-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
Transcription Factors Mat2 and Znf2 Operate Cellular Circuits Orchestrating Opposite- and Same-Sex Mating in
Cryptococcus neoformans is a human fungal pathogen that undergoes a dimorphic transition from a unicellular yeast to multicellular hyphae during opposite sex (mating) and unisexual reproduction (same-sex mating). Opposite - and same-sex mating are induced by similar environmental conditions and involve many shared components, including the conserved pheromone sensing Cpk1 MAPK signal transduction cascade that governs the dimorphic switch in C. neoformans. However, the homeodomain cell identity proteins Sxi1α/Sxi2a encoded by the mating type locus that are essential for completion of sexual reproduction following cell–cell fusion during opposite-sex mating are dispensable for same-sex mating. Therefore, identification of downstream targets of the Cpk1 MAPK pathway holds the key to understanding molecular mechanisms governing the two distinct developmental fates. Thus far, homology-based approaches failed to identify downstream transcription factors which may therefore be species-specific. Here, we applied insertional mutagenesis via Agrobacterium-mediated transformation and transcription analysis using whole genome microarrays to identify factors involved in C. neoformans differentiation. Two transcription factors, Mat2 and Znf2, were identified as key regulators of hyphal growth during same - and opposite-sex mating. Mat2 is an HMG domain factor, and Znf2 is a zinc finger protein; neither is encoded by the mating type locus. Genetic, phenotypic, and transcriptional analyses of Mat2 and Znf2 provide evidence that Mat2 is a downstream transcription factor of the Cpk1 MAPK pathway whereas Znf2 functions as a more terminal hyphal morphogenesis determinant. Although the components of the MAPK pathway including Mat2 are not required for virulence in animal models, Znf2, as a hyphal morphology determinant, is a negative regulator of virulence. Further characterization of these elements and their target circuits will reveal genes controlling biological processes central to fungal development and virulence.
Published in the journal: . PLoS Genet 6(5): e32767. doi:10.1371/journal.pgen.1000953
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1000953Summary
Cryptococcus neoformans is a human fungal pathogen that undergoes a dimorphic transition from a unicellular yeast to multicellular hyphae during opposite sex (mating) and unisexual reproduction (same-sex mating). Opposite - and same-sex mating are induced by similar environmental conditions and involve many shared components, including the conserved pheromone sensing Cpk1 MAPK signal transduction cascade that governs the dimorphic switch in C. neoformans. However, the homeodomain cell identity proteins Sxi1α/Sxi2a encoded by the mating type locus that are essential for completion of sexual reproduction following cell–cell fusion during opposite-sex mating are dispensable for same-sex mating. Therefore, identification of downstream targets of the Cpk1 MAPK pathway holds the key to understanding molecular mechanisms governing the two distinct developmental fates. Thus far, homology-based approaches failed to identify downstream transcription factors which may therefore be species-specific. Here, we applied insertional mutagenesis via Agrobacterium-mediated transformation and transcription analysis using whole genome microarrays to identify factors involved in C. neoformans differentiation. Two transcription factors, Mat2 and Znf2, were identified as key regulators of hyphal growth during same - and opposite-sex mating. Mat2 is an HMG domain factor, and Znf2 is a zinc finger protein; neither is encoded by the mating type locus. Genetic, phenotypic, and transcriptional analyses of Mat2 and Znf2 provide evidence that Mat2 is a downstream transcription factor of the Cpk1 MAPK pathway whereas Znf2 functions as a more terminal hyphal morphogenesis determinant. Although the components of the MAPK pathway including Mat2 are not required for virulence in animal models, Znf2, as a hyphal morphology determinant, is a negative regulator of virulence. Further characterization of these elements and their target circuits will reveal genes controlling biological processes central to fungal development and virulence.
Introduction
Many fungi undergo dramatic morphological differentiation during their life cycles. The morphological transition between the yeast form and the pseudohyphal form during mating and invasive growth in Saccharomyces cerevisiae has served as a paradigm for developmental biology due to the well-characterized genetics and robust molecular tools in this organism. For example, the mitogen-activated protein kinase (MAPK) cascade regulating the dimorphic switch in S. cerevisiae, often referred to as the pheromone response pathway, provides a framework for studying morphogenesis in a variety of fungal species, including the human fungal pathogen Cryptococcus neoformans [1]–[5]. Homologs of the core components of the MAPK cascade are conserved among evolutionarily distantly related fungi. However, the downstream targets of the MAPK cascade, which are effectors that ultimately evoke species-specific adaptive responses to external or internal signals, are often not conserved. Therefore, the identity of the downstream transcription factors that activate or repress corresponding target genes in different species are often difficult to reveal through a candidate gene approach.
Dimorphism is a prominent feature shared by the majority of pathogenic fungi that can cause systemic mycoses in human and animals, such as Blastomyces dermatitidis, Candida albicans, Coccidioides immitis, Histoplasma capsulatum, Paracoccidioides brasiliensis, Penicillium marneffei, and Sporothrix schenkii. The ability to switch between the unicellular yeast form and the multicellular hyphal form has been actively investigated in these fungi because dimorphism is not only an important aspect of fungal development but also integral to fungal pathogenicity [6]–[19].
Unlike the majority of other human fungal pathogens, C. neoformans has been typically considered as a yeast and not a dimorphic fungus. In addition, C. neoformans belongs to the Basidiomycota and is more closely related to mushrooms in an evolutionary sense than to the dimorphic fungal pathogens mentioned above that belong to the phylum of Ascomycota. This fungus can cause fatal cryptococcal meningitis in predominantly immunocompromised hosts and also, less frequently, in immunocompetent individuals [20]–[23]. It is second only to tuberculosis in mortality burden in AIDS patients worldwide [24].
C. neoformans yeast cells differentiate into a hyphal form during opposite sex mating and same sex mating. This heterothallic fungus has two opposite mating types: a or α, and opposite sex mating initiates when haploid a and α yeast cells undergo cell-cell fusion [25]–[28]. The two parental nuclei remain separated after the cell-cell fusion event and the resulting a-α dikaryon initiates a morphological switch to dikaryotic hyphal growth with clamp cells connecting neighboring hyphal compartments, which ensures the inheritance of both parental nuclei in each hyphal cell [29], [30]. Nuclear fusion followed by meiosis occurs in swollen aerial hyphal tips (basidia). Four chains of basidiospores are subsequently generated [27], [28]. This a-α mating initiated hyphal growth and basidiospore production has been observed in both serotypes A and D of C. neoformans and also in the sibling species C. gattii [25], [26], [31], [32].
Hyphal growth can also occur through same sex mating (also called monokaryotic fruiting) under conditions similar to those that induce a-α mating. Same sex mating involves cells of only one mating type, commonly α, and has been observed under laboratory conditions, mostly in the serotype D lineage [30], [33]–[35] and rarely in serotype A [36]. Hyphae generated during this process contain one nucleus per hyphal compartment with unfused clamp cells [30], [33]–[35], [37]. During same sex mating, nuclear diploidization could be accomplished by either cell-cell fusion between cells of the same mating type (e.g. α-α mating) or endoreplication [30], [38], and this ploidy increase could occur at multiple developmental stages prior to meiosis in the basidia. Reduction to the haploid state through meiosis in the basidia and generation of haploid spores during unisexual reproduction is similar to processes occurring during traditional a-α mating [30].
The homeodomain cell identity protein complex Sxi1α/Sxi2a encoded by the mating type locus is essential for a-α mating. This protein complex initiates dikaryotic specific events and regulates a-α mating after the cell fusion event, but neither Sxi1α nor Sxi2a is required for same sex mating [39], [40] (Lin X and Heitman J, unpublished results). Thus, same sex mating occurs without the Sxi1α or the Sxi2a protein. In S. cerevisiae and the related pathogenic yeast Candida albicans, the a/α cell type and a-α mating is controlled by the a1/α2 homeodomain heterodimeric complex. Remarkably, Candida lusitaniae and Candida guilliermondii, two haploid species with extant complete sexual cycles, are able to undergo sexual reproduction, yet both species lack the α2 gene and C. guilliermondii is also missing the a1 gene. Recent studies on the evolution of mating type determination and sexual reproduction of pathogenic Candida species have revealed considerable plasticity in the configuration of the mating type locus and the related cellular circuits that govern the establishment of cell type identity and promote sexual reproduction [41]–[44]. By analogy, same sex mating in C. neoformans could involve unique cellular circuits that evoke same sex mating and thereby bypass the central regulatory role of the Sxi1α/Sxi2a complex in sexual reproduction.
The morphological transition from the yeast to the hyphal form during both opposite and same sex mating is governed by the Cpk1 MAPK (mitogen-activated protein kinase) pheromone response signaling pathway [4]. This MAPK pathway controlling development is structurally and functionally conserved among different fungi, including S. cerevisiae [1]–[5], [45]. This pathway involves sequential activation of PAK (p21-activated kinase Ste20), MEKK (MAPK kinase kinase Ste11), MEK (MAPK kinase Ste7), and MAPK (Cpk1) [4]. The C. neoformans homologs of this cascade have been identified and shown to effect the dimorphic transition during mating [4], [46]–[48].
In S. cerevisiae, the downstream transcription factor of the pheromone response MAPK cascade is the homeodomain protein Ste12 [49], [50]. STE12 homologs have also been identified in C. neoformans and are encoded by the mating type locus [51]–[54]. Although overexpression of STE12α induces pheromone production and deletion of the gene results in defective monokaryotic fruiting [51]–[53], disruption of STE12 does not abolish pheromone sensing or opposite sex mating in contrast to other components of the MAPK cascade [4], [52], [53](Figure S1). Thus in C. neoformans Ste12 does not appear to be the sole or major target of the Cpk1 pathway, and instead likely functions in a branched or parallel signaling pathway [4]. In the ascomyceteous dimorphic fungus Penicillium marneffei, the Ste12 homolog stlA is also dispensable for dimorphic switching [55]. Studies of mating pathways in other fungal species also indicate that the key downstream transcription factors are often not conserved across different fungal lineages. Interestingly, HMG domain proteins are frequently the downstream transcription factors in pheromone sensing and mating cascades. Examples include Ste11 in the ascomyceteous yeast Schizosaccharomyces pombe [56] and Prf1 in the basidiomycetous dimorphic plant pathogen Ustilago maydis [57], [58]. C. neoformans and U. maydis are evolutionarily related, yet deletion of the PRF1 homolog in several C. neoformans strain backgrounds does not affect opposite or same sex mating (Lin X, Kraus P, Hicks J, and Heitman J, unpublished results), indicating this HMG protein is not the MAPK target in C. neoformans, further supporting the species-specific nature of effector transcription factors.
Genes encoded by the mating type locus (>20) of C. neoformans play central roles in dimorphic hyphal growth. For example, the mating type locus encodes several key mating elements, including the pheromones (MFα, MFa), pheromone receptors (Ste3α and Ste3a), and components of the Cpk1 MAPK pathway such as Ste11 and Ste20 [30], [40], [47], [48], [59]–[62]. Whereas the Cpk1 MAPK pathway controls both a-α and α-α mating, the Sxi1α/Sxi2a complex is only specifically required for a-α mating. No distinct molecules specifically involved in α-α mating have been discovered and whether the Cpk1 MAPK pathway regulates a-α mating through the Sxi1α/Sxi2a complex also remained to be tested. Moreover, no apparent candidate downstream effector of the Cpk1 MAPK pathway appears to be encoded by the mating type locus.
Dimorphism and pathogenicity are intimately related in many dimorphic fungi and genes involved in dimorphism often regulate virulence [6]–[19]. For example, in C. albicans, a common human pathogen related to S. cerevisiae, the Cph1 (Ste12 homolog) MAPK pathway contributes to virulence [18], [63]–[65]. Similarly, the Prf1 MAPK pathway regulates both dimorphic growth and pathogenicity in the plant pathogen U. maydis [57], [66], [67]. Whether such a relationship also exists in C. neoformans needs to be tested.
The objectives of this study are to identify transcription factors downstream of the pheromone sensing Cpk1 MAPK pathway and to examine (1) if they play distinct roles in a-α and α-α mating and (2) if they are required for Cryptococcus virulence. Here, novel transcription factors involved in dimorphic hyphal growth of C. neoformans were identified via genetic and genomic approaches. First, genes highly expressed in a hyperfilamentous strain were identified by microarray analysis. Second, genes required for filamentation were identified by isolating mutants locked in the yeast phase following insertional mutagenesis. These approaches led to the identification of two transcription factors: Mat2 and Znf2, which are key regulators of a-α and α-α mating. Mat2 is an effector transcription factor of the Cpk1 MAPK pathway, whereas Znf2 functions as a more terminal hyphal morphology determinant. Like other components of the Cpk1 pathway, Mat2 is dispensable for virulence. Interestingly, deletion of Znf2 locks cells in the yeast phase and also enhances virulence. Our results suggest that although components in the signal transduction pathway may not regulate virulence, the inherent ability to grow in different morphotypes does affect Cryptococcus pathogenicity. Together, these findings provide a foundation to elucidate the circuits evoking two different modes of sexual reproduction in C. neoformans and to investigate the relationship between dimorphism and virulence in this ubiquitous human pathogen.
Results
Identification of the HMG protein Mat2 orchestrating hyphal growth via insertional mutagenesis
Because the transcription factors controlled by pheromone sensing MAPK pathways are species-specific and difficult to identify, we employed insertional mutagenesis via Agrobacterium-mediated transformation (AMT), an approach successfully applied in C. neoformans to identify genes regulating virulence traits [68]–[70]. Here this approach was applied to identify genes required for dimorphic hyphal growth. As the Cpk1 MAPK pathway regulates both a-α mating and α-α unisexual mating processes, and disruption of its components severely compromises dimorphic hyphal growth, we hypothesized that the transcription factor target of this pathway would be critical for filamentation during both processes. Because only one cell type is involved in unisexual reproduction, we screened for mutants defective in the transition from yeast to hyphae during α-α unisexual reproduction.
A total of 3600 insertional mutants were generated in a hyperfilamentous strain (XL280α) background. XL280α is an F1 progeny from a cross between strains B3501α and B3502a that share ∼75% genetic contents with each other [35]. The complete genome sequences of both B3501 and B3502 (congenic with JEC21 and isogenic with JEC20) are known [71]. Strain XL280α produces abundant hyphae on a variety of filamentation inducing media (SLAD, Filament Agar, and V8 agar) (data not shown) [35]. The insertional mutants generated were incubated on V8 agar medium at room temperature in the dark and screened for filamentation defects by microscopic examination. Six mutants that consistently displayed no hyphal growth on filamentation inducing media were selected. The insertion sites of these mutants were identified using inverse PCR and sequencing as described previously [68]. Sequences obtained were used in BLAST searches of the C. neoformans genome databases for the serotype D reference strain JEC21 to identify the insertion site and consequently, the disrupted genes (Table 1). The MAPK kinase Ste7 that signals directly upstream of the MAP kinase Cpk1 was identified via this approach (Table 1).
Tab. 1. Genes necessary for filamentation based on insertional mutant phenotype. 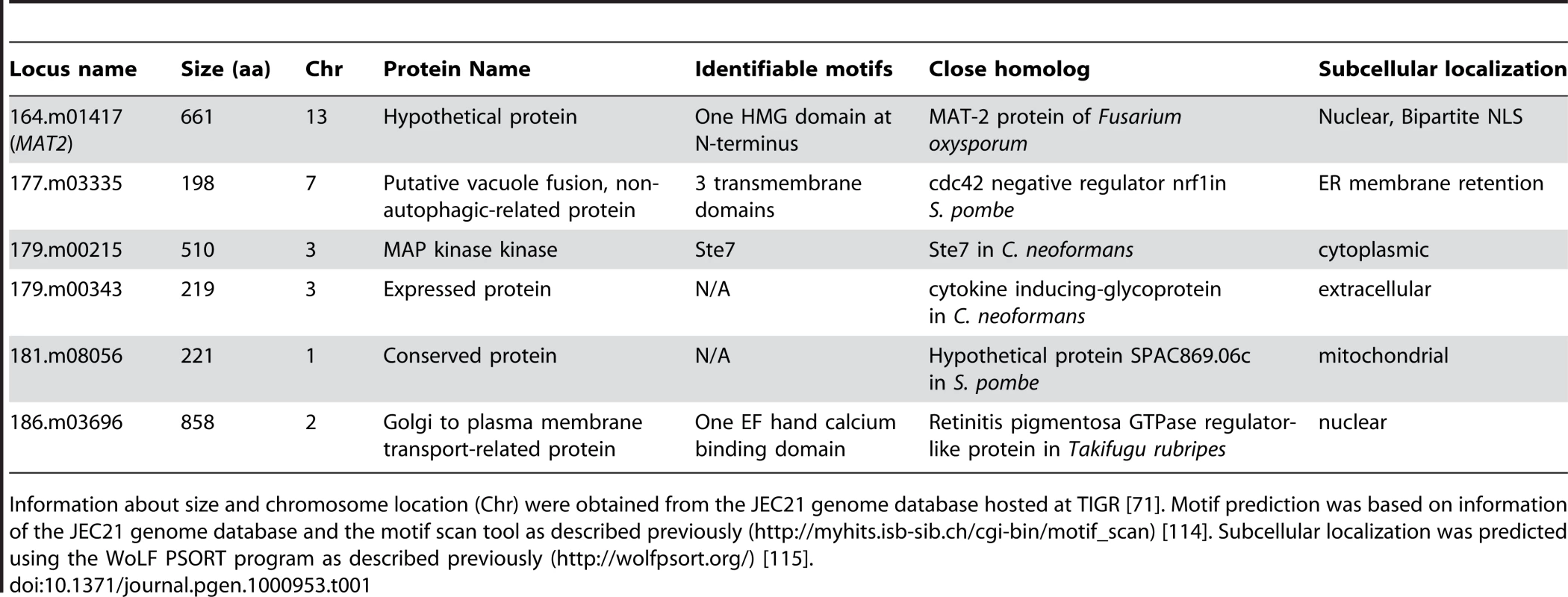
Information about size and chromosome location (Chr) were obtained from the JEC21 genome database hosted at TIGR [71]. Motif prediction was based on information of the JEC21 genome database and the motif scan tool as described previously (http://myhits.isb-sib.ch/cgi-bin/motif_scan) [114]. Subcellular localization was predicted using the WoLF PSORT program as described previously (http://wolfpsort.org/) [115]. Of the six genes identified, only 164.m01417 is likely to encode a transcription factor due to the presence of an HMG domain. The N-terminal HMG domain of the predicted protein shares similarity with the Fusarium oxysporum MAT-2 protein and was therefore named Cryptococcus MAT2. The MAT2 gene is located on chromosome 13 and is unlinked to the mating type locus located on chromosome 4. Because HMG domain proteins have been found to be central transcription factors governning mating in many fungi [72], and because MAT-2 genes are well-known regulators of mating encoded by the mating type locus in many other fungal species [73]–[75], the Cryptococcus MAT2 gene was chosen for further characterization in this study.
A zinc finger protein, Znf2, is highly expressed during hyphal growth
We hypothesized that, like the components of the Cpk1 MAPK cascade, the downstream transcription factors of this pathway function as positive regulators of a-α and α-α mating, and are upregulated during transitions to hyphal growth. Therefore, genes highly expressed in the hyperfilamentous strain XL280α compared to a non-filamentous strain XL34α under filamentation inducing conditions were identified by microarray profiling [76]. Like strain XL280α, strain XL34α is also an F1 progeny from the cross between strains B3510α and B3502a [35]. However, XL34α remains in the yeast form even under hyphal inducing conditions and is non-filamentous (data not shown). The comparison of the transcriptome profiles of XL280α and XL34α revealed 24 genes that exhibited three-fold or higher expression levels in XL280α compared to XL34α (Table 2). Not surprisingly, eight genes are encoded by the α mating type locus. The CPK1 gene encoding the pheromone sensing MAP kinase unlinked to the mating type locus was also highly expressed. Three genes encoding potential transcription factors were identified: ZNF1α located in the mating type locus, ZNF2 resident in another genomic region, and the well-characterized cell identity gene SXI1α located in the mating type locus. Because znf1 mutations in several serotype D genetic backgrounds including both mating types did not abolish cell fusion or hyphal formation during a-α mating, or filamentation during same sex mating (Lin X and Heitman J, unpublished results), Znf1 is apparently not essential for Cryptococcus dimorphic hyphal growth. The SXI1α gene is known to be specifically required during a-α mating and to be dispensable for filamentation during α-α mating [39], [40] (Lin X and Heitman J, unpublished results); thus this gene does not encode a direct transcription factor effector for the Cpk1 MAPK pathway. Therefore, the remaining candidate is the gene encoding a protein with four N-terminal zinc finger C2H2 domains. Because the mammalian Znf2.2 protein is the closest homolog, this putative zinc finger protein was named Cryptococcus Znf2. As is the case for the MAT2 gene, the ZNF2 gene is located on a different chromosome (chromosome 8) from the MAT locus (chromosome 4). The high level of ZNF2 expression in XL280α was corroborated by northern blot analysis (Figure S2).
Tab. 2. Genes highly expressed during filamentous growth in XL280α versus in XL34α. 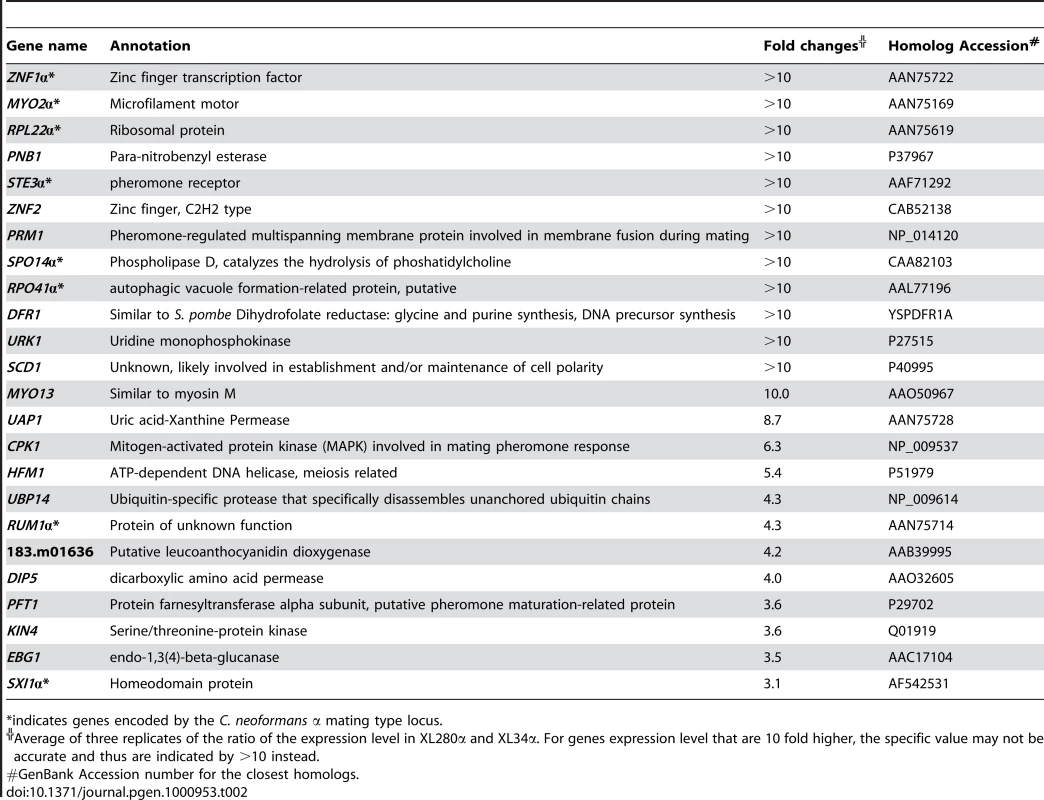
*indicates genes encoded by the C. neoformans α mating type locus. Deletions of MAT2 and ZNF2 abolish filamentation during a–α and α–α mating
To establish the roles of the MAT2 and ZNF2 genes in dimorphic hyphal growth, both genes were deleted via biolistic transformation in the serotype D reference strain JEC21α and strain XL280α backgrounds. In contrast to the hyperfilamentous strain XL280α, which produces filaments surrounding the entire colony periphery on all filamentation inducing media tested, JEC21α only filaments sporadically. Deletion of either the MAT2 or the ZNF2 gene blocked filamentation during α-α same sex mating on filament agar (low nitrogen) in both genetic backgrounds (Figure 1A). Similar results were obtained with other filamentation inducing media such as SLAD (low nitrogen) or V8 medium (contains inositol that induces mating [77]) (data not shown).
Fig. 1. Deletion of either the MAT2 or the ZNF2 gene impairs filamentation during a-α and α-α mating. 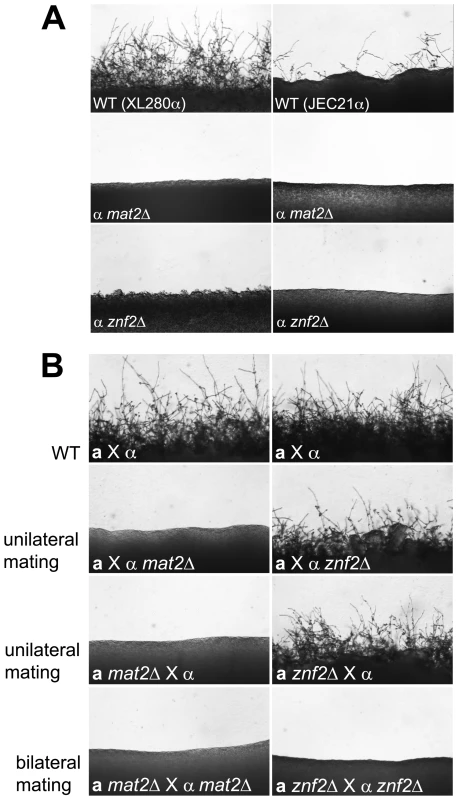
(A) JEC21α, XL280α, and corresponding mat2Δ and znf2Δ mutants (XL XL926, XL576, XL942, and XL574) were individually incubated on V8 medium for 1 week in the dark at 22°C to examine the ability to differentiate. (B) Appropriate α and a mating partners of wild type (JEC21 and JEC20), and mat2Δ (XL926 and XL961) and znf2Δ (XL576 and XL879) mutants in the JEC21α background were mixed and co-cultured on filamentation agar medium for 48 hours in the dark at 22°C to examine filamentation during unilateral and bilateral matings. a-α mating of C. neoformans is initiated when haploid cells of opposite mating types (a and α) fuse with each other and produce dikaryotic hyphae. Because a-α mating involves two partners of opposite mating types, both unilateral (a mutant strain crossed with a wild type partner) and bilateral matings (a mutant strain crossed with a mutant partner) for mat2Δ and znf2Δ mutants were examined to establish their roles in a-α mating. No mating hyphae were produced by mat2Δ mutants in either unilateral (α mat2Δ × a, or α × a mat2Δ) or bilateral matings (α mat2Δ × a mat2Δ) (Figure 1B). This indicates that the presence of a functional MAT2 gene in both the α and the a partner is required for a-α mating to occur. Upon prolonged incubation (>2 weeks), unilateral matings occasionally produced a few sporadic mating filaments and thus the block to mating is not absolute (data not shown). A similar phenotype has also been observed in mutants of the Cpk1 MAPK pathway [4]. In contrast, with znf2Δ mutants, mating hyphae and basidiospores were produced during unilateral matings (α znf2Δ × a, or α × a znf2Δ), albeit at a slightly reduced level compared to matings between wild type partners (α × a). However, filamentation was entirely abolished in bilateral matings (α znf2Δ × a znf2Δ) (Figure 1B), even following prolonged incubation. This indicates that znf2Δ mutants are bilaterally sterile but the presence of one functional ZNF2 gene in one of the two partners is sufficient for mating to proceed and Znf2 from either partner can compensate for the absence in the other partner.
Mat2 is required for cell–cell fusion whereas Znf2 is required for hyphal morphogenesis during mating
a-α mating involves several steps: cell fusion between a and α partners, dikaryotic hyphae formation, basidium formation, and sporulation. Because mutation of either MAT2 or ZNF2 blocked hyphal formation during bilateral matings, these two genes could be critical for cell-cell fusion, the initiation of hyphal morphogenesis, or both. To define the step(s) in α-a mating during which Mat2 and Znf2 function, their roles in cell fusion were examined. Yeast cells of genetically marked wild type α and a strains, α mat2Δ and a mat2Δ mutants, and α znf2Δ and a znf2Δ mutants were paired and incubated on V8 agar for 15 hours in the dark at room temperature. The cocultured cells were collected and plated on appropriate media to select for fusion products (see “Materials and Methods” for details)(Figure 2). No fusion products were detected from the bilateral pairing of α mat2Δ and a mat2Δ mutants. We further tested the efficiency of cell fusion from the unilateral pairing of a wild type partner and mat2Δ mutants (α mat2Δ + a or α + a mat2Δ) under the same experimental conditions. Again, no cell fusion products were recovered. These observations indicate that Mat2 is required for cell-cell fusion events and both partners must carry a functional MAT2 gene in order for cell fusion to occur. This phenotype is again reminiscent of that of Cpk1 MAPK cascade mutants [4].
Fig. 2. Znf2 is required for hyphal formation after cell-cell fusion during a-α mating. 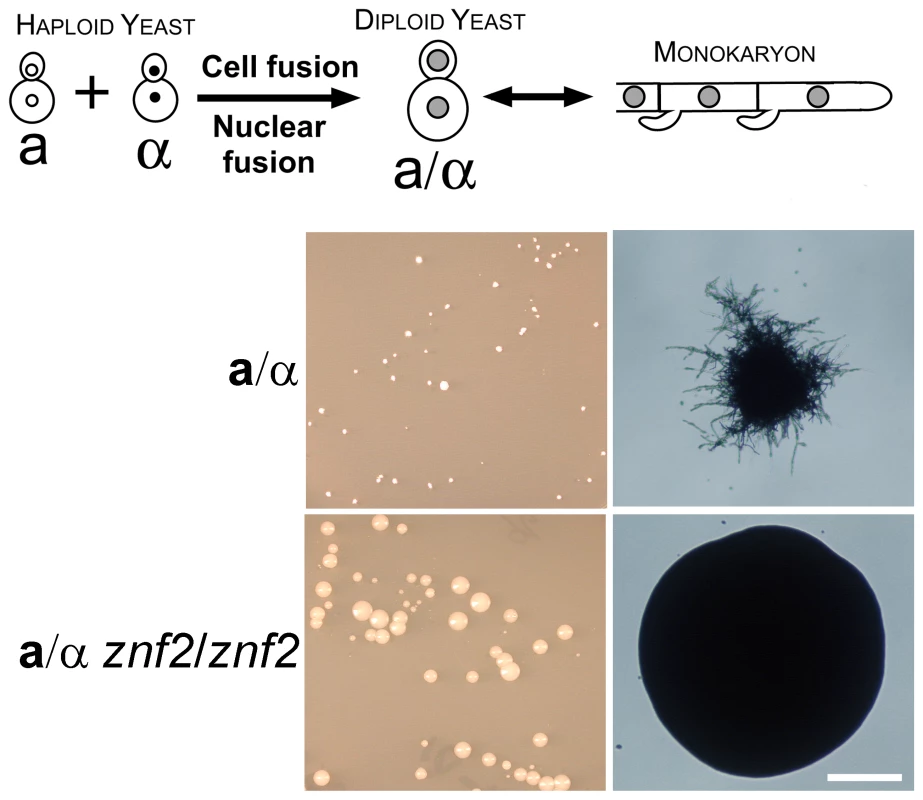
The schematic diagram depicts the mating process of wild-type strains. Haploid α and a yeast cells were co-incubated and the cell-cell fusion products, when selected at high temperatures, will undergo nuclear fusion and become heterozygous α/a diploid yeast cells. When the environment becomes favorable for filamentation (by lowering the temperature), the α/a diploid cells will undergo a morphological transition, produce hyphae, and eventually undergo meiosis and sporulate (not shown here). Appropriately marked α and a strains of wild type (XL877 and XL878) and znf2Δ mutants (XL874 and XL875) were paired, mixed, and co-cultured on V8 medium for 15 hours at room temperature in the dark. The cocultures were collected and transferred to YNB minimal medium to select for fusion products as shown in the left panel. Microscopic images of the colony derived from the fusion event are shown in the right panel. Scale bar, 200 micrometers. In contrast, the bilateral pairing of α znf2Δ and a znf2Δ mutants yielded 600% more fusion products than the control, indicating that Znf2 is not necessary for cell fusion, and may normally repress cell fusion during α-a matings. Interestingly, while the fused wild type diploids (a/α) produced very small colonies and then readily filamented at ambient temperatures (Figure 2), the fused mutant a/α diploids (znf2Δ/znf2Δ) derived from XL875α (znf2::NATr ade2) and XL874a (znf2::NATr lys1) mutants did not produce any filaments and continued growing as budding yeast, yielding large yeast colonies under the same conditions (Figure 2). This result indicates that Znf2 is critical for hyphal formation and functions after the cell-cell fusion event during a-α mating. As formation of conjugation tubes is necessary for cell-cell fusion during bisexual mating [27], the observation that znf2Δ cells can undergo cell fusion successfully but are unable to form hyphae suggests that the mechanisms to produce conjugation tubes are not identical with those to produce true hyphae.
To determine if Znf2 also functions after the cell-cell fusion event during same sex mating (α-α mating), mutant α/α diploids (znf2Δ/znf2Δ) were generated by fusion between two auxotrophically marked α mutants XL872α (znf2::NATr ade2 lys1) and XL873α (znf2::NATr ura5). The α/α homozygous diploid mutant (znf2Δ/znf2Δ) again only grew in the yeast form (data not shown), similar to the a/α heterozygous diploid mutant (znf2Δ/znf2Δ) described above. Taken together, the key role of Znf2 is to enable or support hyphal morphogenesis after cell fusion.
Mat2 regulates pheromone sensing and response, whereas Znf2 does not
The Cpk1 MAPK pathway mediates pheromone sensing and response during mating [4]. To determine whether Mat2 and Znf2 are important for pheromone production during mating, transcription of the MFα pheromone gene in wild type, mat2Δ, znf2Δ, and ste7Δ mutants was monitored during bilateral a-α matings. The ste7Δ mutants were chosen as representative of the Cpk1 MAPK cascade as Ste7 is the MEK upstream of the MAPK Cpk1, and also because this gene was identified in our insertional mutagenesis screen (Table 1). As shown in Figure 3A, no pheromone transcript was detected in the wild type prior to the initiation of mating, but its level increased at 6 hours post-coincubation, reached the highest level at 15 hours post-coincubation, and returned to a lower basal level by 24 hours. A similar pheromone expression pattern of wild type cells during mating has been observed independently (Griffith B, Fraser J, and Heitman J, unpublished results). In contrast, no MFα transcript was detected in either the ste7Δ or the mat2Δ mutants over the time course studied, indicating that Mat2 and Ste7 are required for pheromone induction. This phenotype of the mat2Δ and the ste7Δ mutants is in accord with the known roles of the Cpk1 MAPK pathway in pheromone sensing and response. MFα expression in the znf2Δ mutant followed a pattern similar to wild type, with an increased level of pheromone production during early stages of mating followed by reduced levels. Interestingly, at all time points examined (except at the zero time point at which no pheromone expression was observed), a higher level of MFα was observed in the znf2Δ mutant. The elevated level of pheromone expression during bilateral matings in the znf2Δ mutants may be responsible for the increased efficiency in cell fusion observed. Taken together, our evidence indicates that Mat2 likely functions as a direct target of the Cpk1 MAPK pathway, whereas Znf2 functions further downstream.
Fig. 3. Mat2 is required for pheromone sensing and production, whereas Znf2 is dispensable for the response during a-α mating. 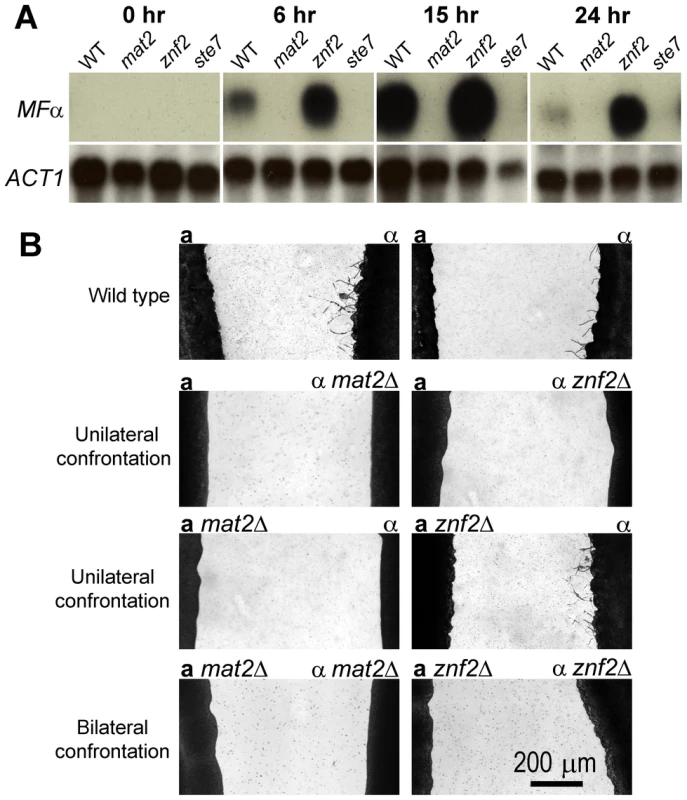
(A) Northern blot analysis of the expression pattern of the MFα gene during a-α mating in wild type, mat2Δ, znf2Δ, and ste7Δ mutants in JEC21 background at 0 hours, 6 hours, 15 hours, and 24 hours post coinoculation of the a and α mating partners. The expression level of the actin gene (ACT1) serves as a control. (B) Confrontation assays of the effect of mat2 and znf2 mutations on the ability of a and α cells to produce and respond to pheromones. Scale bar, 200 micrometers. Confrontation assays were performed to assess the effect of mat2 and znf2 mutations on the ability of a and α cells to produce and respond to pheromones. When a and α cells are grown in close proximity but not mixed, α cells produce conjugation tubes or monokaryotic hyphae in response to a cells/MFa pheromone, whereas a cells predominately become enlarged in response to α cells/MFα pheromone [59]. In addition to inducing a cell responses, MFα pheromone also provides a positive autocrine feedback to the pheromone response pathway in α cells to promote cell fusion [78]. As shown in Figure 3B, a mat2Δ cells failed to induce wild type α cells to produce conjugation tubes or filaments, indicative of defects in pheromone production. The α mat2Δ cells did not produce any filaments when confronted with wild type a cells, reflecting defects also in response to pheromone produced by the confronting mating partner. The phenotype of mat2Δ mutants was identical to that of ste7Δ mutants (Figure 3B). These observations are consistent with the northern blot analysis showing no detectable MFα pheromone gene expression during bilateral matings of mat2Δ or ste7Δ mutants and are also consistent with previous findings about the critical roles of components of the Cpk1 MAPK pathway in pheromone production and response [4]. In contrast, the a znf2Δ mutant cells successfully induced wild type α cells to produce filaments, indicating no defects in pheromone production. However, the α znf2Δ cells did not produce any conjugation tubes or hyphae when confronted with wild type a cells. Based on the northern blot analysis and cell fusion assays, α znf2Δ cells have no defect in producing or responding to pheromone. Thus, the defect of the znf2Δ mutant likely reflects an inability to form hyphae. Again, these lines of evidence support the hypothesis that Mat2 functions as the direct transcription factor effector of the Cpk1 MAPK pathway, whereas Znf2 is a more terminal morphology determinant.
To examine the epistatic relationship between ZNF2 and the Cpk1 MAPK pathway, a znf2Δ cpk1Δ double mutant was isolated following a genetic cross between the znf2Δ mutant and the cpk1Δ mutant. This double mutant (XL1131α) showed severe impaired unilateral mating when coincubated with the wild type mating partner JEC20a (data not shown), similar to the cpk1Δ single mutant [4]. This observation again suggests that Znf2 functions downstream of Cpk1.
To further test the relationship between Mat2, Znf2, the Cpk1 pathway, and the Sxi1α/2a complex, we examined the expression pattern of the CPK1, MAT2, SXI1α, and ZNF2 genes during bilateral matings in the following strain pairs: α × a, α znf2Δ × a znf2Δ, α mat2Δ × a mat2Δ, α ste7Δ × a ste7Δ, and sxi1αΔ × sxi2aΔ, which were cocultured on V8 medium (pH 7.0) for 24 hr. As shown in Figure 4, minimal or no ZNF2 or SXI1α transcripts were detectable in the ste7Δ and mat2Δ mutants, indicating that the Cpk1 MAPK pathway regulates the transcription of ZNF2 and SXI1α. The transcription level of the CPK1 gene was downregulated in the mat2Δ and possibly also in ste7Δ mutants, suggesting that mutations in the Cpk1 MAPK pathway may reduce transcription of other components in the pathway, consistent with positive feedback regulation of the pheromone sensing pathway. The expression levels of SXI1α and possibly CPK1 were slightly reduced in the znf2Δ mutants, suggesting that ZNF2 may also normally positively regulate the expression of these genes. On the other hand, the transcription levels of MAT2 and CPK1 were not affected in the sxi1αΔ/sxi2aΔ mutants, suggesting that the Sxi1α/Sxi2a complex does not regulate the Cpk1 MAPK pathway. In addition, no significant change in the transcription level of ZNF2 was observed in the sxi1αΔ/sxi2aΔ mutants, indicating that the Sxi1α/Sxi2a complex does not regulate Znf2 at the transcriptional level. Similar gene expression results in these mutants during bilateral matings were also observed in the genome wide expression studies (Tables S1, S2, S3). Again, these gene expression results support the conclusion that Mat2 is part of the Cpk1 MAPK pathway and together they regulate the expression of ZNF2 and SXI1α/2a during mating. Znf2 also may play a modest role in regulating the transcription of CPK1 and SXI1α. In contrast, deletion of SXI1α shows no effect on the transcription the MAPK pathway or ZNF2.
Fig. 4. Znf2 and the Sxi1α/2a complex do not regulate Mat2 or the Cpk1 pathway at the transcript level. 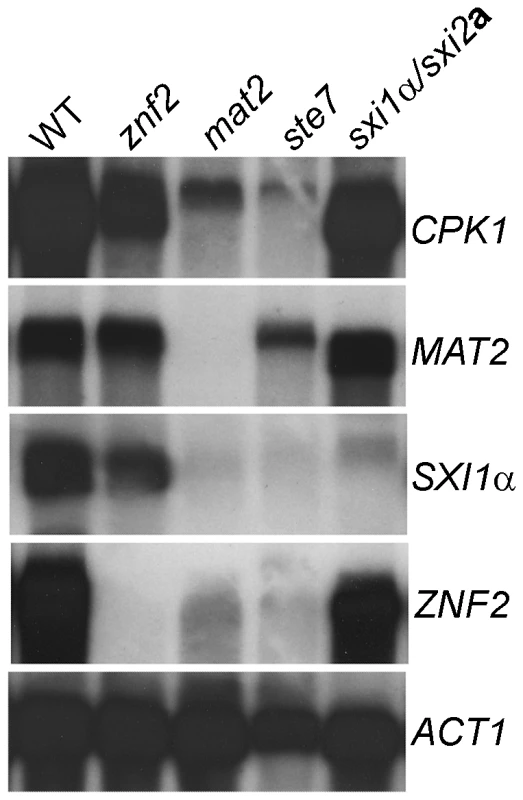
The expression pattern of the CPK1, MAT2, SXI1α, and ZNF2 genes during bilateral matings in the following strain pairs (α × a, α znf2Δ × a znf2Δ, α mat2Δ × a mat2Δ, α ste7Δ × a ste7Δ, and sxi1αΔ × sxi2aΔ in JEC21 background) that had been cocultured on V8 medium (pH = 7.0) for 24 hr. Znf2 evokes hyphal formation, whereas Mat2 is dispensable
As both Mat2 and Znf2 are required for hyphal growth during a-α and α-α mating, it was not evident whether abolished hyphal growth in the mat2Δ and znf2Δ mutants was attributable to their role in hyphal morphogenesis per se or to an involvement in relaying signals to stimulate the initiation of hyphal formation. For example, the homeodomain cell identity protein complex Sxi1α/Sxi2a initiates hyphal growth after the cell-cell fusion event during a-α mating [39], [40], but neither Sxi1α nor Sxi2a is required for formation of hyphae as sxi1αΔ or sxi2aΔ mutants can still filament during same sex mating [39], [40] (Lin X and Heitman J, unpublished results). Thus, the Sxi1α/Sxi2a complex stimulates mating hyphal formation [39] by relaying stimuli, but is not required for hyphal morphogenesis per se.
Because Mat2 is required for cell-cell fusion, examining its role in hyphal formation during mating after the cell-cell fusion step is challenging. To circumvent the cell-cell fusion step, we decided to take advantage of the previous observation that haploid cells carrying the Sxi1α/Sxi2a complex mimic a/α diploids or a-α dikaryotic cells and can filament in response to temperature and environmental cues [39], [40], [69], [79] (Figure 5, top panel). Transforming a cells with the α cell identity gene SXI1α, or α cells with the a cell identity gene SXI2a generates haploids with the cell-identity complex derived from a and α cells. Here we transformed mutants of a mating type with the SXI1α gene under the control of the constitutively active GPD1 promoter. As a control, the presence of PGPD1-SXI1α in ste7Δ, mat2Δ, or znf2Δ mutants in the α cell type did not stimulate filament production (data not shown). As shown in Figure 5, transforming the a ste7Δ mutant with PGPD1-SXI1α enabled the originally non-filamentous a cells to produce hyphae (wild type JEC20a does not produce hyphae). Similarly, the presence of the PGPD1-SXI1α gene in a mat2Δ cells also stimulated formation of hyphae (Figure 5). In contrast, no filaments were observed when PGPD1-SXI1α was transformed into the a znf2Δ cells (Figure 5).
Fig. 5. Mat2 and the Cpk1 MAPK cascade are not required for hyphal morphogenesis whereas Znf2 is a hyphal morphology determinant. 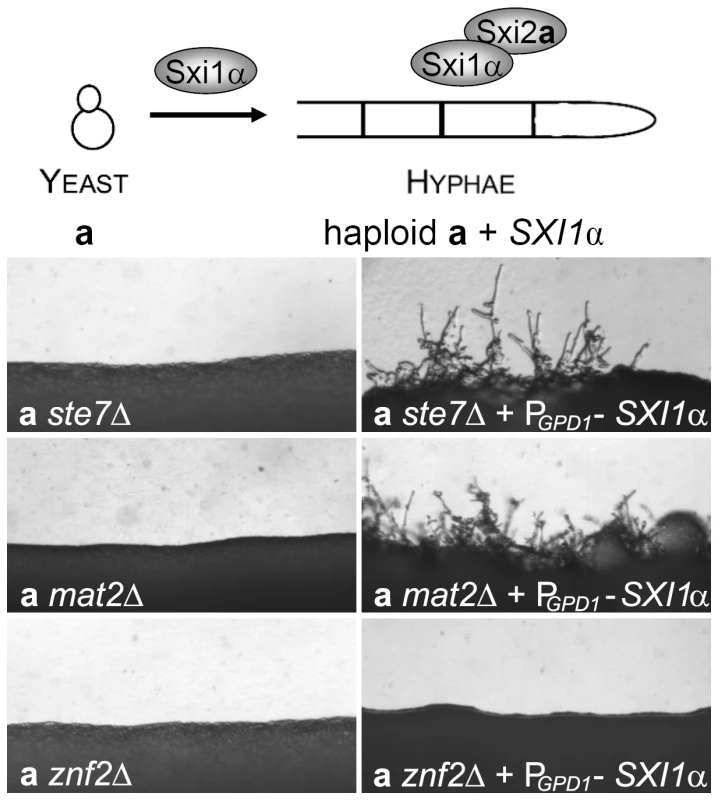
The schematic diagram shows that if haploid a cells that harbor the SXI2a gene in their genome are transformed with the SXI1α gene (indicated by the Sxi1α above the arrow), both the SXI1α and SXI2a genes will be expressed in the a cells. The protein complex formed by these two cell-identity proteins renders the haploid a cells competent to produce hyphae like a/α diploid or a-α dikaryons without the requirement for the fusion of α and a cells. The a cells with ste7Δ, mat2Δ, or znf2Δ mutation in JEC21 background and the corresponding mutants bearing the PGPD1-SXI1α transgene were cultured on filamentation agar medium and incubated in the dark for 48 hours at 22°C. These observations indicate that Mat2, like Ste7, is not necessary for hyphal morphogenesis per se but is required for relaying the pheromone stimulus to initiate hyphal formation. Conversely, Znf2 is not required for sensing pheromone cues that stimulate hyphal growth, but it is a terminal determinant of hyphal morphogenesis. That said, Znf2 does influence pheromone sensing and cell fusion as the cell fusion efficiency and the pheromone transcript level are higher in the znf2Δ mutants compared to the wild type control. However, these differences caused by ZNF2 deletion are quantitative instead of qualitative. In contrast, its function is hyphal growth is absolutely required.
Mat2 is a key element of the Cpk1 MAPK pathway, while Znf2 functions in distinct but overlapping pathways
Genes transcriptionally regulated by the two novel transcription factors were examined at the whole genome level employing the C. neoformans genome wide 70 mer oligonuleotide microarray based on the JEC21α genome sequence. Transcripts produced by ste7Δ, mat2Δ, and znf2Δ mutants of both mating types in the JEC21 (α)/JEC20(a) backgrounds during bilateral matings after 15 hours of co-incubation on V8 medium were compared to those produced by the wild type (JEC21 × JEC20). Genes differentially expressed in the mat2Δ mutants under this condition are almost identical with those in the ste7Δ mutants (Figure 6)(Table S1). Discrepancies in a few genes might be caused by variations in of the microarray experiments. This result further supports the conclusion that Mat2 is a direct downstream transcription factor of the Cpk1 MAPK pathway. Many down regulated genes encode products known to be involved in mating, including those involved in pheromone synthesis and processing, pheromone receptors, G proteins upstream of the MAPK pathway, the MAP kinase Cpk1, and the homodomain protein Sxi1α (Figure 6)(Table S1). Down-regulation of these genes is consistent with the northern hybridization analysis presented earlier. Genes involved in morphogenesis were the second largest group that showed transcriptional differences compared to wild type, including several genes in the septin family and other cell cytoskeletal genes (cell wall components). Genes likely involved in transport, transcription, or development were also identified.
Fig. 6. Gene expression profiles of ste7Δ, mat2Δ, and znf2Δ mutants during bilateral mating. 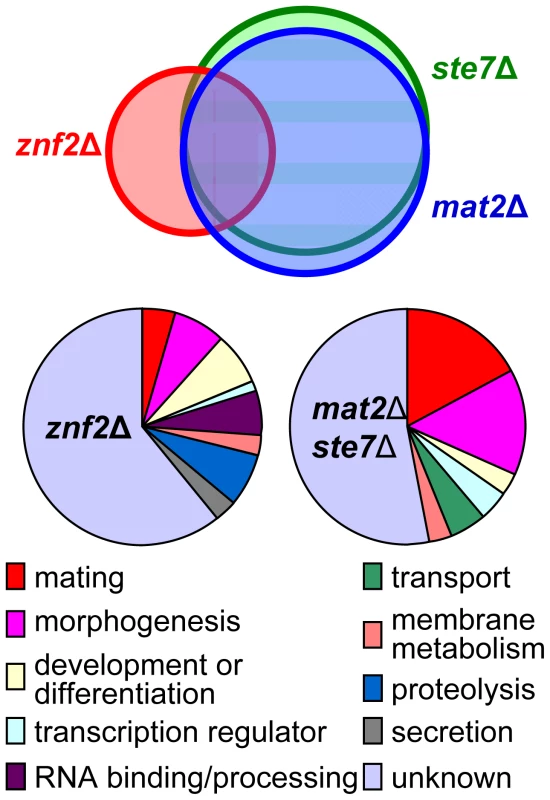
The top panel shows the nearly identical profiles between ste7Δ and mat2Δ mutants with 96% overlap in contrast to only 47% congruence with genes differentially expressed in znf2Δ mutants. The bottom panel shows a classification of the genes that were differentially expressed in the mutants based on GO ontology. In contrast, fewer genes were found to be differentially expressed in the znf2Δ mutants during bilateral mating (Figure 6)(Table S2). The most striking difference in the expression profile of znf2Δ mutants compared to those of the ste7Δ or the mat2Δ mutants was that genes involved in the pheromone sensing and response were not significantly differently expressed in the znf2Δ mutants when compared to the wild type (Table S2). Additionally, genes involved in cellular transport were not significantly changed, and fewer genes involved in morphogenesis were identified. This observation supports the conclusion that Znf2 does not regulate the pheromone sensing pathway and functions as a more terminal hyphal morphogenesis determinant. It is interesting to note that more genes functioning in RNA binding or processing, protease activities, and secretion were differentially expressed in the znf2Δ mutant compared to the ste7Δ or the mat2Δ mutants. There are many uncharacterized genes with no defined homologs, and these genes cannot at present to be assigned to any specific process.
To examine the expression profiles of these mutants during unisexual mating, transcripts produced by ste7Δ, mat2Δ, and znf2Δ mutants in the XL280(α) background during unisexual mating after 24 hours of incubation on V8 medium were compared to those produced by the wild type strain XL280. Although different numbers of genes were selected compared to the those described above due to the difference in quality of the microarray data, about 48% of the genes that were selected for differential expression in the ste7 mutant during unisexual mating in the XL280 background were identical to those selected in the α ste7 × a ste7 bilateral mating in the JEC21/JEC20 background (highlighted in Table S3). Similarly, 42% of the genes for the mat2 mutant and 48% for the znf2 mutant during unisexual mating in the XL280 background were identical to those selected in the α mat2 × a mat2 and α znf2 × a znf2 bilateral matings in the JEC21/JEC20 background respectively (Table S3). Again, genes involved in the pheromone response pathway were not significantly differently expressed in the znf2Δ mutant but were in the ste7Δ and mat2Δ mutants. Genes that were dramatically suppressed in the mutants compared to the corresponding wild type controls tend to be consistently represented under both conditions.
Mat2 is dispensable for virulence, while Znf2 is a negative regulator of pathogenicity
Dimorphic transitions between yeast and hyphae have been linked to virulence in several pathogenic fungi. To determine if Mat2 and Znf2 are necessary for Cryptococcus pathogenicity, the impact of gene deletions on Cryptococcus virulence potential was examined in a murine inhalation model of cryptococcosis. Because serotype A is in general more prevalent and more virulent than serotype D [80]–[82], the roles of Mat2 and Znf2 in virulence were examined in the highly virulent serotype A H99 background. Disruption of the MAT2 and the ZNF2 genes in strain H99 caused similar defects during a-α mating as observed in serotype D backgrounds (data not shown), indicating conserved functions of these two proteins in the two serotypes. Because Mat2 is required for cell fusion and the mat2Δ mutant is unable to complete unilateral mating, the linkage between the mutation and the phenotype was only confirmed for znf2Δ via genetic crosses (data not shown). Introducing a wild type copy of ZNF2 ectopically into the znf2Δ mutant restored the mating defect, indicating that the phenotype was indeed caused by mutation in the ZNF2 gene (data not shown). To assess the impact of the deletion of MAT2 or ZNF2 on Cryptococcus virulence potential, animals were intranasally infected with yeast cells of the deletion mutants (ste7Δ, mat2Δ, and znf2Δ) and wild type H99 and their survival was monitored. As shown in Figure 7, the ste7Δ and mat2Δ mutants were of equivalent virulence compared to the wild type control. This result is consistent with previous observations that components of the Cpk1 MAPK pathway are dispensable for Cryptococcus virulence in murine models of cryptococcosis [4]. That disruption of MAT2 exerted no or minimal effect on virulence is in accord with the model that Mat2 is a downstream transcription factor of the Cpk1 MAPK pathway.
Fig. 7. Mat2 is dispensable for virulence, while Znf2 is a negative regulator of pathogenicity. 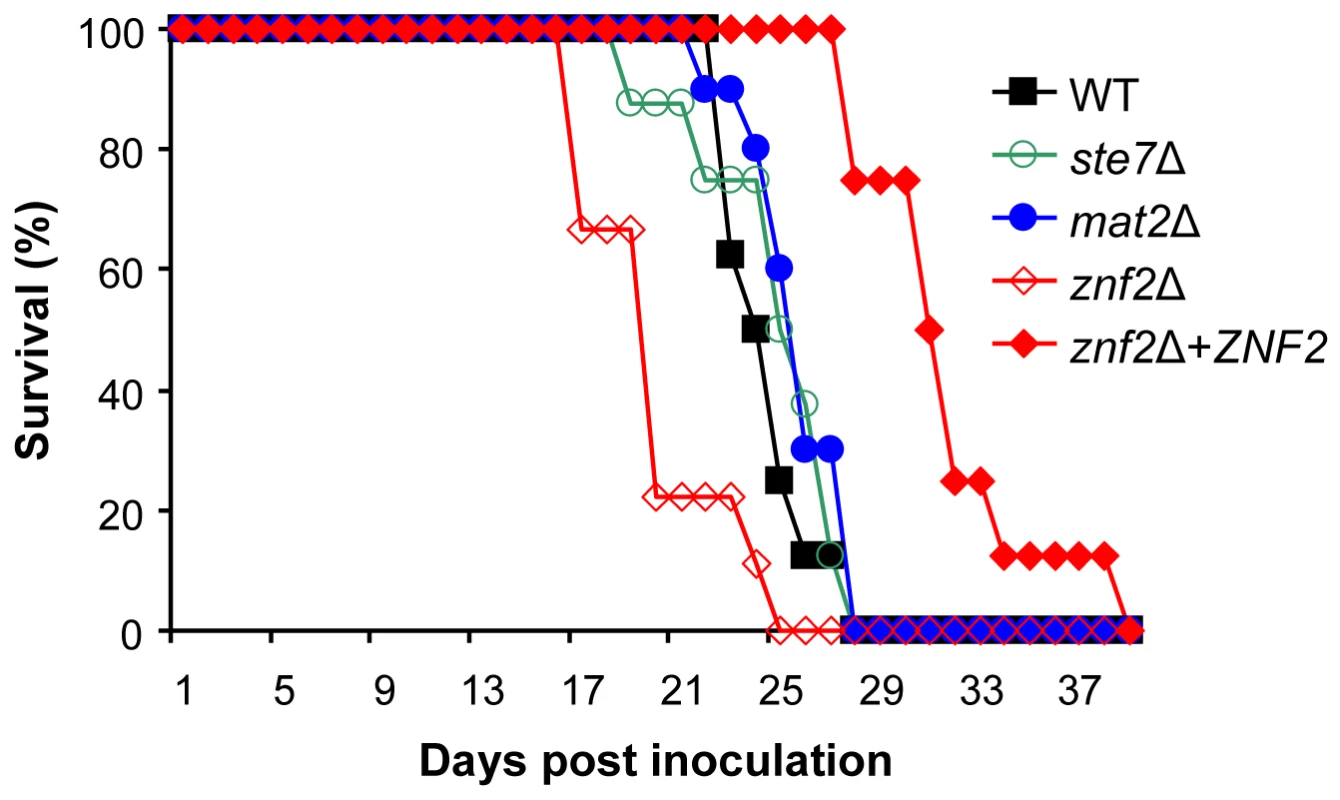
Animals (10 each group) were intranasally infected with 5×104 yeast cells of the wild type (H99), ste7Δ (YSB345), mat2Δ (XL1598), znf2Δ (XL1601), and znf2Δ-ZNF2 (XL1643) strains. The survival rates of animals were plotted against time after inoculation, and P values compared to the wild type control are: ste7Δ (P = 0.68), mat2Δ (P = 0.048), znf2Δ (P = 0.0026), znf2Δ+ZNF2 (P = 0.00034). In contrast, a modestly enhanced virulence was observed for the znf2Δ mutant and the difference from the wild type is statistically significant (Figure 7A). This virulence experiment was repeated with a modestly higher infectious dose and a similar pattern was observed (Figure S3). The complemented strain, in which the ZNF2 gene with its native promoter and terminator was ectopically integrated into the genome, was modestly less virulent than the wild type strain. Because the integration site of the wild type copy of ZNF2 occurs in an intergenic region (data not shown), it is unlikely that other genes regulating virulence were affected. The observation that the introduced wild type copy of ZNF2 restored the mating ability of the znf2Δ mutant suggests that the reduced virulence in this strain is likely due to a position effect, which may increase the activity of Znf2. This result suggests that Znf2 acts as a negative regulator of virulence and that there might be an inverse relationship between ZNF2 activity and virulence potential. This hypothesis warrants additional testing in future investigations.
Mat2 and Znf2 do not regulate defined virulence traits in vitro
To determine if the virulence potential of the mat2Δ and znf2Δ mutants correlates with their in vitro phenotypes with respect to well-characterized virulence traits of Cryptococcus, the mutants and control wild type strains were assayed for melanization, capsule production, and the ability to grow at mammalian body temperature 37°C. Disruption of MAT2 or ZNF2 did not cause any apparent alteration in these traits in all of the backgrounds tested (Figure S4). Our results indicate that Znf2 regulates Cryptococcus virulence via means other than the well-defined characterized virulence factors examined.
Discussion
Several lines of evidence presented in this study converge to implicate Mat2, an HMG transcription factor, and Znf2, a zinc finger transcription factor, as central components governing opposite and same sex mating in C. neoformans. The congruence of the transcriptional profiles of ste7Δ and mat2Δ mutants and their parallel phenotypes in vitro and in vivo provide evidence that these two proteins function in a common pathway. Based on previous observations that HMG proteins serve as the downstream transcription factors in the mating pathway in diverse fungal species [72], [83] (Figure 8B), we hypothesize that Mat2 is a direct target of Cpk1 in Cryptococcus (Figure 8). The well-known transcription factor Ste12 is unlikely to be a direct major downstream target of Cpk1 based on the observations that deletion of STE12 only modestly reduces but does not abolish either opposite sex mating or same-sex mating (Figures S1, S5). However, a direct interaction between Cpk1 and Mat2 has yet to be confirmed. Similarly, the relationship between the MAPK pathway/Mat2 and Ste12 remains to be illustrated.
Fig. 8. Mat2 and Znf2 operate cellular circuits orchestrating opposite- and same-sex mating in C. neoformans. 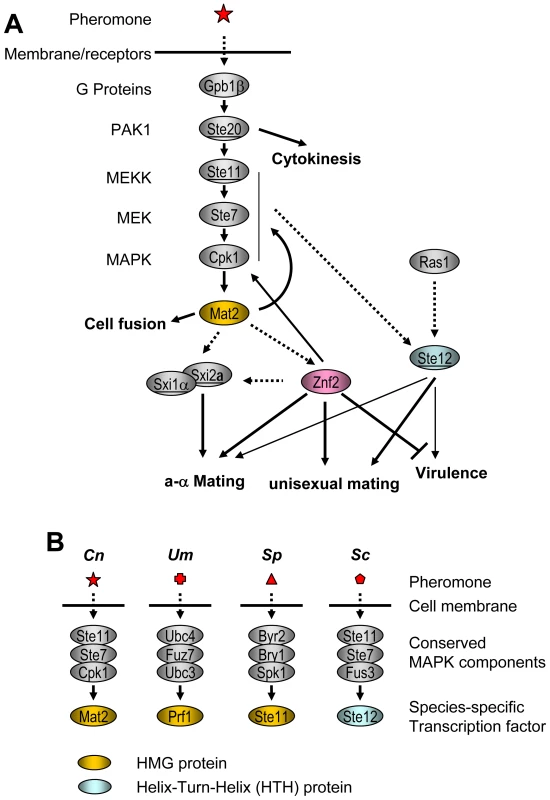
(A) Mat2 is depicted as the direct target of the MAPK pathway and it further serves to regulate the homodomain complex and Znf2. Genes encoded by the MAT locus are underlined. The dotted arrow indicates predicted interactions. (B) HMG domain proteins serve as the transcription factor downstream of the MAPK in the pheromone sensing pathways of divergent fungal species. Cn: C. neoformans. Um: U. maydis. Sp: S. pombe. Sc: S. cerevisiae. In comparison, our evidence indicates that Znf2 is required for hyphal morphogenesis during both unisexual mating and bisexual mating, and is not critical for the pheromone response (Figure 8A). Znf2 may regulate the cell identity homeodomain protein Sxi1α at the transcriptional level during mating.
In dimorphic fungi, the link between dimorphism and virulence has been recognized for decades and is under intensive investigation [6]–[19]. C. neoformans, on the other hand, has been typically considered as a yeast [6]. In addition, because the two central pathways involved in mating dimorphic hyphal growth (the Cpk1 MAPK pathway and the Sxi1α/Sxi2a complex) have been shown to have no or minimal impact on virulence, the association between dimorphism and virulence in C. neoformans has not been a focus of research efforts [6]. However, based on the evidence adduced in this study, we submit that the impaired dimorphic mating hyphal growth in the mutants of the Cpk1 MAPK pathway or the Sxi1α/Sxi2a complex may be attributable to defects in sensing extra-cellular or intra-cellular cues that stimulate initiation of filamentation rather than impaired ability to undergo hyphal growth. For example, disruption of the SXI1α gene affects a-α mating hyphal growth but filamentation during same sex mating still occurs [40]. Here we show that disruption of STE7 and MAT2 markedly impairs hyphal growth during both a-α and α-α mating, but with the activation of Sxi1α/Sxi2a, hyphal growth can be partially restored (Figure 5). Studies in C. albicans and U. maydis have also shown that disruption of the pheromone-sensing pathway results in defective hyphal growth under some but not all conditions [18], [84]–[88]. These studies echo the same message that mutations in signal transduction pathways do not abrogate the inherent ability of a strain to undergo hyphal growth. If it is the ultimate inherent ability to undergo dimorphic morphogenesis, not relay the signal, that affects virulence in C. neoformans, then it is not surprising that Mat2, like other components in the Cpk1 MAPK signal transduction pathway or the Sxi1α/Sxi2a complex, is dispensable for virulence.
Because Cryptococcus hyphae are rarely observed in human or animal tissues [37], [89]–[94], and the host conditions suppress hyphal growth, it is possible that cells locked in the yeast form, either naturally or by genetic manipulation (e.g. deletion of ZNF2), will show enhanced virulence in animal models of cryptococcosis, whereas cells locked in a filamentous form would be avirulent. This hypothesis is supported by previous observations that the filamentous form shows drastically reduced pathogenicity in animal models when used as the infectious inoculum [6], [95]–[98] and the filamentous form can immunize animals against subsequent challenge of virulent C. neoformans in the yeast form [99]–[101]. Thus, studies on cell shape and virulence in this organism could have significant clinical impact. However, the exact molecular mechanisms by which Znf2 determines the dimorphic transition to hyphal form and affects virulence potential remain elusive and may reflect distinct processes that Znf2 regulates. Based on the gene expression studies of the znf2Δ mutants during mating (Figure 6 and Tables S2, S3), Znf2 may regulate (1) lysis of protein or carbohydrate, (2) RNA processing, (3) tracking, or (4) lipid metabolism. Identification and investigation of the direct targets of this transcription factor will be essential to understand the functions of Znf2 in morphogenesis and virulence, and will be the focus of our future research.
The recent discovery that C. albicans can also undergo same sex mating underscores the importance of transitions in modes of sexual reproduction in pathogenic microbes [42], [102]. That two divergent human pathogens both underwent independent transitions enabling same sex mating to occur illustrates the plasticity of sexual reproduction and its contribution to their evolution. Given that same sex mating arose independently in these two divergent human fungal pathogens suggests that other examples likely remain to be discovered in the fungal kingdom, and possibly also in other more divergent eukaryotes such as pathogenic parasites [103].
The ultimate questions of how unisexual and bisexual reproduction evolved and how they are regulated in C. neoformans remain to be answered. Previous studies on the genetic circuits controlling mating in S. cerevisiae may prove illustrative. The a/α diploid S. cerevisiae cell type, instructed by the cell identity a1/α2 complex, is competent to undergo meiosis and produce meiotic progeny. By contrast, a/a and α/α diploid yeast cells are normally incompetent to undergo meiosis. However, mutations in the meiosis repressor gene RME1 can bypass the normal requirement for a1/α2 and enable a/a rme1/rme1 and α/α rme1/rme1 mutants to undergo meiosis at a lower efficiency [104]–[107]. Thus, mutation of a single gene bypasses the normal requirement for contributions from the two mating type alleles in the genetic circuit that enables diploid S. cerevisiae cells to engage in sexual reproduction. Analogous genetic changes in the regulatory circuit that normally orchestrates opposite sex mating of C. neoformans may have enabled the evolution of modified circuits enabling same sex mating, and in so doing bypassed the normal requirement for either Sxi1α or Sxi2a.
The nature of signaling pathways and differentiation cascades as linear pathways vs. branched networks may influence the developmental plasticity of the evolution of morphogenesis and sexual reproduction. We hypothesize that a branched network governs sexual reproduction of C. neoformans and that same sex mating may employ some of the components in the circuits governing opposite sex mating. In essence, mutations and epistatic interactions arose that allowed the cell to bypass the normal requirement for Sxi1α/Sxi2a for sexual reproduction, and other parallel (MAPK/Mat2 or Ste12) or downstream morphogenesis determinants (Znf2), subsumed a central essential role indicating cell fate and the ability to undergo sexual reproduction (Figure 8).
Materials and Methods
Ethics statement
All the animal work was performed according to the guidelines of NIH and Duke University Institutional Animal Care and Use Committee (IACUC).
Strains and growth conditions
Strains used in this study and their sources are listed in Table S4. Cells were maintained on rich YPD (1% yeast extract, 2% BactoPeptone, and 2% dextrose) or minimum YNB medium (Yeast Nitrogen Base Medium, Difco, Detroit, MI) media. For marker screening, YPD+NAT and YPD+NEO media were employed for strains with dominant drug resistance markers while synthetic complete medium minus adenine (SC-ade), uracil (SC-ura), or lysine (SC-lys) were utilized for strains with auxotrophic markers. Mating and cell fusion assays were conducted on V8 solid medium (pH = 7.0 or pH = 5.0) [108] in the dark at 22°C. Filamentation agar [34] or synthetic low ammonium dextrose (SLAD) agar (YNB without amino acids plus 50 µM ammonium sulfate) were used for monokaryotic fruiting and confrontation assays.
Generation of insertional mutants and screening for mutants defective in filamentation
Insertional mutagenesis via Agrobacterium mediated transformation in XL280α was performed essentially as described previously [68]. Briefly, A. tumefaciens strain LBA4404 containing a pPZP-NATcc plasmid [70] was grown for 48 h at 25°C in Luria-Bertani medium with kanamycin in shaking cultures. Bacterial cells were washed twice with sterile water and suspended in induction medium with 100 µM acetosyringone (600 nm of 0.15) and incubated for another 6 hours. C. neoformans XL280 cells grown overnight in YPD were washed in induction medium and resuspended at 107 cells/ml. Equal aliquots (200 µl) of C. neoformans and A. tumefaciens were mixed, plated onto induction medium agar, and incubated at room temperature for 3 days. The cells were scraped from the plates and transferred to YPD medium with nourseothricin (NAT) and cefotaxime (each at 100 µg/ml). 3600 transformants were examined for filamentation on V8 juice solid medium (pH = 7.0) after incubation for 7 days at room temperature in the dark. Mutants that exhibited no hyphal growth were selected. To identify the insertion sites in selected mutants, genomic DNA was digested with a restriction enzyme, purified, and self-ligated. PCR amplicons from the ligation using primers AI076/AI077 were sequenced as described previously [68], [70]. Sequences of the flanking regions were used to BLAST the C. neoformans serotype D JEC21 genome database to determine the T-DNA insertion sites and subsequently the mutated genetic loci.
RNA purification and microarray analysis
For the initial search for transcription factors, total RNA was purified from strains XL280 and XL34 after incubation on V8 medium (pH 7.0) at room temperature for 24 hours using TRIzol Reagent according to the manufacturer's instructions (Invitrogen). Cy3 and Cy5 -labeled cDNA was generated by incorporating amino-allyl-dUTP during reverse transcription of 10 µg of total RNA as described previously [76] and competitively hybridized to a partial genome array generated previously in the Heitman laboratory [76]. After hybridization, arrays were scanned with a GenePix 4000B scanner (Axon Instruments, http://www.axon.com) and analyzed by using GenePix Pro version 4.0 and BRB array tools (developed by Richard Simon and Amy Peng Lam at the National Cancer Institute; http://linus.nci.nih.gov/BRB-ArrayTools.html) as described previously [38], [109].
For later transcriptional profiling of various mutants, total RNA was extracted from bilateral matings (α × a, α ste7Δ × a ste7Δ, α mat2Δ × a mat2Δ, and α znf2Δ × a znf2Δ) after incubation on V8 medium (pH = 7.0) at 22°C for the indicated time. They were either processed for Northern Blot analysis or for the whole genome microarray analysis as described above except that they were hybridized to a C. neoformans whole genome 70 mer oligo array with additional serotype-specific and mating-type–specific 70 mer oligos for genes in the MAT locus designed by Brian Griffith and James Fraser in the Heitman lab [38], [109]. Information about the array can be found at the following Web site: http://genome.wustl.edu/services/microarray/cryptococcus_neoformans). The array was analyzed similarly as described above.
Northern blots
RNA was separated on agarose gels blotted to nylon membrane. Redi-Prime II kit (Amersham) was used to generate probes. For MFα pheromone gene expression analysis, total RNA was extracted from cocultures of mating partners on V8 medium (pH = 7.0) at 0, 6 hr, 15 hr, and 24 hr after incubation in the dark at room temperature. The C. neoformans actin gene transcript served as a control. For the time course examination of MFα expression level, total RNA was used. For the examination of expression pattern of the CPK1, MAT2, SXI1α, and ZNF2 genes during bilateral matings, mRNA was used. mRNA purification was performed using the PolyATtract mRNA Isolation System III (Fisher) according to the manufacture's instruction.
Genomic DNA preparations
Strains were grown in 50 ml YPD medium at 30°C overnight with shaking. The cells were washed three times with distilled water and harvested by centrifugation at 4000×g for 8 minutes. The cell pellet was frozen immediately at −80°C, lyophilized overnight, and stored at −20°C until genomic DNA was prepared using the CTAB protocol as described previously [110].
Gene disruption and complementation
To disrupt the MAT2 or ZNF2 gene, an overlap PCR product was generated with the NAT or NEO marker amplified from plasmid pAI1 or pJAF1 [32], [68] and 5′ and 3′ flanking sequences of the MAT2 or the ZNF2 locus from strain JEC21α (967 bp and 859 bp, respectively). The PCR product was directly introduced into strains JEC21α, XL280α, JEC20a, or XL254a by biolistic transformation [111]. Mutants in which the gene had been replaced by homologous recombination were screened by PCR and confirmed by Southern blotting. The same approach with the NEO marker was used to produce deletion mutants in the serotype A H99 background with primers designed based on the H99 genome sequence. For complementation, an overlap PCR product with the NEO or NAT marker and the wild-type ZNF2 gene containing its native promoter and terminator from strain JEC21 or H99 was generated. The PCR product was directly introduced into znf2Δ mutants in various backgrounds. Isogenic MATa strains with the ZNF2 deletion were obtained by selecting resistant MATa progeny from a cross between the mutant α strains and the corresponding congenic pair strains (JEC20a or KN99a).
Mating and self-filamentation assays
For matings, mating partners (a and α) isolates were grown on YPD medium separately overnight at 30°C and then cocultured together on V8 medium (pH = 7.0 for serotype D strains, pH = 5.0 for serotype A strains) in the dark at 22°C. Each partner was also grown alone on the same V8 medium plate as a control. Matings were examined microscopically for formation of mating hyphae and chains of basidiospores. Random basidiospores were isolated using a micromanipulator. Their mating type was examined by mating with reference strains JEC20 (a) or JEC21 (α). For self-filamentation assays (only for serotype D strains), cells were patched on V8 medium alone and hyphae formation was examined microscopically.
Cell fusion assay
Strains were grown on YPD agar for 2 days, resuspended in sterile water, quantitated in a spectrophotometer, and diluted to 107 cells/ml. Equal numbers of mating partners were mixed and 10 microliters of the mixture were dropped on V8 juice agar medium (pH = 7.0). After 15 hours of incubation in the dark at room temperature, cells were removed, washed, and plated on media to select fusion products at room temperature. CFU were counted to measure the efficiency of cell fusion. The znf2Δ mutant strains XL874a (znf2::NATr lys1) and XL875α (znf2::NATr ade2) were used for fusion assay and the corresponding control pairs were XL878a (lys1) and XL877α (ade2). Fused prototrophic products were selected on minimum YNB medium. The colony morphology of the fusion product from wild type and the znf2Δ mutant was also observed microscopically.
The mat2Δ mutants used for fusion assay were XL926α (mat2::NATr) and XL961a (mat2::NEOr). The corresponding control pairs were JEC21α marked with NATr and JEC20a marked with NEOr. Fused products were selected on YPD+NAT+NEO medium. To assay cell fusion during unilateral matings, XL926α (mat2::NATr) was paired with JEC20a marked with NEOr, and XL961a (mat2::NEOr) was paired with JEC21α marked with NATr. Again, the fusion products were selected on YPD+NAT+NEO medium.
Confrontation assay
Strains (a and α) were streaked in close proximity but not touching each other on Filamentation agar or V8 medium and incubated in the dark at 22°C. Formation of conjugation tubes or monokaryotic hyphae was examined after 24 hours.
Ploidy determination by fluorescence flow cytometry
Cells were processed for flow cytometry as described previously [81]. Briefly, cells were harvested after overnight growth in YPD medium, washed once in PBS buffer, and fixed in 1 ml of 70% ethanol overnight at 4°C. Fixed cells were washed once with 1 ml of NS buffer (10 mM Tris–HCl (pH = 7.6), 250 mM sucrose, 1 mM EDTA (pH = 8.0), 1 mM MgCl2, 0.1 mM CaCl2, 0.1 mM ZnCl2) and then stained with propidium iodide (10 mg/ml) in 200 µl of NS buffer containing RNaseA (1 mg/ml) at 4°C for 4–16 h. Then 50 µl of stained cells was diluted into 2 ml of 50 mM Tris–HCl (pH = 8.0) and sonicated for 1 min. Flow cytometry was performed on 10,000 cells and analyzed on the FL1 channel with a Becton–Dickinson FACScan.
In vitro assay of virulence factors
Virulence traits were assayed as previously described [81]. Briefly, yeast cells were grown in YPD medium overnight and washed three times with water. Cell density was determined by optical density at 600 nm and cells were serially diluted (×10). To analyze growth at different temperatures, cells were spotted on YPD medium and incubated at the indicated temperatures for 48 hours. To examine melanin production, cells were spotted on melanin-inducing media containing L-DOPA (L-dihydroxyphenylalanine, 100 mg/L) [112] and incubated at 22°C in the dark for 6 days. Melanization was observed as the colony developed a brown color. To characterize capsule production, equal numbers of C. neoformans cells were transferred to Dulbecco's Modified Eagle Medium (DMEM) (Invitrogen, California) and grown for three days at 37°C. Cells were then suspended in India ink. The capsule excludes ink particles and was visualized as a white halo surrounding the yeast cell.
Murine inhalation model of cryptococcosis
Animals were infected essentially as previously described [113]. For strains in the serotype A H99 background, groups of 6 - to 8-week-old female A/J mice were anesthetized by intraperitoneal injection of Phenobarbital (∼0.035 mg/g) and they were infected intranasally with 5×104 Cryptococcus cells in 50 µl PBS. The inocula of yeast cells were confirmed by CFU after serial dilutions. After inoculation of fungal cells, animals were monitored twice daily, and those showing signs of severe morbidity (weight loss, extension of the cerebral portion of the cranium, abnormal gait, paralysis, seizures, convulsions, or coma) were sacrificed by CO2 inhalation. The survival rates of animals were plotted against time, and P values were calculated with the Mann-Whitney U test. For strains in various serotype D backgrounds, DBA mice were used and the inoculum was increased to 1×106 Cryptococcus cells per animal. Other procedures were the same as described above.
Supporting Information
Zdroje
1. BanuettF
1998 Signalling in the yeasts: an informational cascade with links to the filamentous fungi. Microbiol Mol Biol Rev 62 249 274
2. DohlmanHG
SlessarevaJE
2006 Pheromone signaling pathways in yeast. Sci STKE 2006 cm6
3. SchwartzMA
MadhaniHD
2004 Principles of MAP kinase signaling specificity in Saccharomyces cerevisiae. Annu Rev Genet 38 725 748
4. DavidsonRC
NicholsCB
CoxGM
PerfectJR
HeitmanJ
2003 A MAP kinase cascade composed of cell type specific and non-specific elements controls mating and differentiation of the fungal pathogen Cryptococcus neoformans. Mol Microbiol 49 469 485
5. BardwellL
CookJG
InouyeCJ
ThornerJ
1994 Signal propagation and regulation in the mating pheromone response pathway of the yeast Saccharomyces cerevisiae. Dev Biol 166 363 379
6. LinX
2009 Cryptococcus neoformans: morphogenesis, infection, and evolution. Infection, Genetics and Evolution 9 401 416
7. AndrianopoulosA
2002 Control of morphogenesis in the human fungal pathogen Penicillium marneffei. Int J Med Microbiol 292 331 347
8. Kwon-ChungKJ
BennettJE
1992 Medical mycology Philadelphia Lea & Febiger
9. GreerDL
1978 Basidiomycetes as agents of human infections: a review. Mycopathologia 65 133 139
10. BraunBR
JohnsonAD
1997 Control of filament formation in Candida albicans by the transcriptional repressor TUP1. Science 277 105 109
11. CsankC
SchroppelK
LebererE
HarcusD
MohamedO
1998 Roles of the Candida albicans mitogen-activated protein kinase homolog, Cek1p, in hyphal development and systemic candidiasis. Infect Immun 66 2713 2721
12. LoHJ
KohlerJR
DiDomenicoB
LoebenbergD
CacciapuotiA
1997 Nonfilamentous C. albicans mutants are avirulent. Cell 90 939 949
13. StoldtVR
SonnebornA
LeukerCE
ErnstJF
1997 Efg1p, an essential regulator of morphogenesis of the human pathogen Candida albicans, is a member of a conserved class of bHLH proteins regulating morphogenetic processes in fungi. EMBO J 16 1982 1991
14. MarescaB
KobayashiGS
2000 Dimorphism in Histoplasma capsulatum and Blastomyces dermatitidis. Contrib Microbiol 5 201 216
15. KleinBS
TebbetsB
2007 Dimorphism and virulence in fungi. Curr Opin Microbiol 10 314 319
16. LimaRF
Santos BritoMM
SchafferGM
LimaOC
Borba CdeM
2004 Evaluation of the in vitro and in vivo dimorphism of Sporothrix schenckii, Blastomyces dermatitidis, and Paracoccidioides brasiliensis isolates after preservation in mineral oil. Can J Microbiol 50 445 449
17. DhillonNK
SharmaS
KhullerGK
2003 Signaling through protein kinases and transcriptional regulators in Candida albicans. Crit Rev Microbiol 29 259 275
18. LiuH
2002 Co-regulation of pathogenesis with dimorphism and phenotypic switching in Candida albicans, a commensal and a pathogen. Int J Med Microbiol 292 299 311
19. LopezCE
2006 Dimorphism and pathogenesis of Histoplasma capsulatum. Rev Argent Microbiol 38 235 242
20. CasadevallA
PerfectJR
1998 Cryptococcus neoformans Washington, D.C. ASM Press
21. MitchellTG
PerfectJR
1995 Cryptococcosis in the era of AIDS–100 years after the discovery of Cryptococcus neoformans. Clin Microbiol Rev 8 515 548
22. Pukkila-WorleyR
MylonakisE
2008 Epidemiology and management of cryptococcal meningitis: developments and challenges. Expert Opin Pharmacother 9 1 10
23. ChenJ
VarmaA
DiazMR
LitvintsevaAP
WollenbergKK
2008 Cryptococcus neoformans strains and infection in apparently immunocompetent patients, China. Emerg Infect Dis 14 755 762
24. ParkBJ
Refocusing on a re-emergent disease: The current global burden of cryptococcal meningitis among persons living with HIV/AIDS; 2008; Washington, DC. Abstract M-1848
25. Kwon-ChungKJ
1975 A new genus, Filobasidiella, the perfect state of Cryptococcus neoformans. Mycologia 67 1197 1200
26. Kwon-ChungKJ
1976 A new species of Filobasidiella, the sexual state of Cryptococcus neoformans B and C serotypes. Mycologia 68 943 946
27. McClellandCM
ChangYC
VarmaA
Kwon-ChungKJ
2004 Uniqueness of the mating system in Cryptococcus neoformans. Trends Microbiol 12 208 212
28. AlspaughJA
DavidsonRC
HeitmanJ
2000 Morphogenesis of Cryptococcus neoformans. Contrib Microbiol 5 217 238
29. IwasaM
TanabeS
KamadaT
1998 The two nuclei in the dikaryon of the homobasidiomycete Coprinus cinereus change position after each conjugate division. Fungal Genet Biol 23 110 116
30. LinX
HullCM
HeitmanJ
2005 Sexual reproduction between partners of the same mating type in Cryptococcus neoformans. Nature 434 1017 1021
31. NielsenK
CoxGM
WangP
ToffalettiDL
PerfectJR
2003 Sexual cycle of Cryptococcus neoformans var. grubii and virulence of congenic a and a isolates. Infect Immun 71 4831 4841
32. FraserJA
SubaranRL
NicholsCB
HeitmanJ
2003 Recapitulation of the sexual cycle of the primary fungal pathogen Cryptococcus neoformans var. gattii: implications for an outbreak on Vancouver Island, Canada. Eukaryot Cell 2 1036 1045
33. ErkeKH
1976 Light microscopy of basidia, basidiospores, and nuclei in spores and hyphae of Filobasidiella neoformans (Cryptococcus neoformans). J Bacteriol 128 445 455
34. WickesBL
MayorgaME
EdmanU
EdmanJC
1996 Dimorphism and haploid fruiting in Cryptococcus neoformans: association with the alpha-mating type. Proc Natl Acad Sci USA 93 7327 7331
35. LinX
HuangJC
MitchellTG
HeitmanJ
2006 Virulence attributes and hyphal growth of C. neoformans are quantitative traits and the MATa allele enhances filamentation. PLoS Genet 2 e187 doi:10.1371/journal.pgen.0020187
36. BuiT
LinX
MalikR
HeitmanJ
CarterD
2008 Isolates of Cryptococcus neoformans from infected animals reveal genetic exchange in unisexual, a mating type populations. Eukaryot Cell 7 1771 1780
37. ToddRL
HerrmannWW
1936 The life cycle of the organism causing yeast meningitis. J Bacteriol 32 89 103
38. LinX
LitvintsevaA
NielsenK
PatelS
KapadiaZ
2007 aADa hybrids of Cryptococcus neoformans: Evidence of same sex mating in nature and hybrid fitness. PLoS Genet 3 e186 doi:10.1371/journal.pgen.0030186
39. HullCM
BoilyMJ
HeitmanJ
2005 Sex-specific homeodomain proteins Sxi1a and Sxi2a coordinately regulate sexual development in Cryptococcus neoformans. Eukaryot Cell 4 526 535
40. HullCM
DavidsonRC
HeitmanJ
2002 Cell identity and sexual development in Cryptococcus neoformans are controlled by the mating-type-specific homeodomain protein Sxi1a. Genes Dev 16 3046 3060
41. ButlerG
RasmussenMD
LinMF
SantosMA
SakthikumarS
2009 Evolution of pathogenicity and sexual reproduction in eight Candida genomes. Nature 459 657 662
42. AlbyK
SchaeferD
BennettRJ
2009 Homothallic and heterothallic mating in the opportunistic pathogen Candida albicans. Nature 460 890 893
43. ReedyJL
FloydAM
HeitmanJ
2009 Mechanistic plasticity of sexual reproduction and meiosis in the Candida pathogenic species complex. Curr Biol 19 891 899
44. HuangG
SrikanthaT
SahniN
YiS
SollDR
2009 CO2 regulates white-to-opaque switching in Candida albicans. Current Biology 19 1 5
45. HerskowitzI
1995 MAP kinase pathways in yeast: for mating and more. Cell 80 187 197
46. ClarkeDL
WoodleeGL
McClellandCM
SeymourTS
WickesBL
2001 The Cryptococcus neoformans STE11a gene is similar to other fungal mitogen-activated protein kinase kinase kinase (MAPKKK) genes but is mating type specific. Mol Microbiol 40 200 213
47. WangP
NicholsCB
LengelerKB
CardenasME
CoxGM
2002 Mating-type-specific and nonspecific PAK kinases play shared and divergent roles in Cryptococcus neoformans. Eukaryot Cell 1 257 272
48. NicholsCB
FraserJA
HeitmanJ
2004 PAK kinases Ste20 and Pak1 govern cell polarity at different stages of mating in Cryptococcus neoformans. Mol Biol Cell 15 4476 4489
49. GavriasV
AndrianopoulosA
GimenoCJ
TimberlakeWE
1996 Saccharomyces cerevisiae TEC1 is required for pseudohyphal growth. Mol Microbiol 19 1255 1263
50. MadhaniHD
FinkGR
1997 Combinatorial control required for the specificity of yeast MAPK signaling. Science 275 1314 1317
51. WickesBL
EdmanU
EdmanJC
1997 The Cryptococcus neoformans STE12a gene: a putative Saccharomyces cerevisiae STE12 homologue that is mating type specific. Mol Microbiol 26 951 960
52. YueC
CavalloLM
AlspaughJA
WangP
CoxGM
1999 The STE12a homolog is required for haploid filamentation but largely dispensable for mating and virulence in Cryptococcus neoformans. Genetics 153 1601 1615
53. ChangYC
WickesBL
MillerGF
PenoyerLA
Kwon-ChungKJ
2000 Cryptococcus neoformans STE12a regulates virulence but is not essential for mating. J Exp Med 191 871 882
54. ChangYC
PenoyerLA
Kwon-ChungKJ
2001 The second STE12 homologue of Cryptococcus neoformans is MATa-specific and plays an important role in virulence. Proc Natl Acad Sci U S A 98 3258 3263
55. BornemanAR
HynesMJ
AndrianopoulosA
2001 An STE12 homolog from the asexual, dimorphic fungus Penicillium marneffei complements the defect in sexual development of an Aspergillus nidulans steA mutant. Genetics 157 1003 1014
56. SugimotoA
IinoY
MaedaT
WatanabeY
YamamotoM
1991 Schizosaccharomyces pombe ste11+ encodes a transcription factor with an HMG motif that is a critical regulator of sexual development. Genes Dev 5 1990 1999
57. HartmannHA
KahmannR
BolkerM
1996 The pheromone response factor coordinates filamentous growth and pathogenicity in Ustilago maydis. EMBO J 15 1632 1641
58. HartmannHA
KrugerJ
LottspeichF
KahmannR
1999 Environmental signals controlling sexual development of the corn smut fungus Ustilago maydis through the transcriptional regulator Prf1. Plant Cell 11 1293 1306
59. HullCM
HeitmanJ
2002 Genetics of Cryptococcus neoformans. Annu Rev Genet 36 557 615
60. TscharkeRL
LazeraM
ChangYC
WickesBL
Kwon-ChungKJ
2003 Haploid fruiting in Cryptococcus neoformans is not mating type alpha-specific. Fungal Genet Biol 39 230 237
61. LengelerKB
FoxDS
FraserJA
AllenA
ForresterK
2002 Mating-type locus of Cryptococcus neoformans: a step in the evolution of sex chromosomes. Eukaryot Cell 1 704 718
62. KarosM
ChangYC
McClellandCM
ClarkeDL
FuJ
2000 Mapping of the Cryptococcus neoformans MATa locus: presence of mating type-specific mitogen-activated protein kinase cascade homologs. J Bacteriol 182 6222 6227
63. MitchellAP
1998 Dimorphism and virulence in Candida albicans. Curr Opin Microbiol 1 687 692
64. Sanchez-MartinezC
Perez-MartinJ
2001 Dimorphism in fungal pathogens: Candida albicans and Ustilago maydis–similar inputs, different outputs. Curr Opin Microbiol 4 214 221
65. LiuH
2001 Transcriptional control of dimorphism in Candida albicans. Curr Opin Microbiol 4 728 735
66. MullerP
AichingerC
FeldbruggeM
KahmannR
1999 The MAP kinase kpp2 regulates mating and pathogenic development in Ustilago maydis. Mol Microbiol 34 1007 1017
67. Mendoza-MendozaA
EskovaA
WeiseC
CzajkowskiR
KahmannR
2009 Hap2 regulates the pheromone response transcription factor prf1 in Ustilago maydis. Mol Microbiol Epub ahead of print
68. IdnurmA
ReedyJL
NussbaumJC
HeitmanJ
2004 Cryptococcus neoformans virulence gene discovery through insertional mutagenesis. Eukaryotic Cell 3 420 429
69. IdnurmA
HeitmanJ
2005 Light controls growth and development via a conserved pathway in the fungal kingdom. PLoS Biol 3 e95 doi:10.1371/journal.pbio.0030095
70. WaltonFJ
IdnurmA
HeitmanJ
2005 Novel gene functions required for melanization of the human pathogen Cryptococcus neoformans. Mol Microbiol 57 1381 1396
71. LoftusBJ
FungE
RoncagliaP
RowleyD
AmedeoP
2005 The genome of the basidiomycetous yeast and human pathogen Cryptococcus neoformans. Science 307 1321 1324
72. CasseltonLA
2002 Mate recognition in fungi. Heredity 88 142 147
73. CoppinE
DebuchyR
ArnaiseS
PicardM
1997 Mating types and sexual development in filamentous ascomycetes. Microbiol Mol Biol Rev 61 411 428
74. LinX
HeitmanJ
2007 Mechanisms of homothallism in fungi.
HeitmanJ
KronstadJ
TaylorJ
CasseltonLA
Sex in fungi: molecular determination and evolutionary implications: American Society of Microbiology
75. HeitmanJ
KronstadJ
TaylorJ
CasseltonLA
2007 Sex in fungi : molecular determination and evolutionary implications Washington, D.C. ASM Press xxv 542
76. KrausPR
BoilyMJ
GilesSS
StajichJE
AllenA
2004 Identification of Cryptococcus neoformans temperature-regulated genes with a genomic-DNA microarray. Eukaryot Cell 3 1249 1260
77. XueC
TadaY
DongX
HeitmanJ
2007 The human fungal pathogen Cryptococcus can complete its sexual cycle during a pathogenic association with plants. Cell Host & Microbe 1 263 273
78. ShenWC
DavidsonRC
CoxGM
HeitmanJ
2002 Pheromones stimulate mating and differentiation via paracrine and autocrine signaling in Cryptococcus neoformans. Eukaryot Cell 1 366 377
79. SiaRA
LengelerKB
HeitmanJ
2000 Diploid strains of the pathogenic basidiomycete Cryptococcus neoformans are thermally dimorphic. Fungal Genet Biol 29 153 163
80. BarchiesiF
CogliatiM
EspostoMC
SpreghiniE
SchimizziAM
2005 Comparative analysis of pathogenicity of Cryptococcus neoformans serotypes A, D and AD in murine cryptococcosis. J Infect 51 10 16
81. LinX
NielsenK
PatelS
HeitmanJ
2008 Impact of mating type, serotype, and ploidy on the virulence of Cryptococcus neoformans. Infect Immun 76 2923 2938
82. LinX
HeitmanJ
2006 The biology of the Cryptococcus neoformans species complex. Annu Rev Microbiol 60 69 105
83. IdnurmA
WaltonFJ
FloydA
HeitmanJ
2008 Identification of the sex genes in an early diverged fungus. Nature 451 193 196
84. LiuH
KohlerJ
FinkGR
1994 Suppression of hyphal formation in Candida albicans by mutation of a STE12 homolog. Science 266 1723 1726
85. KohlerJR
FinkGR
1996 Candida albicans strains heterozygous and homozygous for mutations in mitogen-activated protein kinase signaling components have defects in hyphal development. Proc Natl Acad Sci U S A 93 13223 13228
86. SinghP
GhoshS
DattaA
1997 A novel MAP-kinase kinase from Candida albicans. Gene 190 99 104
87. LebererE
HarcusD
BroadbentID
ClarkKL
DignardD
1996 Signal transduction through homologs of the Ste20p and Ste7p protein kinases can trigger hyphal formation in the pathogenic fungus Candida albicans. Proc Natl Acad Sci U S A 93 13217 13222
88. BanuettF
HerskowitzI
1996 Discrete developmental stages during teliospore formation in the corn smut fungus, Ustilago maydis. Development 122 2965 2976
89. WilliamsonJD
SilvermanJF
MallakCT
ChristieJD
1996 Atypical cytomorphologic appearance of Cryptococcus neoformans: a report of five cases. Acta Cytol 40 363 370
90. RipponJW
1988 Medical mycology : the pathogenic fungi and the pathogenic actinomycetes Philadelphia Saunders 797
91. WarrenNG
HazenKC
1999 Candida, Cryptococcus, and other yeasts of medical importance.
MurrayPR
BaronEJ
PfallerMA
Manual of clinical microbiology. 7th ed Washington, D.C. ASM Press 1184 1199
92. BemisDA
KrahwinkelDJ
BowmanLA
MondonP
Kwon-ChungKJ
2000 Temperature-sensitive strain of Cryptococcus neoformans producing hyphal elements in a feline nasal granuloma. J Clin Microbiol 38 926 928
93. FreedER
DumaRJ
ShadomyHJ
UtzJP
1971 Meningoencephalitis due to hyphae-forming Cryptococcus neoformans. Am J Clin Pathol 55 30 33
94. AnandiV
BabuPG
JohnTJ
1991 Infection due to Cryptococcus neoformans of unusual morphology in a patient with AIDS. Mycoses 34 377 379
95. ShadomyHJ
UtzJP
1966 Preliminary studies on a hypha-forming mutant of Cryptococcus neoformans. Mycologia 58 383 390
96. ShadomyHJ
LurieHI
1971 Histopathological observations in experimental cryptococcosis caused by a hypha-producing strain of Cryptococcus neoformans (Coward strain) in mice. Sabouraudia 9 6 9
97. ZimmerBL
HempelHO
GoodmanNL
1983 Pathogenicity of the hyphae of Filobasidiella neoformans. Mycopathologia 81 107 110
98. LurieHI
ShadomyHJ
1971 Morphological variations of a hypha-forming strain of Cryptococcus neoformans (Coward strain) in tissues of mice. Sabouraudia 9 10 14
99. FromtlingRA
BlackstockR
HallNK
BulmerGS
1979 Kinetics of lymphocyte transformation in mice immunized with viable avirulent forms of Cryptococcus neoformans. Infect Immun 24 449 453
100. FromtlingRA
BlackstockR
HallNK
BulmerGS
1979 Immunization of mice with an avirulent pseudohyphal form of Cryptococcus neoformans. Mycopathologia 68 179 181
101. FromtlingRA
BlackstockR
BulmerGS
1980 Immunization and passive transfer in immunity in murine cryptococcosis.
KuttinES
BaumGL
Human and animal mycology : proceedings of the VII Congress of ISHAM Amsterdam; Princeton; New York Excerpta Medica; distributors for the USA, Elsevier North-Holland 122 124
102. HeitmanJ
2009 Microbial genetics: Love the one you're with. Nature 460 807 808
103. PoxleitnerMK
CarpenterML
MancusoJJ
WangCJ
DawsonSC
2008 Evidence for karyogamy and exchange of genetic material in the binucleate intestinal parasite Giardia intestinalis. Science 319 1530 1533
104. CovitzPA
HerskowitzI
MitchellAP
1991 The yeast RME1 gene encodes a putative zinc finger protein that is directly repressed by a1-a2. Genes Dev 5 1982 1989
105. CovitzPA
MitchellAP
1993 Repression by the yeast meiotic inhibitor RME1. Genes Dev 7 1598 1608
106. ShimizuM
LiW
CovitzPA
HaraM
ShindoH
1998 Genomic footprinting of the yeast zinc finger protein Rme1p and its roles in repression of the meiotic activator IME1. Nucleic Acids Res 26 2329 2336
107. MitchellAP
HerskowitzI
1986 Activation of meiosis and sporulation by repression of the RME1 product in yeast. Nature 319 738 742
108. Kwon-ChungKJ
BennettJE
RhodesJC
1982 Taxonomic studies on Filobasidiella species and their anamorphs. Antonie Van Leeuwenhoek 48 25 38
109. LinX
PatelS
LitvintsevaAP
FloydA
MitchellTG
2009 Diploids in the Cryptococcus neoformans serotype A population homozygous for the α mating type originate via unisexual mating. PLoS Pathog 5 e1000283 doi:10.1371/journal.ppat.1000283
110. PitkinJW
PanaccioneDG
WaltonJD
1996 A putative cyclic peptide efflux pump encoded by the TOXA gene of the plant-pathogenic fungus Cochliobolus carbonum. Microbiology 142 1557 1565
111. ToffalettiDL
RudeTH
JohnstonSA
DurackDT
PerfectJR
1993 Gene transfer in Cryptococcus neoformans by use of biolistic delivery of DNA. J Bacteriol 175 1405 1411
112. ChaskesS
TyndallRL
1978 Pigment production by Cryptococcus neoformans and other Cryptococcus species from aminophenols and diaminobenzenes. J Clin Microbiol 7 146 152
113. CoxGM
MukherjeeJ
ColeGT
CasadevallA
PerfectJR
2000 Urease as a virulence factor in experimental cryptococcosis. Infect Immun 68 443 448
114. HuloN
BairochA
BulliardV
CeruttiL
CucheBA
2008 The 20 years of PROSITE. Nucleic Acids Res 36 D245 249
115. HortonP
ParkKJ
ObayashiT
FujitaN
HaradaH
2007 WoLF PSORT: protein localization predictor. Nucleic Acids Res 35 W585 587
116. Kwon-ChungKJ
KozelTR
EdmanJC
PolacheckI
EllisD
1992 Recent advances in biology and immunology of Cryptococcus neoformans. J Med Vet Mycol 30 Suppl 1 133 142
117. HeitmanJ
AllenB
AlspaughJA
Kwon-ChungKJ
1999 On the origins of congenic MATa and MATa strains of the pathogenic yeast Cryptococcus neoformans. Fungal Genet Biol 28 1 5
118. MooreTD
EdmanJC
1993 The alpha-mating type locus of Cryptococcus neoformans contains a peptide pheromone gene. Mol Cell Biol 13 1962 1970
119. PerfectJR
LangSD
DurackDT
1980 Chronic cryptococcal meningitis: a new experimental model in rabbits. Am J Pathol 101 177 194
120. BahnYS
HicksJK
GilesSS
CoxGM
HeitmanJ
2004 Adenylyl cyclase-associated protein Aca1 regulates virulence and differentiation of Cryptococcus neoformans via the cyclic AMP-protein kinase A cascade. Eukaryot Cell 3 1476 1491
Štítky
Genetika Reprodukční medicína
Článek Epistasis of Transcriptomes Reveals Synergism between Transcriptional Activators Hnf1α and Hnf4αČlánek SMA-10/LRIG Is a Conserved Transmembrane Protein that Enhances Bone Morphogenetic Protein SignalingČlánek B1 SOX Coordinate Cell Specification with Patterning and Morphogenesis in the Early Zebrafish EmbryoČlánek Bulk Segregant Analysis by High-Throughput Sequencing Reveals a Novel Xylose Utilization Gene fromČlánek Abundant Quantitative Trait Loci Exist for DNA Methylation and Gene Expression in Human Brain
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2010 Číslo 5- Kazuistika – Perspektivy využití precizované medicíny v rámci personalizované specifické terapie onkologických pacientů
- Nobelova cena za chemii pro genetické nůžky: Objev, který změní naši budoucnost?
- Technologie na bázi RNA v klinické praxi: od přebarvených petúnií k terapii vzácných a dosud jen obtížně léčitelných chorob u lidí
- „Nepředstavovali jsme si, že náš výzkum povede přímo ke vzniku nových léků, dokonce ještě za našeho života“
- Bezplatné služby pro diagnostiku ATTRv amyloidózy pro kardiology
-
Všechny články tohoto čísla
- Digital Quantification of Human Eye Color Highlights Genetic Association of Three New Loci
- Common Genetic Variants near the Brittle Cornea Syndrome Locus Influence the Blinding Disease Risk Factor Central Corneal Thickness
- Age- and Temperature-Dependent Somatic Mutation Accumulation in
- All About Mitochondrial Eve: An Interview with Rebecca Cann
- Aging and Chronic Sun Exposure Cause Distinct Epigenetic Changes in Human Skin
- Transposed Genes in Arabidopsis Are Often Associated with Flanking Repeats
- A Survey of Genomic Traces Reveals a Common Sequencing Error, RNA Editing, and DNA Editing
- Gene Transposition Causing Natural Variation for Growth in
- The Polyproline Site in Hinge 2 Influences the Functional Capacity of Truncated Dystrophins
- Epistasis of Transcriptomes Reveals Synergism between Transcriptional Activators Hnf1α and Hnf4α
- Integration of Light Signals by the Retinoblastoma Pathway in the Control of S Phase Entry in the Picophytoplanktonic Cell
- The Proprotein Convertase Encoded by () Is Required in Corpora Cardiaca Endocrine Cells Producing the Glucose Regulatory Hormone AKH
- Sgs1 and Exo1 Redundantly Inhibit Break-Induced Replication and Telomere Addition at Broken Chromosome Ends
- A MATE-Family Efflux Pump Rescues the 8-Oxoguanine-Repair-Deficient Mutator Phenotype and Protects Against HO Killing
- The Relationship among Gene Expression, the Evolution of Gene Dosage, and the Rate of Protein Evolution
- SMA-10/LRIG Is a Conserved Transmembrane Protein that Enhances Bone Morphogenetic Protein Signaling
- The Nuclear Receptor DHR3 Modulates dS6 Kinase–Dependent Growth in
- Involvement of Global Genome Repair, Transcription Coupled Repair, and Chromatin Remodeling in UV DNA Damage Response Changes during Development
- B1 SOX Coordinate Cell Specification with Patterning and Morphogenesis in the Early Zebrafish Embryo
- Linkage and Association Mapping of Flowering Time in Nature
- Bulk Segregant Analysis by High-Throughput Sequencing Reveals a Novel Xylose Utilization Gene from
- Abundant Quantitative Trait Loci Exist for DNA Methylation and Gene Expression in Human Brain
- Ablation of Whirlin Long Isoform Disrupts the USH2 Protein Complex and Causes Vision and Hearing Loss
- Characterization of Oxidative Guanine Damage and Repair in Mammalian Telomeres
- DNA Adenine Methylation Is Required to Replicate Both Chromosomes Once per Cell Cycle
- Genome-Wide Copy Number Variation in Epilepsy: Novel Susceptibility Loci in Idiopathic Generalized and Focal Epilepsies
- FACT Prevents the Accumulation of Free Histones Evicted from Transcribed Chromatin and a Subsequent Cell Cycle Delay in G1
- GC-Biased Evolution Near Human Accelerated Regions
- Liver and Adipose Expression Associated SNPs Are Enriched for Association to Type 2 Diabetes
- Myeloid Cell-Restricted Insulin Receptor Deficiency Protects Against Obesity-Induced Inflammation and Systemic Insulin Resistance
- The Mating Type Locus () and Sexual Reproduction of : Insights into the Evolution of Sex and Sex-Determining Chromosomal Regions in Fungi
- B-Cyclin/CDKs Regulate Mitotic Spindle Assembly by Phosphorylating Kinesins-5 in Budding Yeast
- Post-Replication Repair Suppresses Duplication-Mediated Genome Instability
- Genome Sequence of the Plant Growth Promoting Endophytic Bacterium sp. 638
- Transcription Factors Mat2 and Znf2 Operate Cellular Circuits Orchestrating Opposite- and Same-Sex Mating in
- The Use of Orthologous Sequences to Predict the Impact of Amino Acid Substitutions on Protein Function
- Mutation in Archain 1, a Subunit of COPI Coatomer Complex, Causes Diluted Coat Color and Purkinje Cell Degeneration
- Shelterin-Like Proteins and Yku Inhibit Nucleolytic Processing of Telomeres
- Affecting Function Causes a Dilated Heart in Adult
- Manipulation of Behavioral Decline in with the Rag GTPase
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Common Genetic Variants near the Brittle Cornea Syndrome Locus Influence the Blinding Disease Risk Factor Central Corneal Thickness
- All About Mitochondrial Eve: An Interview with Rebecca Cann
- The Relationship among Gene Expression, the Evolution of Gene Dosage, and the Rate of Protein Evolution
- SMA-10/LRIG Is a Conserved Transmembrane Protein that Enhances Bone Morphogenetic Protein Signaling
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Autoři: MUDr. Petr Výborný, CSc., FEBO
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



