-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
Top novinky
ReklamaWall Teichoic Acids of Limit Recognition by the Drosophila Peptidoglycan Recognition Protein-SA to Promote Pathogenicity
The cell wall of Gram-positive bacteria is a complex network of surface proteins, capsular polysaccharides and wall teichoic acids (WTA) covalently linked to Peptidoglycan (PG). The absence of WTA has been associated with a reduced pathogenicity of Staphylococcus aureus (S. aureus). Here, we assessed whether this was due to increased detection of PG, an important target of innate immune receptors. Antibiotic-mediated or genetic inhibition of WTA production in S. aureus led to increased binding of the non-lytic PG Recognition Protein-SA (PGRP-SA), and this was associated with a reduction in host susceptibility to infection. Moreover, PGRP-SD, another innate sensor required to control wild type S. aureus infection, became redundant. Our data imply that by using WTA to limit access of innate immune receptors to PG, under-detected bacteria are able to establish an infection and ultimately overwhelm the host. We propose that different PGRPs work in concert to counter this strategy.
Published in the journal: . PLoS Pathog 7(12): e32767. doi:10.1371/journal.ppat.1002421
Category: Research Article
doi: https://doi.org/10.1371/journal.ppat.1002421Summary
The cell wall of Gram-positive bacteria is a complex network of surface proteins, capsular polysaccharides and wall teichoic acids (WTA) covalently linked to Peptidoglycan (PG). The absence of WTA has been associated with a reduced pathogenicity of Staphylococcus aureus (S. aureus). Here, we assessed whether this was due to increased detection of PG, an important target of innate immune receptors. Antibiotic-mediated or genetic inhibition of WTA production in S. aureus led to increased binding of the non-lytic PG Recognition Protein-SA (PGRP-SA), and this was associated with a reduction in host susceptibility to infection. Moreover, PGRP-SD, another innate sensor required to control wild type S. aureus infection, became redundant. Our data imply that by using WTA to limit access of innate immune receptors to PG, under-detected bacteria are able to establish an infection and ultimately overwhelm the host. We propose that different PGRPs work in concert to counter this strategy.
Introduction
The complex cell surface of bacteria has been directly or indirectly associated with different strategies that bacterial pathogens use to interact with the host. These include acquisition of specific adhesion factors, formation of biofilms, adaptation to an intracellular environment, production of a protective capsular polysaccharide or evasion of innate immune defences (e.g. lysozyme) [1]. The host counters these strategies by targeting conserved molecules (pathogen associated molecular patterns or PAMPs), unique in bacteria, that are either present at the bacterial surface or are released by bacteria as they attempt to establish infection. Bacterial PAMPs include Peptidoglycan (PG), a heterogeneous polymer of glycan chains cross-linked by short peptides of variable length and amino acid composition [2]. Although PG recognition is essential to trigger an inflammatory response, this macromolecule may not be easily accessible for recognition at the surface of bacteria.
In Gram-positive bacteria, PG is buried within a complex cell surface consisting of different molecules [3]–[5]. Such molecules include surface proteins, covalently linked or tightly associated with PG, capsular polysaccharides, usually required for the ability of different bacteria to cause disease [6] and wall teichoic acids (WTA), phosphate-rich glycopolymers involved in the resistance of bacteria to environmental stress and regulation of bacterial division [7]. It is not clear therefore, how the host would be able to sense bacterial PG buried within such complex structures. One hypothesis is that the innate immune system recognises soluble PG fragments that are released from the bacterial cell surface through the activity of enzymes produced by bacteria (such as autolysins) or by the host (such as lysozyme) [2], [8]. However, certain bacteria have the ability to modify their PG, turning it more resistant to the action of such enzymes [9], thus preventing the release of small soluble fragments capable of triggering an innate immune response in the host. This may be the case for Listeria monocytogenes that has the ability to de-N-acetylate its PG allowing them to survive the action of lysozyme and evade the host innate immune system [10]. Another hypothesis is that the components of the host innate immune system are able to bind directly to PG present within the bacterial cell surface. As discussed earlier, PG is decorated with a variety of large molecules that may sterically block access of host receptors to the underlying PG. In Gram-positive bacteria, cell wall glycopolymers, including WTA may play this role [1]. The role of WTA protecting the PG from recognition would have important implications regarding the onset of infection by major human pathogens such as Staphylococcus aureus (S. aureus) [1]. Recently, it has been shown that different components, present at the cell wall of S. aureus bacteria, may determine the survival of infected Drosophila. Specifically, S. aureus strains impaired in the expression of enzymes involved with the metabolism of cell wall components were unable to kill flies [11]. Moreover, it has been proposed that D-alanylation of the WTA produced by S. aureus may inhibit the recognition of PG by host receptors. This inhibitory effect was observed in vitro not only when WTA was covalently attached to polymeric PG but, surprisingly, also when WTA was covalently attached to monomeric PG [12].
The fruit fly Drosophila melanogaster recognises Gram-positive bacteria by either direct binding to PG or its smallest components [13]. Based on in vitro data [14] and infection studies of mutants [14], [15], the current working hypothesis is that a flexible system of pattern recognition receptors (PRRs) can be deployed by the host immune system to detect Lysine-type PG from different Gram-positive bacterial pathogens. Two Peptidoglycan Recognition Proteins (PGRPs), namely PGRP-SA and PGRP-SD are major components of this system [15], [16]. Depending on the bacterium, each, or both of these PGRPs – along with Gram-Negative Binding Protein1 (GNBP1) [17] – interacts with PG and activate a downstream proteolytic cascade, which culminates in Toll receptor signalling. The signal reaches the cytoplasmic NF-κB/I-κB complex via a receptor/adaptor complex comprising dMyD88, Tube and the IRAK homologue Pelle. At that point the I-κB homologue Cactus is phosphorylated and targeted for degradation while the NF-κB homologue Dif is free to enter the nucleus of host cells and regulate target genes [18]. Prominent among these genes, is a group of potent antimicrobial peptides (AMPs), which are synthesised by the fat body and secreted into the haemolymph. An AMP frequently used as a read-out for the Toll pathway is Drosomycin (Drs). AMPs and local melanization, along with the phagocytic activity of haemocytes constitute respectively the humoral and cellular arm of the fruit fly response to infection [18].
Here, we report for the first time that Drosophila PGRP-SA, a non-lytic PGRP was able to bind intact live bacteria in vivo. Access to PG was limited by the presence of WTA: binding of PGRP-SA to various live Gram-positive bacteria was minimal, but binding to purified PG, stripped of covalent modifications (including WTA) was far greater. Through inhibiting WTA synthesis, either by the addition of an antibiotic or genetically, we were able to potentiate detection of these bacteria by PGRP-SA. For S. aureus, this correlated with a reduced ability of the bacteria to proliferate within the host, and a reduced susceptibility of the host to infection in a PGRP-SA/GNBP1 dependent manner. We also observed that PGRP-SD, essential for sensing wild type S. aureus, became redundant as WTA levels were reduced. Overall, our results suggest that WTA may be part of a general mechanism used by Gram-positive bacteria, which limits the access of innate receptors to PG, thereby enabling bacteria to evade detection and establish infection.
Results
To address the question of whether Gram-positive bacteria counter host recognition by limiting access of innate sensors to PG, we constructed a fluorescent derivative of the fruit fly Lys-type PG receptor, PGRP-SA (mCherry-PGRP-SA). This construct and an untagged version (rPGRP-SA) were expressed in Escherichia coli and the resulting proteins were purified. As shown in the supplementary material (Figure S1A), injection of mCherry-PGRP-SA, or rPGRP-SA, into PGRP-SA deficient flies restored Drs-GFP production induced by infection with Micrococcus luteus (M. luteus). Endogenous Drs expression was also restored as confirmed by qPCR (Figure S1B). These observations were consistent with our previous results when using a recombinant PGRP-SA expressed in the lepidopteran cell line Sf9 [19]. Taken together, these results showed that the fluorescently tagged PGRP-SA and the untagged versions are functional and capable of restoring an innate immune response in PGRP-SA deficient flies.
Initially, we used rPGRP-SA and mCherry-PGRP-SA in co-precipitation experiments in order to study binding to PG from different Gram-positive bacteria. Both bound with similar affinity to PG purified from M. luteus, Enterococcus faecalis (E. faecalis), and S. aureus (data not shown and Figure 1A, respectively). For details of PG composition of these bacteria see Figure S2. Importantly, this indicated that the mCherry-tag appeared not to interfere with PGRP-SA binding, and thus, demonstrated that both proteins were able to bind Lys-type PG of different composition. We therefore assessed in vitro, the binding of mCherry-PGRP-SA to the surface of live bacteria harvested during exponential growth phase. Notably, the binding of the recombinant protein to live bacteria exhibited a range of different affinities in contrast to their respective purified PG. Binding to live E. faecalis and S. aureus was significantly reduced, when compared to binding to M. luteus (Figure 1B). However, the binding levels of PGRP-SA to the purified PG from these bacteria were similar (Figure 1A). We also noticed that while mCherry-PGRP-SA was capable of binding the entire surface of M. luteus cells, it bound at specific sites at the surface of S. aureus cells, similar to what has been described recently for mammalian bactericidal PGRPs [20]. These results suggested that although the three types of bacterial PG were similarly recognized by PGRP-SA, the presence of other components found at the surface of live bacteria might have prevented PGRP-SA from finding its PG ligand.
Fig. 1. Differential binding of PGRP-SA to the surface of live Gram-positive bacteria. 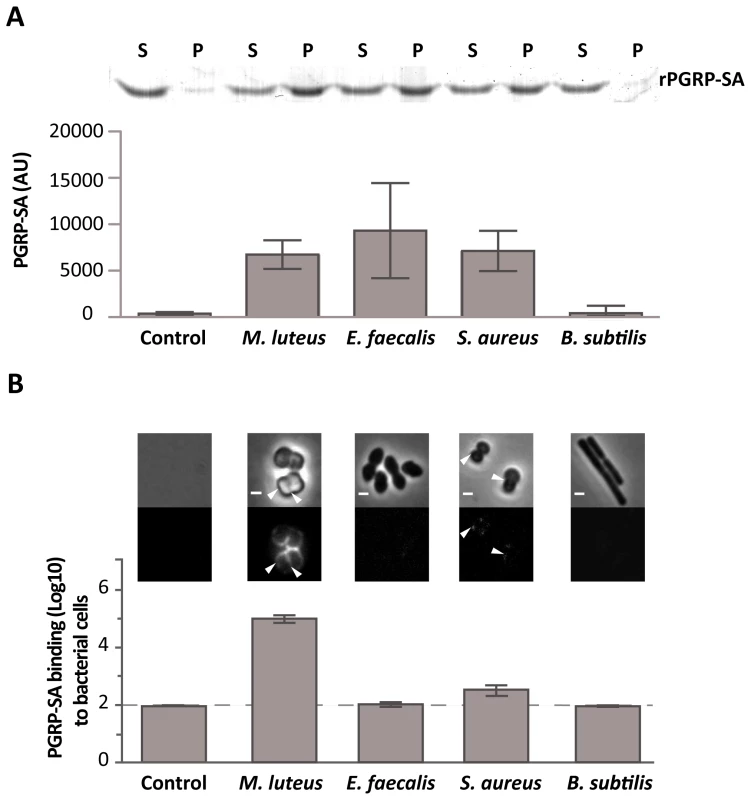
(A) PGRP-SA and PG co-precipitation assay. Lys-type PG from M. luteus, E. faecalis, S. aureus, and DAP-type PG from B. subtilis (this acts as a negative substrate control for PGRP-SA binding, which recognizes Lys-type PG), was incubated with rPGRP-SA for 30 minutes. Unbound rPGRP-SA remained in the supernatant fraction upon centrifugation (S). rPGRP-SA bound to the insoluble PG was co-precipitated and found in the pellet fraction (P). Quantified data (performed using ImageJ software) was plotted as mean values with 95% confidence limits: very little co-precipitation of rPGRP-SA occurred in the absence of PG (labelled Control) or in the presence of B. subtilis DAP-type PG; however, PGRP-SA was co-precipitated similarly (One-way ANOVA, P>0.05) and at higher levels with the PG from M. luteus, E. faecalis, or S. aureus. The data shown (mean with 95% confidence intervals) was obtained from 4 independent co-precipitation experiments. (B) mCherry-PGRP-SA was incubated with bacteria cells harvested in exponential phase, washed with PBS and visualized using fluorescence microscopy. Grey panels are phase-contrast images of bacterial cells (white scale bar represents 1 µm), and black panels mCherry-PGRP-SA binding: white arrowheads highlight binding to the lateral cell surface or the region of cell division. The total fluorescence of mCherry-PGRP-SA bound to a bacterium (covering all lateral and cell division regions, and including background) was quantified for each species (n = 50), and represented as the median (with 25% and 75% inter-quartile range). Dashed-line indicates the level of the background signal, control. Kruskal-Wallis analysis with Dunn's multiple comparison post-test did not reveal significant differences (P>0.05) between mCherry-PGRP-SA binding to E. faecalis and B. subtilis, which were indistinguishable from the control. However, the protein bound more to S. aureus and M. luteus relative to the control, with the latter exhibiting highest binding (P<0.05 in all cases). The cell surface of a Gram-positive bacterium is a complex structure consisting of a thick layer of PG, surface proteins and glycopolymers such as capsular polysaccharides and WTA. As previous studies had shown that certain PG-binding proteins, such as bacterial autolysins, have a higher affinity for the surface of bacterial strains lacking WTA [21]–[23], it was decided to investigate whether presence of WTA could be preventing PGRP-SA from binding to the surface of live bacteria. Further support for the choice of WTA came from the fact that different Gram-positive bacteria can produce WTA with a variable composition [24]–[26]. M. luteus, for which mCherry-PGRP-SA displayed the highest affinity, does not produce WTA [24], [27], (Figure S2C). To test whether WTA mediated the differential binding of PGRP-SA, we cultured bacteria in the presence of tunicamycin, thereby inhibiting their ability to synthesize WTA. At lower, sub-inhibitory concentrations as those used in this study, tunicamycin specifically inhibits TagO [28]: a glycosyltransferase that specifically localizes to the division septum of S. aureus [29] and is required for the initial step of WTA biosynthesis, namely, the transfer of GlcNAc to the C55-P lipid anchor bactoprenol. We observed higher levels of mCherry-PGRP-SA binding to the newly synthesized cell material, located at the division septum, when Gram-positive bacteria cells were treated with tunicamycin (Figure 2A). S. aureus and S. saprophyticus exhibited a similar increase in binding, 63× and 84× respectively, whilst E. faecalis binding increased 8×. It should be noted that the effect of tunicamycin in these bacteria was not the same. While addition of the antibiotic resulted in binding of mCherry-PGRP-SA to the entire cell surface of S. aureus, binding was observed predominantly at the division septum in S. saprophyticus and exclusively at this region in E. faecalis. We attribute these differences to how and where the new cell wall synthesis occurs in these bacteria. Nevertheless, the results described above suggested that WTA in different bacteria might protect PG from exposure to host receptors.
Fig. 2. WTA reduce PGRP-SA binding at the bacterial cell surface. 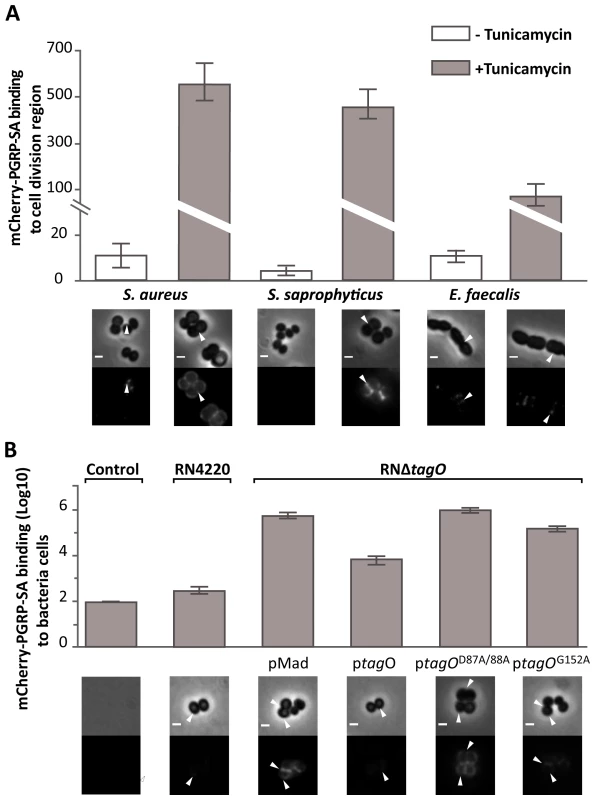
Grey panels are phase-contrast images of bacterial cells (white scale bar represents 1 µm), and black panels mCherry-PGRP-SA binding; white arrowheads highlight binding to the lateral cell surface or region of cell division. The binding of mCherry-PGRP-SA to individual bacterial cells (n = 50) was quantified, and represented as the median (with 25% and 75% inter-quartile range). (A) mCherry-PGRP-SA binding to Gram-positive bacteria grown with or without tunicamycin, an inhibitor of WTA synthesis. mCherry-PGRP-SA binding to the cell division region, rather than total binding, was measured because binding at the former was consistently enhanced for all treated bacteria species. Mann-Whitney U tests were used to compare differences for treated and untreated between each type of bacteria (P<0.05 in all cases). (B) RNΔtagO mutant background rescued with variants of the tagO gene – expressed from a replicative pMAD vector – produce varying levels of WTA, given as a% relative to the wild type RN4220: pMAD vector (0%), ptagO (90%), ptagOD87A/D88A (0%), ptagOG152A (22%). Total binding of mCherry-PGRP-SA to the surface of live bacteria increases as the levels of WTA are reduced. Kruskal-Wallis analysis followed by Dunn's multiple comparison post-test, revealed significant differences for all comparisons (P<0.05) except for that between PGRP-SA binding to pMAD and ptagOD87A/D88A. To confirm that WTA were indeed required to reduce access of PGRP-SA at the cell surface, we quantified the binding of mCherry-PGRP-SA to S. aureus mutants that produced varying amounts of WTA due to mutations in the tagO gene [29]. We chose S. aureus because it is a major human pathogen with a well-characterised WTA synthetic pathway [30], [31]. A complete absence of WTA, which occurs when tagO is entirely deleted (RNΔtagO pMAD), or when two highly conserved residues have been mutated (RNΔtagO ptagOD87A/D88A), resulted in equivalently enhanced levels of mCherry-PGRP-SA binding, when compared to the wild type strain (∼2×103 and ∼3.3×103-fold respectively, Figure 2B). To verify that the observed result was indeed due to the loss of WTA, we expressed wild type tagO in the RNΔtagO background (RNΔtagO ptagO): this rescued the loss of WTA (WTA levels restored to 90% of wild type levels) [29], and reduced mCherry-PGRP-SA binding to levels close to those observed for the wild type strain (Figure 2B). A tagO mutant that could only support production of a reduced amount of WTA (RNΔtagO ptagOG152A; 24% levels of WTA compared to wild type) exhibited an intermediate level of mCherry-PGRP-SA binding relative to all strains (6×102-fold increase relative to the wild type strain, Figure 2B). Overall, our data indicated that WTA found in the cell wall of different live Gram-positive bacteria restricted PGRP-SA from binding their PG, and in S. aureus this occurs in a dose dependent manner.
We next wanted to examine whether increased PGRP-SA binding – due to a lack of WTA – affected the ability of bacteria to survive in an in vivo system. We chose D. melanogaster because it is a well-established model for dissecting pattern recognition in innate immunity [18]. We know for example that in vitro, three PRRs – PGRP-SD/PGRP-SA/GNBP1 – form a ternary complex for binding to the PG of S. aureus [14]. As a first approach wild type and mutant S. aureus strains were injected into wild type flies and also into flies defective for PGRP-SD or PGRP-SA. We then determined the number of CFUs 6 and 17 hours post-infection; the latter time point being when the first flies succumb to infection (Figure 3 and S5). All flies were inoculated with low and statistically identical numbers of bacteria (∼102 CFUs per fly; Figure 3, Time 0). Our rationale was to induce infections that were comparable and that could evolve over time. For example, flies generally succumb to bacterial infection when their numbers increase beyond 106 CFUs per fly [18], [32], and therefore, high initial loads (e.g. 104–105 CFUs per fly) may overwhelm the host and consequently may not be informative regarding the course of an infection. We observed that wild type S. aureus (NCTC8325-4) CFUs increased in all fly backgrounds over the period of infection to numbers that were statistically separable, with PGRP-SA deficient flies carrying the heaviest load (Figure 3). In contrast, the numbers of the S. aureus mutant, which lacked WTA (NCTCΔtagO) [29], did not significantly increase in the wild type or PGRP-SD mutant background. However, the number of NCTCΔtagO bacteria in the PGRP-SA mutant was significantly higher at both the 6 and 17 hours time points (Figure 3). Two-way ANOVA revealed a significant interaction between the bacteria and fly strains, which was due to the large increase of NCTCΔtagO bacteria in the PGRP-SA mutant. Together, these data indicated that WTA were fundamental for S. aureus to counter recognition by PGRP-SA, and consequently, the bacteria were able to increase their number during the initial course of infection.
Fig. 3. PGRP-SA is fundamental for controlling bacterial numbers in flies infected with a S. aureus mutant that lacks WTA. 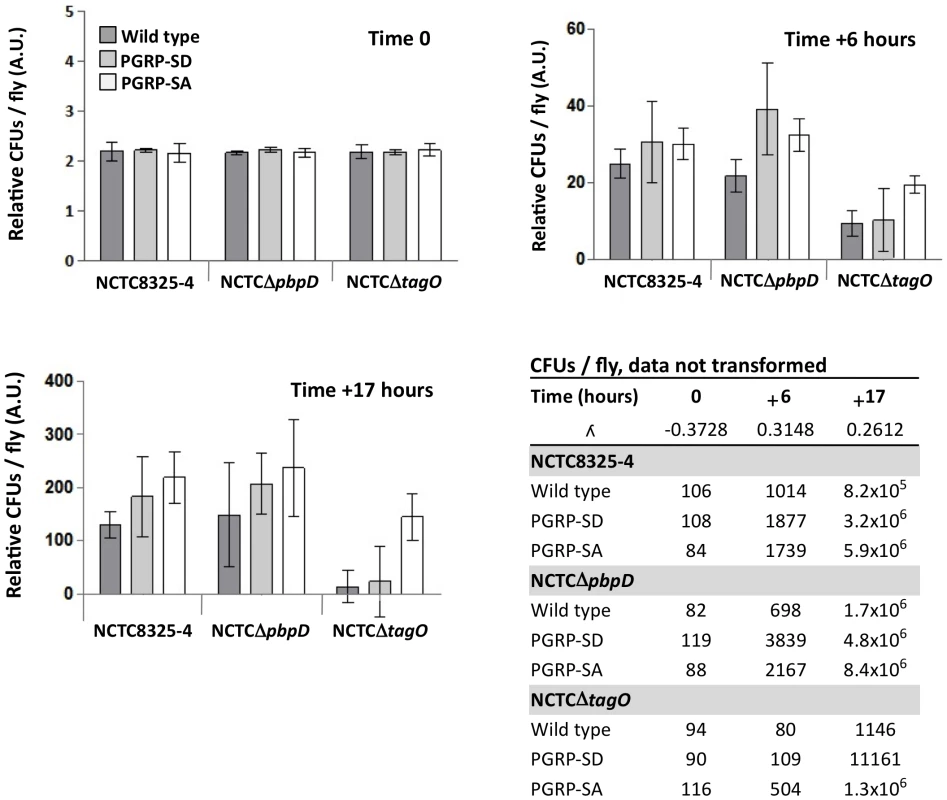
Wild type flies, and those lacking PGRP-SD or PGRP-SA, were infected with different S. aureus strains: NCTC8325-4 is the wild type; NCTCΔpbpD is a mutant that produces WTA but has a PG similar to NCTCΔtagO, both exhibiting reduced cross-linking; NCTCΔtagO lacks WTA. The table gives the mean CFUs per fly (from 3 independent experiments). For each time point, the CFUs per fly data set was transformed via a Box-Cox transformation (which returns a λ number, where data-point = data-pointλ – 1/λ) and represented as means with 95% confidence intervals. Flies were inoculated with a low (∼100 CFUs per fly) and comparable number of bacteria (Time 0; Two-way ANOVA did not reveal significant differences, P>0.05), and CFUs per fly were determined at 6 and 17 hours post-infection. In contrast to NCTC8325-4 and NCTCΔpbpD, the number of NCTCΔtagO bacteria did not significantly increase in the wild type or PGRP-SD mutant background during the period of infection (Table); however, in the PGRP-SA mutant the number of bacteria increased significantly for all strains (P<0.05, Repeated Measures One-way ANOVA). Two-way ANOVA of the CFUs data at Time +17 hours revealed a significant interaction (P<0.05) between the bacteria and fly strains, which was due to the large increase of NCTCΔtagO CFUs in the PGRP-SA mutant, whilst differences in CFUs were similar for NCTC8325-4 and NCTCΔpbpD. One-way ANOVA and 95% Tukey's HSD intervals were used to look for factor differences at this time. For each fly background NCTC8325-4 and NCTCΔpbpD CFUs were equivalent (P>0.05). NCTCΔtagO CFUs in the wild type and PGRP-SD backgrounds were similar (P>0.05), but separated from all other values (P<0.05). In the PGRP-SA mutant, NCTCΔtagO CFUs reached levels seen with the other bacteria in wild type and PGRP-SD flies. The negative error bars for the NCTCΔtagO infection occur because of large variation of the biological repeats. This is consistent with the fact that NCTCΔtagO occasionally causes a lethal infection in both the wild type and PGRP-SD backgrounds. We have previously observed that PG produced by NCTCΔtagO bacteria has reduced levels of cross-linking relative to the wild type strain [29]. To evaluate whether this contributed to the inability of NCTCΔtagO bacteria to increase their number in wild type or PGRP-SD mutant flies, we assessed mCherry-PGRP-SA binding to NCTCΔpbpD and determined CFUs at 6 and 17 hours. NCTCΔpbpD is a derivative of NCTC8325-4 in which pbpD (the gene encoding to penicillin binding protein 4, PBP4) has been deleted. Deletion of pbpD results in a strain that produces PG with a similar level of cross-linking to that found in NCTCΔtagO [29], but which still produces WTA. The inability of NCTCΔpbpD and NCTCΔtagO to produce a highly crosslinked PG did not interfere with bacteria growth in culture, as its duplication time at 30°C was very similar to the parental NCTC8325-4 strain (Figure S3B). In both experiments, NCTCΔpbpD behaved as the wild type bacteria. Firstly, binding mcherry-PGRP-SA similarly (Figure S3C) and secondly, for each fly background attaining numbers that were statistically inseparable from those for NCTC8325-4 (Figure 3, Time +17 hours).
To assess whether the developing trend in bacterial numbers at 17 hours post-infection resolved into differences in how flies survive, we monitored the number of flies alive at 24 hour intervals over 3 days. In addition, we infected GNBP1 mutant flies, because GNBP1 has been postulated to work as part of a complex with PGRP-SA [14], [17]. Survival curves for a particular fly background when infected with either NCTC8325-4 or NCTCΔpbpD were statistically inseparable, except for those obtained for the wild type background, where flies succumbed more to NCTCΔpbpD (Figure 4; 62% and 38% survival at 72 hours post-infection, respectively). Nearly all PGRP-SA and GNBP1 mutant flies had died by 24 hours, whereas ∼40% of PGRP-SD mutant flies survived beyond this time point, succumbing to infection around 48 hours (∼5% of flies surviving). In contrast, ∼95% of wild type and PGRP-SD mutant flies survived the NCTCΔtagO infection up to 72 hours (furthermore, taking CFUs at this time-point revealed that NCTCΔtagO had been eliminated from these flies, 0 CFUs per fly). The majority of PGPR-SA and GNBP1 flies had succumbed to infection by 48 hours (3% of flies surviving). A similar trend in survival outcome was observed with NCTC8325-4 after treatment with tunicamycin (Figure 4). These data confirmed that WTA were indeed required to counter host immunity, because without them, infection could be controlled in a PGRP-SA/GNBP1 dependent manner. Differences in CFUs were apparent 6 hours post-infection suggesting that recognition and reduction of propagation or killing of bacteria, occurs rapidly following infection. Interestingly, these results also showed that a requirement for PGRP-SD was bypassed when WTA are removed and PGRP-SA has far greater access to PG.
Fig. 4. PGRP-SA and not PGRP-SD is required to control infection by S. aureus mutant lacking WTA. 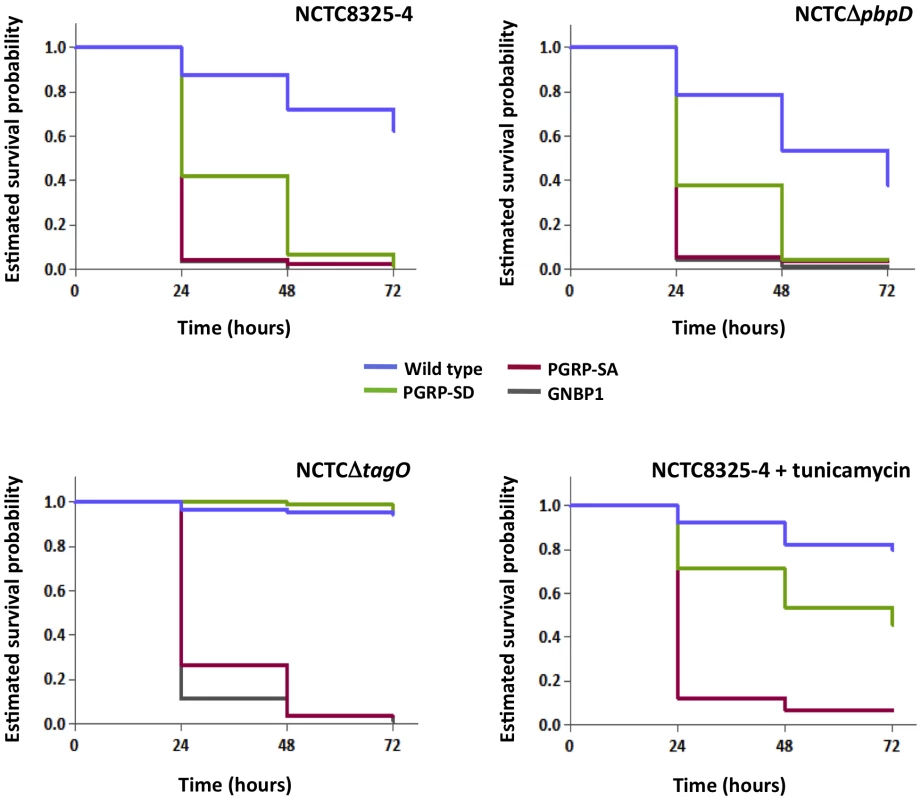
Flies assayed for survival were injected concurrently with those for determining CFUs. The survival of infected flies (n = 90) was monitored at 24-hour intervals for three days, and estimates of survival plotted (for clarity, 95% confidence intervals have been omitted). For each fly background – except wild type – survival curves were statistically inseparable for flies infected with NCTC8325-4 or NCTCΔpbpD (log-rank test, P>0.05). PGRP-SD, PGRP-SA and GNBP1 mutant flies succumbed strongly to infection by 72 hours, whereas wild type survived up to ∼60%. In contrast, wild type and PGRP-SD mutant flies were barely susceptible to infection with NCTCΔtagO, however, PGRP-SA and GNBP1 flies succumbed strongly; a similar survival trend was seen when flies were infected with tunicamycin-treated NCTC8325-4 (GNBP1 mutant flies were not infected for this experiment). To further demonstrate the necessity for WTA to protect PG from host recognition, we monitored survival of flies infected with the aforementioned TagO point mutations (Figure 2B and Figure 5). In these experiments, we wanted to rule out unknown causes that may occur due to the absence of the TagO protein per se, and also, lessen adverse effects that may occur due to a complete lack of WTA. The survival trend for flies infected with RNΔtagO pMAD, that lacks tagO and carries an empty pMAD plasmid vector (vector control), was similar to that for NCTCΔtagO: the PGRP-SA mutant succumbed rapidly, whereas the PGRP-SD mutant and wild type flies generally survived, their curves being statistically inseparable (Figure 5). The injection of the complemented strain (RNΔtagO ptagO) resulted in survival outcomes that were characteristic of NCTC8325-4, with PGRP-SD mutant and wild type flies succumbing to the infection, with their curves being statistically separated (Figure 5). Notably, wild type and PGRP-SD mutant flies infected with RNΔtagO ptagOG152A (which produces ∼24% WTA relative to RNΔtagO ptagO but produces similar levels of the TagO protein) [29] survived to intermediary levels (Figure 5). Overall, survival of wild type flies decreased as WTA levels increased (with a concomitant decrease in PGRP-SA binding, Figure 2B), and likewise for the PGRP-SD mutant; with the difference between wild type and PGRP-SD mutant survival successively increasing. In contrast, survival of PGRP-SA mutant flies was independent of WTA levels, with flies succumbing strongly for all infections in a statistically inseparable manner (Figure 5). These data confirmed that it was indeed in vivo protection of PG by WTA against the consequences of PGRP-SA binding, and furthermore, suggested that a requirement for PGRP-SD gradually became redundant as WTA levels decreased.
Fig. 5. The levels of WTA modulate the requirement for PRRs. 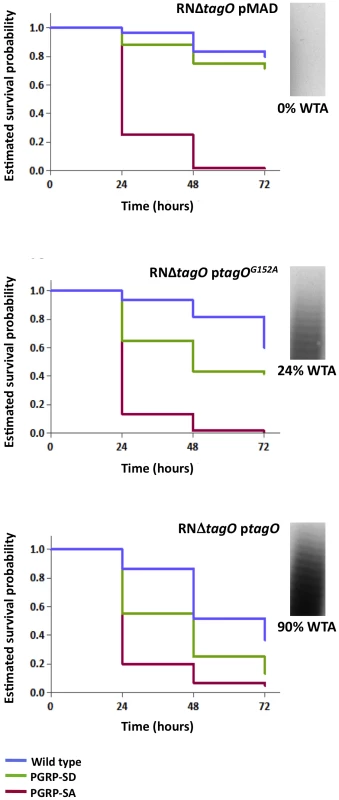
Survival of infected flies (n = 60) was monitored at 24 hours intervals for three days, and estimates of survival constructed from the raw data. Flies were infected with S. aureus mutants that produce different levels of WTA (percentage of WTA produced by each strain was quantified as the signal intensity of bands of WTA in the native gels, and it was normalized against the corresponding value for the wild type – considered as 100%): RNΔtagO pMAD lacks WTA; RNΔtagO ptagO produces 90% WTA relative to the parental RN4220; and RNΔtagO ptagOG152A produces 24% WTA relative to the parental RN4220 strain. Wild type flies succumb successively to infection as the levels of WTA increase (log-rank test, P<0.05), likewise for the PGRP-SD mutant. In addition, survival of wild type and PGRP-SD mutant flies increasingly separates for each of the bacterial mutants: wild type versus PGRP-SD, P = 0.2452 (log-rank test, RNΔtagO pMAD); P = 0.0053 (RNΔtagO ptagOG152A); P = 0.0001 (RNΔtagO ptagO). For all infections, PGRP-SA mutant flies succumb equally to infection (log-rank test, P>0.05). It has been reported previously that D-alanylation of WTA is also required for the pathogenicity of S. aureus [11]; D-alanylation is a process that incorporates D-alanine residues into the glycerol-/ribitol-phosphate backbone of WTA, thereby reducing the negative charge of the polymer [33]. We examined therefore, whether a S. aureus mutant that lacks the D-alanylation pathway (RNΔdltABCD) bound mCherry-PGRP-SA equivalently to RNΔtagO. Binding of, mCherry-PGRP-SA to RNΔdltABCD was similar to the binding to the wild type bacteria (Figure S3). This prompted us to assess how RNΔdltABCD affected survival of the wild type, PGRP-SD and PGRP-SA mutant flies. In contrast to RNΔtagO, PGRP-SA mutant flies did not succumb strongly to RNΔdltABCD infection, with 83% surviving at 72 hours post-infection (Figure S4); furthermore, survival was statistically inseparable for the different fly backgrounds (Figure S4). These data demonstrated that D-alanylation is not necessary for WTA to limit the access of PGRP-SA, that neither PGRP-SD nor PGRP-SA were required to control the RNΔdltABCD infection and consequently, the reduced killing effect of RNΔdltABCD had nothing to do with recognition.
Discussion
The results shown here indicate that in respect to Gram-positive bacteria, where the cell wall is not concealed by outer membrane (e.g. staphylococci), pathogen recognition, via recognition of PG, is tightly linked to host survival. Our studies bring forward the notion that one of the strategies used by pathogens to reduce recognition is to restrict accessibility to inflammatory non-self components of the cell wall. Specifically, the results here show that presence of WTA in a range of Gram-positive bacteria impaired PGRP-SA binding. The use of tunicamycin to abolish WTA synthesis dramatically improved receptor recognition of bacteria as well as host survival of flies infected antibiotic treated S. aureus. Genetically deleting a major component of the WTA synthesis (TagO) in S. aureus also increased PGRP-SA binding leading to increased host survival. It should also be noted that, rPGRP-SA was capable of binding in vitro significantly better to WTA-free PG than to WTA-linked PG that were purified from wild type S. aureus bacteria, treated with trypsin to remove any attached surface proteins and adjusted to the same concentration of PG (Figure S2). This observation confirmed the results obtained with live bacteria and allowed us to eliminate the notion that deletion of tagO gene may influence the amount of protein present at the cell surface and that this change in protein levels was influencing the binding of PGRP-SA. Effectively during the course of this work we have removed WTA from PG by treatment with antibiotic, by deletion of the tagO gene and finally we have chemically removed them from PG. In all the cases binding of PGRP-SA to PG has increased.
S. aureus produces WTA composed of about 40 ribitol phosphate-repeating units modified with N-acetylglucosamine (GlcNAc) and D-alanine [7]. The latter modification is mediated by the D-Alanine ligase DltA and partially neutralizes the negative charge of the cell surface thus reducing attraction of cationic AMPs [33]. ΔdltA mutants are more susceptible to killing by cationic AMPs and neutrophils in vitro and have markedly reduced virulence in several animal infection models including Drosophila [11], [34]. In one of these studies [11], Tabuchi and colleagues showed that S. aureus producing WTA without D-alanylation were impaired in their ability to kill Drosophila. Surprisingly, the ΔdltA mutant was more impaired in the ability to kill flies than an independently generated tagO mutant [11]; the latter according to the authors had the same pathogenicity as wild type S. aureus [11], contrary to our findings.
There is a crucial point to be made in reference to this however, which is at the heart of our experimental design and gives physiological relevance to our results. We propose that WTA are important to reduce S. aureus recognition by the host and thus help the pathogen increase its numbers inside the fly. The host uses PGRP-SA to control bacterial numbers and the more PGRP-SA binds to the cell wall (see Figure 2B) the more the bacterial load is controlled (as seen by comparing CFUs between wild type NCTC8325-4 S. aureus and NCTCΔtagO in Figure 3A). In PGRP-SA mutants the control mechanism is absent and NCTCΔtagO was able to proliferate and kill the host (Figure 3B). We were able to observe this because we started from a low bacterial load (102 cells/initial infection/fly) and followed the progress of pathogen load inside the host. Tabuchi et al. injected 104–106 cells per fly for all bacterial strains used [11]. In our hands this concentration overwhelmed the host from the beginning and it is not surprising that these authors were unable to resolve statistical differences in host survival.
In order to rule out possible pleiotropic effects produced by the inactivation of the tagO due to the insertion of non-replicative plasmids or reversion of the mutation by elimination of the plasmid from the chromosome, we have specifically deleted the tagO gene in a manner that left no resistance marker in the bacterial chromosome and thus minimized possible alterations on the transcription of neighbouring genes. Finally, in order to increase the confidence of our results, we have complemented the tagO null mutant with plasmids that allowed the expression of a partially active (TagOG152A), TagO protein and have statistically analyzed the estimated host survival probability curves obtained. Finally we should emphasize that deletion of the tagO gene in NCTC8325-4 strain (an agr positive strain) and in RN4220 (an agr negative strains) resulted in similar outcomes (Figure S3) - a reduced pathogenicity in the Drosophila infection model and the production of a bacterial cell surface that was better recognized by mCherry-PGRP-SA.
In parallel experiments we have also generated a ΔdltA deletion mutant (this study) as well as a deletion of the ΔdltA operon (ΔdltABCD) [29] and found that both were indeed less pathogenic than wild type S. aureus (Figure S4), similar to what was previously reported [11]. However, this reduced pathogenicity was also observed in PGRP-SA and PGRP-SD single mutant flies (in contrast to ΔtagO). This indicated that the non-pathogenicity of ΔdltA was not linked to recognition by PGRP-SA or PGRP-SD.
We propose that increased “visibility” of PG to PGRP-SA when WTA were removed, dramatically improved survival of the host. However, alternative interpretations of our results may exist. In the following section we will attempt to challenge and rule them out:
-
We have recently reported that removal of WTA has an impact on PG cross-linking and consequently on the susceptibility to host lysozyme [29]. The possibility that the increased host survival may be the result of decreased pathogen resistance to the lysozyme constitutively expressed in the fly (due to the reduced PG cross-linking in the S. aureus tagO null mutant) rather than removal of a physical entity (WTA), which blocked access to PG, was ruled out as follows. We generated an S. aureus mutant unable to produce high-level PG cross-linking but capable of producing regular levels of WTA, by deleting the pbpD gene [29]. The pbpD gene encodes to PBP4 which is responsible for the final stages of PG maturation and results in highly cross-linked PG. As shown in Figure 4, bacteria that produce PG with a low level of cross-linking, but normal levels of WTA, were able to kill wild type flies similarly to the parental S. aureus strain. In addition, similar amounts of mCherry-PGRP-SA bound to the surface of both wild type bacteria and NCTCΔpbpD (Figure S3C). These results indicate that in ΔtagO increased recognition by mCherry-PGRP-SA and the inability to kill flies is due to the absence of the WTA and not due to modifications in PG cross-linking.
-
The hypothesis that the absence of teichoic acids could turn S. aureus bacteria more susceptible to enzymes present in the haemolymph of Drosophila, such as lysozyme-like enzymes, which would make the bacteria unable to kill flies, was also considered and ruled out. In accordance with previous reports [34] we have verified that the S. aureus tagO null mutant is as resistant to lysozyme as the parental strain. The tagO null mutant only becomes susceptible to lysozyme when an additional mutation in the oat gene, encoding a protein responsible for PG O-acetylation, is introduced (data not shown). Most importantly, injection of S. aureus tagO null mutant into PGRP-SA mutant flies was lethal, indicating that the S. aureus tagO null mutant bacteria were able to multiply in the haemolymph of flies if undeterred by PGRP-SA.
-
The possibility that PGRP-SA is responsible for directly killing bacteria lacking WTA was also ruled-out as there was no alteration in the growth rate of S. aureus tagO null mutant when grown in the presence of recombinant PGRP-SA (data not shown). PGRP-SA is believed to be non-lytic [35]. Nevertheless, this is a working hypothesis and has not been formally proven. In contrast, an unusual L,D-carboxypeptidase activity has been observed towards PG of some Gram-negative bacteria [36]. At the present moment, we cannot exclude that a protein existing in the haemolymph is capable of mediating killing of S. aureus tagO in complex with PGRP-SA. In accordance to the latter hypothesis we have previously shown that PGRP-SA enhances the weak endomuramidase activity of GNBP1 for PG of M luteus, the cell wall of which (like tagO), is devoid of WTA [37].
-
The possibility that the absence of WTA could turn S. aureus bacteria more susceptible to AMPs (produced as a consequence of the recognition of an invading pathogen) was also tested. Injection of S. aureus tagO null mutant into mutant flies affected in the ability to produce AMPS, such as Dif1-key1, spzrm7 and spz1 was not lethal to the flies, indicating that the S. aureus tagO null mutant bacteria were being eliminated in a way that was dependent on recognition by PGRP-SA but not dependent upon activation of the production of AMPs (Figure S6). At the moment we are unable to identify how Drosophila flies are killing invading S. aureus tagO null mutant bacteria. It is possible that bacteria, upon recognition by PGRP-SA, are more easily phagocytised or that, as in Tenebrio molitor [38], PGRP-SA binding recruits the local melanization cascade, triggering such a response.
Our results underline an important aspect of pathogen recognition by the host, which remains relatively unexplored. Namely, how does the host recognition machinery respond to changes in the surface of bacteria? Here we manipulated the amount of WTA on the cell surface of S. aureus. Previously, two host PGRPs, PGRP-SA and PGRP-SD were found to be involved in recognition of wild type S. aureus [14], [15]. We found here that when WTA were genetically removed, the requirement for PGRP-SD was abolished. Flies deficient for PGRP-SD had estimated survival probabilities comparable to wild type flies following infection by S. aureus ΔtagO or ΔtagOptagOD87/D88A. When a small amount of WTA was left on the surface through the residual activity of the S. aureus ΔtagOptagOG152A then PGRP-SD mutants were less able to survive infection. However this sensitivity was not as pronounced as when infected with S. aureus ΔtagOptagO, the strain with reconstituted wild type levels of WTA. Previous studies have established that PGRP-SD does not bind Gram-positive Lys-type PG [14], [39]. However, in its presence, PGRP-SA was able to bind substantially better to cell wall from S. aureus and S. saprophyticus [14]. Our results, combined with the latter observation, support a role for PGRP-SD in neutralizing the effect of WTA obstructing access to PG. The alternate hypothesis that PGRP-SD may directly recognize WTA, and is therefore not necessary when flies are infected with bacteria that lack teichoic acids, is also a possibility.
The role of teichoic acids in concealing PG at the surface of Gram-positive bacteria may be also effective in preventing recognition by innate immune sensors of other organisms. It is now established that insect PGRPs have mammalian homologues and mice and humans express four genes encoding members of this family [35]. Our results correlate with data, which attributed a significantly reduced virulence of tagO mutants in cotton rat nasal colonisation model [40] as well as a mouse endophthalmitis model [41] and suggest a mechanism for how this may happen: absence of teichoic acids may render PG at the bacteria surface more exposed to the host immune system.
Materials and Methods
Microbial and fly strains
Isogenic wild type flies (Bloomington #25174) were used as the wild type control. For the survival and bacterial Colony Forming Unit (CFU) experiments, and DD1 flies for assaying Drs levels visually or via qPCR; the latter carries a Drs-GFP and a Diptericin-lacz reporter [42]. The PGRP-SA and PGRP-SD mutant backgrounds are, respectively: flies with the semmelweis mutation in PGRP-SA [16] and a 1499 bp deletion in PGRP-SD (PGRP-SDΔ3) [15]. The spzrm7 [43] and spz1 [44] Toll pathway mutant backgrounds, and the Dif1-key1 [45] Toll-IMD pathways double mutant background, were used to assess survival of flies deficient for AMPs. All fly stocks were reared at 25°C. Bacterial strains are listed below. S. aureus strains were grown in tryptic soy broth medium (TSB; Difco) supplemented with antibiotic (erythromycin 10 µg/ml; Sigma-Aldrich) when required. E. faecalis was grown in brain heart infusion medium (BHI; Fluka). M. luteus was grown in Luria-Bertani medium (LB; Difco). Bacteria were plated from -80°C stocks every 7 days Growth of all bacteria cultures were done at 30°C as S. aureus mutants impaired in the synthesis of teichoic acids are thermosensitive [46].
S. aureus strains
NCTC8325-4 (S. aureus reference strain from R. Novick); NCTCΔtagO (NCTC8325-4 tagO null mutant [29]); RN4220 (Restriction deficient derivative of S. aureus NCTC8325-4 that can be electroporated); RNΔtagO (RN4220 tagO null mutant [29]); RNΔtagOpMAD (RNΔtagO transformed with pMAD [29] – shuttle vector with a thermosensitive origin of replication for Gram-positive bacteria); RNΔtagO ptagO (RnΔtagO transformed with ptagO [29]); RNΔtagO ptagOD87A/D88A (RNΔtagO transformed ptagOD87A/D88A [29]); RNΔtagO ptagOG152A; RNΔtagO transformed with ptagOG152A, [29]); RNΔdltABCD (RN4220 dltABCD null mutant [29]); RNΔdltABCD (RN4220 dltABCD null mutant [29]); RNΔdltA (RN4220 dltA null mutant, this study). M. luteus strain: DMS20030 [47]; E. faecalis strain: JH2-2 [48]; B. subtilis strain MB24 [49].
Construction of the RNΔdltA null mutant
To delete the dltA gene from the chromosome of S. aureus RN4220 we started by amplifying two 0.55 Kb DNA fragments from the genome of S. aureus NCTC 8325-4 strain, corresponding to the upstream (primers 5′-AGATCTgaatgtatatatttgcgctgatg-3′ and 5′-gtaaaatcaccatatggaatcatattaagtctccctcattagaactc-3′) and downstream (primers 5′ - gagttctaatgagggagacttaatatgattccatatggtgattttac-3′ and 5′-GAATTCcgaaacgtttgtaacgatcg-3′) regions of the dltA gene. The two fragments were joined by overlap PCR using primers P33 and P36 and the resulting PCR product was digested with BglII and EcoRI and cloned into the pMAD vector, producing the plasmid pΔdltA. This plasmid was sequenced and electroporated into S. aureus RN4220 strain. Insertion and excision of pΔdltA into the chromosome of RN4220 was performed as previously described [29] with the exception of the incubation temperature after excision of the plasmid, which was 30°C (instead of 43°C) due to the thermosensitive nature of the cells lacking D-alanylation. Deletion of dltA was confirmed by PCR, and the resulting strain was named RNΔdltA.
Survival experiments and determination of CFUs
Overnight 10 ml cultures of bacteria were washed and resuspended in an equal volume of sterile phosphate buffered saline (PBS), and further diluted 1/1000. Healthy looking adult flies from uncrowded bottles, 2–4 days old, were injected in the thorax with 32 nl of a bacterial cell suspension or PBS using a nanoinjector (Nanoject II, Drummond Scientific). For determination of CFUs, injected flies (6 females) were crushed immediately in media appropriate for the bacteria injected and the homogenates were diluted and plated on tryptic soy agar-media (TSA). The plates were incubated at 30°C for 20–30 hours and the colony forming units (CFUs) per fly were measured by counting the number of colonies on each plate, the CFUs per fly were used to adjust the initial dose of bacteria injected to approximately 100 CFUs per fly. For the time course (0, 6, 17 hours) determination of CFUs, each value represents an arithmetic average derived from three biological repeat experiments (n = 3). Flies for survival and PGRP-SA mutant rescue assays were inoculated concurrently with those for determining CFUs, with ten or fifteen flies of each sex injected per bacteria-fly strain combination (or PBS-fly strain); each combination being repeated independently three times (n = 3). Following injection, flies were transferred to 30°C and survival assessed every 24 hours over a period of 3 days. Since the trends in survival were the same (i.e. survival curves were positioned similarly relative to one another) for each independent biological repeat, the data for each bacteria-fly strain combination was added (n = 60 or n = 90) and estimates of survival curves constructed. Flies injected with PBS were mostly unaffected for all fly backgrounds.
Purification of recombinant rPGRP-SA and mCherry-PGRP-SA from E.coli
A truncated version of PGRP-SA (in which the N-terminal sorting sequence was replaced with a T7 tag, and a poly-histidine tag was added to the C-terminus) was expressed in E. coli and purified using cobalt affinity resin (Talon; BD Biosciences) under denaturing conditions. A mCherry tagged derivative, mCherry-PGRP-SA was produced using the same procedure. Proteins were stored in 20 mM Tris-HCl pH 8.0 and 150 mM NaCl.
Protein functionality assays
Functionality assays of the rPGRP-SA and mCherry-PGRP-SA proteins were performed as previously described [14]. Drs-GFP expression was monitored after 24 hours of the M. luteus infection through the production of fluorescent signal produced by the infected flies; and by qPCR using as template RNA extracted from 6 infected female flies, similar to what was previously described [50].
Purification of peptidoglycan
Peptidoglycan was prepared from exponentially growing cultures of S. aureus, B. subtilis, M. luteus, and E. faecalis as previously described [13].
PGRP-SA-peptidoglycan co-precipitation assay
50 µg of recombinant PGRP-SA was incubated with 0.2 mg of peptidoglycan and 17 µg of BSA (New England Biolabs) in 20 mM Tris-HCL pH 8.0 and 300 mM NaCl in a final volume of 300 µl. Incubation was at 25°C with agitation for 30 minutes. Peptidoglycan and co-precipitated proteins were harvested by centrifugation, washed twice with 20 mM Tris-HCl pH 8.0, 300 mM NaCl and then resuspended in 1× SDS loading buffer, boiled for 5 minutes and run on 12% SDS PAGE mini gels. An aliquot of the supernatant, representing unbound protein, was also run. Gels were stained with Coommasie stain, destained and imaged using an ImageScanner (Amersham Biosciences/GE Healthcare). Quantifications of bands performed using ImageJ software [51]; each value represents an arithmetic average derived from three biological repeat experiments (n = 3).
mCherry-PGRP-SA binding to bacteria
Bacteria were grown to mid-exponential phase. Washed cell cultures in PBS (500 µl) were incubated with 50 µl of mCherry-PGRP-SA (2 mg/ml in 150 mM NaCl, 20 mM Tris pH 8.0) for 5 minutes on ice. The cells were washed twice with PBS and harvested at 4°C (3000 rpm, 10 minutes). Finally the bacteria were resuspended in 20 µl PBS. A drop of this culture was placed on a PBS, 1% agarose slide and visualised. Images were obtained using a Zeiss Axio ObserverZ1 microscope equipped with a Photometrics CoolSNAP HQ2 camera (Roper Scientific using Metamorph software, Meta Imaging series 7.5) and analyzed using ImageJ software.
WTA extraction
WTA were extracted by alkaline hydrolysis from overnight cultures were analyzed by native polyacrylamide gel electrophoresis and visualized by combined alcian blue silver staining, as previously described [52]. ImageJ software [51] was used to quantify the percentage of WTA produced by each strain as previously described [29]. The signal intensity of each lane was quantified and normalized against the corresponding value for the wild type (considered as 100%).
WTA inhibition
Tunicamycin minimum inhibitory concentration (MIC) assays were performed as previously described [28]. Overnight cultures of bacteria were grown in antibiotic free medium or in the presence of a subinhibitory concentration of tunicamycin (0.8 ug/ml for E. faecalis – 17× less than the MIC - and 0.4 ug/ml for S. aureus and S. saprophyticus – 32× less than MIC), that doesn't interfere with the bacterial growth rate. For mCherry-PGRP-SA binding assays, overnight cultures were diluted 1∶100 into fresh medium, with or without tunicamycin at the appropriate concentration, and were grown until mid-exponential phase. For survival experiments, we used S. aureus overnight culture grown with tunicamycin as above described.
Data analysis
As nonparametric tests lack statistical power with small samples, when required, data sets with three biological repeats (n = 3) were transformed to give a normal distribution (Lilliefors test, P>0.05) and then checked for equal variance (Levene's test, P>0.05); subsequently, data was analysed using parametric tests.
Binding assays
Data for the PGRP-SA-peptidoglycan co-precipitation assay was normal with equal variance, thus not transformed; One-way ANOVA was applied to the data. For the mCherry-PGRP-SA binding to bacteria assays data (n = 50) was non-normal but with equal variance, therefore nonparametric Kruskal-Wallis test followed by Dunn's multiple comparison was applied.
CFUs
The complete CFU data set exhibited neither normality nor equal variance, and attempts to rectify this by transforming the data failed. Therefore, the data was separated into 6 groups, which were independently transformed via a Box-Cox transformation (Box-Cox returns a λ number, where a transformed data-point = data-pointλ – 1/λ) to give a normal distribution with equal variance, and statistical analysis performed as described. Firstly, for each bacterial strain (groups 1–3, graphical representations not shown), Repeated Measures Two-way ANOVA was used to look for differences over time and between the fly backgrounds. However, due to interactions between these two factors, Repeated Measures One-way ANOVA with 95% Tukey's HSD Intervals was used to look for differences over time for each particular bacteria strain and fly background combination (i.e. 9 separate tests, data for each was normally distributed with equal variance). Secondly, at each time point (groups 4–6, Figure 3), Two-Way ANOVA was used to look for differences between the bacterial strains and between the fly backgrounds; where there was an interaction between these two factors, One-way ANOVA with 95% Tukey's HSD Intervals was used to look for differences between the fly backgrounds for a particular bacterial strain.
Fly survival
Estimated survival curves were constructed from the raw data sets and the Log-rank (Mantel-Cox) test used to determine statistical significance between the curves. For clarity in display, 95% confidence intervals have been omitted from the graphs. All data was plotted and analyzed using GraphPad Prism 5 (GraphPad Software, Inc.) or MATLAB R2009a.
Supporting Information
Zdroje
1. FosterTJ 2005 Immune evasion by staphylococci. Nat Rev Microbiol 3 948 958
2. ChaputCBonecaIG 2007 Peptidoglycan detection by mammals and flies. Microbes Infect 9 637 647
3. VollmerWBlanotDde PedroMA 2008 Peptidoglycan structure and architecture. FEMS Microbiol Rev 32 149 167
4. SchleiferKHKandlerO 1972 Peptidoglycan types of bacterial cell walls and their taxonomic implications. Bacteriol Rev 36 407 477
5. ScottJRBarnettTC 2006 Surface proteins of gram-positive bacteria and how they get there. Annu Rev Microbiol 60 397 423
6. KadiogluAWeiserJNPatonJCAndrewPW 2008 The role of Streptococcus pneumoniae virulence factors in host respiratory colonization and disease. Nat Rev Microbiol 6 288 301
7. WeidenmaierCPeschelA 2008 Teichoic acids and related cell-wall glycopolymers in Gram-positive physiology and host interactions. Nat Rev Microbiol 6 276 287
8. HumannJLenzLL 2009 Bacterial peptidoglycan degrading enzymes and their impact on host muropeptide detection. J Innate Immun 1 88 97
9. VollmerW 2008 Structural variation in the glycan strands of bacterial peptidoglycan. FEMS Microbiol Rev 32 287 306
10. BonecaIGDussurgetOCabanesDNahoriMASousaS 2007 A critical role for peptidoglycan N-deacetylation in Listeria evasion from the host innate immune system. Proc Natl Acad Sci U S A 104 997 1002
11. TabuchiYShiratsuchiAKurokawaKGongJHSekimizuK 2010 Inhibitory role for D-alanylation of wall teichoic acid in activation of insect Toll pathway by peptidoglycan of Staphylococcus aureus. J Immunol 185 2424 2431
12. KurokawaKGongJHRyuKHZhengLChaeJH 2011 Biochemical characterization of evasion from peptidoglycan recognition by Staphylococcus aureus D-alanylated wall teichoic acid in insect innate immunity. Dev Comp Immunol 35 835 839
13. FilipeSRTomaszALigoxygakisP 2005 Requirements of peptidoglycan structure that allow detection by the Drosophila Toll pathway. EMBO Rep 6 327 333
14. WangLGilbertRJAtilanoMLFilipeSRGayNJ 2008 Peptidoglycan recognition protein-SD provides versatility of receptor formation in Drosophila immunity. Proc Natl Acad Sci U S A 105 11881 11886
15. BischoffVVignalCBonecaIGMichelTHoffmannJA 2004 Function of the drosophila pattern-recognition receptor PGRP-SD in the detection of Gram-positive bacteria. Nat Immunol 5 1175 1180
16. MichelTReichhartJMHoffmannJARoyetJ 2001 Drosophila Toll is activated by Gram-positive bacteria through a circulating peptidoglycan recognition protein. Nature 414 756 759
17. GobertVGottarMMatskevichAARutschmannSRoyetJ 2003 Dual activation of the Drosophila toll pathway by two pattern recognition receptors. Science 302 2126 2130
18. LemaitreBHoffmannJ 2007 The host defense of Drosophila melanogaster. Annu Rev Immunol 25 697 743
19. WangLWeberANAtilanoMLFilipeSRGayNJ 2006 Sensing of Gram-positive bacteria in Drosophila: GNBP1 is needed to process and present peptidoglycan to PGRP-SA. EMBO J 25 5005 5014
20. KashyapDRWangMLiuLHBoonsGJGuptaD 2011 Peptidoglycan recognition proteins kill bacteria by activating protein-sensing two-component systems. Nat Med 17 676 683
21. SchlagMBiswasRKrismerBKohlerTZollS 2010 Role of staphylococcal wall teichoic acid in targeting the major autolysin Atl. Mol Microbiol 75 864 873
22. GrundlingAMissiakasDMSchneewindO 2006 Staphylococcus aureus mutants with increased lysostaphin resistance. J Bacteriol 188 6286 6297
23. SteenABuistGLeenhoutsKJEl KhattabiMGrijpstraF 2003 Cell wall attachment of a widely distributed peptidoglycan binding domain is hindered by cell wall constituents. J Biol Chem 278 23874 23881
24. DavisonALBaddileyJ 1963 The Distribution of Teichoic Acids in Staphylococci. J Gen Microbiol 32 271 276
25. SwobodaJGCampbellJMeredithTCWalkerS 2010 Wall teichoic acid function, biosynthesis, and inhibition. Chembiochem 11 35 45
26. WangYHuebnerJTzianabosAOMartirosianGKasperDL 1999 Structure of an antigenic teichoic acid shared by clinical isolates of Enterococcus faecalis and vancomycin-resistant Enterococcus faecium. Carbohydr Res 316 155 160
27. SaltonMRJ 1994 The bacterial cell envelope - a historical perspective. GhuysenJ-MHakenbeckR Bacterial Cell Wall Elsevier 1 22
28. CampbellJSinghAKSanta MariaJPJrKimYBrownS 2011 Synthetic lethal compound combinations reveal a fundamental connection between wall teichoic acid and peptidoglycan biosyntheses in Staphylococcus aureus. ACS Chem Biol 6 106 116
29. AtilanoMLPereiraPMYatesJReedPVeigaH 2010 Teichoic acids are temporal and spatial regulators of peptidoglycan cross-linking in Staphylococcus aureus. Proc Natl Acad Sci U S A 107 18991 18996
30. BrownSZhangYHWalkerS 2008 A revised pathway proposed for Staphylococcus aureus wall teichoic acid biosynthesis based on in vitro reconstitution of the intracellular steps. Chem Biol 15 12 21
31. ArcherGL 1998 Staphylococcus aureus: a well-armed pathogen. Clin Infect Dis 26 1179 1181
32. GalacMRLazzaroBP 2011 Comparative pathology of bacteria in the genus Providencia to a natural host, Drosophila melanogaster. Microbes Infect 13 673 683
33. PeschelAOttoMJackRWKalbacherHJungG 1999 Inactivation of the dlt operon in Staphylococcus aureus confers sensitivity to defensins, protegrins, and other antimicrobial peptides. J Biol Chem 274 8405 8410
34. BeraABiswasRHerbertSKulauzovicEWeidenmaierC 2007 Influence of wall teichoic acid on lysozyme resistance in Staphylococcus aureus. J Bacteriol 189 280 283
35. DziarskiRGuptaD 2006 The peptidoglycan recognition proteins (PGRPs). Genome Biol 7 232
36. ChangCIPili-FlourySHervéMParquetCChelliahY 2004 A Drosophila pattern recognition receptor contains a peptidoglycan docking groove and unusual L,D-carboxypeptidase activity. PLoS Biol 2 e277
37. WangLWeberANAtilanoMLFilipeSRGayNJ 2006 Sensing of Gram-positive bacteria in Drosophila:GNBP1 is needed to process and present peptidoglycan to PGRP-SA. EMBO J 25 5005 14
38. ParkJWJeBRPiaoSInamuraSFujimotoY 2006 A synthetic peptidoglycan fragment as a competitive inhibitor of the melanization cascade. J Biol Chem 281 7747 7755
39. LeonePBischoffVKellenbergerCHetruCRoyetJ 2008 Crystal structure of Drosophila PGRP-SD suggests binding to DAP-type but not lysine-type peptidoglycan. Mol Immunol 45 2521 2530
40. WeidenmaierCKokai-KunJFKulauzovicEKohlerTThummG 2008 Differential roles of sortase-anchored surface proteins and wall teichoic acid in Staphylococcus aureus nasal colonization. Int J Med Microbiol 298 505 513
41. SuzukiTCampbellJSwobodaJGWalkerSGilmoreMS 2011 Role of wall teichoic acids in Staphylococcus aureus endophthalmitis. Invest Ophthalmol Vis Sci 52 3187 3192
42. FerrandonDJungACCriquiMLemaitreBUttenweiler-JosephS 1998 A drosomycin-GFP reporter transgene reveals a local immune response in Drosophila that is not dependent on the Toll pathway. EMBO J 17 1217 1227
43. LemaitreBNicolasEMichautLReichhartJMHoffmannJA 1996 The dorsoventral regulatory gene cassette spatzle/Toll/cactus controls the potent antifungal response in Drosophila adults. Cell 86 973 983
44. LindsleyDLZimmGG 1992 The Genome of Drosophila melanogaster. San Diego Academic Press
45. RutschmannSKilincAFerrandonD 2002 Cutting edge: the toll pathway is required for resistance to gram-positive bacterial infections in Drosophila. J Immunol 168 1542 1546
46. Vergara-IrigarayMMaira-LitranTMerinoNPierGBPenadesJR 2008 Wall teichoic acids are dispensable for anchoring the PNAG exopolysaccharide to the Staphylococcus aureus cell surface. Microbiology 154 865 877
47. WieserMDennerEBKampferPSchumannPTindallB 2002 Emended descriptions of the genus Micrococcus, Micrococcus luteus (Cohn 1872) and Micrococcus lylae (Kloos, et al. 1974). Int J Syst Evol Microbiol 52 629 637
48. JacobAEHobbsSJ 1974 Conjugal transfer of plasmid-borne multiple antibiotic resistance in Streptococcus faecalis var. zymogenes. J Bacteriol 117 360 372
49. HenriquesAOGlaserPPiggotPJMoranCPJr 1998 Control of cell shape and elongation by the rodA gene in Bacillus subtilis. Mol Microbiol 28 235 247
50. GlittenbergMTSilasSMacCallumDMGowNALigoxygakisP 2011 Wild-type Drosophila melanogaster as an alternative model system for investigating the pathogenicity of Candida albicans. Dis Model Mech 4 504 514
51. AbramoffMMagalhaesPRamS 2004 Image processing with ImageJ. Biophoton Int 11 36 42
52. MeredithTCSwobodaJGWalkerS 2008 Late-stage polyribitol phosphate wall teichoic acid biosynthesis in Staphylococcus aureus. J Bacteriol 190 3046 3056
Štítky
Hygiena a epidemiologie Infekční lékařství Laboratoř
Článek Genesis of Mammalian Prions: From Non-infectious Amyloid Fibrils to a Transmissible Prion DiseaseČlánek Role of Permissive Neuraminidase Mutations in Influenza A/Brisbane/59/2007-like (H1N1) VirusesČlánek Allelic Variation on Murine Chromosome 11 Modifies Host Inflammatory Responses and Resistance toČlánek Multifaceted Regulation of Translational Readthrough by RNA Replication Elements in a TombusvirusČlánek Latent KSHV Infection of Endothelial Cells Induces Integrin Beta3 to Activate Angiogenic PhenotypesČlánek Controlling Viral Immuno-Inflammatory Lesions by Modulating Aryl Hydrocarbon Receptor Signaling
Článek vyšel v časopisePLOS Pathogens
Nejčtenější tento týden
2011 Číslo 12- Perorální antivirotika jako vysoce efektivní nástroj prevence hospitalizací kvůli COVID-19 − otázky a odpovědi pro praxi
- Mozkové metastázy nejsou absolutní kontraindikací antiagregační léčby
- Parazitičtí červi v terapii Crohnovy choroby a dalších zánětlivých autoimunitních onemocnění
- Vakcíny proti klíšťové encefalitidě
- Kdy je nejlepší očkovat
-
Všechny články tohoto čísla
- Inhibition of Apoptosis and NF-κB Activation by Vaccinia Protein N1 Occur via Distinct Binding Surfaces and Make Different Contributions to Virulence
- Genesis of Mammalian Prions: From Non-infectious Amyloid Fibrils to a Transmissible Prion Disease
- Kaposi's Sarcoma Herpesvirus microRNAs Target Caspase 3 and Regulate Apoptosis
- Nutritional Immunology: A Multi-Dimensional Approach
- Role of Permissive Neuraminidase Mutations in Influenza A/Brisbane/59/2007-like (H1N1) Viruses
- Vaccinomics and Personalized Vaccinology: Is Science Leading Us Toward a New Path of Directed Vaccine Development and Discovery?
- Symbiont Infections Induce Strong Cytoplasmic Incompatibility in the Tsetse Fly
- Allelic Variation on Murine Chromosome 11 Modifies Host Inflammatory Responses and Resistance to
- Computational and Biochemical Analysis of the Effector AvrBs2 and Its Role in the Modulation of Type Three Effector Delivery
- Granzyme B Inhibits Vaccinia Virus Production through Proteolytic Cleavage of Eukaryotic Initiation Factor 4 Gamma 3
- Association of Activating KIR Copy Number Variation of NK Cells with Containment of SIV Replication in Rhesus Monkeys
- Fungal Virulence and Development Is Regulated by Alternative Pre-mRNA 3′End Processing in
- versus the Host: Remodeling of the Bacterial Outer Membrane Is Required for Survival in the Gastric Mucosa
- Follicular Dendritic Cell-Specific Prion Protein (PrP) Expression Alone Is Sufficient to Sustain Prion Infection in the Spleen
- Autophagy Protein Atg3 is Essential for Maintaining Mitochondrial Integrity and for Normal Intracellular Development of Tachyzoites
- Longevity and Composition of Cellular Immune Responses Following Experimental Malaria Infection in Humans
- Sequential Adaptive Mutations Enhance Efficient Vector Switching by Chikungunya Virus and Its Epidemic Emergence
- Acquisition of Pneumococci Specific Effector and Regulatory Cd4 T Cells Localising within Human Upper Respiratory-Tract Mucosal Lymphoid Tissue
- The Meaning of Death: Evolution and Ecology of Apoptosis in Protozoan Parasites
- Deficiency of a Niemann-Pick, Type C1-related Protein in Is Associated with Multiple Lipidoses and Increased Pathogenicity
- Feeding Cells Induced by Phytoparasitic Nematodes Require γ-Tubulin Ring Complex for Microtubule Reorganization
- Eight RGS and RGS-like Proteins Orchestrate Growth, Differentiation, and Pathogenicity of
- Prion Uptake in the Gut: Identification of the First Uptake and Replication Sites
- Nef Decreases HIV-1 Sensitivity to Neutralizing Antibodies that Target the Membrane-proximal External Region of TMgp41
- Multifaceted Regulation of Translational Readthrough by RNA Replication Elements in a Tombusvirus
- A Temporal Role Of Type I Interferon Signaling in CD8 T Cell Maturation during Acute West Nile Virus Infection
- The Membrane Fusion Step of Vaccinia Virus Entry Is Cooperatively Mediated by Multiple Viral Proteins and Host Cell Components
- HIV-1 Capsid-Cyclophilin Interactions Determine Nuclear Import Pathway, Integration Targeting and Replication Efficiency
- Neonatal CD8 T-cell Hierarchy Is Distinct from Adults and Is Influenced by Intrinsic T cell Properties in Respiratory Syncytial Virus Infected Mice
- Two Novel Transcriptional Regulators Are Essential for Infection-related Morphogenesis and Pathogenicity of the Rice Blast Fungus
- Five Questions about Non-Mevalonate Isoprenoid Biosynthesis
- The Human Cytomegalovirus UL11 Protein Interacts with the Receptor Tyrosine Phosphatase CD45, Resulting in Functional Paralysis of T Cells
- Wall Teichoic Acids of Limit Recognition by the Drosophila Peptidoglycan Recognition Protein-SA to Promote Pathogenicity
- A Novel Role for the NLRC4 Inflammasome in Mucosal Defenses against the Fungal Pathogen
- Inflammasome-dependent Pyroptosis and IL-18 Protect against Lung Infection while IL-1β Is Deleterious
- CNS Recruitment of CD8+ T Lymphocytes Specific for a Peripheral Virus Infection Triggers Neuropathogenesis during Polymicrobial Challenge
- Latent KSHV Infection of Endothelial Cells Induces Integrin Beta3 to Activate Angiogenic Phenotypes
- A Receptor-based Switch that Regulates Anthrax Toxin Pore Formation
- Targeting of Heparin-Binding Hemagglutinin to Mitochondria in Macrophages
- Chikungunya Virus Neutralization Antigens and Direct Cell-to-Cell Transmission Are Revealed by Human Antibody-Escape Mutants
- Ce-Duox1/BLI-3 Generated Reactive Oxygen Species Trigger Protective SKN-1 Activity via p38 MAPK Signaling during Infection in
- Structural Elucidation and Functional Characterization of the Effector Protein ATR13
- Controlling Viral Immuno-Inflammatory Lesions by Modulating Aryl Hydrocarbon Receptor Signaling
- SAMHD1-Deficient CD14+ Cells from Individuals with Aicardi-Goutières Syndrome Are Highly Susceptible to HIV-1 Infection
- Acid Stability of the Hemagglutinin Protein Regulates H5N1 Influenza Virus Pathogenicity
- Cryo Electron Tomography of Herpes Simplex Virus during Axonal Transport and Secondary Envelopment in Primary Neurons
- A Novel Human Cytomegalovirus Locus Modulates Cell Type-Specific Outcomes of Infection
- Juxtamembrane Shedding of AMA1 Is Sequence Independent and Essential, and Helps Evade Invasion-Inhibitory Antibodies
- Pathogenesis and Host Response in Syrian Hamsters following Intranasal Infection with Andes Virus
- IRGM Is a Common Target of RNA Viruses that Subvert the Autophagy Network
- Epstein-Barr Virus Evades CD4 T Cell Responses in Lytic Cycle through BZLF1-mediated Downregulation of CD74 and the Cooperation of vBcl-2
- Quantitative Multicolor Super-Resolution Microscopy Reveals Tetherin HIV-1 Interaction
- Late Repression of NF-κB Activity by Invasive but Not Non-Invasive Meningococcal Isolates Is Required to Display Apoptosis of Epithelial Cells
- Polar Flagellar Biosynthesis and a Regulator of Flagellar Number Influence Spatial Parameters of Cell Division in
- Epstein-Barr Virus Nuclear Antigen 3C Stabilizes Gemin3 to Block p53-mediated Apoptosis
- The Enteropathogenic (EPEC) Tir Effector Inhibits NF-κB Activity by Targeting TNFα Receptor-Associated Factors
- Toward an Integrated Model of Capsule Regulation in
- A Systematic Screen to Discover and Analyze Apicoplast Proteins Identifies a Conserved and Essential Protein Import Factor
- A Host Small GTP-binding Protein ARL8 Plays Crucial Roles in Tobamovirus RNA Replication
- Comparative Pathobiology of Fungal Pathogens of Plants and Animals
- Synergistic Roles of Eukaryotic Translation Elongation Factors 1Bγ and 1A in Stimulation of Tombusvirus Minus-Strand Synthesis
- Engineered Immunity to Infection
- Inflammatory Monocytes and Neutrophils Are Licensed to Kill during Memory Responses
- Sialidases Affect the Host Cell Adherence and Epsilon Toxin-Induced Cytotoxicity of Type D Strain CN3718
- Eurasian-Origin Gene Segments Contribute to the Transmissibility, Aerosol Release, and Morphology of the 2009 Pandemic H1N1 Influenza Virus
- SARS Coronavirus nsp1 Protein Induces Template-Dependent Endonucleolytic Cleavage of mRNAs: Viral mRNAs Are Resistant to nsp1-Induced RNA Cleavage
- Identification and Characterization of a Novel Non-Structural Protein of Bluetongue Virus
- Functional Analysis of the Kinome of the Wheat Scab Fungus
- Norovirus Regulation of the Innate Immune Response and Apoptosis Occurs via the Product of the Alternative Open Reading Frame 4
- PLOS Pathogens
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Controlling Viral Immuno-Inflammatory Lesions by Modulating Aryl Hydrocarbon Receptor Signaling
- Fungal Virulence and Development Is Regulated by Alternative Pre-mRNA 3′End Processing in
- Cryo Electron Tomography of Herpes Simplex Virus during Axonal Transport and Secondary Envelopment in Primary Neurons
- Epstein-Barr Virus Nuclear Antigen 3C Stabilizes Gemin3 to Block p53-mediated Apoptosis
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Autoři: prof. MUDr. Pavel Horák, CSc., doc. MUDr. Ludmila Brunerová, Ph.D., doc. MUDr. Václav Vyskočil, Ph.D., prim. MUDr. Richard Pikner, Ph.D., MUDr. Olga Růžičková, MUDr. Jan Rosa, prof. MUDr. Vladimír Palička, CSc., Dr.h.c.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



