-
Články
Reklama
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaGenome-Wide Association Study of White Blood Cell Count in 16,388 African Americans: the Continental Origins and Genetic Epidemiology Network (COGENT)
Total white blood cell (WBC) and neutrophil counts are lower among individuals of African descent due to the common African-derived “null” variant of the Duffy Antigen Receptor for Chemokines (DARC) gene. Additional common genetic polymorphisms were recently associated with total WBC and WBC sub-type levels in European and Japanese populations. No additional loci that account for WBC variability have been identified in African Americans. In order to address this, we performed a large genome-wide association study (GWAS) of total WBC and cell subtype counts in 16,388 African-American participants from 7 population-based cohorts available in the Continental Origins and Genetic Epidemiology Network. In addition to the DARC locus on chromosome 1q23, we identified two other regions (chromosomes 4q13 and 16q22) associated with WBC in African Americans (P<2.5×10−8). The lead SNP (rs9131) on chromosome 4q13 is located in the CXCL2 gene, which encodes a chemotactic cytokine for polymorphonuclear leukocytes. Independent evidence of the novel CXCL2 association with WBC was present in 3,551 Hispanic Americans, 14,767 Japanese, and 19,509 European Americans. The index SNP (rs12149261) on chromosome 16q22 associated with WBC count is located in a large inter-chromosomal segmental duplication encompassing part of the hydrocephalus inducing homolog (HYDIN) gene. We demonstrate that the chromosome 16q22 association finding is most likely due to a genotyping artifact as a consequence of sequence similarity between duplicated regions on chromosomes 16q22 and 1q21. Among the WBC loci recently identified in European or Japanese populations, replication was observed in our African-American meta-analysis for rs445 of CDK6 on chromosome 7q21 and rs4065321 of PSMD3-CSF3 region on chromosome 17q21. In summary, the CXCL2, CDK6, and PSMD3-CSF3 regions are associated with WBC count in African American and other populations. We also demonstrate that large inter-chromosomal duplications can result in false positive associations in GWAS.
Published in the journal: . PLoS Genet 7(6): e32767. doi:10.1371/journal.pgen.1002108
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1002108Summary
Total white blood cell (WBC) and neutrophil counts are lower among individuals of African descent due to the common African-derived “null” variant of the Duffy Antigen Receptor for Chemokines (DARC) gene. Additional common genetic polymorphisms were recently associated with total WBC and WBC sub-type levels in European and Japanese populations. No additional loci that account for WBC variability have been identified in African Americans. In order to address this, we performed a large genome-wide association study (GWAS) of total WBC and cell subtype counts in 16,388 African-American participants from 7 population-based cohorts available in the Continental Origins and Genetic Epidemiology Network. In addition to the DARC locus on chromosome 1q23, we identified two other regions (chromosomes 4q13 and 16q22) associated with WBC in African Americans (P<2.5×10−8). The lead SNP (rs9131) on chromosome 4q13 is located in the CXCL2 gene, which encodes a chemotactic cytokine for polymorphonuclear leukocytes. Independent evidence of the novel CXCL2 association with WBC was present in 3,551 Hispanic Americans, 14,767 Japanese, and 19,509 European Americans. The index SNP (rs12149261) on chromosome 16q22 associated with WBC count is located in a large inter-chromosomal segmental duplication encompassing part of the hydrocephalus inducing homolog (HYDIN) gene. We demonstrate that the chromosome 16q22 association finding is most likely due to a genotyping artifact as a consequence of sequence similarity between duplicated regions on chromosomes 16q22 and 1q21. Among the WBC loci recently identified in European or Japanese populations, replication was observed in our African-American meta-analysis for rs445 of CDK6 on chromosome 7q21 and rs4065321 of PSMD3-CSF3 region on chromosome 17q21. In summary, the CXCL2, CDK6, and PSMD3-CSF3 regions are associated with WBC count in African American and other populations. We also demonstrate that large inter-chromosomal duplications can result in false positive associations in GWAS.
Introduction
Proliferation and differentiation of hematopoietic stem cells into mature white blood cells (WBC) in the bone marrow, followed by release into the circulation of mature WBC, is a highly regulated process [1]. WBC comprise several subtypes including neutrophils, lymphocytes, monocytes, eosinophils, and basophils. These cells play an essential role in innate and adaptive immunity against invading microorganisms. They are also involved in the pathogenesis of various acute and chronic diseases. The circulating numbers of leukocytes can be influenced by stress, infection, or inflammation. Total WBC and neutrophil counts also differ by ethnicity, with levels 10–20% lower among African American than European American populations [2], [3]. This difference is due to a common African-derived “null” variant (rs2814778) of the Duffy Antigen Receptor for Chemokines (DARC) gene, which also confers selective advantage against malaria [4]–[6]. By abolishing expression of DARC on red blood cells, the Duffy null variant may alter the concentration and distribution of chemokines in the blood and tissue [7]–[10], thereby regulating neutrophil production and migration.
Several clinically distinct forms of congenital neutropenia are inherited as rare, monogenic disorders [11]. Genetic polymorphisms more common in the population, including those that reside in the region of 17q21 harboring the CSF3 gene, were recently associated with circulating total WBC and WBC subtype counts in European and Japanese populations [12]–[15]. Yet these common polymorphisms account for only a fraction of the reported 50–60% heritability of WBC count [16]–[18]. In addition, the contribution of these or other loci to variation in total WBC or WBC subtypes have yet to be thoroughly evaluated through current genome-wide association approaches in other populations, such as African Americans. To identify additional polymorphisms associated with WBC and its subtypes (neutrophils, lymphocytes, monocytes, eosinophils, basophils), we therefore performed a large, multi-cohort genome wide association study (GWAS) of typed and imputed SNPs in African Americans, with follow-up in additional ethnic samples of European and Japanese ancestry.
Results
We performed GWA analysis of total WBC in an African-American discovery sample of 16,388 individuals from 7 population-based cohorts from the Continental Origins and Genetic Epidemiology Network (COGENT). The characteristics of each cohort are summarized in Table 1. Following stringent genotyping and imputation quality control procedures, a total of at least 2.4 million autosomal SNPs were available for analysis in each cohort (Table S1). Summary-level study results were combined by using inverse variance-weighted meta-analysis. The genomic-control corrected QQ plot for the combined African-African GWA analysis is shown in Figure 1. As summarized in Table 2, Table S2, and the Manhattan plot in Figure 2, three regions on chromosomes 1q23, 4q13, and 16q22 reached genome-wide significance at the threshold of P<2.5×10−8. These 3 loci are described in further detail below. Additional GWA analyses were performed on a subset of up to 7,477 COGENT African American participants with data available on WBC subtype counts (neutrophils, lymphocytes, monocytes, eosinophils and basophils) (Figures S1 and S2, Tables S3, S4, S5, S6, S7). Apart from the association of the chromosome 1q23 DARC locus with neutrophils and monocytes [6] (see below and Table 3), there were no new genome-wide significant associations (all P>2.5×10−8) for these phenotypes. African American cohort-specific results for index SNPs newly discovered or confirmed to be associated with WBC phenotypes are summarized in Figure S3 (total WBC), Figure S4 (neutrophil count), and Table S8.
Fig. 1. Quantile–quantile plot of P-values in meta-analysis for total white blood count. 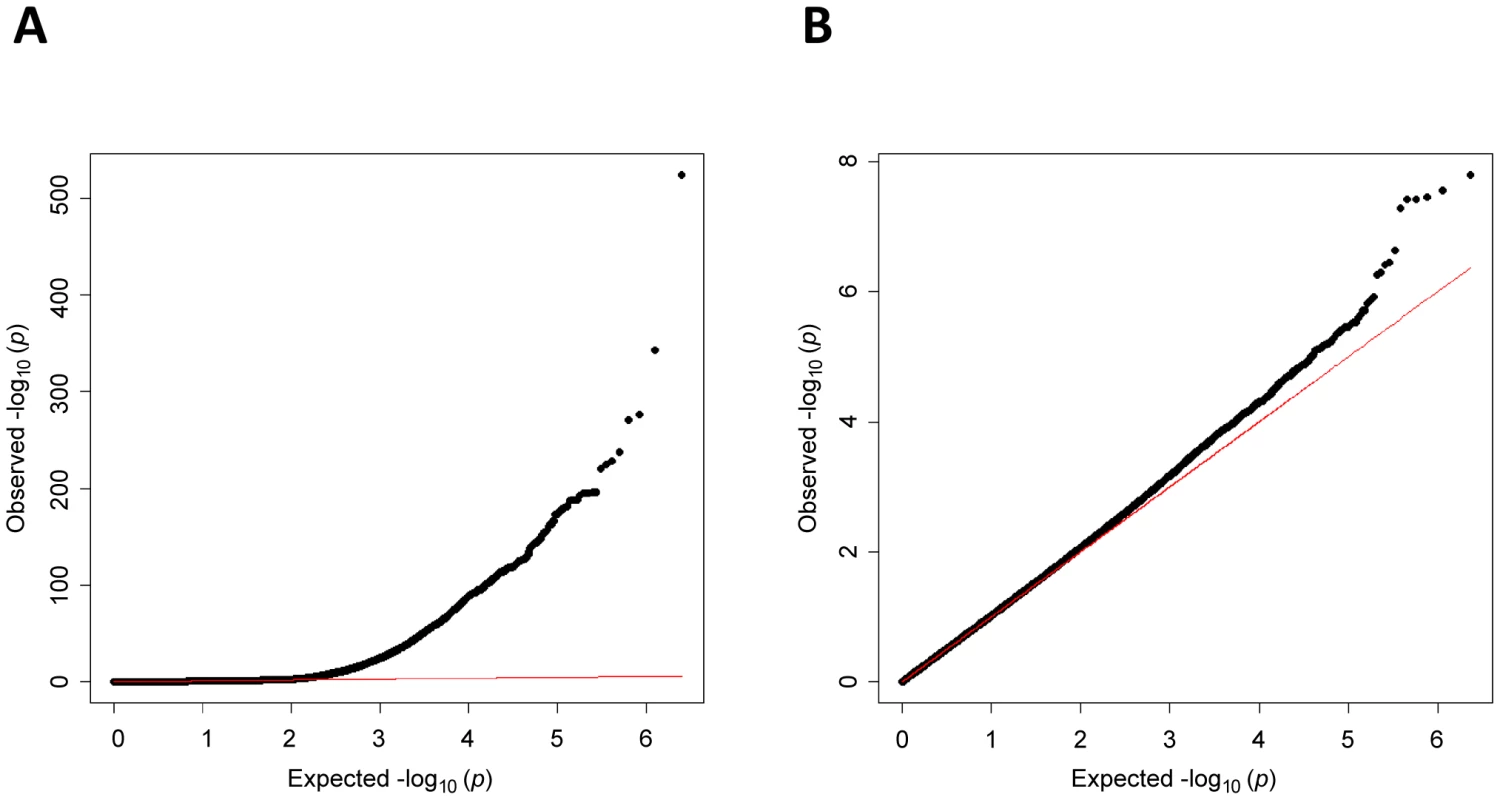
Panel A includes all markers, panel B excludes markers on chromosome 1. Horizontal axis indicates expected −log10 P-values. Vertical axis indicates observed −log10 P-values. The red line represents y = x. The marked deviation from expectations in panel A is due to markers on chromosome 1 near the DARC locus. The individual study ancestry-corrected inflation factors with (without) chromosome 1 markers were 1.06 (1.04) for ARIC, 1.02 (1.01) for CARDIA, 1.11 (1.08) for JHS, 1.03 (1.01) for Health ABC, 1.09 (1.05) for WHI, 1.04 (1.00) for GeneSTAR, and 1.01 (0.98) for HANDLS. The overall inflation factor prior to correction was 1.05 (0.98). Fig. 2. Manhattan plot of meta-analysis P-values in GWAS for total WBC count. 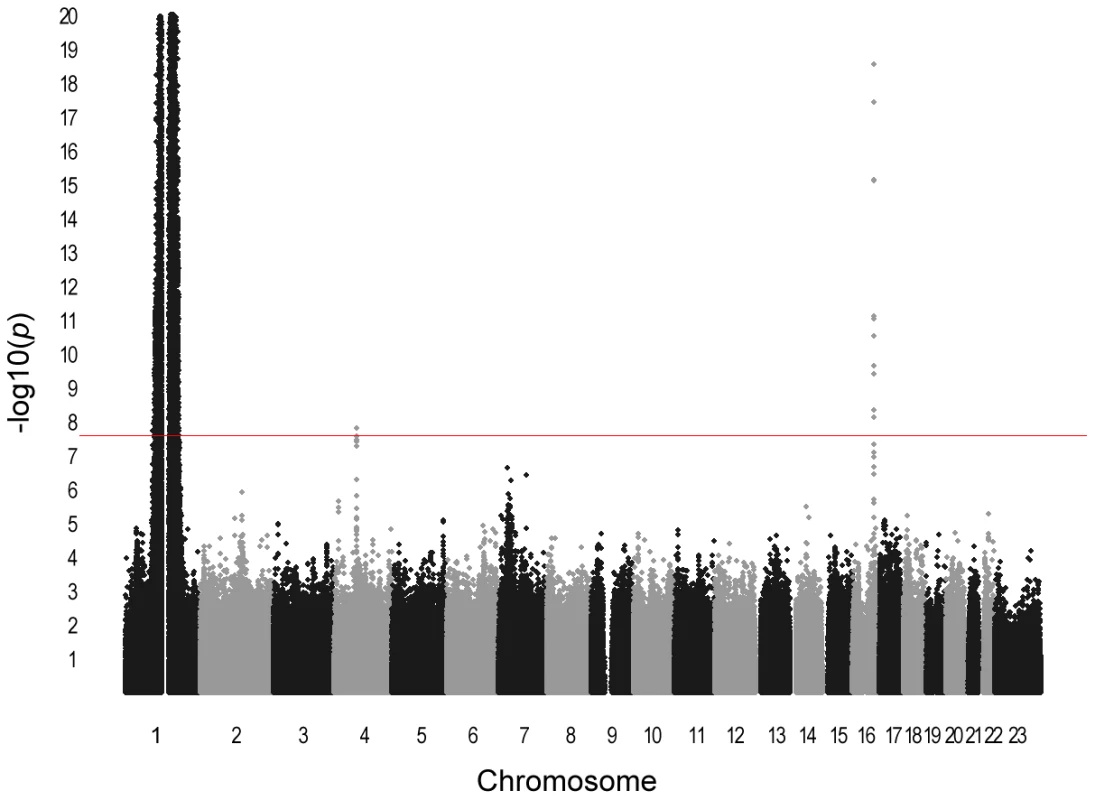
Horizontal axis indicates chromosomal position. Vertical axis indicates −log10 P-values from inverse variance-weighted fixed effects meta-analysis. The red horizontal line indicates the genome-wide significance threshold of P = 2.5×10−8. Association signals are present at 1q23, 4q13, and 16q22. The P-values for the broad chromosome 1 signal are truncated at 10−20. This region spans nearly 90 Mb on both arms of chromosome 1 and results artifactually in two apparently distinct peaks because of the lack of genotyped or imputed SNPs around the centromere. Tab. 1. Characteristics of Continental Origins and Genetic Epidemiology Networks (COGENT) African-American discovery GWAS participants (n = 16,388). 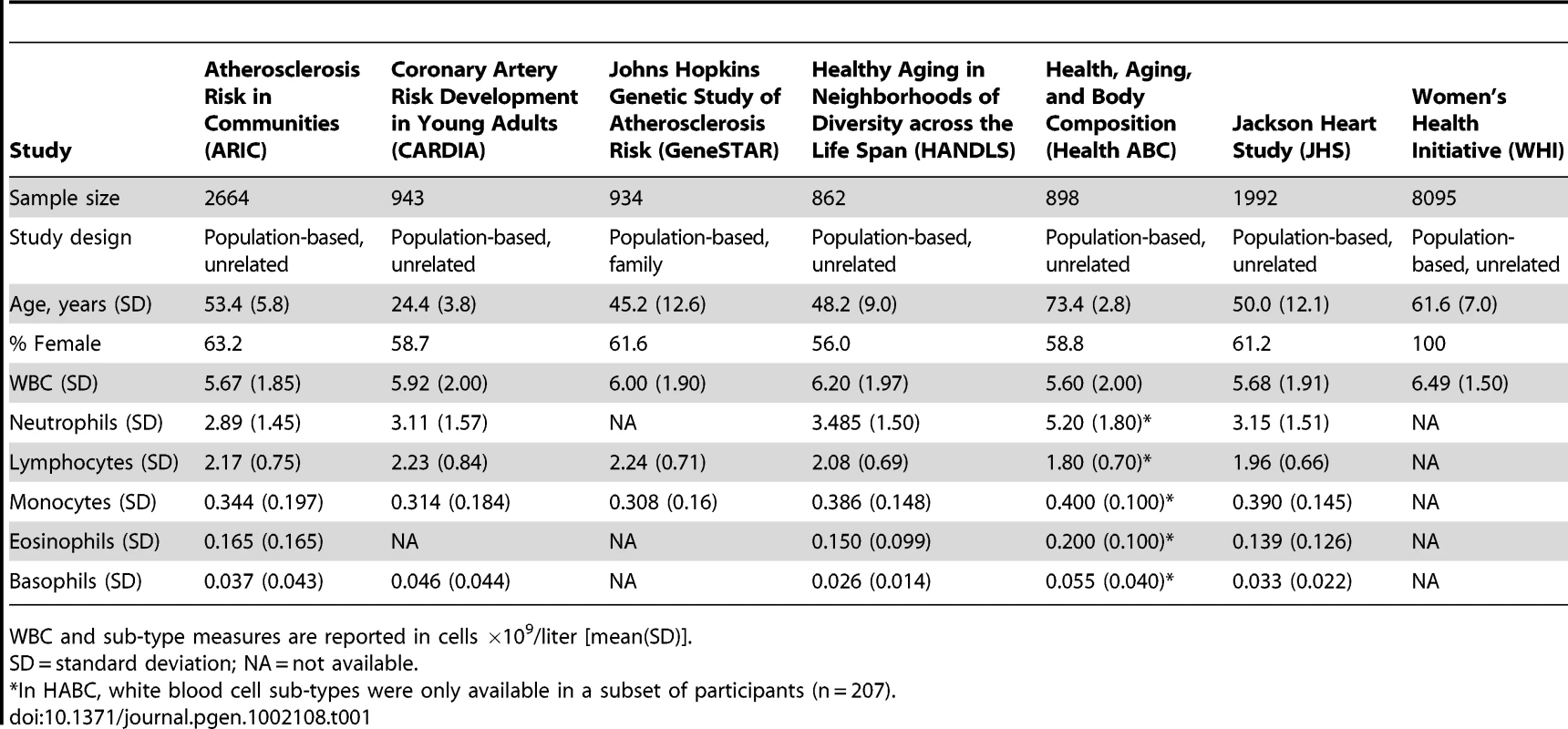
WBC and sub-type measures are reported in cells ×109/liter [mean(SD)]. Tab. 2. Results of genome-wide significant SNPs for total white blood cell count. 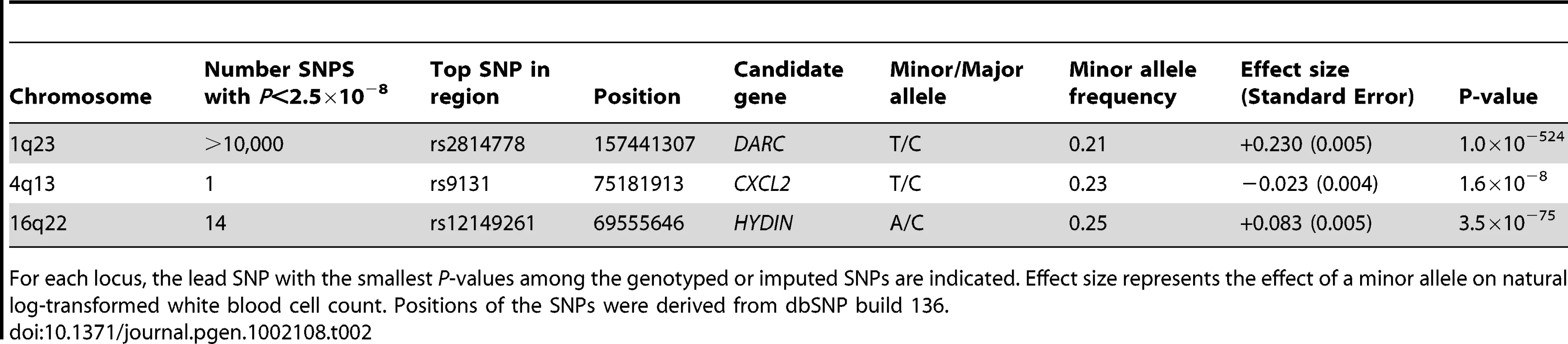
For each locus, the lead SNP with the smallest P-values among the genotyped or imputed SNPs are indicated. Effect size represents the effect of a minor allele on natural log-transformed white blood cell count. Positions of the SNPs were derived from dbSNP build 136. Tab. 3. Meta-analysis results of genome-wide significant SNPs for white blood cell count subtypes. 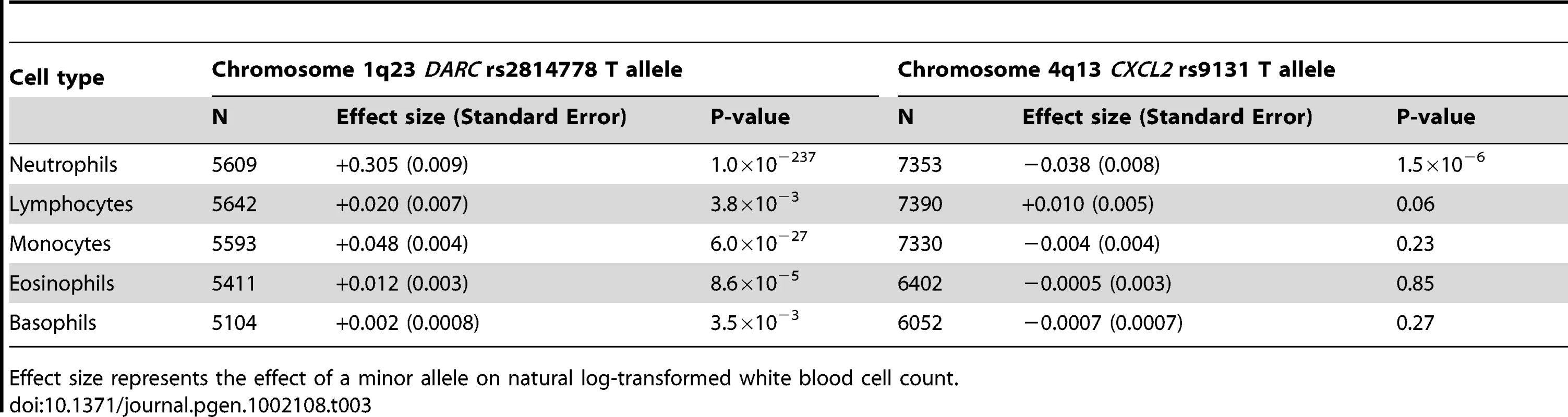
Effect size represents the effect of a minor allele on natural log-transformed white blood cell count. Validation of DARC region on chromosome 1q23 as WBC–associated locus in African Americans
The GWA association signal on chromosome 1 is comprised of a broad peak encompassing 4,649 genotyped and imputed SNPs that exceeded the threshold of genome-wide significance. This region spans nearly 90 Mb on both arms of chromosome 1 (90,385,392–177,814,914 bp) and is approximately centered around the centromere. This results artifactually in two apparently distinct peaks in the Manhattan plot (Figure 2) because of the lack of genotyped or imputed SNPs around the centromere. Based on the 99% confidence interval of the distribution of test statistics, the strongest region of association is concentrated between position 155,127,086 and 160,217,075 on the short arm of chromosome 1 (P = 10−154 to 10−524). This region is centered around the DARC gene locus on 1q23.2. DARC contains rs2814778 (the Duffy null allele), previously identified as the likely causal chromosome 1q WBC-associated polymorphism in an admixture mapping study performed in the JHS and Health ABC cohorts, and confirmed in ARIC [4], [5]. As previously reported [4], [5], the DARC rs2814778 association with WBC is most consistent with a dominant rather than an additive model (P for dominance deviation <10−40). For example, in the largest cohort (WHI), the mean age - and global ancestry-adjusted WBC count was 4,823±1,004/µl in homozygotes for the African null allele, 6,307±1,006/µl in heterozygotes, and 6,563±1013/µl in homozygotes for the European wild-type allele.
Because the magnitude of the DARC rs2814778 polymorphism association might obscure any additional association signals present on chromosome 1, we repeated the GWAS analysis conditioning on the Duffy null rs2814778 polymorphism. All chromosome 1 SNPs which were significantly associated with WBC prior to rs2814778 adjustment became non-significant conditional on rs2814778 genotype (data not shown). When the association analysis was conducted separately for each white cell subtype, the DARC rs2814778 polymorphism was most strongly associated with the number of circulating neutrophils (P<10−236) (Table 3), but was also associated with the numbers of circulating monocytes (P<10−26), and to a lesser extent, lymphocytes, eosinophils, and basophils.
HYDIN region association on chromosome 16q22 is most likely due to genotyping artifact
On chromosome 16q22, 13 SNPs spanning a ∼250 kb region (bp 69474507–69726247) that includes part of the large HYDIN gene locus were significantly associated with WBC. The lead SNP in the HYDIN region was rs12149261 (minor allele frequency or MAF 25%), an intronic polymorphism. The HYDIN association signal was confined to genotyped SNPs on the Affy6.0 array (ARIC, CARDIA, JHS, WHI). SNPs in this region were absent from the Illumina platform (Health ABC, GeneSTAR, HANDLS) and also absent from HapMap 2, thereby limiting imputation in the latter 3 cohorts.
Further examination of the sequence context in this region revealed that the HYDIN gene encompasses a large, recently duplicated segment of the genome, with a nearly identical 360-kb paralogous segment inserted on chromosome 1q21 [19], [20]. The chromosome 1q21 paralogue of the chromosome 16q22 segmental duplication is absent from build 36 of the NCBI human genome assembly. Nonetheless, 1q21 falls within the region encompassing the DARC association signal for WBC. Using genome-wide Affymetrix 6.0 genotype data from the ARIC African-American cohort, we determined the r-squared (pair-wise LD) between rs12149261 and every other typed SNP in the genome. There was reduced local LD within the chromosome 16 duplicated region, relative to the surrounding chromosome 16 SNP (Figure S5). Three SNPs had r-squared values of >0.20 with rs12149261: one located 20 kb away on chromosome 16 in the HYDIN gene (rs1774524; r2 = 0.27), and two located on chromosome 1 at ∼120 Mb near the HYDIN paralogue (rs12087334 and rs4659245; r2 = 0.25 and 0.22, respectively). Moreover, combined analysis of the 4 cohorts typed on the Affymetrix GWA platform showed that the chromosome 16q22 association signal at rs12149261 (P = 2.12×10−18) was completely abolished after conditioning on chromosome 1 DARC rs2814778 (P = 0.36). While defects in the HYDIN gene result in hydrocephalus [19], [20], this genomic region has not previously been associated with WBC. Together, these results demonstrate that the chromosome 16 HYDIN association finding is most likely a probe cross-hybridization artifact due to inter-chromosomal sequence similarity with the duplicated segment on chromosome 1q21 near the DARC region and that the polymorphisms associated with WBC in the studies using the Affymetrix arrays actually map to the chromosome 1 region.
Discovery of a novel CXCL2 association finding on chromosome 4q13 and replication in other ethnic populations
A novel SNP association on chromosome 4q13 was identified in our African-American WBC discovery GWAS. The lead SNP rs9131 is located in the 3′ UTR of the CXCL2 gene, which encodes a macrophage-derived chemotactic cytokine for polymorphonuclear leukocytes. In African Americans, the minor T allele (MAF = 23%) was associated with lower WBC. Several additional SNPs in the chromosome 4 chemokine gene cluster had P-values ranging from 10−5 to 10−7, including rs2367291 located upstream of CXCL1 (Figure 3A) Further adjustment for rs9131, however, abolished these associations (data not shown). Based on HapMap phase 2 and 1000 genomes data, rs9131 is in perfect LD with 7 other inter-genic SNPs in this region. Analysis of the subset of COGENT study participants with data available for number of circulating white cell subtypes indicated the rs9131 association was confined to neutrophils (Table 3).
Fig. 3. Population-specific −log P values for total WBC association at the chromosome 4q13 CXCL2 locus. 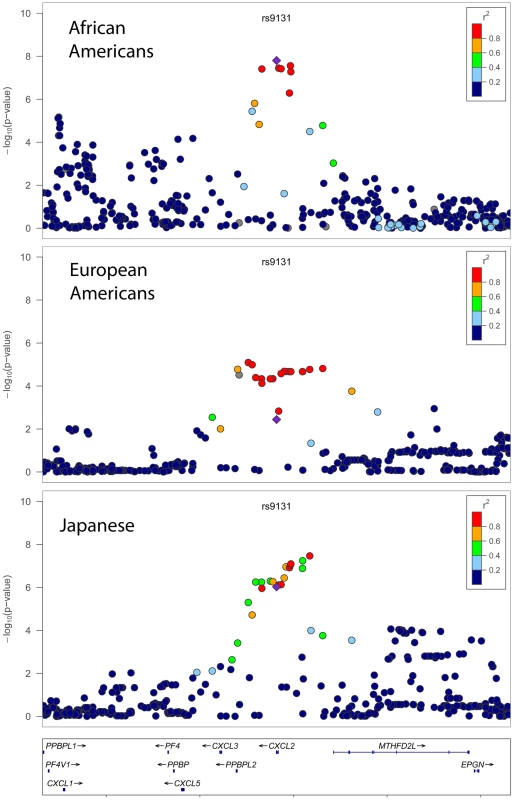
Shown is a 500 kb window of chromosome 4q13 centered at rs9131 (purple square). Plots were generated for African Americans (top panel), European Americans (middle panel), Japanese (bottom panel), using LocusZoom. The color of each SNP indicates the level of pairwise linkage disequilibrium (r-squared) relative to the index SNP rs9131. R-squared values were calculated from HapMap (release 22) YRI (top panel), CEU (middle panel), or CHB+JPT (bottom panel) populations. SNPs with missing LD information are shown in grey. To assess the role of the newly identified CXCL2 association in other ethnic populations, we performed in silico replication using 3 samples: 3,551 Hispanic-American women from WHI-SHARe, 19,509 European-American participants from the CHARGE consortium, and 14,767 Japanese subjects from RIKEN. In Europeans, Hispanics, and Japanese, the T allele of rs9131 (frequency = 65%, 62%, and 46%, respectively) was associated with lower WBC (P = 0.004, 0.002, and 9.4×10−7, respectively), as was seen in African Americans (P = 2×10−8). The direction and magnitude of association was consistent across racial/ethnic groups: 0.009±0.003, 0.018±0.006, and 0.013±0.003 natural log units lower in Europeans, Hispanics, and Japanese, respectively, compared to 0.023±0.004 natural log units lower WBC count in the African-American discovery sample. Pooling the results across populations using a random effects meta-analysis gave a combined effect estimate (beta for lnWBC) of −0.015 (95%CI = −0.009 to −0.021) for rs9131. The P for Cochrane's Q test for heterogeneity was 0.04, with an I2 of 64%. In contrast, there was no evidence that the chromosome 1 DARC region was associated with WBC count in either European or Japanese populations (data not shown).
Regional plots comparing the SNP association and linkage disequilibrium patterns across CXCL2 on chromosome 4 in African Americans, Europeans, and Japanese 4 are shown in Figure 3. In Europeans and Japanese, several additional SNPs in the CXCL2 region of chromosome 4 had stronger association WBC signals than rs9131. Specifically, rs16850408, which is located in an inter-genic region between CXCL2 and the pro-platelet basic protein-like 2 gene (PPBPL2), was most strongly associated with WBC (P = 8.04×10−6) in Europeans. The r-squared between rs16850408 and rs9131 is 0.76 in European and 0.3 in African HapMap samples. In Japanese, rs7686861 located in the intergenic region between CXCL2 and MTHFD2L (methylenetetrahydrofolate dehydrogenase 2-like) was the lead SNP (P = 3.4×10−8). The r-squared between rs7686861 and rs9131 is 0.21 in Asian and 0.23 in African HapMap samples. To further narrow the locus of WBC count association, we performed a sample size-weighted meta-analysis of the CXCL2 region across all 3 ethnic groups. The cross-population association signal mapped to a 75 kb region (positions 75,155,842–75,231,250), which contains CXCL2 and no other genes in the chromosome 4q13 region. The top SNPs included rs1371799 (P = 1.7×10−17) as well as several others located within the CXCL2 promoter and 5′ flanking region (Figure 4).
Fig. 4. Trans-population meta-analysis results for total WBC count at the chromosome 4q13 CXCL2 locus. 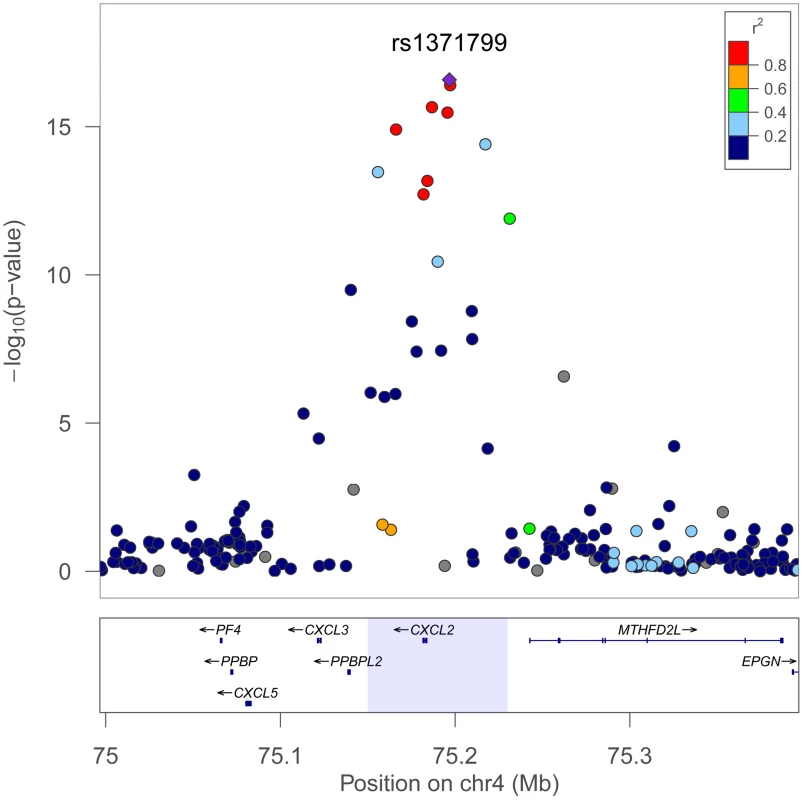
The strongest association signal is localized to an LD bin of several SNPs within the CXCL2 promoter and 5′ flanking region, including rs1371799 (purple triangle). Meta-analysis was performed using Fisher's method to combine P-values across African, European, and Japanese populations. The 99% confidence interval for the cross-population association signal mapped to a 75 kb region shaded in light blue (lower panel). Plot was generated using LocusZoom. Linkage disequilibrium is shown for the African population. Assessment of other previously discovered WBC–associated loci in African-Americans
Several GWAS loci have been published from European or Japanese cohorts, including those associated with WBC (GSDMA-ORMDL3-PSMD3-CSF3, HSB1L-MYB, CDSN-PSORS1C1, CDK6, and RAP1B), neutrophil count (PSMD3-CSF3, PLCB4), and eosinophil count (IL1RL1, IKZF2, GATA2, IL5, SH2B3) [12]–[15]. Table 4 shows the association results of these same loci in our African-American sample, for the originally reported index SNP. Extending the association analyses to SNPs in LD with the index SNP (r2≥0.5 in HapMap CEU or CHB+JPT) did not reveal any additional associations (data not shown). For the chromosome 17 PSMD3–CSF3 region, the T allele of rs4065321 reported to be associated with lower WBC in Japanese was similarly associated with lower total WBC in African-Americans (P = 1×10−4). Most of the African-American WBC-associated SNPs in this region were intronic to PSMD3, while one SNP (rs7224260) is located in the 3′ flanking region of CSF3. The T allele of CDK6 rs445 was associated with lower total WBC (Table 4), and also with lower neutrophil count in 7,392 African Americans (beta −0.0249±0.0049; p = 1.7×10−7). The remaining European and Japanese WBC-association genomic regions listed in Table 4 showed little evidence of replication in African Americans.
Tab. 4. Assessment in African-Americans of loci previously associated with leukocyte traits in Caucasians and/or Japanese. 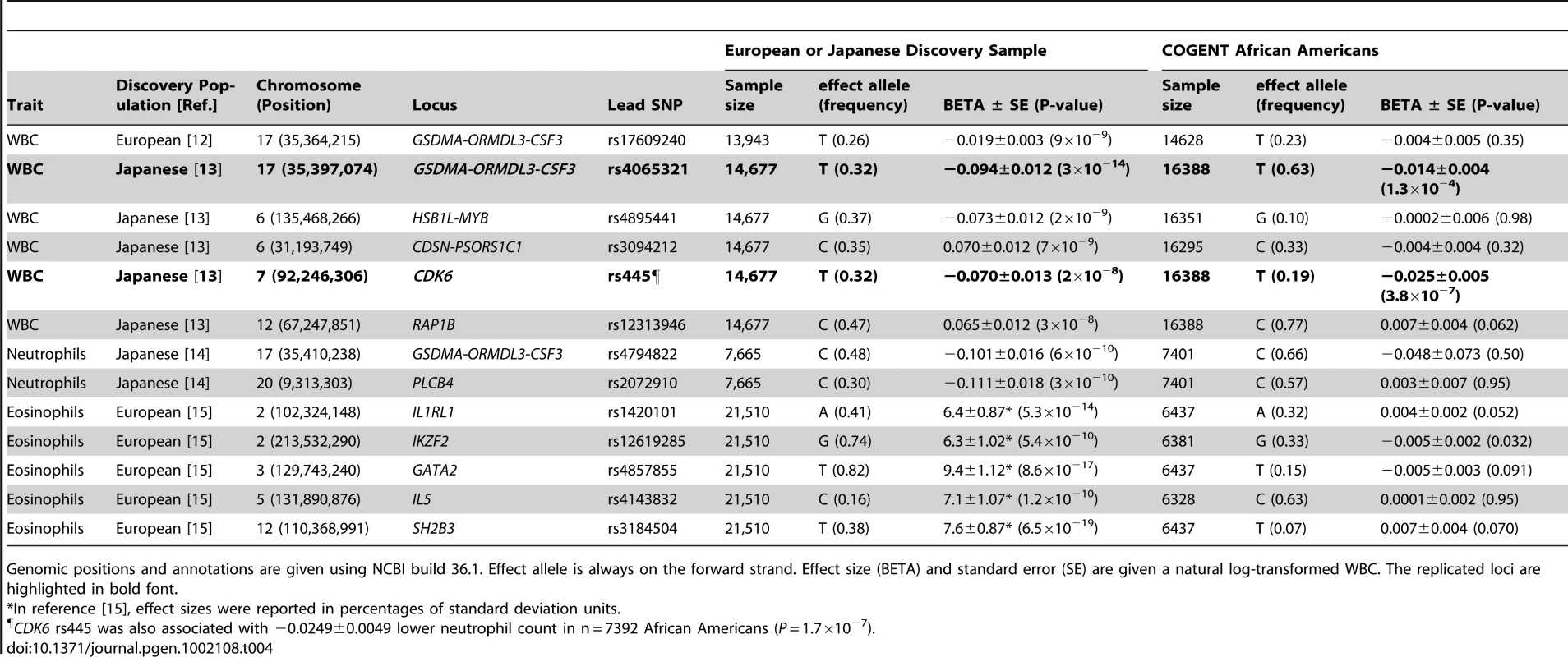
Genomic positions and annotations are given using NCBI build 36.1. Effect allele is always on the forward strand. Effect size (BETA) and standard error (SE) are given a natural log-transformed WBC. The replicated loci are highlighted in bold font. Effect of locus-specific ancestry on newly and previously reported WBC–associated SNPs
In recently admixed populations, it is possible that confounding of a SNP association may occur as a result of local as well as global differences in genetic ancestry between study participants [21]. Therefore, we repeated the association analyses for any newly reported African American or previously reported European and Japanese genome-wide significant WBC-associated loci, additionally adjusting for estimated local ancestry in our COGENT African American study participants. We performed these locus-specific ancestry conditional analyses in a subset of 13,694 participants from each of the 4 cohorts genotyped on Affymetrix 6.0 (WHI, ARIC, CARDIA, and JHS). After meta-analyzing the African American cohort-specific results, there was essentially no difference between the local ancestry adjusted versus global ancestry-adjusted associations at any of the WBC-associated loci (Table S9). However, when we performed an additional association analysis for each lead SNP stratifying on the estimated local number of European versus African chromosomes, the CDK6 rs445 and PSMD3-CSF3 rs4065321 WBC associations were stronger on a local European ancestral background than on an African background (Table S9). Notably, the CDK6 and PSMD3-CSF3 loci are also the only two previously reported WBC associations that we were able to replicate in our African American sample. For European and Japanese WBC-associated loci that didn't replicate in our African American sample, there was no evidence of any differential association according to local ancestral background or proportion of European ancestry in the AA sample (data not shown).
Heritability of WBC phenotypes in African Americans and proportion of variance explained
Polygenic heritability was estimated for unadjusted and age - and sex-adjusted total WBC, neutrophil, lymphocyte, and monocyte count using 236 African-American pedigrees from the GeneSTAR study (Table S10). All WBC phenotypes showed significant heritability (P<0.001). The heritability estimates ranged from 48–49% for total WBC and neutrophil count to ∼29% for monocyte count. The proportion of total variance explained by DARC rs2814778+CXCL2 rs9131+CDK6 rs445+PSMD3-CSF3 rs4065321 in the COGENT African American cohorts ranged from 16% to 24% for WBC, 20% to 25% for neutrophils, and 2% to 7% for monocytes.
Since multiple, independent variants at the same locus may account for some of the “missing heritability” of complex traits [22], we repeated the association tests for all genotyped SNPs within 500 kb of the DARC, CXCL2, CDK6, and PSMD3-CSF3 gene regions for WBC association, conditioning on the lead SNP in each region. None of the 4 loci contained additional SNPs associated with WBC at P<2.5×10−5 (a Bonferroni-corrected significance threshold calculated from the 2,000 SNPs tested in these 4 regions).
Discussion
Recently the African null allele of rs2814778 at the Duffy Antigen Receptor for Chemokines locus on chromosome 1 was found to be associated with lower total leukocyte and neutrophil counts in African Americans [4]–[6]. By screening 16,388 African-American participants, we have confirmed the strong DARC association. We also identified a second chemokine-related gene region associated with lower WBC, with the lead SNP rs9131 located in the CXCL2 gene. Independent evidence of the novel CXCL2 association was present in other ethnic populations, including ∼3,500 Hispanic Americans, ∼15,000 Japanese, and ∼20,000 European Americans. Two additional WBC loci recently identified through GWAS of European or Japanese populations (CDK6 gene region on chromosome 7 and PSMD3-CSF3 region on chromosome 17 [12]–[14] were associated with WBC traits in African Americans. We also demonstrate that large inter-chromosomal duplications can result in false positive associations in GWAS as was shown for HYDIN.
Our estimate of heritability for total WBC and neutrophil count in African Americans was close to 50%, which is similar to that reported in European populations [16]–[18]. While our GWAS has identified a few, select loci to be associated with WBC count in African Americans, the proportion of variation explained for WBC and neutrophil count was still less than 25%, and considerably lower for the remaining WBC subtypes. Therefore it seems likely that in addition to the DARC and CXCL2 loci, other yet-to-be identified loci exist. Alternatively, genetic factors may account for a lower percentage of the variance in WBC count than suggested by heritability estimates and perhaps environmental factors should be more broadly considered. Other factors may have limited our ability to identify genetic mechanisms underlying these traits, including phenotype measurement error and reduced sample size and power for the WBC subtype GWA analyses. Multiple rare genetic variants or gene-gene and gene-environment interaction may also account for some of the inter-individual variation of these hematologic traits.
Myelopoiesis is regulated by a number of cytokines, chemokines, growth factors, and their receptors. The cytokine granulocyte colony-stimulating factor (G-CSF), encoded by the CSF3 gene on chromosome 17, is critically involved in granulopoiesis by stimulating proliferation, differentiation, and survival of neutrophil precursors [23] and by regulating the rate of release of neutrophils from the bone marrow under non-inflammatory conditions [24]. During infection or inflammation, neutrophil, monocyte and eosinophil mobilization from the bone marrow can occur through the systemic and/or local action of several chemokines, which stimulate chemotaxis across the bone marrow sinusoidal endothelium. G-CSF stimulates neutrophil mobilization and release by down-regulating signaling of stromal-derived factor 1 (CXCL12) through its receptor CXCR4, which serves as a bone marrow retention signal for mature neutrophils [23], [25]. In contrast, the chemokines CXCL1 and CXCL2, by binding to CXCR2, promote rapid release of neutrophils from the bone marrow, thereby elevating blood neutrophil counts during infection or during G-CSF-induced neutrophil mobilization [25]–[27].
DARC is selectively expressed on red blood cells and venular endothelial cells and binds several pro-inflammatory chemokines of both the CXC and CC subfamilies. Endothelial DARC facilitates leukocyte recruitment and trans-endothelial migration, thereby contributing to inflammatory disease pathogenesis and severity in animal models [28]–[30]. Erythrocyte DARC has been proposed to act as a chemokine scavenger, sink or reservoir, maintaining basal plasma chemokine concentrations, though the biological relevance of this sink function remains unclear [6]–[9]. The African Duffy null variant disrupts a DARC promoter binding site for the transcription factor GATA-1, and results in complete absence of DARC from erythrocytes without affecting endothelial DARC expression [31]. Duffy-negative individuals are protected from P. vivax malaria [32], [33] and have been reported to have a survival advantage in leukopenic HIV-infected persons of African descent [34]. Interestingly, during systemic inflammation neutrophils from DARC-deficient mice exhibit impaired chemotaxis toward CXCL2 that appears to result from altered plasma chemokine levels and down-regulation of neutrophil CXCR2 expression [29]. It is conceivable that a homeostatic role of DARC in CXCL1/CXCL2 - CXCR2 chemokine ligand-receptor interactions during inflammation may also extend to the setting of neutrophil release from the bone marrow under both basal and inflammatory conditions.
Nucleotide diversity can vary substantially across populations due to different evolutionary histories and migration patterns. Generally, nucleotide diversity is greatest and linkage disequilibrium lowest among African populations. By leveraging the extent of variation in LD patterns between populations, localization of causal variants can be improved by analyzing multiple ethnic groups [35]–[38]. By combining WBC count association results from the CXCL2 region across African Americans, European Americans, and Japanese, we were able to narrow the association signal to the CXCL2 promoter and 5′ flanking region.
The multi-gene region on chromosome 17q21.1 has now been associated with WBC or neutrophil count in Europeans [12], Japanese [13], [14], and African Americans. The index SNPs originally reported (rs17609240, rs4065321, rs4794822, rs2305481) for these traits are in strong to moderate LD in Europeans and Japanese (r2 = 0.5 to 1.0), spanning an LD block that includes several genes (GSDMA, ORMDL3, PSMD3, CSF3, MED24, SNORD124, and THRA). The lower extent of LD in African-Americans suggests finer localization of the rs4065321 WBC-associated signal to the region containing PSMD3 and CSF3. Other variants in this region have been associated with childhood-onset asthma [39]. CSF3, which encodes G-CSF, constitutes the most likely biologic candidate in this region responsible for phenotypic variation in WBC. However, the functional SNPs responsible for variation in WBC phenotypes remain to be identified. Expression (eQTL) analysis demonstrated that the SNP associated with neutrophil count by Okada et al was associated with PSMD3 expression, rather than CSF3 expression [14]. PSMD3 encodes one of the non-ATPase subunits of the 19S regulator of 26S proteasome, which is involved in regulation of the cell cycle through the ubiquitin–proteasome pathway.
The current analysis also replicated the association between WBC count and a region on chromosome 7 containing the gene for CDK6, or cyclin-dependent kinase 6, another regulator of cell cycle progression known to be expressed in proliferating hematopoietic progenitor cells [40]. Through its interaction with the transcription factor Runx1, CDK6 inhibits terminal granulocytic differentiation [41]. For the chromosome 7 WBC locus, rs445 is located within the first intron of CDK6, and represents the lead SNP in both Japanese [13] and our African American sample. There is no other variant in strong LD (r-squared>0.8) with rs445 in any HapMap or 1000 Genomes population. Therefore it is possible that CDK6 rs445 may represent the actual causal variant. Other polymorphisms within the CDK6 gene have been associated with susceptibility to rheumatoid arthritis [42] and height [43].
Benign neutropenia is defined as an absolute neutrophil count (ANC) of less than 1.5×109 cells/L on repeated occasions [2], [44]. It occurs in up to 40% of individuals of African descent [2] and is present in ∼5% of adult African Americans compared to <1% of European Americans [3]. The benign neutropenia of African Americans is characterized by normal myeloid maturation, but slightly reduced numbers of bone marrow myeloid progenitors [45], [46] and reduced numbers of mature neutrophils that can be released from bone marrow stores [47]. Despite having slightly lower steady-state bone marrow CD34+ hematopoietic progenitor cells, African Americans paradoxically appear to have enhanced peripheral blood stem-cell mobilization in response to administration of G-CSF compared to whites [44], [48]. The genetic determinants of these features of G-CSF-induced stem cell mobilization remain to be determined.
In summary, polymorphisms within DARC on chromosome 1 and CXCL2 on chromosome 4, and near CDK6 on chromosome 7 and CSF3 on chromosome 17, are associated with WBC in African Americans. These findings contribute to our understanding of genetic factors underlying variation in WBC within and between populations and highlight the importance of common genetic variants in genomic regions encoding chemokine ligands and receptors to regulation of myelopoiesis and circulating leukocyte counts in human populations. Further localization and characterization of the functional variants responsible for these WBC and neutrophil associations could help to inform clinical approaches to cancer-associated neutropenia or hematopoietic stem cell mobilization.
Methods
Subjects
The subjects participating in the GWAS consisted of a total of 16,388 self-identified African-American individuals from 7 population-based cohorts (ARIC, CARDIA, JHS, WHI, HANDLS, Health ABC, and GeneSTAR) that belong to the Continental Origins and Genetic Epidemiology Network (COGENT). Detailed descriptions of each participating COGENT cohort, their quality control practices and study-level analyses are provided in the Text S1. Clinical information of the subjects was collected by self-report and clinical examination. All participants provided written informed consent as approved by local Human Subjects Committees. We excluded study participants on the basis of pregnancy, cancer, or AIDS diagnosis at the time of blood count measurement.
WBC phenotype data
Certified staff obtained fasting blood samples at the baseline clinic visit. Samples for complete blood count (CBC) analysis were obtained by venipuncture and collected into tubes containing ethylenediaminetetraacetic acid (EDTA). Total circulating WBC count and cell subtype counts were performed at local clinical laboratories using automated hematology cell counters and standardized quality assurance procedures [4], [6], [49]–[51]. Total WBC count was reported in millions of cells per ml, and was recorded in all 16,388 study participants. Information on WBC subtype was available only in a subset of 7,477 (45.6%) participants from ARIC, CARDIA, JHS, HANDLS, GeneSTAR, and Health ABC. WBC differentials were performed by clinically certified hematology laboratories. The absolute numbers of each type of WBC were calculated by multiplying the proportion of the WBC count comprised by each cell type by the total WBC measure. To evaluate normality of the phenotypes for subsequent regression analyses, we performed Box-Cox likelihood ratio tests on raw WBC phenotypes. On this basis, all WBC traits were natural log transformed to normalize the distributions of the phenotypic data.
Genotype data and quality control
Genome-wide genotyping was performed within each COGENT cohort using methods described under Text S1. DNA samples with a genome-wide genotyping success rate <90%, duplicate discordance or sex mismatch, genetic ancestry outliers (as determined by cluster analysis performed using principal component analysis or multi-dimensional scaling), SNPs with genotyping success rate <95%, monomorphic SNPs, SNPs with minor allele frequency (MAF) <1%, and SNPs that map to several genomic locations were removed from the analyses. Significantly associated SNPs were examined for strong deviations from Hardy–Weinberg equilibrium and/or raw genotype data examined for abnormal clustering. Participants and SNPs passing basic quality control were imputed to >2.2 million SNPs based on HapMap2 haplotype data using a 1∶1 mixture of Europeans (CEU) and Africans (YRI) as the reference panel. Details of the genotype imputation procedure are described further under Text S1. Prior to discovery meta-analyses, SNPs were excluded if imputation quality metrics (equivalent to the squared correlation between proximal imputed and genotyped SNPs) were less than 0.50.
Data analysis
For all cohorts, genome-wide association (GWA) analysis for quantitative WBC traits was performed using linear regression adjusted for covariates, implemented in either PLINK v1.07 [52] or MACH2QTL v1.08. Allelic dosage at each SNP was used as the independent variable, adjusted for primary covariates of age, age-squared, sex, and clinic site (if applicable). To adjust for population stratification and global admixture, the principal components were also incorporated as covariates in the regression models (see Text S1). For GeneSTAR, family structure was accounted for in the association tests using linear mixed effects (LME) models implemented in R [53]. Although the JHS has a small number of related individuals, extensive analyses showed that results were concordant using linear regression or LME, after genomic control. Therefore, results are presented for JHS using linear regression. For imputed genotypes, we used dosage information (i.e. a value between 0.0–2.0 calculated using the probability of each of the three possible genotypes) in the regression model implemented in PLINK and MACH2QTL (for cohorts with unrelated individuals) or the Maximum Likelihood Estimation (MLE) routines (for GeneSTAR).
For each WBC phenotype, meta-analyses were conducted using inverse-variance weighted fixed-effects models to combine beta coefficients and standard errors from study level regression results for each SNP to derive a combined p-value and effect estimates. Study level results were corrected for genomic inflation factors (λ) by incorporating study specific λ estimates into the scaling of the standard errors (SE) of the regression coefficients by multiplying the SE by the squate-root of the genomic inflation factor. The inflation factors for all completed analyses are presented in Table S1. Meta-analyses were implemented in the software METAL [54] and were performed independently by another analyst to confirm results. Between-study heterogeneity of results was assessed by using Cochran's Q statistic and the I2 inconsistency metric. For each genome-wide significant or replicated locus, cohort specific-results and overall WBC effect estimates and confidence intervals are summarized using forest plots (Figure S3 and S4). The mean and standard deviation WBC count for each genotype class is provided in Table S8.
To maintain an overall type 1 error rate of 5%, a threshold of α = 2.5×10−8 was used to declare genome-wide statistical significance. This threshold has been suggested for African ancestry populations based on estimates of ∼2 million independent common variant tests in African genomes [55].
Given the nonlinear nature of the original phenotype, we performed a sensitivity analysis of whether our results are robust to the assumption of an additive genetic model. We repeated the GWA analysis for the WHI, ARIC, CARDIA, JHS cohorts, the four largest African American cohorts (n = 13,694) using a 2 degree of freedom genotypic model as well as a dominance deviation test, and meta-analyzed the results using METAL.
To assess in the COGENT African-Americans WBC trait-associated loci previously reported in Europeans or Japanese, we evaluated the African-American meta-analysis results for each index SNP in the regions reported, including consistency of direction of effect, and assessed statistical significance by a simple Bonferroni adjustment based on the total number of SNPs assessed using a 2-sided hypothesis test. In addition, we performed a more exploratory assessment of all SNPs within a 500 kb window that were correlated in African Americans with the European or Japanese index SNP in HapMap CEU or CHB+JPT (r2≥0.5). We adjusted these exploratory regional analyses for multiple testing based on the effective number of SNPs, taking into account pairwise linkage disequilibrium patterns.
To further assess the potential existence of multiple, independent variants influencing a trait at the same locus (allelic heterogeneity), regression analyses were repeated, conditional on the most strongly associated (index) SNP in that region. Each study repeated the primary GWA analysis, additionally adjusting for the lead SNP in each region under the appropriate regression models. The cohort-specific results were then meta-analyzed in the same way as for the primary GWA study using METAL.
Replication and fine-mapping of new WBC association signals
Replication of novel association findings was performed using GWA data in 3 other ethnic populations: 3,551 Hispanic American women from WHI, 14,767 Japanese from RIKEN, and 19,509 European Americans from CHARGE. Further details of each study population are provided under Text S1. Both genotyped and imputed SNP data were available in the European and Japanese samples, while only genotyped SNP data were available in the Hispanic Americans. To further localize the causal variant responsible for the CXCL2-WBC association, we extended the association analysis to include all genotyped and imputed SNPs within a 500 kb region centered at rs9131, the SNP most strongly associated with WBC count in African Americans. We then performed a trans-population meta-analysis of each SNP in this region by combining test statistics from the African American (COGENT), European (CHARGE), and Japanese (RIKEN) association analyses using Fisher's method [56], which may have some advantages over the standard meta-analytic approach in this setting [37]. Nonetheless, we also performed a standard inverse variance-weighted meta-analysis using either fixed or random effects [57], and obtained results similar to Fisher's method.
Local ancestry analyses
For between-study GWA platform consistency, we estimated locus-specific ancestry using Affymetrix 6.0 genotyped SNP data from the 4 largest African-American cohorts (WHI, ARIC, JHS, CARDIA), which constitute ∼85% of our total COGENT African American sample. For each African American, locus-specific ancestry (probabilities of whether an individual has 0, 1, or 2 alleles of African ancestry at each locus) was estimated using a Hidden Markov Model and local haplotype structure to detect transitions in ancestry along the genome [58], [59]. Phased haplotype data from the HapMap CEU and YRI individuals were used as reference panels. To assess the impact of local ancestry on any genome-wide SNP associations, each of the 4 cohorts repeated each SNP genotype-WBC phenotype linear regression model, adjusting for local ancestry proportion as a covariate. In addition, we stratified the SNP genotype-WBC phenotype association test on the number of estimated local European chromosomes (≥1 versus <1) to compare whether variants in genome-wide significant regions have the same versus different effect on African and European ancestral population backgrounds. The cohort-specific results of these analyses were combined using METAL.
Heritability and proportion of variance explained
In the GeneSTAR family study, variance components models in the ASSOC subroutine of S.A.G.E. [60] were used to derive maximum likelihood estimates of polygenic (narrow-sense) heritability (σ2g) using natural-log transformed unadjusted or covariate-adjusted phenotype data. The statistical significance of the heritability estimate was obtained using a likelihood ratio test. In each of the 7 COGENT African American cohorts, the fraction of variance explained was estimated using the formula: 2pq×β2, where p is the frequency of the effect allele of the SNP, q = 1−p, and β is the additive effect in each population estimated by standardizing WBC to have standard deviation 1.
Supporting Information
Zdroje
1. MetcalfD 2008 Hematopoietic cytokines. Blood 111 485 491
2. HaddyTBRanaSRCastroO 1999 Benign ethnic neutropenia: what is a normal absolute neutrophil count? J Lab Clin Med 133 15 22
3. HsiehMMEverhartJEByrd-HoltDDTisdaleJFRodgersGP 2007 Prevalence of neutropenia in the U.S. population: age, sex, smoking status, and ethnic differences. Ann Intern Med 146 486 492
4. NallsMAWilsonJGPattersonNJTandonAZmudaJM 2008 Admixture mapping of white cell count: genetic locus responsible for lower white blood cell count in the Health ABC and Jackson Heart studies. Am J Hum Genet 82 81 87
5. ReichDNallsMAKaoWHAkylbekovaELTandonA 2009 Reduced neutrophil count in people of African descent is due to a regulatory variant in the Duffy antigen receptor for chemokines gene. PLoS Genet 5 e1000360 doi:10.1371/journal.pgen.1000360
6. LoKSWilsonJGLangeLAFolsomARGalarneauG 2010 Genetic association analysis highlights new loci that modulate hematological trait variation in Caucasians and African Americans. Hum Genet In press
7. DawsonTCLentschABWangZCowhigJERotA 2000 Exaggerated response to endotoxin in mice lacking the Duffy antigen/receptor for chemokines (DARC). Blood 96 1681 1684
8. LeeJSWurfelMMMatute-BelloGFrevertCWRosengartMR 2006 The Duffy antigen modifies systemic and local tissue chemokine responses following lipopolysaccharide stimulation. J Immunol 177 8086 8094
9. ReutershanJHarryBChangDBagbyGJLeyK 2009 DARC on RBC limits lung injury by balancing compartmental distribution of CXC chemokines. Eur J Immunol 39 1597 1607
10. SchnabelRBBaumertJBarbalicMDupuisJEllinorPT 2010 Duffy antigen receptor for chemokines (Darc) polymorphism regulates circulating concentrations of monocyte chemoattractant protein-1 and other inflammatory mediators. Blood 115 5289 5299
11. DaleDCLinkDC 2009 The many causes of severe congenital neutropenia. N Engl J Med 360 3 5
12. SoranzoNSpectorTDManginoMKuhnelBRendonA 2009 A genome-wide meta-analysis identifies 22 loci associated with eight hematological parameters in the HaemGen consortium. Nat Genet 41 1182 1190
13. KamataniYMatsudaKOkadaYKuboMHosonoN 2010 Genome-wide association study of hematological and biochemical traits in a Japanese population. Nat Genet 42 210 215
14. OkadaYKamataniYTakahashiAMatsudaKHosonoN 2010 Common variations in PSMD3-CSF3 and PLCB4 are associated with neutrophil count. Hum Mol Genet 15; 19 2079 2085
15. GudbjartssonDFBjornsdottirUSHalapiEHelgadottirASulemP 2009 Sequence variants affecting eosinophil numbers associate with asthma and myocardial infarction. Nat Genet 41 342 347
16. WhitfieldJBMartinNG 1985 Genetic and environmental influences on the size and number of cells in the blood. Genet Epidemiol 2 133 144
17. EvansDMFrazerIHMartinNG 1999 Genetic and environmental causes of variation in basal levels of blood cells. Twin Res 2 250 257
18. PiliaGChenWMScuteriAOrruMAlbaiG 2006 Heritability of cardiovascular and personality traits in 6,148 Sardinians. PLoS Genet 2 e132 doi:10.1371/journal.pgen.0020132
19. DoggettNAXieGMeinckeLJSutherlandRDMundtMO 2006 A 360-kb inter-chromosomal duplication of the human HYDIN locus. Genomics 88 762 771
20. Brunetti-PierriNBergJSScagliaFBelmontJBacinoCA 2008 Recurrent reciprocal 1q21.1 deletions and duplications associated with microcephaly or macrocephaly and developmental and behavioral abnormalities. Nat Genet 40 1466 1471
21. WangXZhuXQinHCooperRSEwensWJ 2011 Adjustment for local ancestry in genetic association analysis of admixed populations. Bioinformatics 27 670 677
22. Lango-AllenHEstradaKLettreGBerndtSIWeedonMN 2010 Hundreds of variants clustered in genomic loci and biological pathways affect human height. Nature 467 832 8
23. ChristopherMJLinkDC 2007 Regulation of neutrophil homeostasis. Curr Opin Hematol Jan 3 8
24. SemeradCLLiuFGregoryADStumpfKLinkDC 2002 G-CSF is an essential regulator of neutrophil trafficking from the bone marrow to the blood. Immunity 17 413 23
25. EashKJGreenbaumAMGopalanPKLinkDC 2010 CXCR2 and CXCR4 antagonistically regulate neutrophil trafficking from murine bone marrow. J Clin Invest 120 2423 31
26. WengnerAMPitchfordSCFurzeRCRankinSM 2008 The coordinated action of G-CSF and ELR+CXC chemokines in neutrophil mobilization during acute inflammation. Blood 111 42 49
27. Nguyen-JacksonHPanopoulosADZhangHLiHSWatowichSS 2010 STAT3 controls the neutrophil migratory response to CXCR2 ligands by direct activation of G-CSF-induced CXCR2 expression and via modulation of CXCR2 signal transduction. Blood 115 3354 63
28. ZarbockASchmolkeMBockhornSGScharteMBuschmannK 2007 The Duffy antigen receptor for chemokines in acute renal failure: a facilitator of renal chemokine presentation. Crit Care Med 35 2156 2163
29. PruensterMMuddeLBombosiP 2009 The Duffy antigen receptor for chemokines transports chemokines and supports their promigratory activity. Nat Immunol 10 101 108
30. ZarbockABishopJMüllerH 2010 Chemokine homeostasis vs. chemokine presentation during severe acute lung injury: the other side of the Duffy antigen receptor for chemokines. Am J Physiol Lung Cell Mol Physiol 298 L462 L471
31. PeiperSCWangZXNeoteKMartinAWShowellHJ 1995 The Duffy antigen/receptor for chemokines (DARC) is expressed in endothelial cells of Duffy negative individuals who lack the erythrocyte receptor. J Exp Med 181 1311 1317
32. MillerLHMasonSJClydeDFMcGinnissMH 1976 The resistance factor to Plasmodium vivax in blacks. The Duffy-blood-group genotype, FyFy. N Engl J Med 295 302 304
33. HorukRChitnisCEDarbonneWCColbyTJRybickiA 1993 A receptor for the malarial parasite Plasmodium vivax: the erythrocyte chemokine receptor. Science 261 1182 1184
34. HeWNeilSKulkarniHWrightEAganBK 2008 Duffy antigen receptor for chemokines mediates trans-infection of HIV-1 from red blood cells to target cells and affects HIV-AIDS susceptibility. Cell Host Microbe 4 52 62
35. PulitSLVoightBFde BakkerPI 2010 Multiethnic genetic association studies improve power for locus discovery. PLoS ONE 5 e12600 doi:10.1371/journal.pone.0012600
36. ZaitlenNPaşaniucBGurTZivEHalperinE 2010 Leveraging genetic variability across populations for the identification of causal variants. Am J Hum Genet 86 23 33
37. TeoYYOngRTSimXTaiESChiaKS 2010 Identifying candidate causal variants via trans-population fine-mapping. Genet Epidemiol 34 653 64
38. RosenbergNAHuangLJewettEMSzpiechZAJankovicI 2010 Genome-wide association studies in diverse populations. Nat Rev Genet 11 356 66
39. MoffattMFGutIGDemenaisFStrachanDPBouzigonE 2010 A large-scale, consortium-based genomewide association study of asthma. N Engl J Med 363 1211 21
40. MeyersonMHarlowE 1994 Identification of G1 kinase activity for cdk6, a novel cyclin D partner. Mol Cell Biol 14 2077 2086
41. FujimotoTAndersonKJacobsenSENishikawaSINerlovC 2007 Cdk6 blocks myeloid differentiation by interfering with Runx1 DNA binding and Runx1-C/EBPalpha interaction. EMBO J 26 2361 70
42. RaychaudhuriSRemmersEFLeeATHackettRGuiducciC 2008 Common variants at CD40 and other loci confer risk of rheumatoid arthritis. Nat Genet 40 1216 23
43. SoranzoNRivadeneiraFChinappen-HorsleyUMalkinaIRichardsJB 2009 Meta-analysis of genome-wide scans for human adult stature identifies novel Loci and associations with measures of skeletal frame size. PLoS Genet 5 e1000445 doi:10.1371/journal.pgen.1000445
44. HsiehMMTisdaleJFRodgersGPYoungNSTrimbleEL 2010 Neutrophil count in African Americans: lowering the target cutoff to initiate or resume chemotherapy? J Clin Oncol 28 1633 1637
45. HollowellJGvan AssendelftOWGunterEWLewisBGNajjarM 2005 Hematological and iron-related analytes–reference data for persons aged 1 year and over: United States, 1988–94. Vital Health Stat 11 1 156
46. RezvaniKFlanaganAMSarmaUConstantinoviciNBainBJ 2001 Investigation of ethnic neutropenia by assessment of bone marrow colony-forming cells. Acta Haematol 105 32 37
47. MasonBALessinLSchechterGP 1979 Marrow granulocyte reserves in black Americans. Hydrocortisone-induced granulocytosis in the “benign” neutropenia of the black. Am J Med 67 201 205
48. VasuSLeitmanSFTisdaleJFHsiehMMChildsRW 2008 Donor demographic and laboratory predictors of allogeneic peripheral blood stem cell mobilization in an ethnically diverse population. Blood 112 2092 2100
49. ShimakawaTBildDE 1993 Relationship between hemoglobin and cardiovascular risk factors in young adults. J Clin Epidemiol 46 1257 1266
50. QayyumRBeckerDMYanekLRMoyTFBeckerLC 2008 Platelet inhibition by aspirin 81 and 325 mg/day in men versus women without clinically apparent cardiovascular disease. Am J Cardiol 2008 101 1359 63
51. MargolisKLMansonJEGreenlandPRodaboughRJBrayPF 2005 Leukocyte count as a predictor of cardiovascular events and mortality in postmenopausal women: the Women's Health Initiative Observational Study. Arch Intern Med 165 500 508
52. PurcellSNealeBTodd-BrownKThomasLFerreiraMA 2007 PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81 559 575
53. ChenMHYangQ 2010 GWAF: an R package for genome-wide association analyses with family data. Bioinformatics 26 580 1
54. WillerCJLiYAbecasisGR 2010 METAL: fast and efficient meta-analysis of genomewide association scans. Bioinformatics 26 2190 1
55. Pe'erIYelenskyRAltshulerDDalyMJ 2008 Estimation of the multiple testing burden for genomewide association studies of nearly all common variants. Genet Epidemiol 32 381 5
56. FisherRA 1925 Statistical Methods for Research Workers London Oliver & Boyd
57. DerSimonianRLairdN 1986 Meta-analysis in clinical trials. Controlled Clinical Trials 7 177 188
58. PriceALTandonAPattersonNBarnesKCRafaelsN 2009 Sensitive detection of chromosomal segments of distinct ancestry in admixed populations. PLoS Genet 5 e1000519 doi:10.1371/journal.pgen.1000519
59. TangHCoramMWangPZhuXRischN 2006 Reconstructing genetic ancestry blocks in admixed individuals. Am J Hum Genet 79 1 12
60. SAGE 2009 Statistical Analysis for Genetic Epidemiology, Release 5.4.2: http://darwin.cwru.edu/
Štítky
Genetika Reprodukční medicína
Článek Local Absence of Secondary Structure Permits Translation of mRNAs that Lack Ribosome-Binding SitesČlánek Independent Chromatin Binding of ARGONAUTE4 and SPT5L/KTF1 Mediates Transcriptional Gene SilencingČlánek Trade-Off between Bile Resistance and Nutritional Competence Drives Diversification in the Mouse GutČlánek FGF Signaling Regulates the Number of Posterior Taste Papillae by Controlling Progenitor Field SizeČlánek Mammalian BTBD12 (SLX4) Protects against Genomic Instability during Mammalian SpermatogenesisČlánek Identification of Nine Novel Loci Associated with White Blood Cell Subtypes in a Japanese PopulationČlánek Differential Effects of and Risk Variants on Association with Diabetic ESRD in African AmericansČlánek Dynamic Chromatin Localization of Sirt6 Shapes Stress- and Aging-Related Transcriptional Networks
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2011 Číslo 6- Je „freeze-all“ pro všechny? Odborníci na fertilitu diskutovali na virtuálním summitu
- Kazuistika – Perspektivy využití precizované medicíny v rámci personalizované specifické terapie onkologických pacientů
- Nobelova cena za chemii pro genetické nůžky: Objev, který změní naši budoucnost?
- Technologie na bázi RNA v klinické praxi: od přebarvených petúnií k terapii vzácných a dosud jen obtížně léčitelných chorob u lidí
- „Nepředstavovali jsme si, že náš výzkum povede přímo ke vzniku nových léků, dokonce ještě za našeho života“
-
Všechny články tohoto čísla
- Local Absence of Secondary Structure Permits Translation of mRNAs that Lack Ribosome-Binding Sites
- Statistical Inference on the Mechanisms of Genome Evolution
- Revisiting Heterochromatin in Embryonic Stem Cells
- A Two-Stage Meta-Analysis Identifies Several New Loci for Parkinson's Disease
- Identification of a Sudden Cardiac Death Susceptibility Locus at 2q24.2 through Genome-Wide Association in European Ancestry Individuals
- Genomic Prevalence of Heterochromatic H3K9me2 and Transcription Do Not Discriminate Pluripotent from Terminally Differentiated Cells
- Epistasis between Beneficial Mutations and the Phenotype-to-Fitness Map for a ssDNA Virus
- Recurrent Chromosome 16p13.1 Duplications Are a Risk Factor for Aortic Dissections
- Telomere DNA Deficiency Is Associated with Development of Human Embryonic Aneuploidy
- Genome-Wide Association Study of White Blood Cell Count in 16,388 African Americans: the Continental Origins and Genetic Epidemiology Network (COGENT)
- Unexpected Role for DNA Polymerase I As a Source of Genetic Variability
- Transportin-SR Is Required for Proper Splicing of Genes and Plant Immunity
- How Chromatin Is Remodelled during DNA Repair of UV-Induced DNA Damage in
- Independent Chromatin Binding of ARGONAUTE4 and SPT5L/KTF1 Mediates Transcriptional Gene Silencing
- Two Evolutionary Histories in the Genome of Rice: the Roles of Domestication Genes
- Natural Allelic Variation Defines a Role for : Trichome Cell Fate Determination
- Multiple Common Susceptibility Variants near BMP Pathway Loci , , and Explain Part of the Missing Heritability of Colorectal Cancer
- Pathogenic Mechanism of the FIG4 Mutation Responsible for Charcot-Marie-Tooth Disease CMT4J
- A Functional Variant in Promoter Modulates Its Expression and Confers Disease Risk for Systemic Lupus Erythematosus
- Drift and Genome Complexity Revisited
- Chromosomal Macrodomains and Associated Proteins: Implications for DNA Organization and Replication in Gram Negative Bacteria
- Trade-Off between Bile Resistance and Nutritional Competence Drives Diversification in the Mouse Gut
- Pathways of Distinction Analysis: A New Technique for Multi–SNP Analysis of GWAS Data
- Web-Based Genome-Wide Association Study Identifies Two Novel Loci and a Substantial Genetic Component for Parkinson's Disease
- Chk2 and p53 Are Haploinsufficient with Dependent and Independent Functions to Eliminate Cells after Telomere Loss
- Exome Sequencing Identifies Mutations in High Myopia
- Distinct Functional Constraints Partition Sequence Conservation in a -Regulatory Element
- CorE from Is a Copper-Dependent RNA Polymerase Sigma Factor
- A Single Sex Pheromone Receptor Determines Chemical Response Specificity of Sexual Behavior in the Silkmoth
- FGF Signaling Regulates the Number of Posterior Taste Papillae by Controlling Progenitor Field Size
- Maps of Open Chromatin Guide the Functional Follow-Up of Genome-Wide Association Signals: Application to Hematological Traits
- Increased Susceptibility to Cortical Spreading Depression in the Mouse Model of Familial Hemiplegic Migraine Type 2
- Differential Gene Expression and Epiregulation of Alpha Zein Gene Copies in Maize Haplotypes
- Parallel Adaptive Divergence among Geographically Diverse Human Populations
- Genetic Analysis of Genome-Scale Recombination Rate Evolution in House Mice
- Mechanisms for the Evolution of a Derived Function in the Ancestral Glucocorticoid Receptor
- Mammalian BTBD12 (SLX4) Protects against Genomic Instability during Mammalian Spermatogenesis
- Interferon Regulatory Factor 8 Regulates Pathways for Antigen Presentation in Myeloid Cells and during Tuberculosis
- High-Resolution Analysis of Parent-of-Origin Allelic Expression in the Arabidopsis Endosperm
- Specific SKN-1/Nrf Stress Responses to Perturbations in Translation Elongation and Proteasome Activity
- Graded Nodal/Activin Signaling Titrates Conversion of Quantitative Phospho-Smad2 Levels into Qualitative Embryonic Stem Cell Fate Decisions
- Genome-Wide Analysis Reveals PADI4 Cooperates with Elk-1 to Activate Expression in Breast Cancer Cells
- Trait Variation in Yeast Is Defined by Population History
- Meiosis-Specific Loading of the Centromere-Specific Histone CENH3 in
- A Genome-Wide Survey of Imprinted Genes in Rice Seeds Reveals Imprinting Primarily Occurs in the Endosperm
- Multiple Regulatory Mechanisms to Inhibit Untimely Initiation of DNA Replication Are Important for Stable Genome Maintenance
- SIRT1 Promotes N-Myc Oncogenesis through a Positive Feedback Loop Involving the Effects of MKP3 and ERK on N-Myc Protein Stability
- Bacteriophage Crosstalk: Coordination of Prophage Induction by Trans-Acting Antirepressors
- Role of the Single-Stranded DNA–Binding Protein SsbB in Pneumococcal Transformation: Maintenance of a Reservoir for Genetic Plasticity
- Genomic Convergence among ERRα, PROX1, and BMAL1 in the Control of Metabolic Clock Outputs
- Genome-Wide Association of Bipolar Disorder Suggests an Enrichment of Replicable Associations in Regions near Genes
- Identification of Nine Novel Loci Associated with White Blood Cell Subtypes in a Japanese Population
- DNA Ligase III Promotes Alternative Nonhomologous End-Joining during Chromosomal Translocation Formation
- Differential Effects of and Risk Variants on Association with Diabetic ESRD in African Americans
- Finished Genome of the Fungal Wheat Pathogen Reveals Dispensome Structure, Chromosome Plasticity, and Stealth Pathogenesis
- Dynamic Chromatin Localization of Sirt6 Shapes Stress- and Aging-Related Transcriptional Networks
- Extracellular Matrix Dynamics in Hepatocarcinogenesis: a Comparative Proteomics Study of Transgenic and Null Mouse Models
- Integrating 5-Hydroxymethylcytosine into the Epigenomic Landscape of Human Embryonic Stem Cells
- Vive La Différence: An Interview with Catherine Dulac
- Multiple Loci Are Associated with White Blood Cell Phenotypes
- Nuclear Accumulation of Stress Response mRNAs Contributes to the Neurodegeneration Caused by Fragile X Premutation rCGG Repeats
- A New Mutation Affecting FRQ-Less Rhythms in the Circadian System of
- Cryptic Transcription Mediates Repression of Subtelomeric Metal Homeostasis Genes
- A New Isoform of the Histone Demethylase JMJD2A/KDM4A Is Required for Skeletal Muscle Differentiation
- Genetic Determinants of Lipid Traits in Diverse Populations from the Population Architecture using Genomics and Epidemiology (PAGE) Study
- A Genome-Wide RNAi Screen for Factors Involved in Neuronal Specification in
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Recurrent Chromosome 16p13.1 Duplications Are a Risk Factor for Aortic Dissections
- Statistical Inference on the Mechanisms of Genome Evolution
- Genome-Wide Association Study of White Blood Cell Count in 16,388 African Americans: the Continental Origins and Genetic Epidemiology Network (COGENT)
- Chromosomal Macrodomains and Associated Proteins: Implications for DNA Organization and Replication in Gram Negative Bacteria
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



