-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
The Importance of Evaluating the Development of Oct Findings During Conservative Treatment of Vitreomacular Traction Complicated by Macular Hole Formation
Authors: J. Dítě; J. Štěpánková; M. Česká Burdová; D. Dotřelová
Authors place of work: Oční klinika dětí a dospělých 2. lékařské fakulty Univerzity Karlovy a Fakultní nemocnice v Motole, přednosta: prof. MUDr. Dagmar Dotřelová, CSc.
Published in the journal: Čes. a slov. Oftal., 74, 2018, No. 6, p. 240-244
Category: Kazuistika
doi: https://doi.org/10.31348/2018/6/4Summary
Macular hole (MH) was considered untreatable condition for a long time until Kelly and Wendel described the posibility of surgical treatment with pars plana vitrectomy (PPV) in the nineties of the twentieth century. Since then have both surgical instruments and operation techniques developed rapidly and PPV has become the mainstay of MH therapy. Only stage I according to Gass classification which equates vitreomacular traction (VMT) according to Vitreomacular Traction Study group classification is routinely treated conservatively with observation only. It is however necessary to assess each case individually as some small and favourable MHs may close spontaneously.
Here we present a case report of 59 years old woman with mild myopia and best corrected visual acuity (BCVA) 6/6 bilaterally. She presented on July 2017 with blurry vision in her right eye and was diagnosed with bilateral VMT according to optical coherence tomography. BCVA was 6/9 in her right eye and 6/6 in her left eye. During five weeks a full thickness MH developed in left eye that was accompanied by a decrease in BCVA to 6/12p. Considering favourable size and configuration of MH and the fact that the patient preferred conservative approach, we postponed the surgery, observed the MH and recommended bed rest. The MH closed spontaneously during five weeks with resulting BCVA 6/6. In her right eye the VMT released during four months with resulting BCVA 6/6 as well.
The authors of the study declare that the formation and theme of the professional report and its publication are not in conflict of interest or supported by any pharmaceutical company.
Keywords:
Retina – macular hole – vitreomacular traction – spontaneous closure
INTRODUCTION
Idiopathic Macular Hole (MH) is a pathology in which the progression of a retinal defect occurs in the region of the fovea, afflicting the entire thickness of the neuroretina, which does not originate as the consequence of a trauma or other pathological process. According to a study by McCannel et al. from 2009, in the Caucasian race the incidence of MH is within the range of 7.8 people and 8.7 eyes per 100 000 of the population per year [13]. Of the identified cases, 72% of MH were described in women, and 50% of the patients were within the age range of 65-74 years [13].
MH was first described by Knapp in 1869, the first case was recorded in a patient following a blunt trauma to the eye, and in the following decades the prevailing opinion was that the predominant majority of macular holes originated in this manner [12]. During the course of the 20th century it was discovered that the majority of MHs do not occur as a consequence of trauma or secondarily on the basis of another pathological process [2]. For these MHs the term idiopathic or later primary MH was coined. A significant advance in the study and description of MH was made by Gass, who in 1988 presented a classification dividing macular holes into 4 stages according to the finding on the ocular fundus, and at the same time defined the tractional forces of the contracting vitreous body acting directly in the fovea, as the key pathophysiological moment of their origin [6, 8]. His classification was later supplemented with regard to the finding of optical coherence tomography (OCT) in the individual stages (table 1).
Tab. 1. Gass classification of idiopathic macular hole (modified according to OCT findings) 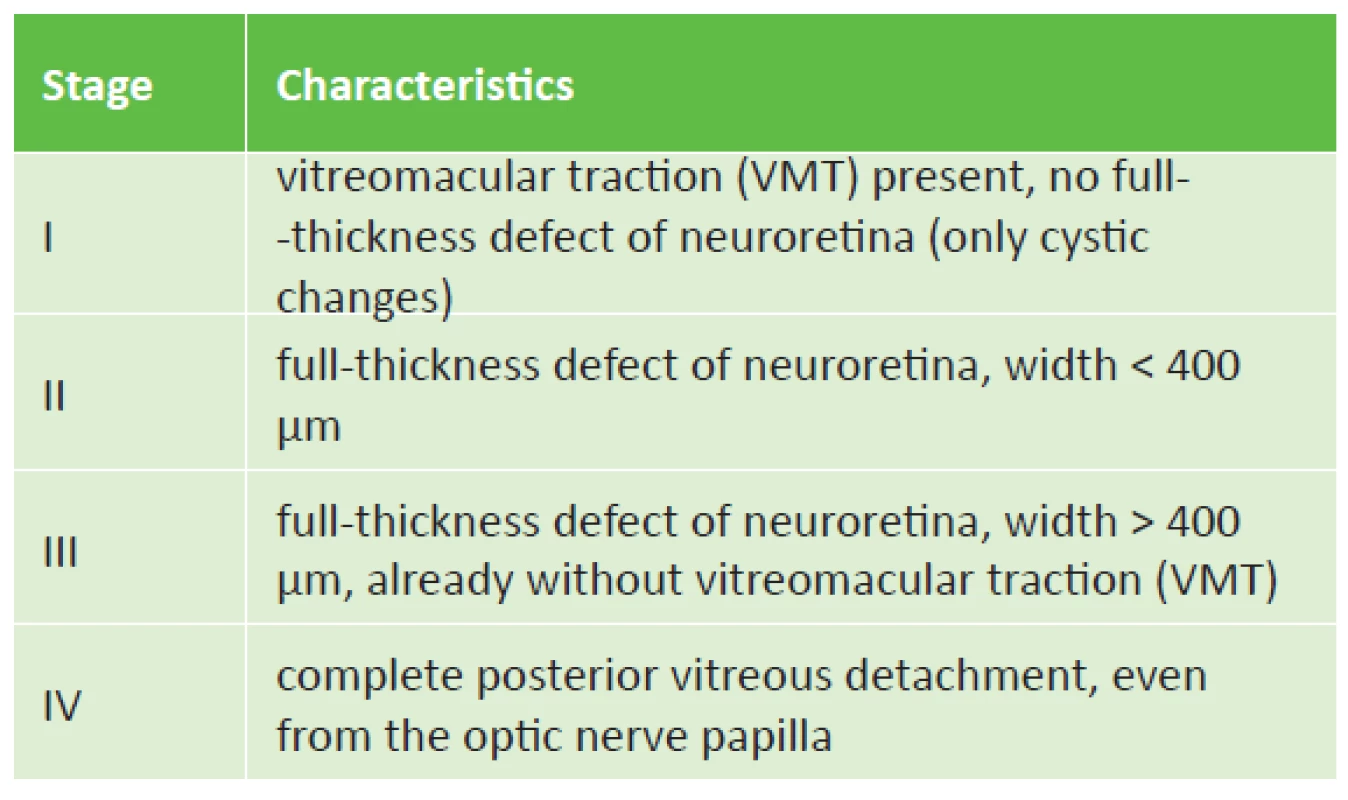
Following on from these discoveries, in 1991 Kelly and Wendel published a cohort of patients in whom they resolved a hitherto untreatable diagnosis surgically, with the aid of pars plana vitrectomy (PPV), with what was then a relatively high rate of success of reattachment of macular hole (58%), although from today's perspective this was accompanied by a relatively high incidence of perioperative and postoperative complications (15%) [10]. Nevertheless, this represented a revolutionary approach. In the following years, together with a rapid advance in instrument equipment, surgical techniques and the experience of operating surgeons, PPV became the gold standard in treatment of idiopathic MH.
With the dramatic development of OCT, pressure gradually began to build for the creation of a new classification system of MH and pathologies of the vitreomacular interface in general, based strictly on the anatomical finding according to OCT. This was achieved in 2013 thanks to the co-operation of a group of vitreoretinal specialists within the framework of The International Vitreomacular Traction Study (IVTS) (table 2) [5].
Tab. 2. IVTS classification of macular hole 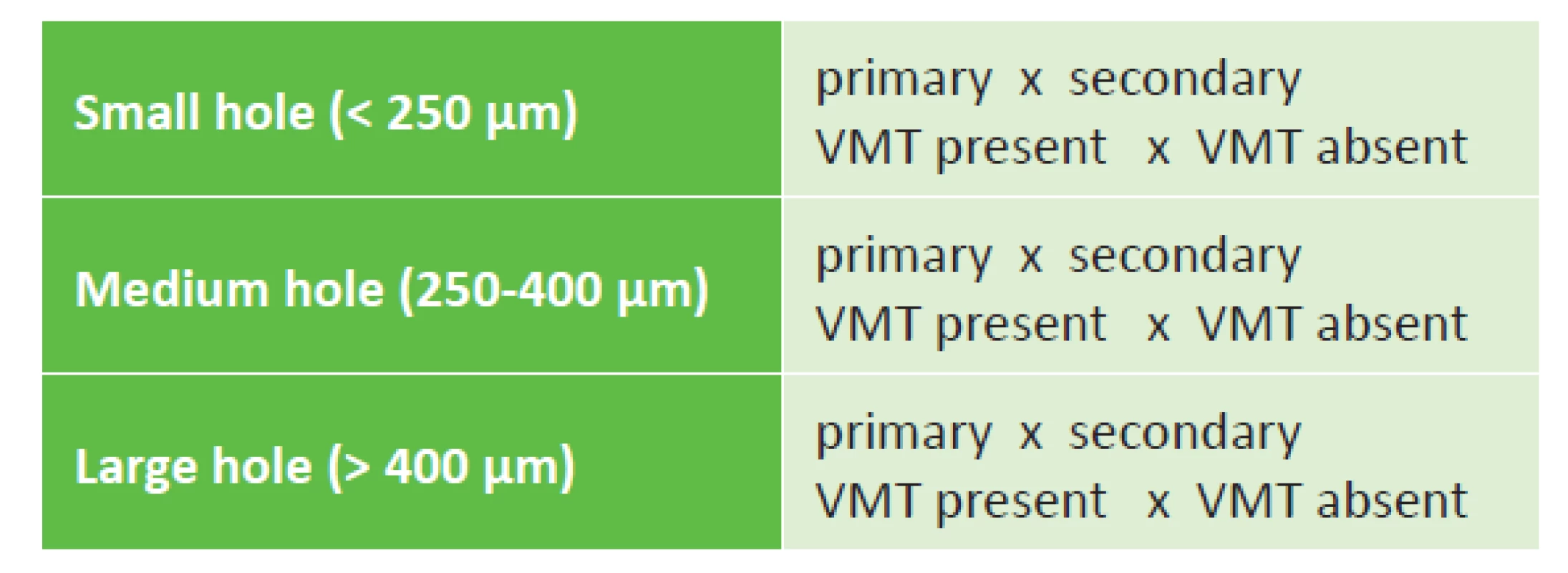
In the majority of cases (approximately 75% in the case of stage II, 55% in the case of stage III and 15% in the case of stage IV according to Gasse) MHs enlarge, and progression of the pathology occurs [7, 11]. For this reason, a conservative approach is recommended only in stage I according to Gasse (corresponding to VMT according to the IVTS classification) – thus in conditions in which the development of fully fledged MH has not yet occurred. In more advanced stages the gold standard of treatment is unequivocally vitrectomy. However, a number of cases are recorded in the literature in which MH closed spontaneously [3, 9, 17].
CASE REPORT
A 59-year-old woman was observed at the Department of Ophthalmology for Children and Adults at the 2nd Faculty of Medicine, Charles University and Motol University Hospital from 2014 for mild myopia -2.0 D and best corrected visual acuity (BCVA) of 6/6 bilaterally. She reported to our centre in July 2017 due to an episode of blurred vision in her right eye, which subsequently spontaneously corrected itself. Initial BCVA was 6/9 in the right eye and 6/6 in the left eye. Upon a biomicroscopic examination on a slit lamp we diagnosed a rounded deposit in the region of the fovea in the right eye, and only an absence of a foveolar reflex in the left eye. Subsequently OCT was performed (instrument RTVue-100, manufacturer Optovue, California, USA) with a finding of perifoveolar ablation of the posterior surface of the vitreous body, with vitreomacular traction syndrome bilaterally, more in the right eye (fig. 1). We recommended bed rest for the patient. A follow-up OCT three weeks later demonstrated a similar finding, a further outpatient follow-up examination was recommended in six weeks.
Fig. 1. Entry OCT image (7/2017, upper – right eye, lower – left eye) 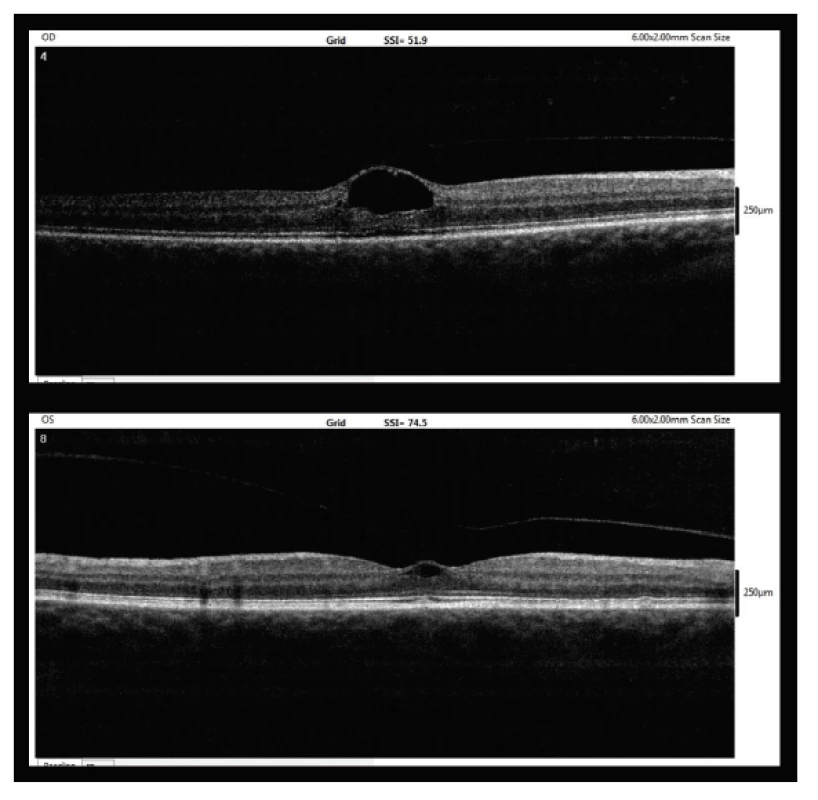
However, after five weeks (9/2017) the patient reported for an unplanned follow-up examination due to a deterioration of vision in the left eye persisting for several days. Vision in the right eye was unchanged, but in the left eye BCVA had decreased to 6/12p. MH was perceptible in the fovea of the left eye upon a biomicroscopic examination of the fundus, with thickening of the vitreous body over it. Examination by OCT confirmed diagnosis of central MD affecting all the layers of the neuroepithelium with a width of 80 µm (fig. 2). On the posterior surface of the displaced posterior vitreous membrane, displaced operculum was perceptible over the MH.
Fig. 2. OCT findings after five weeks (9/2017, upper – right eye, lower – left eye) 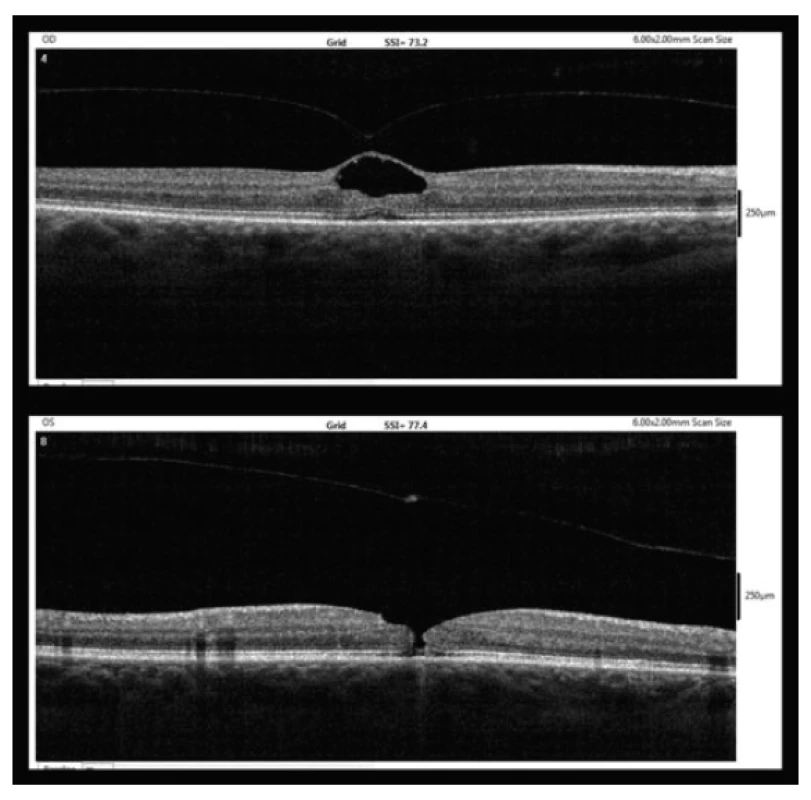
With regard to the fact that the patient was not inclined to undergo an early surgical solution, and also with regard to the prognostically favourable OCT finding – very small MH without VMT, without elevation of edges, without adverse changes of the retinal pigment epithelium (RPE) and preserved structure of the membrana limitans externa (MLE), Müller cells and the line of ellipsoids of the internal segments of the photoreceptors (line of ISe) on the edges of the defect [15, 17] – a regime of strict bed rest lying on the back was recommended, with regular follow-up examinations, and an appointment was also booked for PPV if applicable.
At the next follow-up examination one week after diagnosis of MH, reattachment of the edges of the MH to one another had occurred, with only a slit defect and minor subfoveal ablation of the neuroepithelium (fig. 3). Nepafenac was applied to the left eye in the usual dose (3 mg/ml once per day).
Fig. 3. OCT findings one week after MH diagnosis (upper – right eye, lower – left eye) 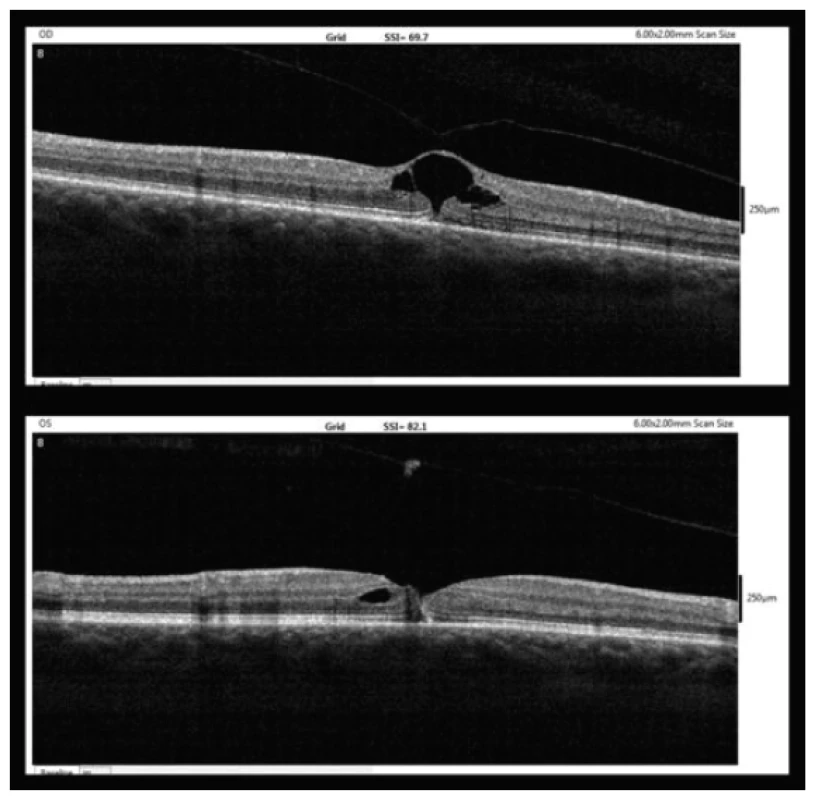
Five weeks after the diagnosis of MH (10/2017), complete reattachment of the edges of the MH to one another was evident, with only minor unevenness on the inner surface of the neuroepithelium (fig. 4). At this point BCVA in the left eye reached 6/6.
Fig. 4. OCT findings five weeks after MH diagnosis (10/2017, upper – right eye, lower – left eye) 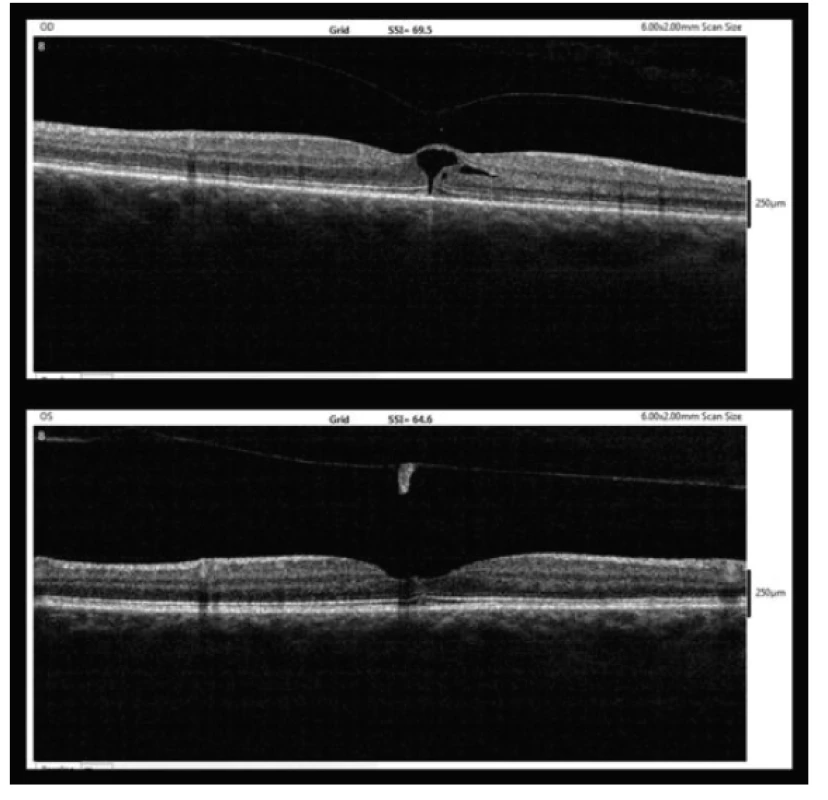
At the same time, in the right eye also the posterior surface of the vitreous body was progressively disattached, with a relaxation of VMT and the return of BCVA to 6/6.
At the last outpatient examination six weeks after diagnosis of MH (3/2018), BCVA was 6/6 bilaterally. On OCT complete ablation of the posterior surface of the vitreous body was present in the right eye in the region of the macula, and normal foveolar depression was restored with normal stratification of all layers of the retina. Also in the left eye the finding was stable. On OCT a closed MH was perceptible with an entirely intact line of ISe, on the inner surface of the retina with unevenness in the place of the loss defect following displacement of the operculum and complete ablation of the posterior surface of the vitreous body in the macula. A displaced operculum was perceptible over the fovea on OCT (fig. 5).
Fig. 5. OCT findings six months after MH diagnosis (3/2018, upper – right eye, lower – left eye) 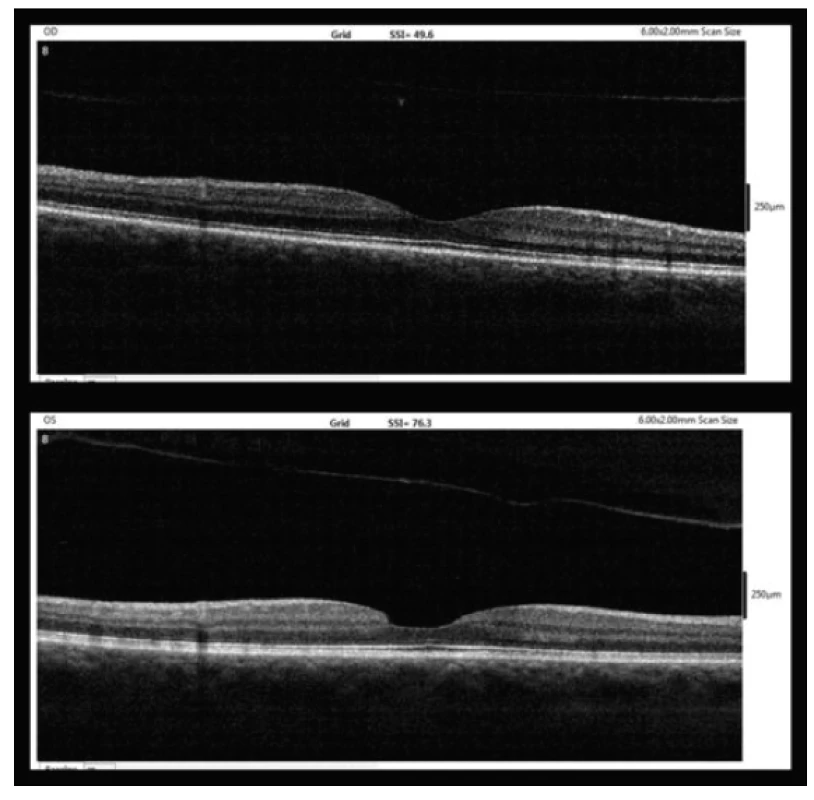
DISCUSSION
Upon the selection of the medical procedure for a patient with a diagnosis of MH, as with other pathologies, the prognosis is of fundamental importance. According to the majority of studies which deal with the natural course and prognosis of MH, especially in the case of MH stage II according to Gasse, progression occurs very frequently and spontaneous closure is rare [3, 7, 11, 17]. For example, the study by Hikichi et al. from 1995 [7] demonstrated 68% probability of progression of size in stage II of MH, in stage III 29% probability and in stage IV 13% probability. A more recent study by Sugiyama et al. described a relatively low proportion (3.5%) of spontaneously closed MHs before the planned performance of vitrectomy, with an average period of 60 days up to the procedure, in which all the spontaneously closed MHs were smaller than 250 μm [17].
In the period before the development of OCT, a study by Kakehashi et al. described the correlation between disruption of VMT and subsequent reattachment of the edges of MH in stage II [9], and a study by Reddy et al. described a series of minor MHs with a size of 50-133 μm, in which visual functions were stable over the course of time, or improved [14].
According to the study by Smiddy et al., there is a possibility of spontaneous closure especially in the case of MH of small dimensions following disruption of VMT, thanks to the reparation capabilities of the glial cells [15]. The recommended indication criteria for intravitreal application of ocriplasmin, i.e. MH < 400 μm with presence of VMT [4, 16], indicate that if disruption of VMT occurs in the case of MH of smaller dimensions, the prognosis is significantly better.
It ensues from the literature that the probability of successful conservative treatment of idiopathic MH is generally very low, but small dimensions of MH and removal of VMT appear to be positive prognostic factors. Similarly important is a preserved structure of Müller cells and other structures of the neuroretina on the edges of the defect [15, 17].
In our case we selected a conservative approach, because the patient, who by coincidence is a doctor, did not wish to undergo a surgical procedure, and because this concerned a relatively prognostically favourable MH of very small dimensions.
This procedure was successful, especially as a consequence of the very small dimensions of the MH, progressive relaxation of VMT, and also as a result of the preserved structure of the Müller cells, MLE and line of ISe on the edges of the defect. The patient was very willing to accept a strict regime of bed rest on her back, in which the pressure of the vitreous body evidently also played a certain role in the healing of this minor MH. The advantage of this procedure is that MH does not require an acute solution, and the prognosis is not worsened if PPV is performed within six months [1].
CONCLUSION
The case report presented here demonstrates that MH of very small dimensions without VMT, with a favourable OCT image, may close spontaneously without a surgical solution, with resulting BCVA of 6/6. Upon selection of a therapeutic procedure in the case of small MH, we recommend individual assessment of the cases according to the OCT finding. In the case of a very favourable finding, it is possible to defer the surgical solution (by a maximum of six months) [1] and attempt conservative treatment.
This case study was presented in the poster section of the XXVI. annual congress of the Czech Ophthalmological Society ČLS JEP [Czech Medical Association of J. E. Purkyně] in Prague (9/2018)
Supported by the project of the Ministry of Health of the conceptual development of a research organisation 00064203 University Hospital MOTOL.
The authors of the study declare that no conflict of interest exists in the compilation, theme and subsequent publication of this professional communication, and that it is not supported by any pharmaceuticals company.
Received by the Editorial Department on: 3 September 2018
Accepted for printing on: 22 November 2018
MUDr. Jakub Dítě
Department of Ophthalmology for Children and Adults, 2nd Faculty of Medicine, Charles University and Motol University Hospital
V Úvalu 84
Prague 5 - Motol, 150 06
Zdroje
1. Bainbridge, J. et al.: Macular holes: vitreoretinal relationships and surgical approaches. Eye (Lond), 22; 2008 : 1301-1309.
2. Coats, G.: The pathology of macular holes. Roy London Hosp Rep; 1907 : 69–96.
3. Chew, EY. et al.: Clinical course of macular holes: the Eye Disease Case-Control Study. Arch Ophthalmol, 117; 1999 : 242-246.
4. Dugel, PU. et al.: Results of the 2-Year Ocriplasmin for Treatment for Symptomatic Vitreomacular Adhesion Including Macular Hole (OASIS) Randomized Trial. Ophthalmology, 123; 2016 : 2232-2247.
5. Duker, JS. et al.: The International Vitreomacular Traction Study Group classification of vitreomacular adhesion, traction, and macular hole. Ophthalmology, 120; 2013 : 2611–2619.
6. Gass, JD.: Idiopathic senile macular hole: its early stages and pathogenesis. Arch Ophthalmol, 106; 1988 : 629–639.
7. Hikichi, T. et al.: Natural outcomes of stage 1, 2, 3, and 4 idiopathic macular holes. Br J Ophthalmol, 79; 1995 : 517–520.
8. Johnson, RN., Gass, JD.: Idiopathic macular holes. Observations, stages of formation, and implications for surgical intervention. Ophthalmology, 95; 1988 : 917-924.
9. Kakehashi, A. et al.: Spontaneous resolution of foveal detachments and macular breaks. Am J Ophthalmol, 120; 1995 : 767-775.
10. Kelly, NE., Wendel, RT.: Vitreous surgery for idiopathic macular holes. Results of a pilot study. Arch Ophthalmol, 109; 1991 : 654-659.
11. Kim, JW. et al.: Baseline characteristics, natural history, and risk factors to progression in eyes with stage 2 macular holes. Results from a prospective randomized clinical trial. Vitrectomy for Macular Hole Study Group. Ophthalmology, 102; 1995 : 1818-1829.
12. Knapp, H.: Ueber isolirte zerreissungen der aderhaut in folge von traument auf dem augapfel. Arch Augenheilk; 1869 : 6–29.
13. McCannel, CA. et al.: Population based incidence of macular holes. Ophthalmology, 116; 2009 : 1366-1369.
14. Reddy, CV. et al.: Microholes of the macula. Arch Ophthalmol, 114; 1996 : 413-416.
15. Smiddy, WE., Flynn, HW.: Pathogenesis of macular holes and therapeutic implications. Am J Ophthalmol, 137; 2004 : 525-537.
16. Stalmans, P. et al.: MIVI-TRUST Study Group. Enzymatic vitreolysis with ocriplasmin for vitreomacular traction and macular holes. N Engl J Med, 367; 2012 : 606-615.
17. Sugiyama, A. et al.: Reappraisal of Spontaneous Closure Rate of Idiopathic Full-Thickness Macular Holes. Open Ophthalmol J, 6; 2012 : 73–74.
Štítky
Oftalmologie
Článek Silent Sinus Syndrome
Článek vyšel v časopiseČeská a slovenská oftalmologie
Nejčtenější tento týden
2018 Číslo 6- Selektivní laserová trabekuloplastika nesnižuje nitroční tlak více než argonová laserová trabekuloplastika
- Progresi glaukomu je třeba hodnotit strukturálními i funkčními parametry
- Ztráta centrálního vidění po filtrujících operacích glaukomu
- Od PGF-2 alfa-isopropyl esteru k latanoprostu: přehled vývoje Xalatanu
- Compliance u pacientů s glaukomem
-
Všechny články tohoto čísla
- Virtiol – Simulation of Quality of Vision with Multifocal and Edof Intraocular Lenses
- Corticosteroid Induced Posterior Subcapsular Cataract
- Ocular Manifestations in Patients with HIV infection
- The Importance of Evaluating the Development of Oct Findings During Conservative Treatment of Vitreomacular Traction Complicated by Macular Hole Formation
- Silent Sinus Syndrome
- Idiopathic Chodoidal Neovascular Membrane in a 12-year-old Girl
- Screening, Treatment and Long-term Observation of Retinopathy of Prematurely Born Children in the Czech Republic
- OČNÍ KLINIKA 1. LÉKAŘSKÉ FAKULTY UNIVERZITY KARLOVY A VŠEOBECNÉ FAKULTNÍ NEMOCNICE V PRAZE SLAVÍ 200 LET OD SVÉHO ZALOŽENÍ
- Vážený a milý pán doc. MUDr. Tomáš Mazalán, CSc.
- Česká a slovenská oftalmologie
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Ocular Manifestations in Patients with HIV infection
- Silent Sinus Syndrome
- Virtiol – Simulation of Quality of Vision with Multifocal and Edof Intraocular Lenses
- Corticosteroid Induced Posterior Subcapsular Cataract
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Autoři: prof. MUDr. Pavel Horák, CSc., doc. MUDr. Ludmila Brunerová, Ph.D., doc. MUDr. Václav Vyskočil, Ph.D., prim. MUDr. Richard Pikner, Ph.D., MUDr. Olga Růžičková, MUDr. Jan Rosa, prof. MUDr. Vladimír Palička, CSc., Dr.h.c.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



