. DongFPirbhaiMZhongYZhongG 2004a Cleavage-dependent activation of a Chlamydia-secreted protease. Mol Microbiol 52 1487 1494
33. ChenDChaiJHartPJZhongG 2009 Identifying catalytic residues in CPAF, a Chlamydia-secreted protease. Arch Biochem Biophys 485 16 23
34. ChenDLeiLFloresRHuangZWuZ 2010 Autoprocessing and self-activation of the secreted protease CPAF in Chlamydia-infected cells. Microb Pathog 49 164 173
35. DongFSharmaJXiaoYZhongYZhongG 2004b Intramolecular dimerization is required for the Chlamydia-secreted protease CPAF to degrade host transcriptional factors. Infect Immun 72 3869 3875
36. DongFPirbhaiMXiaoYZhongYWuY 2005a Degradation of the proapoptotic proteins Bik, Puma, and Bim with Bcl-2 domain 3 homology in Chlamydia trachomatis-infected cells. Infect Immun 73 1861 1864
37. JorgensenIBednarMMAminVDavisBKTingJP 2011 The Chlamydia protease CPAF regulates host and bacterial proteins to maintain pathogen vacuole integrity and promote virulence. Cell Host Microbe 10 21 32
38. LadSPLiJda Silva CorreiaJPanQGadwalS 2007 Cleavage of p65/RelA of the NF-kappaB pathway by Chlamydia. Proc Natl Acad Sci U S A 104 2933 2938
39. RajalingamKSharmaMLohmannCOswaldMThieckO 2008 Mcl-1 is a key regulator of apoptosis resistance in Chlamydia trachomatis-infected cells. PLoS One 3 e3102
40. RajalingamKSharmaMPalandNHurwitzRThieckO 2006 IAP-IAP complexes required for apoptosis resistance of C. trachomatis-infected cells. PLoS Pathog 2 e114
41. FischerUJanickeRUSchulze-OsthoffK 2003 Many cuts to ruin: a comprehensive update of caspase substrates. Cell Death Differ 10 76 100
42. CongYJupelliMGuentzelMNZhongGMurthyAK 2007 Intranasal immunization with chlamydial protease-like activity factor and CpG deoxynucleotides enhances protective immunity against genital Chlamydia muridarum infection. Vaccine 25 3773 3780
43. MurthyAKCongYMurpheyCGuentzelMNForsthuberTG 2006 Chlamydial protease-like activity factor induces protective immunity against genital chlamydial infection in transgenic mice that express the human HLA-DR4 allele. Infect Immun 74 6722 6729
44. SharmaJBosnicAMPiperJMZhongG 2004 Human antibody responses to a Chlamydia-secreted protease factor. Infect Immun 72 7164 7171
45. SkworTKandelRPBasraviSKhanASharmaB 2010 Characterization of humoral immune responses to chlamydial HSP60, CPAF, and CT795 in inflammatory and severe trachoma. Invest Ophthalmol Vis Sci 51 5128 5136
46. OssewaardeJde VriesABestebroerTAnguloA 1996 Application of a Mycoplasma group-specific PCR for monitoring decontamination of Mycoplasma-infected Chlamydia sp. strains. Appl Environ Microbiol 62 328 331
47. HarlowELaneD 1999 Using antibodies: a laboratory manual Cold Spring Harbor Laboratory Press pp 495
48. FischerSFVierJKirschnekSKlosAHessS 2004 Chlamydia inhibit host cell apoptosis by degradation of proapoptotic BH3-only proteins. J Exp Med 200 905 916
49. SunJKintnerJSchoborgRV 2008 The host adherens junction molecule nectin-1 is downregulated in Chlamydia trachomatis-infected genital epithelial cells. Microbiology 154 1290 1299
50. ZhongGFanTLiuL 1999 Chlamydia inhibits interferon gamma-inducible major histocompatibility complex class II expression by degradation of upstream stimulatory factor 1. J Exp Med 189 1931 1938
51. KawanaKQuayleAJFicarraMIbanaJAShenL 2007 CD1d degradation in Chlamydia trachomatis-infected epithelial cells is the result of both cellular and chlamydial proteasomal activity. J Biol Chem 282 7368 7375
52. YuHSchwarzerKForsterMKniemeyerOForsbach-BirkV 2010 Role of high-mobility group box 1 protein and poly(ADP-ribose) polymerase 1 degradation in Chlamydia trachomatis-induced cytopathicity. Infect Immun 78 3288 3297
53. RuppJGieffersJKlingerMvan ZandbergenGWraseR 2007 Chlamydia pneumoniae directly interferes with HIF-1alpha stabilization in human host cells. Cell Microbiol 9 2181 2191
Štítky
Hygiena a epidemiologie Infekční lékařství Laboratoř
Článek vyšel v časopisePLOS Pathogens
Nejčtenější tento týden
2012 Číslo 8- Jak souvisí postcovidový syndrom s poškozením mozku?
- Parazitičtí červi v terapii Crohnovy choroby a dalších zánětlivých autoimunitních onemocnění
- Vakcíny proti klíšťové encefalitidě
- Kdy je nejlepší očkovat
- Možné vedlejší účinky očkování
-
Všechny články tohoto čísla
- Exon Level Transcriptomic Profiling of HIV-1-Infected CD4 T Cells Reveals Virus-Induced Genes and Host Environment Favorable for Viral Replication
- Five Mechanisms of Manipulation by Bacterial Effectors: A Ubiquitous Theme
- Nonhuman Primate Models for HIV Cure Research
- The Ebola Virus Glycoprotein Contributes to but Is Not Sufficient for Virulence
- Trichomonosis, a Common Curable STI, and Prostate Carcinogenesis—A Proposed Molecular Mechanism
- Host Defense and Tolerance: Unique Challenges in the Placenta
- CPAF: A Chlamydial Protease in Search of an Authentic Substrate
- Small Protease Sensitive Oligomers of PrP in Distinct Human Prions Determine Conversion Rate of PrP
- Invariant NKT Cells: Regulation and Function during Viral Infection
- Human Monoclonal Antibody HCV1 Effectively Prevents and Treats HCV Infection in Chimpanzees
- Chemokine Receptor Ccr1 Drives Neutrophil-Mediated Kidney Immunopathology and Mortality in Invasive Candidiasis
- Phagocyte Responses to Protozoan Infection and How Meets the Challenge
- Telomere Length Affects the Frequency and Mechanism of Antigenic Variation in
- A Biofilm-Induced Pathway for Matrix Glucan Delivery: Implications for Drug Resistance
- The HSV-1 Exonuclease, UL12, Stimulates Recombination by a Single Strand Annealing Mechanism
- Inhibition of Fatty Acid Synthase (Fas2) Induces Mitochondrial Cell Death in Serum
- Interferon-alpha Subtype 11 Activates NK Cells and Enables Control of Retroviral Infection
- Transposon-mediated Chromosomal Integration of Transgenes in the Parasitic Nematode and Establishment of Stable Transgenic Lines
- Structural and Biochemical Basis for Development of Influenza Virus Inhibitors Targeting the PA Endonuclease
- Upregulation of Retinal Dehydrogenase 2 in Alternatively Activated Macrophages during Retinoid-dependent Type-2 Immunity to Helminth Infection in Mice
- Measles Immune Suppression: Lessons from the Macaque Model
- Fungi and the Rise of Mammals
- Bacterial Cell Surface Heterogeneity: A Pathogen's Disguise
- Cytoplasmic Entry Induces Fetal Wastage by Disrupting Maternal Foxp3 Regulatory T Cell-Sustained Fetal Tolerance
- PLOS Pathogens
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Invariant NKT Cells: Regulation and Function during Viral Infection
- Host Defense and Tolerance: Unique Challenges in the Placenta
- Nonhuman Primate Models for HIV Cure Research
- Exon Level Transcriptomic Profiling of HIV-1-Infected CD4 T Cells Reveals Virus-Induced Genes and Host Environment Favorable for Viral Replication
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



