-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
Susceptibility of to azithromycin and ceftriaxone in China: A retrospective study of national surveillance data from 2013 to 2016
In an observational study using data from the China Gonococcal Resistance Surveillance Programme, Yueping Yin estimate rates of reduced susceptibility to WHO-recommended antibiotics for Neisseria gonorrhoeae.
Published in the journal: . PLoS Med 15(2): e32767. doi:10.1371/journal.pmed.1002499
Category: Research Article
doi: https://doi.org/10.1371/journal.pmed.1002499Summary
In an observational study using data from the China Gonococcal Resistance Surveillance Programme, Yueping Yin estimate rates of reduced susceptibility to WHO-recommended antibiotics for Neisseria gonorrhoeae.
Introduction
Gonorrhea, caused by Neisseria gonorrhoeae (N. gonorrhoeae), remains one of the most common sexually transmitted diseases (STDs) worldwide. In 2012, the World Health Organization (WHO) estimated 27 million prevalent and 78 million incident cases of gonorrhea among people 15–49 years old. Of these estimated numbers, 42.5% and 45%, respectively, occurred in the Western Pacific Region. China is the most populous country in this region and contributes substantially to the number of people with gonorrhea [1]. China reports approximately 115,000 new cases of gonorrhea annually. The incidence of gonorrhea has increased in recent years, and the infection has become one of the most common notifiable infectious diseases in the country [2]. Gonorrhea causes urethritis, cervicitis, pharyngitis, and anal infection. If left untreated, it can result in complications such as pelvic inflammatory disease in women [3] and infertility in both men and women [4]. Moreover, it increases the risk for HIV acquisition and transmission [5]. Control of gonorrhea in many countries largely relies on detection of cases followed by therapy to cure the infection and eliminate transmission to others. However, successful treatment has been hampered by emerging resistance to each of the antibiotics recommended as first-line therapies [6]. Latest in this trend are resistance and treatment failures for extended-spectrum cephalosporins, including ceftriaxone and cefixime [7,8]. Monotherapy with ceftriaxone (250 mg intramuscularly in a single dose) for uncomplicated gonococcal infections is still the recommended regimen in the current guidelines in China. To halt the development and spread of resistance, dual therapy consisting of ceftriaxone and azithromycin has been recommended as the first-line therapy in international and national guidelines [9–11]. However, the first treatment failure with dual therapy was reported in 2016 in the United Kingdom [12]. To provide evidence for updating China’s national treatment recommendations for gonorrhea, we analyzed data on the susceptibility to azithromycin and ceftriaxone of N. gonorrhoeae isolates collected in the China Gonococcal Resistance Surveillance Programme (China-GRSP).
Methods
Ethical clearance
The study was approved by the Medical Ethics Committee at the Institute of Dermatology, the Chinese Academy of Medical Sciences & Peking Union Medical College, and the National Center for Sexually Transmitted Disease Control (NCSTD) at Nanjing (approval number 2014-LS-026) for the use of anonymized samples and data collected during routine outpatient services to patients attending local dermatology and/or STD clinics.
Study population
The current study was a retrospective study based on clinical isolates and data of patients with confirmed N. gonorrhoea in the China-GRSP. The China-GRSP was launched by the NCSTD as a collaboration project consisting of a few clinics in 1987. China joined the WHO Western Pacific Gonococcal Antimicrobial Susceptibility Programme (GASP) in 1992 and then expanded China-GRSP to more participating clinics in 9 provinces (Guangdong, Guangxi, Hainan, Zhejiang, Sichuan, Tianjin, Xinjiang, Shanghai, and Chongqing) in 2007. In 2013, China-GRSP started collecting data on azithromycin resistance. At each participating clinic, routine medical consultation was provided to patients. Cultures for N. gonorrhoeae were performed in patients with a positive risk assessment. Data from patients with confirmed N. gonorrhoeae from 1 January 2013 to 31 December 2016 were used in this study. The study focused on 7 provinces (Fig 1); Shanghai and Chongqing were not included in this study because these provinces did not collect data about azithromycin resistance. Selected demographic, behavioral, and clinical characteristics of the participants were extracted from outpatient medical records, i.e., from background information collected during the clinic visits.
Fig. 1. Geographic locations of the provinces where N. gonorrhoeae isolates were collected from patients. 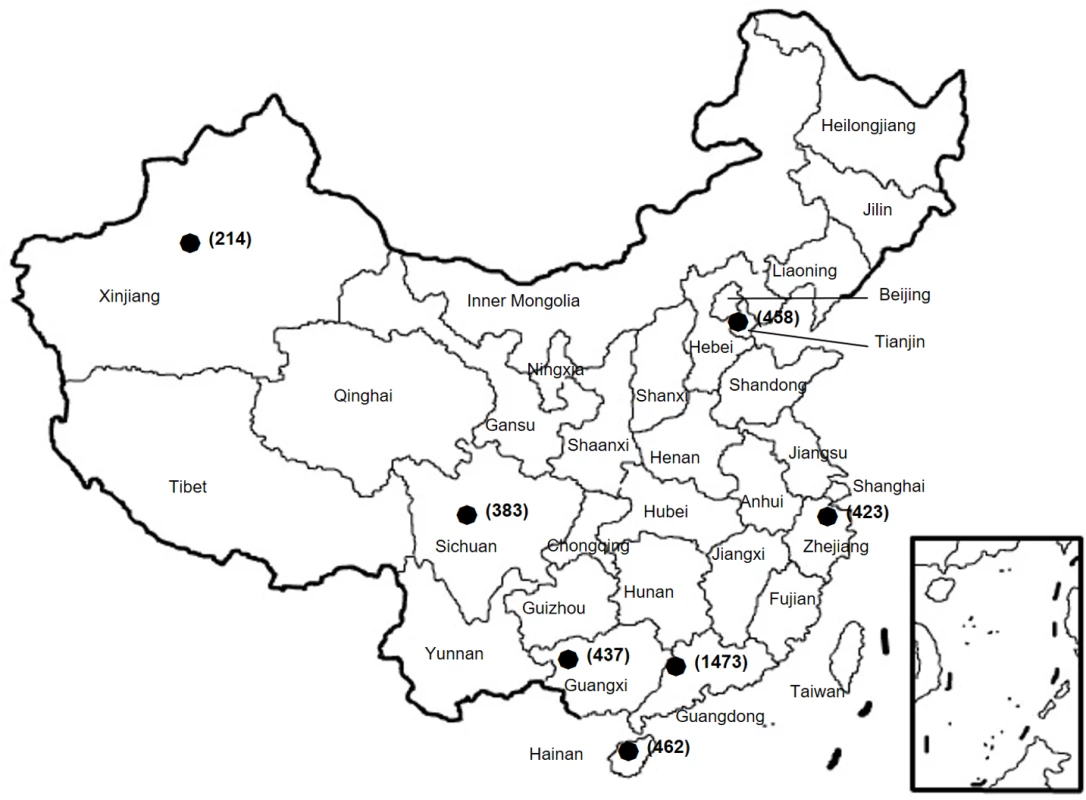
The number of isolates from each province is given in parentheses. Isolation of N. gonorrhoeae
As a patient could be infected at different anatomical sites (cervix/vagina, pharynx, rectum, and urethra), samples from more than 1 site were collected, if indicated. Rectal and pharyngeal samples were generally not obtained from heterosexual males. Samples were inoculated into selective Thayer-Martin (TM) medium and cultured in an incubator at 36°C in 5%–10% CO2 for 24 to 48 hours. With the cultured isolate samples, probable N. gonorrhoeae was identified based on colonial morphology (growth of typical appearing colonies), Gram stain (Gram-negative), and oxidase reaction (oxidase-positive intracellular diplococci in stained smears). Sugar fermentation tests were further performed if the identification was ambiguous. The gonococcal isolates were further sub-cultured from the selective TM medium to a nonselective medium, and the confirmed colonies were suspended in skim milk and frozen to −70°C until shipped to provincial central STD laboratories for antimicrobial susceptibility testing.
Antimicrobial susceptibility testing
Agar dilution antimicrobial susceptibility tests were conducted at the central STD laboratories of the participating provinces according to WHO recommendations [13]. Specifically, the isolates were cultured from frozen stocks onto selective TM medium and sub-cultured on GC medium base agar supplemented with hemoglobin powder and IsoVitaleX Enrichment (BD Diagnostics, Oxford, England) at 36°C with 5% to 10% CO2 for 18 to 20 hours at the central STD laboratories. The colonies were scraped, and suspensions of 107 organisms per milliliter were prepared. Using a multipoint inoculator (104 per point), the suspension was applied to antibiotic-containing medium (GC agar base supplemented with 10% defibrinated sheep blood; Nanjing Bianzhen Biotechnology, Nanjing, China) on a 9-cm diameter plate [14]. The plate was cultured at 36°C with 5% to 10% CO2 for 18 to 24 hours, and the growth of gonococci in different concentrations of antibiotic (0.03, 0.06, 0.125, 0.25, 0.5, 1.0, 2.0, 4.0, and 8.0 mg/l for azithromycin and 0.008, 0.015, 0.03, 0.06, 0.125, 0.25, 0.5, and 1.0 mg/l for ceftriaxone) was observed and recorded. The minimum inhibitory concentration (MIC) was defined as the lowest concentration of an antibiotic that inhibited the growth of gonococci. MIC interpretive criteria were in accordance with the Clinical and Laboratory Standards Institute to define resistance or decreased susceptibility [13,15]. For azithromycin, we categorized MIC values as susceptible (MIC ≤ 0.5 mg/l) or resistant (MIC ≥ 1.0 mg/l). For ceftriaxone, we categorized MICs as susceptible (MIC ≤ 0.06 mg/l) or decreased susceptibility (MIC ≥ 0.125 mg/l). For quality assurance, all the central STD laboratories participated in the external quality assurance program of the WHO Western Pacific GASP through the National STD Reference Laboratory in Nanjing.
Statistical analyses
The development of the statistical analysis plan, including changes inspired by peer review, is described in S1 Text. Those patients whose isolates were successfully cultured and for which MICs for azithromycin and/or ceftriaxone were determined were included in the final analyses. In this study, we calculated the prevalence rates and their 95% confidence intervals (CIs) of N. gonorrhoeae resistance to azithromycin (RTA), decreased susceptibility to ceftriaxone (DSC), and both RTA and DSC (RTA/DSC). Age was described as median and 25%–75% interquartile range (IQR) because the age data followed a left-skewed distribution. MIC was described as geometric mean and 95% CI because MICs were multiple values determined by serial dilutions. For MICs determined as above or below a specific value, we used the midpoint value between this specific value and the value of the next higher or lower dilution for calculating the geometric means. Bivariate analyses were conducted by chi-squared test or chi-squared test for trend. Multinomial logistic regression analysis was conducted to explore the associations of variables with being infected with strains of N. gonorrhoeae with RTA, DSC, or RTA/DSC. Variables with a significance level of P < 0.10 in bivariate analyses were included in a multinomial logistic regression model. Adjusted odds ratios (AORs) and 95% CIs were estimated by adjusting for potential confounding factors that were included in the model. P ≤ 0.05 was set as the level of significance. Statistical analysis was done using SPSS for Windows (version 16.0; SPSS, Chicago, Illinois) and MedCalc for Windows (version 16.8; MedCalc Software, Mariakerke, Belgium).
The current study is reported as per Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines (S1 Checklist).
Results
Baseline characteristics of patients
A total of 3,849 N. gonorrhoeae isolates were collected from 3,849 patients (males, 91.1%; females, 8.9%). Data on repeat infections in individuals were not available due to anonymization of the dataset during the study years. Of the 3,503 males who provided information on sexual orientation (99.9% of the male total), 3,451 (98.5%) were heterosexual and 52 (1.5%) were homosexual or bisexual. The median age was 31 years (IQR 26–40 years) for males and 31 years (IQR 25–43 years) for females. The majority (92.8%) of the participants were of Han ethnicity, and 50.2% were from the provinces along the coast, where more cases of gonorrhea were reported than in other areas [16]. Previous infection with gonorrhea was reported in 15.0% of patients. Among the heterosexual patients, the majority of the isolates were from the urethra (99.4%) in males and the cervix or urethra (96.2%) in females. No patients were found to have a positive N. gonorrhoeae isolate at more than 1 anatomical site.
Azithromycin susceptibility
Out of 3,849 N. gonorrhoeae isolates, 3,827 (99.4%) and 3,849 (100.0%) had MIC results for azithromycin and ceftriaxone, respectively. The geometric mean azithromycin MIC was 0.32 mg/l (95% CI 0.31–0.34 mg/l), with a range of ≤0.05 to ≥8 mg/l. The prevalence of RTA (defined as MIC ≥ 1.0 mg/l) was 18.6% (710/3,827; 95% CI 17.4%–19.8%). The proportions of the isolates with different MICs of azithromycin by year are shown in Fig 2. Neither the mean MIC nor the percentage of resistant strains appeared to change during the 2013–2016 study period, remaining between 0.31 mg/l and 0.34 mg/l and between 17.1% and 20.8%, respectively. Multinomial regression analysis (Table 1) indicates that RTA was significantly associated with younger age (AOR 0.99; 95% CI 0.98–1.00; P = 0.043) and being female (AOR 1.50; 95% CI 1.13–2.00; P = 0.005, compared with males).
Fig. 2. Proportion of <i>N</i>. <i>gonorrhoeae</i> isolates with different minimum inhibitory concentrations (mg/l) for azithromycin, by year. 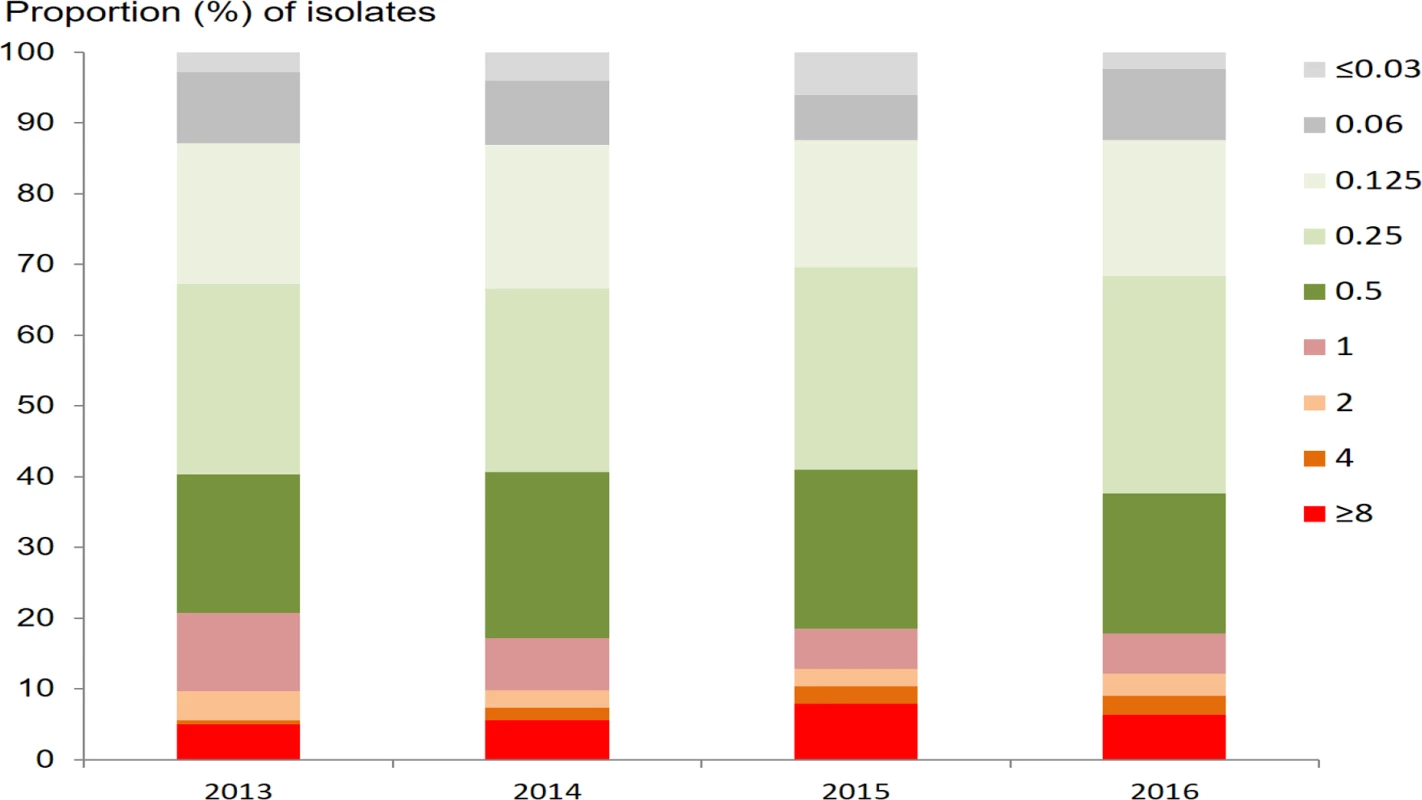
Tab. 1. Associations between resistance to azithromycin or decreased susceptibility to ceftriaxone and demographic and clinical characteristics (multinomial regression analysis*). 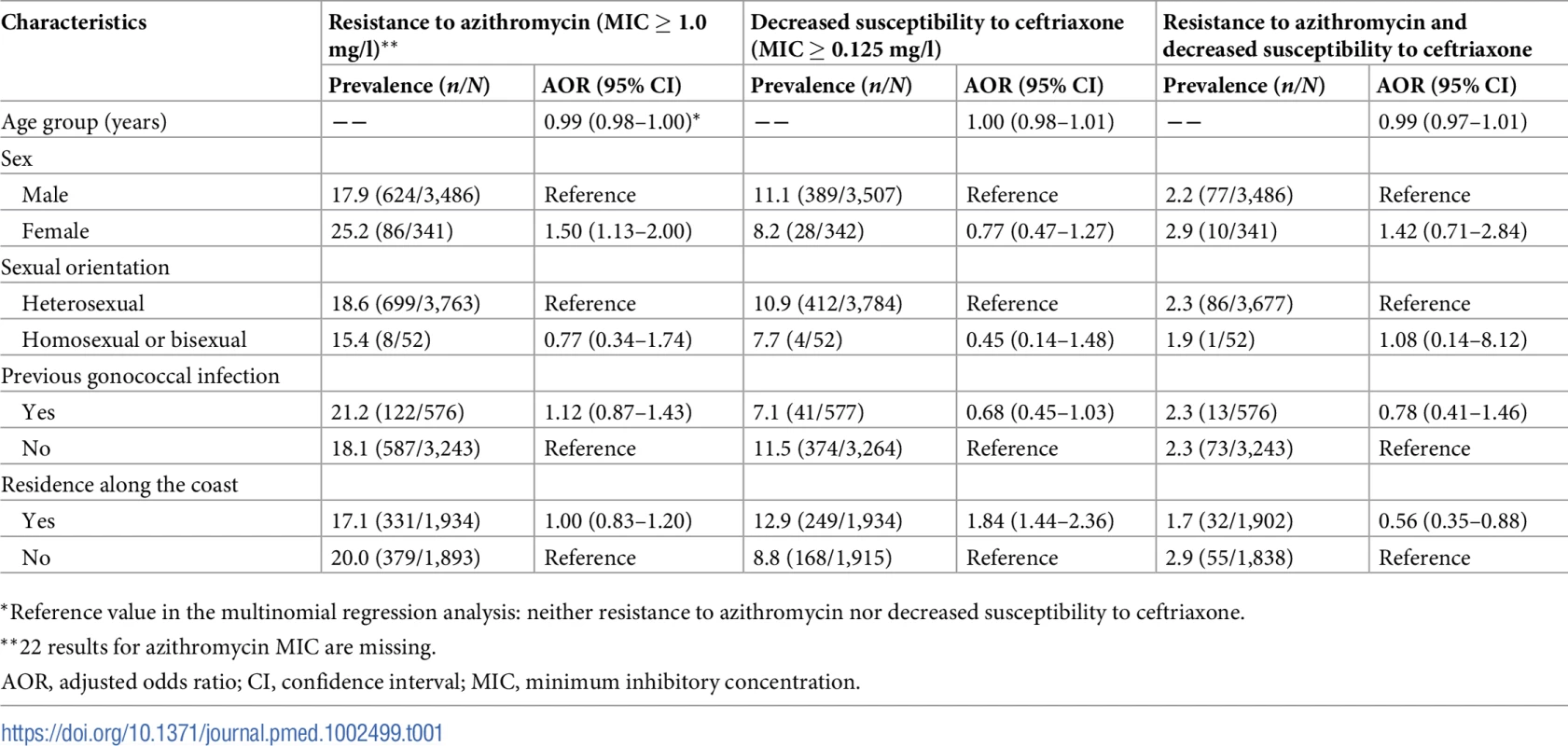
*Reference value in the multinomial regression analysis: neither resistance to azithromycin nor decreased susceptibility to ceftriaxone. Ceftriaxone susceptibility
The geometric mean ceftriaxone MIC was 0.025 mg/l (95% CI 0.024–0.026 mg/l), with a range of ≤0.008 to ≥1.0 mg/l. The prevalence of DSC (defined as MIC ≥ 0.125 mg/l) was 10.8% (417/3,849; 95% CI 9.9%–11.9%). The proportions of isolates with different MICs for ceftriaxone are shown by year in Fig 3. The percentage with DSC fluctuated between 9.7% and 12.2% over the period from 2013 to 2016. In the multinomial analysis (Table 1), DSC was significantly associated with residence in areas along the coast (AOR 1.84; 95% CI 1.44–2.36; P < 0.001, compared with residence in other areas).
Fig. 3. Proportion of <i>N</i>. <i>gonorrhoeae</i> isolates with different minimum inhibitory concentrations (mg/l) for ceftriaxone, by year. 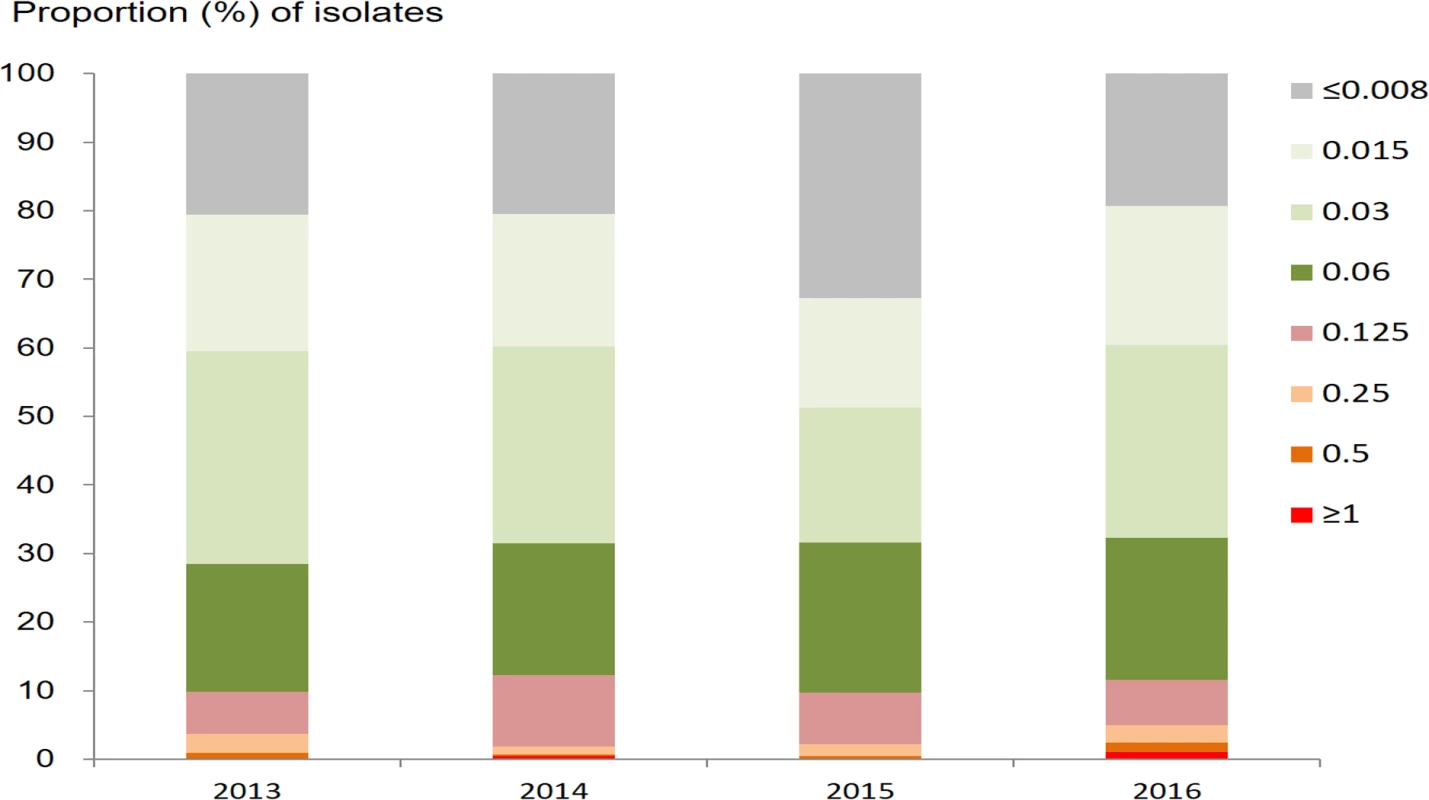
Resistance to azithromycin and decreased susceptibility to ceftriaxone
The proportion of isolates resistant to azithromycin in those with DSC (21.0%, 87/415; 95% CI 17.3%–25.1%) was not statistically different from the proportion of isolates resistant to azithromycin in those susceptible to ceftriaxone (18.3%, 623/3,412; 95% CI 17.0%–19.6%) (chi-squared = 1.79; P = 0.18; Table 2). The prevalence of isolates with RTA/DSC was 2.3% (87/3,827; 95% CI 1.9%–2.8%). The prevalence increased from 1.9% (18/928; 95% CI 1.2%–3.0%) in 2013 to 3.3% (32/981; 95% CI 2.3%–4.6%) in 2016 (chi-squared for trend = 4.78; P = 0.03) (Fig 4). Multinomial regression analysis (Table 1) indicates that residents in areas along the coast are less likely to be infected with strains with RTA/DSC (AOR 0.56; 95% CI 0.35–0.88; P = 0.013).
Fig. 4. Proportion of N. gonorrhoeae isolates with resistance to azithromycin (MIC ≥ 1.0 mg/l) and decreased susceptibility to ceftriaxone (MIC ≥ 0.125 mg/l) from 2013 to 2016. 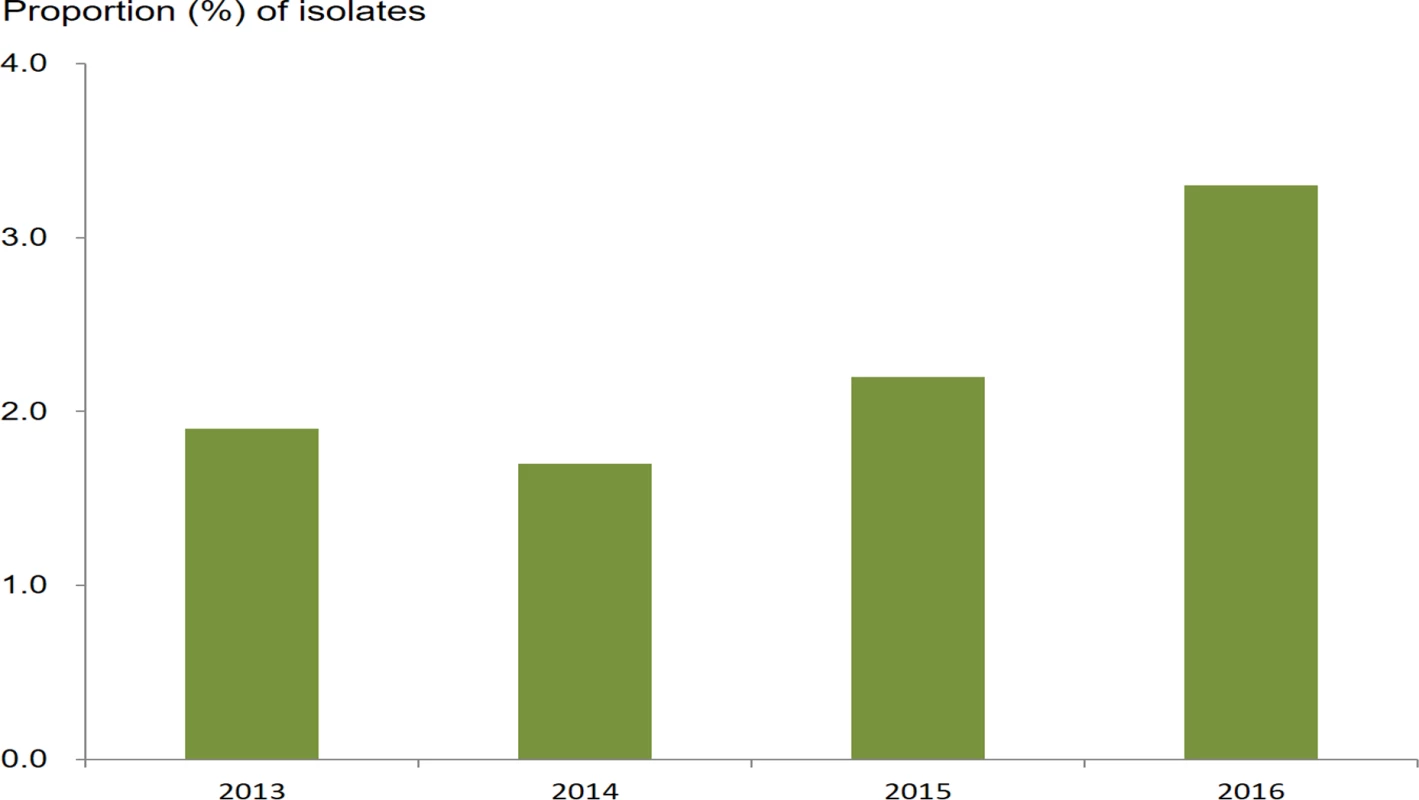
MIC, minimum inhibitory concentration. Tab. 2. Number and percentage of isolates susceptible or resistant to azithromycin by susceptibility breakpoints of ceftriaxone. 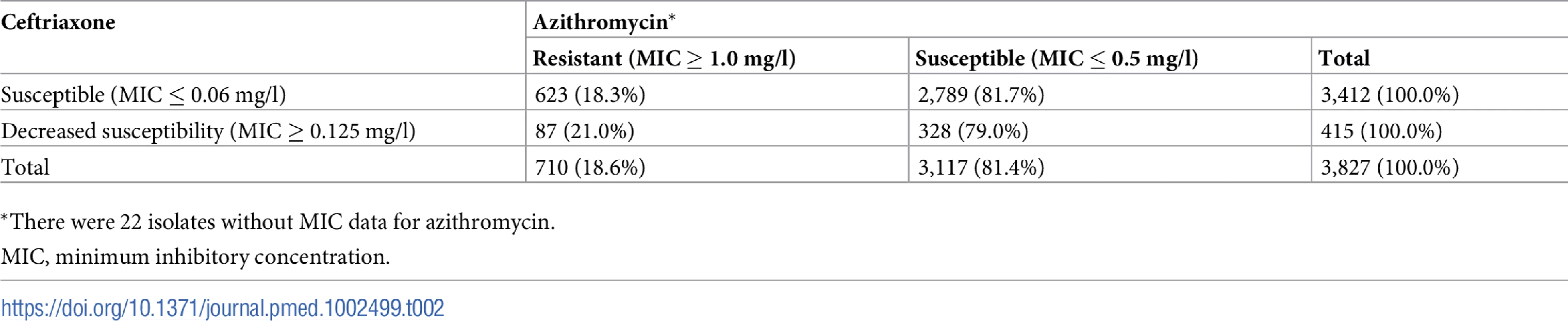
*There were 22 isolates without MIC data for azithromycin. Discussion
The current study represents, to our knowledge, the first national surveillance on trends of susceptibility of N. gonorrhoeae to azithromycin and ceftriaxone over time in China. In addition, associations between susceptibility data and demographic and clinical characteristics of study participants were examined. Our findings indicate that the rate of dual RTA and DSC increased between 2013 and 2016. The prevalences of N. gonorrhoeae RTA and DSC found in this study in China were higher than those reported in many countries, including in Europe and the Americas [17–27] (Table 3). A high prevalence of RTA or DSC has also been reported in Japan, although the sample size of the available study was relatively small (less than 700 isolates in total over 3 years) [27]. It is unclear whether circulating gonococcal strains with altered susceptibility to the key antibiotic agents in China acquired these mutations through domestic selective pressure or whether the strains have been imported. Antimicrobial resistant gonorrhea was documented first in Asia [28,29], then emerged in other areas in the world. Although azithromycin monotherapy is not recommended for gonorrhea treatment in China, this antibiotic is widely used for treatment of chlamydial infection according to the national STD treatment guidelines [30]. The prevalence of chlamydial infection is much higher than that of gonorrhea both among high-risk groups, such as female sex workers [31,32] and men who have sex with men [33], and in the general population [34] in China. Overuse of macrolides for chlamydia and other infections could facilitate emergence of resistance in N. gonorrhoeae [35]. Studies in China have reported that a high percentage (>50%) of outpatient visits result in prescription of antibiotics [36,37].
Tab. 3. Prevalence of resistance to azithromycin or decreased susceptibility to ceftriaxone among N. gonorrhoeae isolates collected from 2013 to 2016 in different countries. 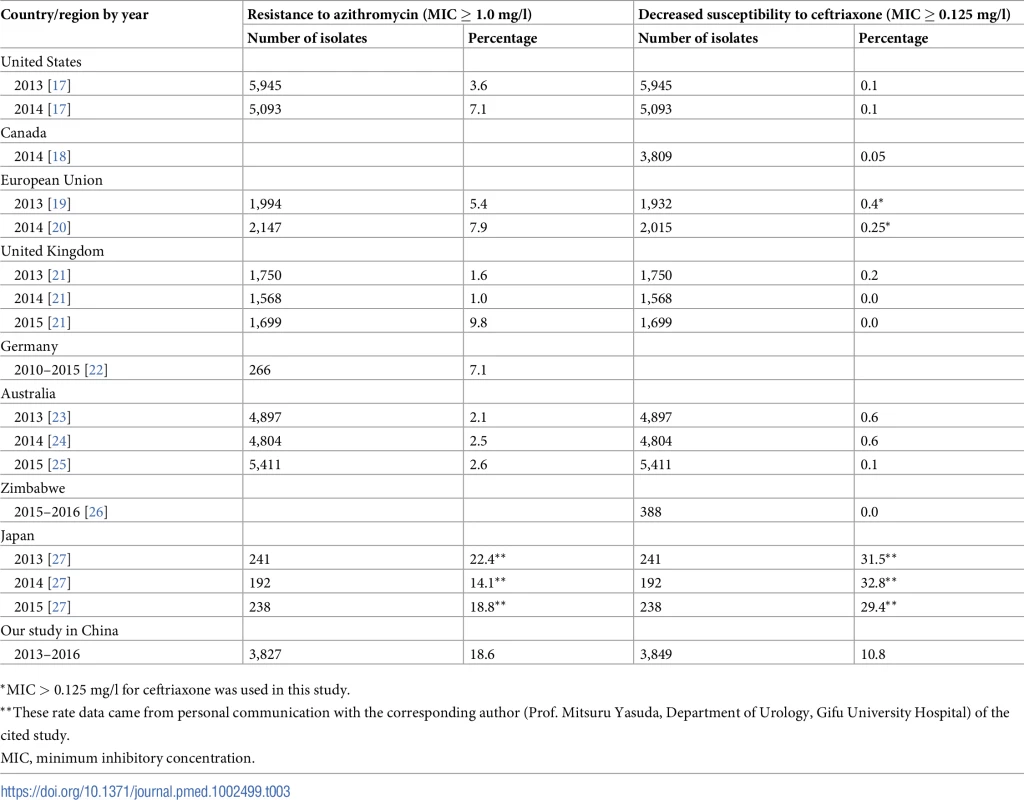
*MIC > 0.125 mg/l for ceftriaxone was used in this study. WHO and many countries have recommended dual antimicrobial therapy with ceftriaxone plus azithromycin for people with symptomatic and asymptomatic gonorrhea [9–11]. However, despite the high prevalence of DSC (more than 10%) in the current study, treatment failure of ceftriaxone has not been documented in China. It is important that clinicians be on high alert to recognize gonorrhea treatment failures so that they can be reported promptly to public health officials. The high and increasing prevalence of RTA/DSC found in the current study suggests the need for further consideration and validation of an appropriate regimen for treatment of gonorrhea in China.
Socioeconomic, behavioral, and clinical factors associated with antimicrobial resistance have been investigated by previous studies in China and other countries [38–40]. Interestingly, the factors independently associated with RTA and DSC were not the same in our study. Female patients had a higher risk of infection with an azithromycin-resistant strain than males. This finding was consistent with that reported in Amsterdam [41] but different from that reported in Shanghai, where male sex was a significant predictor of tetracycline resistance [38]. Our finding of an association of younger age with RTA is different from that reported in Shanghai, where probable resistance to ceftriaxone was most common among those in older age groups [38]. In addition, in our study, DSC was more likely to occur in areas along the coast, but RTA/DSC was more prevalent in other areas. These associations may be related to transmission dynamics linked to the sexual networks of different populations or in different areas. In addition, the resistance patterns are also dependent on the population seeking care in the sentinel clinics. Further studies on genomic epidemiology and sexual networks among people infected with resistant strains are needed. In contrast to the expected association between previous gonorrhea infection—linked to potential exposure to the antibiotics—and antimicrobial resistance, our study indicated that previous infection with gonorrhea was not associated with risk of RTA or DSC.
There are some potential limitations to this study. First, although the current study was a national surveillance survey with a large number of isolates collected from 7 provinces in China, the sample accounts for roughly only 1% of reported cases of gonorrhea during the study period in the country. Geographically, less than a quarter of the 31 provinces in the country participated in the study, and most of them were located in the economically developed areas along the coast. Moreover, the majority of male participants provided urethral specimens (99.4% of men), and the majority of female participants provided cervical or urethral specimens (96.2% of women). Our sample overrepresents men because men are usually symptomatic and easy to identify for specimen collection. Another concern is that most pharyngeal and anal gonorrhea is asymptomatic, and repeat infections or repeat visits of anonymized patients to clinics during the study period may result in the true number of individuals sampled being smaller than the number of “patients” described here, although this difference is likely to be relatively small. In addition, only a small proportion of the study participants were homosexual or bisexual, and therefore the majority of men did not report any anal or pharyngeal sexual intercourse [42]. All of these potential biases may limit the generalizability of the current study to all people infected with gonorrhea in China. Second, those isolates with MIC > 0.5 mg/l for azithromycin or MIC > 0.125 mg/l for ceftriaxone were not further categorized for susceptibility analysis to identify high-level RTA and/or resistance to ceftriaxone. Third, although molecular typing of N. gonorrhoeae was carried out in a few areas and reported previously [43], it was not performed in the present survey. Future studies should include molecular and genomic analyses to investigate gonorrhea transmission and to track the spread of antimicrobial resistance [44]. With regard to the treatment of gonorrhea in China, the national STD guidelines have not yet been updated to include dual therapy with ceftriaxone plus azithromycin for gonorrhea but recommend the use of azithromycin for treating potential infection with chlamydia among patients infected with gonorrhea. Further studies are needed to look into appropriate doses of ceftriaxone and azithromycin as well as use of other antibiotics in China.
In conclusion, the current study was a national study on the susceptibility of N. gonorrhoeae to azithromycin and ceftriaxone with implications for antibiotic choice for treatment of gonorrhea in China. An increasing trend of isolates with simultaneous RTA and DSC will likely impede the introduction of the currently WHO-recommended dual therapy in China. Therefore, evaluation of the efficacy of the dual therapy and the development of novel treatment strategies are urgently warranted in China. In addition, future resources should be dedicated to enhancing antimicrobial resistance surveillance by increasing both the number of laboratories performing susceptibility testing and the quality of susceptibility tests. The development of rapid diagnostics to promptly detect gonococcal infections and identify antimicrobial resistance is also urgently needed.
Supporting Information
Zdroje
1. Newman L, Rowley J, Vander Hoorn S, Wijesooriya NS, Unemo M, Low N, et al. Global estimates of the prevalence and incidence of four curable sexually transmitted infections in 2012 based on systematic review and global reporting. PLoS ONE. 2015;10 : 12:e0143304. doi: 10.1371/journal.pone.0143304 26646541
2. Yang S, Wu J, Ding C, Cui Y, Zhou Y, Li Y, et al. Epidemiological features of and changes in incidence of infectious diseases in China in the first decade after the SARS outbreak: an observational trend study. Lancet Infect Dis. 2017;17 : 716–25. doi: 10.1016/S1473-3099(17)30227-X 28412150
3. McCormack WM. Pelvic inflammatory disease. N Engl J Med. 1994;330 : 115–9. doi: 10.1056/NEJM199401133300207 8259168
4. Hook EW 3rd, Handsfield HH. Gonococcal infections in the adult. In: Holmes KK, Sparling PF, Mardh PA, Lemon SM, Stamm WE, Piot P, et al., editors. Sexually transmitted diseases. 3rd ed. New York: McGraw-Hill; 1999. pp. 451–66.
5. Cohen MS, Hoffman IF, Royce RA, Kazembe P, Dyer JR, Daly CC, et al. Reduction of concentration of HIV-1 in semen after treatment of urethritis: implications for prevention of sexual transmission of HIV-1. AIDSCAP Malawi Research Group. Lancet. 1997;349 : 1868–73. 9217758
6. Unemo M, Shafer WM. Antimicrobial resistance in Neisseria gonorrhoeae in the 21st century: past, evolution, and future. Clin Microbiol Rev. 2014;27 : 587–613. doi: 10.1128/CMR.00010-14 24982323
7. Shimuta K, Unemo M, Nakayama S, Morita-Ishihara T, Dorin M, Kawahata T, et al. Antimicrobial resistance and molecular typing of Neisseria gonorrhoeae isolates in Kyoto and Osaka, Japan, 2010 to 2012: intensified surveillance after identification of the first strain (H041) with high-level ceftriaxone resistance. Antimicrob Agents Chemother. 2013;57 : 5225–32. doi: 10.1128/AAC.01295-13 23939890
8. Ison CA, Hussey J, Sankar KN, Evans J, Alexander S. Gonorrhoea treatment failures to cefixime and azithromycin in England, 2010. Euro Surveill. 2011;16 : 19833. 21492528
9. Workowski KA, Bolan GA, Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm Rep. 2015;64(RR-03):1–137. 26042815
10. Bignell C, Unemo M, European STI Guidelines Editorial Board. 2012 European guideline on the diagnosis and treatment of gonorrhoea in adults. Int J STD AIDS. 2013;24 : 85–92. doi: 10.1177/0956462412472837 24400344
11. World Health Organization. WHO guidelines for the treatment of Neisseria gonorrhoeae. Geneva: World Health Organization; 2016.
12. Fifer H, Natarajan U, Jones L, Alexander S, Hughes G, Golparian D, et al. Failure of dual antimicrobial therapy in treatment of gonorrhea. N Engl J Med. 2016;374 : 2504–6. doi: 10.1056/NEJMc1512757 27332921
13. World Health Organization. Manual for the laboratory identification and antimicrobial susceptibility testing of bacterial pathogens of public health concern in the developing world. Geneva: World Health Organization; 2003.
14. Ye S, Su X, Wang Q, Yin Y, Dai X, Sun H. Surveillance of antibiotic resistance of Neisseria gonorrhoeae isolates in China, 1993–1998. Sex Transm Dis. 2002;29 : 242–5. 11912467
15. US Centers for Disease Control and Prevention. Gonococcal Isolate Surveillance Project (GISP) protocol. Atlanta: US Centers for Disease Control and Prevention; 2016.
16. Gong XD, Yue XL, Jiang N, Teng F, Meng PX, Li J, et al. Epidemiological characteristics and trends of gonorrhea in China from 2000 to 2014. Chin J Dermatol. 2015;48 : 301–6.
17. US Centers for Disease Control and Prevention Division of STD Prevention. Sexually transmitted disease surveillance 2014: Gonococcal Isolate Surveillance Project (GISP) supplement and profiles. Atlanta: US Centers for Disease Control and Prevention; 2016.
18. Martin I, Sawatzky P, Liu G, Allen V, Lefebvre B, Hoang L, et al. Decline in decreased cephalosporin susceptibility and increase in azithromycin resistance in Neisseria gonorrhoeae, Canada. Emerg Infect Dis. 2016;22 : 65–7. doi: 10.3201/eid2201.151247 26689114
19. Cole MJ, Spiteri G, Jacobsson S, Pitt R, Grigorjev V, Unemo M, et al. Is the tide turning again for cephalosporin resistance in Neisseria gonorrhoeae in Europe? Results from the 2013 European surveillance. BMC Infect Dis. 2015;15 : 321. doi: 10.1186/s12879-015-1013-x 26259560
20. European Centre for Disease Prevention and Control. Gonococcal antimicrobial susceptibility surveillance in Europe, 2014. Stockholm: European Centre for Disease Prevention and Control; 2016.
21. Public Health England. Surveillance of antimicrobial resistance in Neisseria gonorrhoeae—key findings from the Gonococcal Resistance to Antimicrobials Surveillance Programme (GRASP). London: Public Health England; 2016.
22. Regnath T, Mertes T, Ignatius R. Antimicrobial resistance of Neisseria gonorrhoeae isolates in south-west Germany, 2004 to 2015: increasing minimal inhibitory concentrations of tetracycline but no resistance to third-generation cephalosporins. Euro Surveill. 2016;21 : 30335.
23. Lahra MM, Australian Gonococcal Surveillance Programme. Australian Gonococcal Surveillance Programme annual report, 2013. Commun Dis Intell Q Rep. 2015;39:E137–45. 26063087
24. Lahra MM, Australian Gonococcal Surveillance Programme. Australian Gonococcal Surveillance Programme annual report, 2014. Commun Dis Intell Q Rep. 2015;39:E347–54. 26620348
25. Lahra MM, Enriquez RP, National Neisseria Network. Australian Gonococcal Surveillance Programme annual report, 2015. Commun Dis Intell Q Rep. 2017;41:E60–7.
26. Latif AS, Gwanzura L, Machiha A, Ndowa F, Tarupiwa A, Gudza-Mugabe M, et al. Antimicrobial susceptibility in Neisseria gonorrhoeae isolates from five sentinel surveillance sites in Zimbabwe, 2015–2016. Sex Transm Infect. 2017 May 5. doi: 10.1136/sextrans-2016-053090 28476914
27. Yasuda M, Hatazaki K, Ito S, Kitanohara M, Yoh M, Kojima M, et al. Antimicrobial susceptibility of Neisseria gonorrhoeae in Japan from 2000 to 2015. Sex Transm Dis. 2017;44 : 149–53. doi: 10.1097/OLQ.0000000000000556 28178112
28. Tapsall J. Neisseria gonorrhoeae and emerging resistance to extended spectrum cephalosporins. Curr Opin Infect Dis 2009;22 : 87–91. doi: 10.1097/QCO.0b013e328320a836 19532086
29. Centers for Disease Control and Prevention (CDC). Cephalosporin susceptibility among Neisseria gonorrhoeae isolates—United States, 2000–2010. MMWR Morb Mortal Wkly Rep. 2011;60 : 873–7. 21734634
30. Wang QQ, Liu QZ, Xu JH. Guidelines of sexually transmitted disease prevention and treatment. Shanghai: Shanghai Science and Technology Publishing House; 2014.
31. Su S, Chow EP, Muessig KE, Yuan L, Tucker JD, Zhang X, et al. Sustained high prevalence of viral hepatitis and sexually transmissible infections among female sex workers in China: a systematic review and meta-analysis. BMC Infect Dis. 2016;16 : 2. doi: 10.1186/s12879-015-1322-0 26732281
32. Chen XS, Yin YP, Liang GJ, Wang QQ, Jiang N, Liu Q, et al. The prevalences of Neisseria gonorrhoeae and Chlamydia trachomatis infections among female sex workers in China. BMC Public Health. 2013;13 : 121. doi: 10.1186/1471-2458-13-121 23390952
33. Fu GF, Jiang N, Hu HY, Mahapatra T, Yin YP, Mahapatra S, et al. The epidemic of HIV, syphilis, chlamydia and gonorrhea and the correlates of sexual transmitted infections among men who have sex with men in Jiangsu, China, 2009. PLoS ONE. 2015;10 : 3:e0118863. doi: 10.1371/journal.pone.0118863 25775451
34. Parish WL, Laumann EO, Cohen MS, Pan S, Zheng H, Hoffman I, et al. Population-based study of chlamydial infection in China: a hidden epidemic. JAMA. 2003;289 : 1265–73. 12633188
35. Unemo M1, Shafer WM. Antibiotic resistance in Neisseria gonorrhoeae: origin, evolution, and lessons learned for the future. Ann N Y Acad Sci. 2011;1230:E19–28. doi: 10.1111/j.1749-6632.2011.06215.x 22239555
36. Yin X, Song F, Gong Y, Tu X, Wang Y, Cao S, et al. A systematic review of antibiotic utilization in China. J Antimicrob Chemother. 2013;68 : 2445–52. doi: 10.1093/jac/dkt223 23800903
37. Wang J, Wang P, Wang X, Zheng Y, Xiao Y. Use and prescription of antibiotics in primary health care settings in China. JAMA Intern Med. 2014;174 : 1914–20. doi: 10.1001/jamainternmed.2014.5214 25285394
38. Trecker MA, Waldner C, Jolly A, Liao M, Gu W, Dillon JA. Behavioral and socioeconomic risk factors associated with probable resistance to ceftriaxone and resistance to penicillin and tetracycline in Neisseria gonorrhoeae in Shanghai. PLoS ONE. 2014;9 : 2:e89458. doi: 10.1371/journal.pone.0089458 24586792
39. Cole MJ, Spiteri G, Town K, Unemo M, Hoffmann S, Chisholm SA, et al. Risk factors for antimicrobial resistant Neisseria gonorrhoeae in Europe. Sex Transm Infect. 2014;41 : 723–9.
40. Goldstein E, Kirkcaldy RD, Reshef D, Berman S, Weinstock H, Sabeti P, et al. Factors related to increasing prevalence of resistance to ciprofloxacin and other antimicrobial drugs in Neisseria gonorrhoeae, United States. Emerg Infect Dis. 2012;18 : 1290–7. doi: 10.3201/eid1808.111202 22840274
41. Wind CM, Schim van der Loeff MF, van Dam AP, de Vries HJ, van der Helm JJ. Trends in antimicrobial susceptibility for azithromycin and ceftriaxone in Neisseria gonorrhoeae isolates in Amsterdam, the Netherlands, between 2012 and 2015. Euro Surveill. 2017;22 : 30431. doi: 10.2807/1560-7917.ES.2017.22.1.30431 28079519
42. Bissessor M, Whiley DM, Lee DM, Snow AF, Fairley CK, Peel J, et al. Detection of Neisseria gonorrhoeae isolates from tonsils and posterior oropharynx. J Clin Microbiol. 2015;53 : 3624–6. doi: 10.1128/JCM.01647-15 26292303
43. Chen SC, Yin YP, Dai XQ, Unemo M, Chen XS. First nationwide study regarding ceftriaxone resistance and molecular epidemiology of Neisseria gonorrhoeae in China. J Antimicrob Chemother. 2016;71 : 92–9. doi: 10.1093/jac/dkv321 26472770
44. De Silva D, Peters J, Cole K, Cole MJ, Cresswell F, Dean G, et al. Whole-genome sequencing to determine transmission of Neisseria gonorrhoeae: an observational study. Lancet Infect Dis. 2016;16 : 1295–303. doi: 10.1016/S1473-3099(16)30157-8 27427203
Štítky
Interní lékařství
Článek vyšel v časopisePLOS Medicine
Nejčtenější tento týden
2018 Číslo 2- Riziko rozvoje závažné infekce po císařském řezu lze snížit provedením antiseptického výplachu vaginy
- Topicky aplikovaný propolis může urychlovat hojení diabetických bércových vředů
- Spolehlivý systém skórování jizev k hodnocení fotografií popálenin
- Intermitentní hladovění v prevenci a léčbě (nejen) civilizačních chorob
- Stárnutí populace: problém, či výzva pro české zdravotnictví a medicínu?
-
Všechny články tohoto čísla
- Setbacks in Alzheimer research demand new strategies, not surrender
- Impact of person-centred care training and person-centred activities on quality of life, agitation, and antipsychotic use in people with dementia living in nursing homes: A cluster-randomised controlled trial
- Susceptibility of to azithromycin and ceftriaxone in China: A retrospective study of national surveillance data from 2013 to 2016
- Population impact of lung cancer screening in the United States: Projections from a microsimulation model
- Prevalence of sexually transmitted infections among young people in South Africa: A nested survey in a health and demographic surveillance site
- The potential impact of case-area targeted interventions in response to cholera outbreaks: A modeling study
- Perfluoroalkyl substances and changes in body weight and resting metabolic rate in response to weight-loss diets: A prospective study
- The 2014–2015 Ebola virus disease outbreak and primary healthcare delivery in Liberia: Time-series analyses for 2010–2016
- Prevalence of sexually transmitted infections and bacterial vaginosis among women in sub-Saharan Africa: An individual participant data meta-analysis of 18 HIV prevention studies
- Preventing cholera outbreaks through early targeted interventions
- Diet during pregnancy and infancy and risk of allergic or autoimmune disease: A systematic review and meta-analysis
- Risk and surrogate benefit for pediatric Phase I trials in oncology: A systematic review with meta-analysis
- PLOS Medicine
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- The potential impact of case-area targeted interventions in response to cholera outbreaks: A modeling study
- Prevalence of sexually transmitted infections among young people in South Africa: A nested survey in a health and demographic surveillance site
- Susceptibility of to azithromycin and ceftriaxone in China: A retrospective study of national surveillance data from 2013 to 2016
- Perfluoroalkyl substances and changes in body weight and resting metabolic rate in response to weight-loss diets: A prospective study
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



