-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praxe
Tsetse EP Protein Protects the Fly Midgut from Trypanosome Establishment
African trypanosomes undergo a complex developmental process in their tsetse fly vector before transmission back to a vertebrate host. Typically, 90% of fly infections fail, most during initial establishment of the parasite in the fly midgut. The specific mechanism(s) underpinning this failure are unknown. We have previously shown that a Glossina-specific, immunoresponsive molecule, tsetse EP protein, is up regulated by the fly in response to gram-negative microbial challenge. Here we show by knockdown using RNA interference that this tsetse EP protein acts as a powerful antagonist of establishment in the fly midgut for both Trypanosoma brucei brucei and T. congolense. We demonstrate that this phenomenon exists in two species of tsetse, Glossina morsitans morsitans and G. palpalis palpalis, suggesting tsetse EP protein may be a major determinant of vector competence in all Glossina species. Tsetse EP protein levels also decline in response to starvation of the fly, providing a possible explanation for increased susceptibility of starved flies to trypanosome infection. As starvation is a common field event, this fact may be of considerable importance in the epidemiology of African trypanosomiasis.
Published in the journal: . PLoS Pathog 6(3): e32767. doi:10.1371/journal.ppat.1000793
Category: Research Article
doi: https://doi.org/10.1371/journal.ppat.1000793Summary
African trypanosomes undergo a complex developmental process in their tsetse fly vector before transmission back to a vertebrate host. Typically, 90% of fly infections fail, most during initial establishment of the parasite in the fly midgut. The specific mechanism(s) underpinning this failure are unknown. We have previously shown that a Glossina-specific, immunoresponsive molecule, tsetse EP protein, is up regulated by the fly in response to gram-negative microbial challenge. Here we show by knockdown using RNA interference that this tsetse EP protein acts as a powerful antagonist of establishment in the fly midgut for both Trypanosoma brucei brucei and T. congolense. We demonstrate that this phenomenon exists in two species of tsetse, Glossina morsitans morsitans and G. palpalis palpalis, suggesting tsetse EP protein may be a major determinant of vector competence in all Glossina species. Tsetse EP protein levels also decline in response to starvation of the fly, providing a possible explanation for increased susceptibility of starved flies to trypanosome infection. As starvation is a common field event, this fact may be of considerable importance in the epidemiology of African trypanosomiasis.
Introduction
African trypanosomes are protozoan parasites that cause sleeping sickness in humans and nagana in domestic livestock in sub-Saharan Africa. An epidemic involving several hundred thousand people that spread through Sudan, the Central African Republic, DRC and Angola in the 1990's, demonstrated how socially and economically devastating these diseases are [1]. Trypanosomes kill more than 3 million cattle annually and those animals that survive display low productivity due to the wasting effects of the disease [2]. The annual losses from trypanosomiasis in cattle amount to more than US $4.5 billion [3]. Trypanosomes, by influencing food production, natural resource utilization and the pattern of human settlement, are thus seen by the African Union as one of the greatest constraints to Africa's socio-economic development [4].
African trypanosomes are cyclically transmitted by tsetse flies (Glossina spp.). Trypanosoma brucei and T. congolense undergo a complex cycle of development in the tsetse beginning almost immediately after ingestion of an infected bloodmeal when trypanosome bloodstream forms (BSF) differentiate to the procyclic form in the fly midgut lumen [5],[6],[7]. For the first three days following infection all flies contain trypanosomes. Between days 4 and 5 trypanosome infections are eliminated from most flies [7] through a process we term self-cure. The identified factors that influence vector competence (the ability to transmit parasites) include the age of the fly, the number of bloodmeals taken and the activation of fly immune processes, with both antimicrobial (host defense) peptides [8], and lectins [9],[10],[11] implicated in parasite-vector interactions. More recently, antioxidants have been shown to increase fly susceptibility when administered to flies in an infective bloodmeal [12]. Most mature tsetse are resistant to trypanosome infection although the mechanisms involved in elimination of trypanosomes from the fly midgut (self-cure) are not understood [13].
As T. brucei BSF trypanosomes transform in the tsetse midgut the trypanosome surface coat changes from variant surface glycoproteins (VSG) to procyclins. At first the procyclins are a mixture of GPEET and EP forms and then expression of GPEET becomes repressed [14]. Our attention has been drawn to a fly protein called tsetse EP (accession number CAC86027), named for the extensive glutamic acid-proline dipeptide repeats that in Glossina morsitans morsitans comprise more than 40% of its length. The repeat section of this molecule shows remarkable sequence identity to the repeat section of the EP form of procyclin surface coat molecules of T. b. brucei [14]. These repeats are very rare in the protein databases and their co-incidence in two species showing such a close biological relationship is remarkable. Our knowledge of tsetse EP is limited although we do know that it is strongly up regulated following fly challenge with Gram-negative bacteria [15] suggesting a possible function in the insect immune response. In addition up regulation of the immune response by injection of E. coli also leads to a significant reduction in trypanosome prevalence [8],[16]. For these reasons we have undertaken a series of experiments to see if these observations are connected. We provide evidence that tsetse EP protein has a powerful role in protecting the tsetse fly midgut from trypanosome infection.
Results
Where is tsetse EP protein produced within the midgut of the fly?
Tsetse midguts were carefully dissected into distinct structural regions (Figure 1, Panel A) to determine the location of tsetse EP mRNA and protein. Tsetse EP transcripts were detected in all sections tested. However, lower levels were consistently observed in the proventriculus (PV) (Figure 1, Panel B). The Western blot (Figure 1, Panel C) overlay of the nigrosine-stained PVDF with the autoluminogram revealed the strong presence of tsetse EP protein in all tissues except for the PV. Given the presence of tsetse EP transcript in the PV we conclude that tsetse EP protein was either not produced in PV, was rapidly turned over in that organ or was rapidly translocated from there into the anterior gut. Similarly, tsetse EP transcript was detected in salivary glands from teneral and fed flies [17] but tsetse EP protein was only weakly detected by immunoblotting suggesting it may be rapidly translocated to midgut.
Fig. 1. Levels of tsetse EP transcripts and proteins in sectioned midguts were determined by semi-quantitative RT-PCR and immunoblotting. 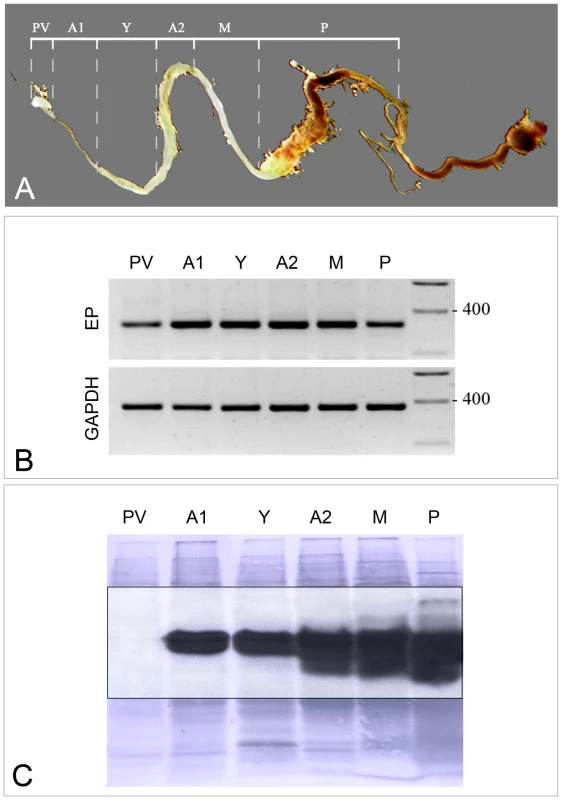
(A) Photograph of tsetse midgut depicting dissection lines. (B) Transcript profiles of midgut sections probed with tsetse EP and GAPDH (control) primers. (C) Immunoblot of midgut tissues (collected from a pool of five male G. m. morsitans) using mAb 247 (anti-EP repeat). PV = proventriculus; A1 = midgut anterior to the bacteriome; Y = bacteriome; A2 = anterior midgut posterior to the bacteriome; M = mid-midgut; P = posterior midgut. Design of dsRNA for tsetse EP knockdown
Tsetse EP protein appears to be ubiquitous in Glossina spp. as its presence was confirmed in eight species of tsetse previously examined by Western blotting with the anti-EP repeat antibody (mAb 247) [15]. Using the available sequence analysis of the amino acid sequence of tsetse EP protein [15],[17] we designed effective double stranded RNA for knockdown experiments (Figure 2). A protein sequence comparison (87% similarity) between two species (G. m. morsitans and G. p. palpalis) revealed that the outstanding sequence difference was in the length of the C-terminal EP repeat region [15]. The tsetse EP protein is probably a preproprotein containing a short (19 mer) hydrophobic, N-terminal signal sequence as predicted by SignalP 3.0 [18]. Amino acids 20–48 appear to be removed from the remaining peptide during an undefined maturation process as determined by mass spectrometry and N-terminal sequencing [17],[19]. The EP rich domain is extremely hydrophilic, and thus almost certainly is highly soluble in aqueous solvents. It is interesting that all 8 of the cysteine residues are situated up stream of the EP rich C-terminus, suggesting that this region may be highly folded. For our experiments, we designed dsRNA to target in RNA interference the homologous region 23 residues downstream from the N-terminus of the mature protein (Figure 2, red highlighted region: GKFASDKCAQEGQ). The dsRNA target varies only slightly between G. m. morsitans and G. p. palpalis (4/39 nucleotides differ and these are shown in yellow lettering in Figure 2). Consequently the same dsRNA construct was used to achieve gene knockdown in both species.
Fig. 2. G. m. morsitans tsetse EP protein. 
The top panel is a schematic of the tsetse EP protein. SP = signal peptide. R AGT indicates the N-terminus (tryptic cleavage site) resulting in the mature protein. The dark blue represents the mature protein and the highlight red region (bracketed with arrows) indicates the target region for RNAi. In the sequence below, the predicted signal peptide is shown in lower case. |A indicates the N-terminus (tryptic cleavage site) resulting in the mature protein. The grey highlighted section indicates the region amplified for RNA interference. The residues in yellow indicate sequence differences in the G. p. palpalis sequence when compared with the sequence of G. m. morsitans. The monoclonal antibody, mAb 247, will recognize EPEPEP, and thus will bind along the entire length of the C-terminal rich region (E195–P306). S* indicates potential O-glycosylation sites. Tsetse EP protein levels are greatly reduced following gene knockdown by RNA interference
During RNAi experiments mRNA levels are often extrapolated to predict protein expression levels. However, this is often misleading as the correlation between transcript abundance and protein expression levels can often vary as much as 30 fold or more, leading to a grossly distorted analysis of a biological system [20] and this may be especially true in the midgut of blood sucking insects where post-transcriptional regulation may be a common phenomenon [21]. Consequently, we measured tsetse EP levels at both the mRNA and protein levels. We show that injection of dsRNA leads to significant reductions in transcript levels compared to controls (Figure 3A). In addition, immunoblot analysis using the anti-EP repeat monoclonal antibody (mAb247) to detect the tsetse EP protein in midguts of knockdown flies showed complete elimination of the endogenous protein following a single injection of 4 or more µg of dsRNA (Figure 3B).
Fig. 3. Tsetse EP mRNA and protein levels following injection of tsetse with dstsetseEP. 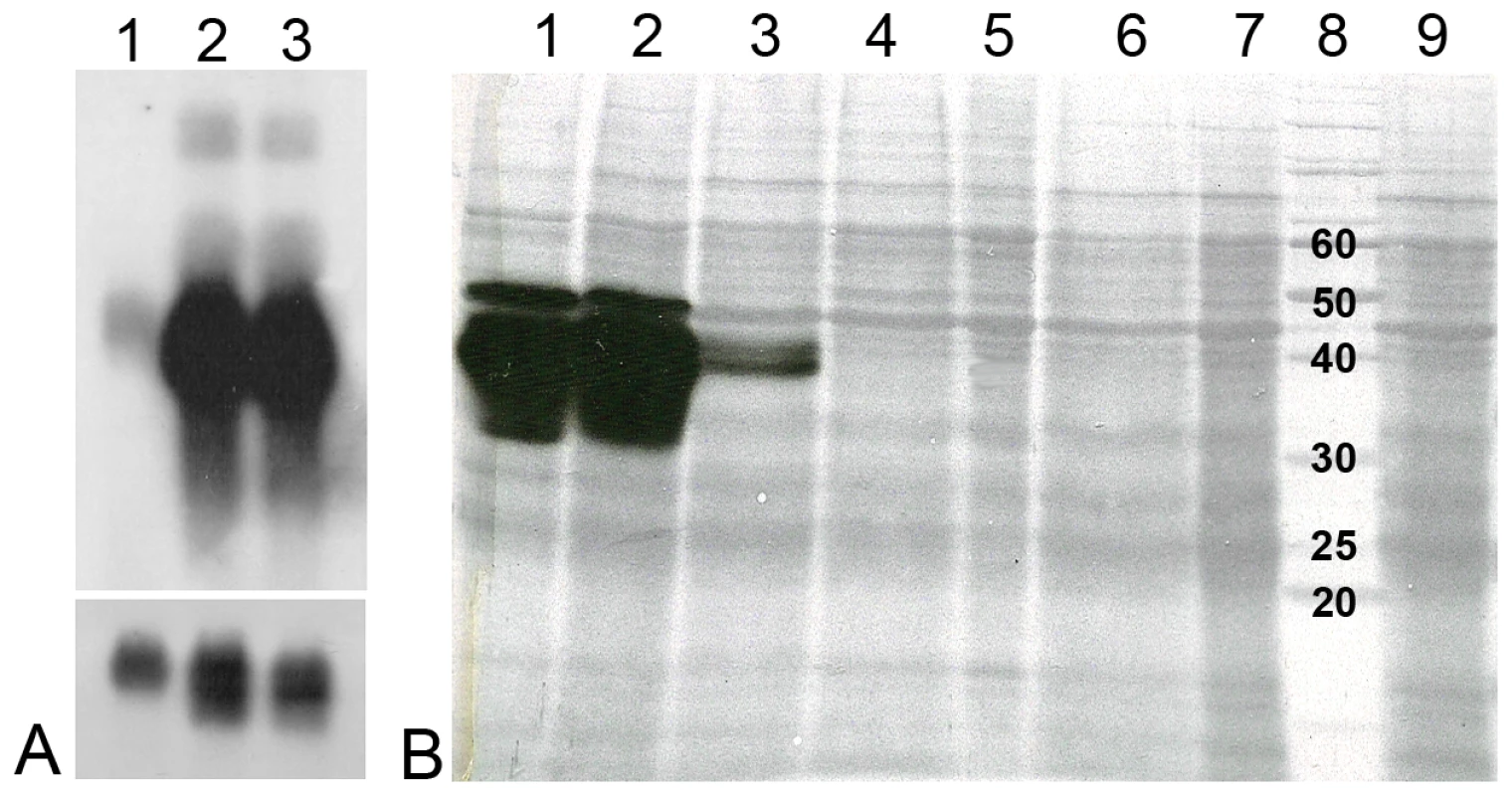
Teneral flies were injected with dsRNA, fed two bloodmeals and then starved for three days prior to dissection. Five midguts were pooled for each sample and the quantity of RNA and protein per lane was adjusted accordingly. (Panel A) Northern blot: lane 1: dstsetse EP knockdown midguts; lane 2: NFW injected midguts; lane 3: control midgut tissue from non-injected flies. The lower section represents GAPDH mRNA loading controls. (Panel B) Immunoblot: Gel-separated midgut proteins (1/2 midgut equivalent) isolated from male G. m. morsitans injected with 2 µL of dsRNA were separated on a 12.5% gel and blotted with mAb 247 (anti-EP repeat). Lane 1: NFW control, lane 2: 8 µg dsAmp control, lane 3: 2 µg dstsetseEP, lane 4: 4 µg dstsetseEP, lane 5: 6 µg dstsetseEP, lane 6: 7 µg dstsetseEP, lane 7: 8 µg dstsetseEP, lane 8: 10 kDa molecular mass ladder, lane 9: 10 µg dstsetseEP. Parasite infection is increased by RNAi-mediated knockdown of tsetse EP protein
We employed a reverse genetics approach to determine if tsetse EP influences parasite establishment in the midgut of the fly. We injected double-stranded RNA (dsRNA) into the thoracic haemocoel of male flies of different ages. Typically the flies were allowed to recover for 36–48 h after injecting dsRNA. This provides enough time for the dsRNA to start silencing tsetse EP protein transcription and for endogenous protein levels to decline [22]. After this point, flies were offered an infective bloodmeal containing virulent strains of either T. b. brucei (TSW196) or T. congolense (1/148) BSF. Seven days after the infectious meal the midguts were dissected, examined microscopically, snap frozen, and the number of infections was recorded (Table 1). A complicating feature of this insect system is a natural decrease in susceptibility in older flies termed the teneral phenomenon. Typically more than 50% of flies establish midgut infections when fed trypanosomes in the first bloodmeal. However, if infected in the second bloodmeal, this susceptibility declines to ∼30% of the population. By the third bloodmeal, tsetse populations are predominantly refractory to infection with typical midgut establishment rates of 10% or less (Table 1). So, we investigated flies with differing feeding histories. Tsetse EP knockdown flies, infected at all feeding time points investigated, showed statistically significant increases in susceptibility to T. b. brucei establishment in the midgut when compared to the controls (Table 1).
Tab. 1. Mean (± S.E.) prevalence of midgut infections in male G. m. morsitans (Gmm) or G. p. palpalis (Gpp) after RNAi knockdown using dstsetse EP. 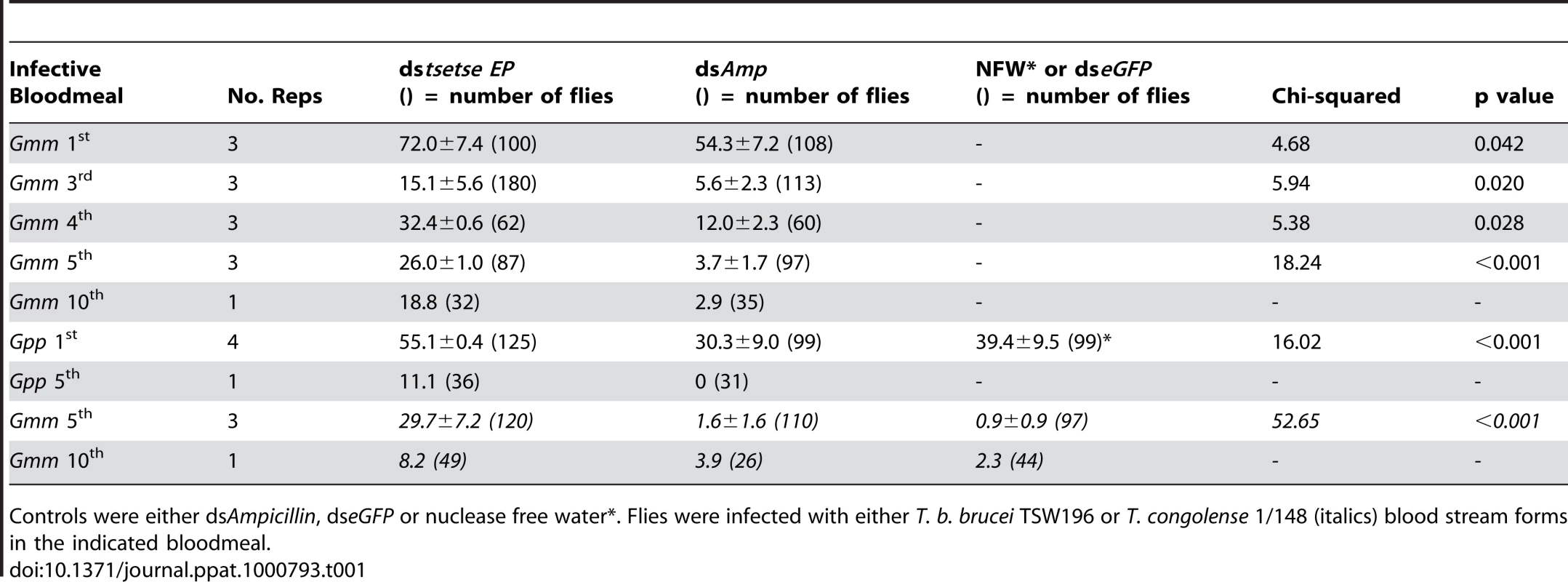
Controls were either dsAmpicillin, dseGFP or nuclease free water*. Flies were infected with either T. b. brucei TSW196 or T. congolense 1/148 (italics) blood stream forms in the indicated bloodmeal. To determine if this phenomenon was present in other tsetse species we also investigated G. p. palpalis. Table 1 shows there are statistically significant increases in T. b. brucei establishment in the midgut of tsetse EP knockdown G. p. palpalis. We also conducted experiments to determine if the phenomenon extending to other trypanosome species. T. congolense also establishes higher midgut infections in EP knockdown flies (Table 1). Based on our current and previous [15] observations, the increase of vector competence to midgut inhabiting trypanosomes in tsetse EP knockdown flies is possibly a genus-wide phenomenon in Glossina.
Tsetse EP protein levels decrease with fly starvation
Male tsetse received 5 bloodmeals prior to starvation. Flies were killed at 24 h time points, up to 7 days after the last blood meal, and individual midguts were assayed by immunoblotting using an anti-EP antibody (mAb247) (Figure 4). After 3 days of starvation a clear decline in tsetse EP protein levels is evident (Figure 4, asterisks). Fat body atrophy was also apparent in these flies when viewed with a dissection microscope. Tsetse EP protein levels increase again in flies 24 hours after feeding following a previous starvation period of 7 days (Figure 4, lane 8). We have no data to show if the starvation-induced decrease in tsetse EP protein is specific or part of a general lowering of protein levels in the midgut in response to starvation.
Fig. 4. Immunoblot film lies below the nigrosine-stained PVDF. 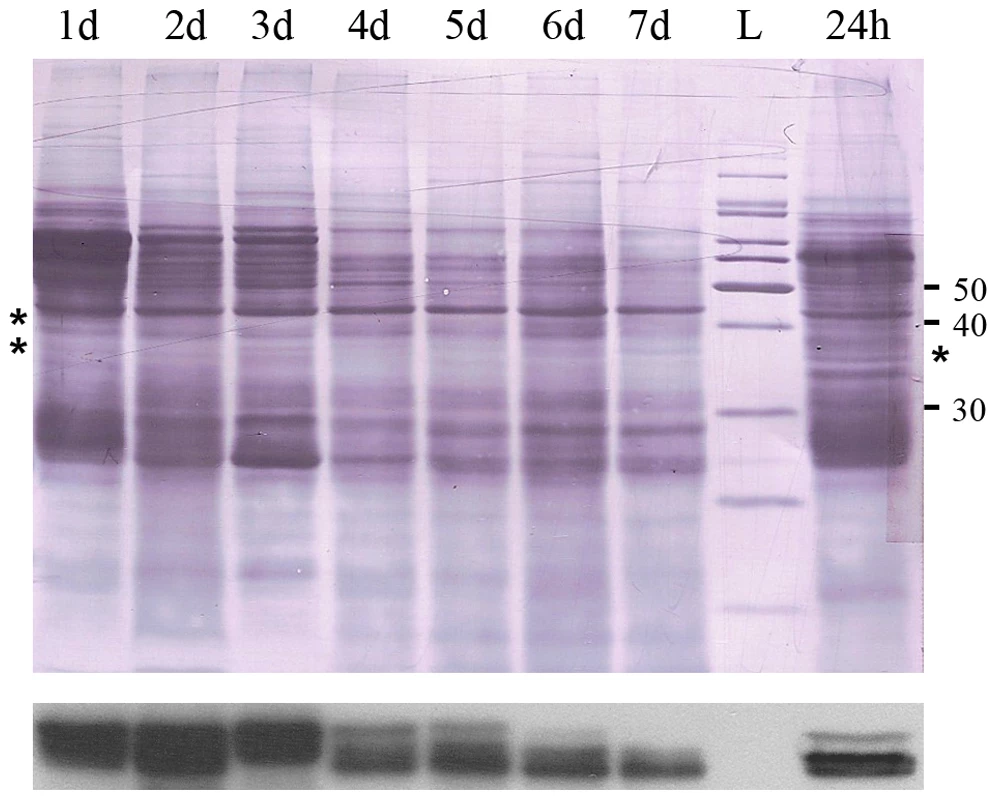
There is a decline in tsetse EP protein levels in midguts over a 7 day starvation period following the 5th bloodmeal. 24 h = Flies starved for 7 days, fed a blood meal and then sacrificed 24 hours later. L = molecular mass ladder. Midgut proteins (1/2 midgut equivalent from pool of 5) were blotted with mAb 247. Discussion
Although RNA interference is an exquisite genetic technique to knockdown target genes, the success in achieving this post-transcriptional silencing appears to be gene-specific with variability due, in part, to the half-life of endogenous target protein and unexpected lethal secondary effects from depletion of gene specific product [23]. Our unpublished observations in Glossina reveal that, for some genes, transcript knockdown cannot be achieved regardless of the construct designed. This may relate to the lack of a spreading mechanism in Diptera and the difficulty of dsRNA reaching cells in complex organs [22]. We have previously shown and confirm here that thoracic injections of microgram amounts of specific dsRNA can effectively depress tsetse EP transcription in the tsetse midgut for up to 2 weeks [22]. Thus, the effect of persistent tsetse EP knockdown on trypanosome midgut establishment (7–10 day experiment) could be confidently measured by microscopic examination. The data we present here shows that a tsetse molecule, tsetse EP protein, plays a role in protecting the midgut from infection with trypanosomes.
Computer analysis of the translated protein sequences from both G. m. morsitans (CAC86027) and G. p. palpalis (AAL82540), using multiple alignment tools and protein prediction algorithms, revealed that these proteins are highly conserved [15]. Including its signal peptide, tsetse EP protein from G. m. morsitans has a mass of 35.7 kDa and appears to form dimers and trimers and potentially larger oligomeric aggregates within the fly [15],[17]. Apart from the EP sequence the tsetse EP protein has no currently defined protein domains [17]. However a possible clue to function may be suggested by the preliminary observation of weak agglutinating activity of the large molecular complex towards freshly collected, washed rabbit red blood cells, suggesting tsetse EP putatively has some lectin activity [17]. In addition it has been demonstrated that tsetse EP protein is strongly up regulated following immune stimulation with E. coli [15] providing good evidence that it is part of the immune response system. Given this it is interesting to note that the Imd immune regulatory pathway mainly responds to gram negative organisms [24] and the Imd pathway has been implicated in the response of dipterans to parasite infections [16],[25].
Although all species of tsetse studied to date express tsetse EP protein [15], orthologues are not found in the Anopheles, Aedes, Apis or Drosophila genomes. A search of non-redundant databases revealed only two eukaryotic protein hits (apart from the procyclins): gi|94390895 [Mus musculus] and gi|109464874 [Rattus norvegicus]. These hypothetical proteins contain significant continuous EP repeat regions: e.g. 115 dipeptide repeats, representing 75% coverage of the rat protein. Unfortunately, no further functional information is available for these proteins. Remarkably, extensive regions of EP repeats (also varying in length) are contained in several procyclins that form the surface coat of procyclic trypanosomes of the T. brucei group [14],[26],[27]. Given the scarcity of EP repeats in organisms the chances of this happening coincidentally in trypanosomes and tsetse flies seem remote.
To examine the possibility that the tsetse EP protein and the EP procyclins from T. b. brucei were involved in antigenic mimicry we investigated another trypanosome species that lacks EP procyclins. The procyclic coat of T. congolense contains no extensive dipeptidyl EP repeats although similar anionic motifs are present [28]. Despite the absence of EP repeats, establishment of T. congolense is similarly affected by tsetse EP protein knockdown (Table 1). Our experiments demonstrate that tsetse EP protein can partially protect against the midgut establishment of trypanosomes from both the Trypanozoon and Nannomonas group trypanosomes and thus, strictly sequence-specific interactions in tsetse and trypanosome are not likely at play.
To assess if trypanosome establishment is altered by tsetse EP gene knockdown in tsetse species other than our G. m. morsitans laboratory model, we tested our RNAi protocol on G. p. palpalis. Knockdown of tsetse EP protein in G. p. palpalis also led to an increase in midgut infections (Table 1), confirming that tsetse EP protein influences trypanosome midgut establishment in both of these major vectors of trypanosomiasis. Given that tsetse EP has been demonstrated in a wide variety of Glossina species [15] this data suggests it may be a genus wide phenomenon.
It has been demonstrated that up regulation of the immune response by injection of E. coli leads to a significant reduction in the ability of trypanosomes to establish in the tsetse midgut [8],[16]. We have already demonstrated that tsetse EP protein is strongly up regulated upon introduction of Gram-negative bacteria into the fly [15]. Our demonstration here that knockdown of tsetse EP leads to increased fly susceptibility suggests that upregulation of tsetse EP protein upon injection of E. coli may be one explanation for the subsequent decrease in the susceptibility of the fly to trypanosomes.
It is interesting to note that older flies in field populations show unexpectedly high levels of susceptibility compared to laboratory reared flies where susceptibility rapidly declines following eclosion [29],[30] (Table 1); the reasons remain unexplained. We have demonstrated here that starvation reduces tsetse EP levels in flies (Figure 4). It has already been demonstrated that starvation of mature flies results in an increase in parasite survival in the midgut [31],[32],[33]. Consequently, starvation, which is likely to be a common phenomenon in the field, could explain the differences in susceptibility seen between field and laboratory populations of flies. The observed reduction of tsetse EP protein expression and loss of parasite resistance upon starvation may have considerable epidemiological significance in African trypanosomiasis.
In summary, this paper provides direct evidence for a tsetse-specific midgut molecule (tsetse EP), which is an antagonist of trypanosome survival in the vector. RNAi-induced knockdown of the midgut-associated, immunoresponsive tsetse EP protein increased the frequency of trypanosome establishment in the fly midgut up to more than six fold. The precise mechanism by which tsetse EP protein influences the refractorial capacity of the midgut remains to be elucidated.
Materials and Methods
Flies and trypanosomes
Tsetse (G. m. morsitans) were maintained in laboratory colony at the Liverpool School of Tropical Medicine (LSTM) at 26°C and 65–70% relative humidity. Glossina palpalis palpalis were supplied as puparia from the International Atomic Energy Agency (IAEA) Entomology Laboratories, Siebersdorf, Austria. Every 48 hours, male flies were fed horse blood through silicone membranes. For infectious bloodmeals blood stream forms (BSF) of Trypanosoma brucei brucei TSW196 MSUS/CI/78/TSW196 [CLONE A], which is a fully fly-transmissible clone and able to undergo genetic exchange [34], and T. congolense 1/148 (Lister 1/148; isolated from a Zebu ox, Dongo River, Nigeria, Godfrey, 1960) were added to sterile defibrinated horse blood (TCS Biosciences Ltd., Buckingham, UK). Typically 200 µL of mouse blood (containing 4×106 parasites) were diluted in 5 mL of horse blood. Flies were dissected 6 days after the infectious bloodmeal. Midguts were dissected in saline on a glass slide and infection status determined by searching 10 random fields by light microscopy (125× magnification).
dsRNA
Double stranded RNA was transcribed using a MEGAscript High Yield T7 Transcription kit (Ambion, Huntingdon, UK). tsetseEP templates were available as clones from the tsetse EST program [35]. A double stranded fragment of the ampicillin resistance gene (dsAMP) was generated using pBluescript II SK+ as template. Template DNA was removed from the transcription reaction by DNase treatment and dsRNA was purified using MEGAclear™ columns (Ambion) and eluted in nuclease free water. Eluates were concentrated in a Christ (Osterode, Germany) 2–18 rotational vacuum concentrator to approximately 5 µg per µL. Primers were designed with the 20 base core T7 promoter sequence at the 5′ end. Primer sequences used were: AmpT7A TAATACGACTCACTATAGGGTTGCCGGGAAGCTAGAGTAAGTA; AmpT7B TAATACGACTCACTATAGGGAACGCTGGTGAAAGTAAAAGATG; EPT7A TAATACGACTCACTATAGGGTTCTGGCAAACCCTCAAT; EPT7B TAATACGACTCACTATAGGGCTACGATAAATATGTCCCTCTAAT.
Borosilicate glass capillaries (2.00 mm outside diameter) were formed into a fine point using a needle puller (PC10; Narishige, Japan). To generate tsetse EP knockdowns, male flies were anaesthetized by chilling and intrathoracically injected with 10 µg (2 µL volume) of dsRNA buffered in nuclease-free water.
RT-PCR
The primers used in semi-quantitative RT-PCR reactions for determination of transcript abundance in tsetse tissues were: Gm GAPDHA CTCAGCTTCTGTGCGTTG (Tm°C 67); Gm GAPDHB AGAGTGCCACCTACGATG (Tm°C 67); GmmEPA ACCGTTCGTTCGCTTTACTAC (Tm°C 47); GmmEPB ACCCGCAGCCGTTTGACTTTC (Tm°C 51).
Total RNA was extracted from individual tissues using Trizol (Invitrogen, Paisley UK) and treated with RQ1 RNase-Free DNase. RNA was quantified using a Nanodrop ND-1000 (Wilmington, DE) spectrophotometer. A Promega Access RT-PCR System (Promega, Southampton, UK) was used for amplification of transcripts. G. m. morsitans GAPDH (Accession number DQ016434) was used to normalize samples. PCR cycling conditions were: 48°C for 45 minutes, 94°C for 2 minutes, followed by 30 cycles of 94°C for 30 seconds, 57°C for 1 minute, 68°C for 2 minutes and a final extension of 68°C for 7 minutes. TsetseEP gives a product of a larger size when genomic DNA (indicative of a putative intron) was used as template (approximately 365 vs 315 bp. respectively) and was used to ensure genomic DNA was removed from experimental templates.
Immunoblots
Immunoblotting using Hybond™-P polyvinylidene difluoride (PVDF) transfer membrane (Amersham Biosciences, Amersham, UK) was performed as previously described [36]. In brief, the primary antibodies used were either a 1∶20 dilution of anti-EP repeat mouse mAb TRBP1/247 [37]. The secondary (detecting) antibody was a 1∶50,000 dilution of horseradish peroxidase conjugated goat anti-mouse IgG/IgM (H+L) (Caltag Laboratories, South San Francisco, CA). Kodak Biomax MR film (Eastman Kodak Company, Rochester, NY) was used to detect chemiluminescence. After development of the autoluminograms, proteins were stained on the PVDF membrane with 0.2% (w/v) nigrosine in PBS. The exposed film was superimposed on the stained PVDF membrane to reveal the precise location of the immunoreactive protein bands in relationship to the entire protein profile and to ensure equivalent protein loading.
Northern blots
Tsetse, which had fed twice, were injected on day 4 post emergence with 7 µg of gene-specific dsRNA (2 µl injection volume). Flies in the control group were injected with nuclease free water. Injected flies were fed again on day 5 and midguts dissected on day 7 were snap frozen in liquid nitrogen in pools of 5. The NorthernMax® formaldehyde-based system for Northern Blots (Ambion) was used. Total RNA (20 µg per lane) was loaded on a 1% formaldehyde-agarose gel. The Strip-EZ™ PCR probe synthesis and removal kit (Ambion) was used to synthesize single stranded DNA probes, which were labeled with [α32P] dATP (MP Biomedicals, Stretton Distributors, UK). Membranes were hybridized overnight at 42°C, given 2×5 minute low stringency washes and 2×15 minute high stringency washes before exposure to Kodak BioMax MR film.
Zdroje
1. PepinJ
MedaHA
2001 The epidemiology and control of human African trypanosomiasis. Advances in Parasitology 49 71 132
2. HurseyBS
2001 The programme against African trypanosomiasis: aims, objectives and achievements. Trends in Parasitology 17 2 3
3. BuddLT
1999 DFID-funded tsetse and trypanosomiasis research and development since 1980 (V. 2. Economic analysis). London Department for International Development
4. KabayoJP
2002 Aiming to eliminate tsetse from Africa. Trends in Parasitology 18 473 475
5. AksoyS
GibsonWC
LehaneMJ
2003 Interactions between tsetse and trypanosomes with implications for the control of trypanosomiasis. Advances in Parasitology 53 1 83
6. Van den AbbeeleJ
ClaesY
van BockstaeleD
Le RayD
CoosemansM
1999 Trypanosoma brucei spp. development in the tsetse fly: characterization of the post-mesocyclic stages in the foregut and proboscis. Parasitology 118 469 478
7. GibsonW
BaileyM
2003 The development of Trypanosoma brucei within the tsetse fly midgut observed using green fluorescent trypanosomes. Kinetoplastid Biology and Disease 2 1
8. HaoZ
KasumbaI
LehaneMJ
GibsonWC
KwonJ
2001 Tsetse immune responses and trypanosome transmission: implications for the development of tsetse-based strategies to reduce trypanosomiasis. Proceedings of the National Academy of Sciences USA 98 12648 12653
9. WelburnSC
MaudlinI
EllisDS
1989 Rate of trypanosome killing by lectins in midguts of different species and strains of Glossina. Medical and Veterinary Entomology 3 77 82
10. MaudlinI
WelburnSC
1987 Lectin mediated establishment of midgut infections of Trypanosoma congolense and Trypanosoma brucei in Glossina morsitans. Tropical Medicine and Parasitology 38 167 170
11. AbubakarLU
BulimoWD
MulaaFJ
OsirEO
2006 Molecular characterization of a tsetse fly midgut proteolytic lectin that mediates differentiation of African trypanosomes. Insect Biochemistry And Molecular Biology 36 344 352
12. MacleodET
MaudlinI
DarbyAC
WelburnSC
2007 Antioxidants promote establishment of trypanosome infections in tsetse. Parasitology 1 5
13. RoditiI
LehaneMJ
2008 Interactions between trypanosomes and tsetse flies. Current Opinion in Microbiology 11 345 351
14. VassellaE
Acosta-SerranoA
StuderE
LeeSH
EnglundPT
2001 Multiple procyclin isoforms are expressed differentially during the development of insect forms of Trypanosoma brucei. Journal of Molecular Biology 312 597 607
15. HainesLR
JacksonAM
LehaneMJ
ThomasJM
YamaguchiAY
2005 Increased expression of unusual EP repeat-containing proteins in the midgut of the tsetse fly (Glossina) after bacterial challenge. Insect Biochemistry And Molecular Biology 35 413 423
16. HuCY
AksoyS
2006 Innate immune responses regulate trypanosome parasite infection of the tsetse fly Glossina morsitans morsitans. Molecular Microbiology 60 1194 1204
17. ChandraM
LinigerM
TetleyL
RoditiI
BarryJD
2004 TsetseEP, a gut protein from the tsetse Glossina morsitans, is related to a major surface glycoprotein of trypanosomes transmitted by the fly and to the products of a Drosophila gene family. Insect Biochemistry And Molecular Biology 34 1163 1173
18. BendtsenJ
NielsenH
von HeijneG
BrunakS
2004 Improved prediction of signal peptides. SignalP 3.0. Journal of Molecular Biology 340 783 785
19. HaddowJD
HainesLR
GoodingRH
OlafsonRW
PearsonTW
2005 Identification of midgut proteins that are differentially expressed in trypanosome-susceptible and normal tsetse flies (Glossina morsitans morsitans). Insect Biochemistry And Molecular Biology 35 425 433
20. GygiS
RochonY
FranzaB
AebersoldR
1999 Correlation between protein and mRNA abundance in yeast. Molecular & Cellular Biology 19 1720 1730
21. HamiltonJV
MunksRJL
LehaneSM
LehaneMJ
2002 Association of midgut defensin with a novel serine protease in the blood-sucking fly Stomoxys calcitrans. Insect Molecular Biology 11 197 205
22. WalsheDP
LehaneSM
LehaneMJ
HainesLR
2009 Prolonged gene knockdown in the tsetse fly Glossina by feeding double stranded RNA. Insect Molecular Biology 18 11 19
23. MedemaR
2004 Optimizing RNA interference for application in mammalian cells. Biochemical Journal 380 595 603
24. LemaitreB
HoffmannJ
2007 The host defense of Drosophila melanogaster. Annual Review of Immunology 25 697 743
25. MeisterS
KanzokS
ZhengX
LunaC
LiT
2005 Immune signalling pathways regulating bacterial and parasite infection of the mosquito Anopheles gambiae. Proceedings Of The National Academy Of Sciences Of The United States Of America 102 11420 11425
26. ButikoferP
RueppS
BoschungM
RoditiI
1997 ‘GPEET’ procyclin is the major surface protein of procyclic culture forms of Trypanosoma brucei brucei strain 427. Biochemical Journal 326 415 423
27. PearsonTW
BeecroftRP
WelburnSC
RueppS
RoditiI
2000 The major cell surface glycoprotein procyclin is a receptor for induction of a novel form of cell death in African trypanosomes in vitro. Molecular and Biochemical Parasitology 111 333 349
28. UtzS
RoditiI
RenggliCK
AlmeidaIC
Acosta-SerranoA
2006 Trypanosoma congolense procyclins: Unmasking cryptic major surface glycoproteins in procyclic forms. Eukaryotic Cell 5 1430 1440
29. WoolhouseMEJ
HargroveJW
1998 On the interpretation of age-prevalence curves for trypanosome infections of tsetse flies. Parasitology 116 149 156
30. MsangiAR
WhitakerCJ
LehaneMJ
1998 Factors influencing the prevalence of trypanosome infection of Glossina pallidipes on the Ruvu flood plain of Eastern Tanzania. Acta Tropica 70 143 155
31. GingrichJB
WardRA
MackenLM
EsserKM
1982 African sleeping sickness: new evidence that mature tsetse flies (Glossina morsitans) can become potent vectors. Transactions of the Royal Society of Tropical Medicine and Hygiene 76 479 481
32. MakumyaviriAM
DemeyF
ClaesY
VerhulstA
Le RayD
1984 Caracterisation de la capacite vectorielle de Glossina morsitans morsitans (Diptera: Glossinidae) vis a vis de Trypanosoma brucei brucei EATRO 1125 (AnTAR 1). Annals de la Societe Belge de Medicine Tropicale 64 365 372
33. KubiC
van den AbbeeleJ
RDED
MarcottyT
DornyP
2006 The effect of starvation on the susceptibility of teneral and non-teneral tsetse flies to trypanosome infection. Medical and Veterinary Entomology 20 388 392
34. GibsonWC
1989 Analysis of a genetic cross between Trypanosoma brucei rhodesiense and T. b. brucei. Parasitology 99 391 402
35. LehaneMJ
AksoyS
GibsonW
KerhornouA
BerrimanM
2003 Adult midgut expressed sequence tags from the tsetse fly Glossina morsitans morsitans and expression analysis of putative immune response genes. Genome Biology 4 art. no.-R63
36. BeecroftRP
RoditiI
PearsonTW
1993 Identification and characterization of an acidic major surface glycoprotein from procyclic stage Trypanosoma congolense. Molecular And Biochemical Parasitology 61 285 294
37. RichardsonJP
BeecroftRP
TolsonDL
LiuMK
PearsonTW
1988 Procyclin - an unusual immunodominant glycoprotein surface-antigen from the procyclic stage of African trypanosomes. Molecular And Biochemical Parasitology 31 203 216
Štítky
Hygiena a epidemiologie Infekční lékařství Laboratoř
Článek vyšel v časopisePLOS Pathogens
Nejčtenější tento týden
2010 Číslo 3- Jak souvisí postcovidový syndrom s poškozením mozku?
- Parazitičtí červi v terapii Crohnovy choroby a dalších zánětlivých autoimunitních onemocnění
- Vakcíny proti klíšťové encefalitidě
- Kdy je nejlepší očkovat
- Možné vedlejší účinky očkování
-
Všechny články tohoto čísla
- All Mold Is Not Alike: The Importance of Intraspecific Diversity in Necrotrophic Plant Pathogens
- Tsetse EP Protein Protects the Fly Midgut from Trypanosome Establishment
- Perforin and IL-2 Upregulation Define Qualitative Differences among Highly Functional Virus-Specific Human CD8 T Cells
- N-Acetylglucosamine Induces White to Opaque Switching, a Mating Prerequisite in
- Origin and Evolution of Sulfadoxine Resistant
- Rapid Evolution of Pandemic Noroviruses of the GII.4 Lineage
- Natural Strain Variation and Antibody Neutralization of Dengue Serotype 3 Viruses
- Fine-Tuning Translation Kinetics Selection as the Driving Force of Codon Usage Bias in the Hepatitis A Virus Capsid
- Structural Basis of Cell Wall Cleavage by a Staphylococcal Autolysin
- Direct Visualization by Cryo-EM of the Mycobacterial Capsular Layer: A Labile Structure Containing ESX-1-Secreted Proteins
- Lipopolysaccharide Is Synthesized via a Novel Pathway with an Evolutionary Connection to Protein -Glycosylation
- MicroRNA Antagonism of the Picornaviral Life Cycle: Alternative Mechanisms of Interference
- Limited Trafficking of a Neurotropic Virus Through Inefficient Retrograde Axonal Transport and the Type I Interferon Response
- Direct Restriction of Virus Release and Incorporation of the Interferon-Induced Protein BST-2 into HIV-1 Particles
- RNAIII Binds to Two Distant Regions of mRNA to Arrest Translation and Promote mRNA Degradation
- Direct TLR2 Signaling Is Critical for NK Cell Activation and Function in Response to Vaccinia Viral Infection
- The Essentials of Protein Import in the Degenerate Mitochondrion of
- Dynamic Imaging of Experimental Induced Hepatic Granulomas Detects Kupffer Cell-Restricted Antigen Presentation to Antigen-Specific CD8 T Cells
- An Accessory to the ‘Trinity’: SR-As Are Essential Pathogen Sensors of Extracellular dsRNA, Mediating Entry and Leading to Subsequent Type I IFN Responses
- Innate Killing of by Macrophages of the Splenic Marginal Zone Requires IRF-7
- Exoerythrocytic Parasites Secrete a Cysteine Protease Inhibitor Involved in Sporozoite Invasion and Capable of Blocking Cell Death of Host Hepatocytes
- Inhibition of Macrophage Migration Inhibitory Factor Ameliorates Ocular -Induced Keratitis
- Membrane Damage Elicits an Immunomodulatory Program in
- Fatal Transmissible Amyloid Encephalopathy: A New Type of Prion Disease Associated with Lack of Prion Protein Membrane Anchoring
- Nucleophosmin Phosphorylation by v-Cyclin-CDK6 Controls KSHV Latency
- A Combination of Independent Transcriptional Regulators Shapes Bacterial Virulence Gene Expression during Infection
- Inhibition of Host Vacuolar H-ATPase Activity by a Effector
- Human Cytomegalovirus Protein pUL117 Targets the Mini-Chromosome Maintenance Complex and Suppresses Cellular DNA Synthesis
- Dispersion as an Important Step in the Biofilm Developmental Cycle
- Kaposi's Sarcoma-Associated Herpesvirus ORF57 Protein Binds and Protects a Nuclear Noncoding RNA from Cellular RNA Decay Pathways
- Differential Regulation of Effector- and Central-Memory Responses to Infection by IL-12 Revealed by Tracking of Tgd057-Specific CD8+ T Cells
- The Human Polyoma JC Virus Agnoprotein Acts as a Viroporin
- Expansion, Maintenance, and Memory in NK and T Cells during Viral Infections: Responding to Pressures for Defense and Regulation
- T Cell-Dependence of Lassa Fever Pathogenesis
- HIV and Mature Dendritic Cells: Trojan Exosomes Riding the Trojan Horse?
- Endocytosis of the Anthrax Toxin Is Mediated by Clathrin, Actin and Unconventional Adaptors
- A Capsid-Encoded PPxY-Motif Facilitates Adenovirus Entry
- Homeostatic Interplay between Bacterial Cell-Cell Signaling and Iron in Virulence
- Serological Profiling of a Protein Microarray Reveals Permanent Host-Pathogen Interplay and Stage-Specific Responses during Candidemia
- YfiBNR Mediates Cyclic di-GMP Dependent Small Colony Variant Formation and Persistence in
- PLOS Pathogens
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Kaposi's Sarcoma-Associated Herpesvirus ORF57 Protein Binds and Protects a Nuclear Noncoding RNA from Cellular RNA Decay Pathways
- Endocytosis of the Anthrax Toxin Is Mediated by Clathrin, Actin and Unconventional Adaptors
- Perforin and IL-2 Upregulation Define Qualitative Differences among Highly Functional Virus-Specific Human CD8 T Cells
- Exoerythrocytic Parasites Secrete a Cysteine Protease Inhibitor Involved in Sporozoite Invasion and Capable of Blocking Cell Death of Host Hepatocytes
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



