-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
THE VALUE OF CLINICAL DIAGNOSIS OF DIGITAL GLOMUS TUMORS
Autoři: E. Cigna; B. Carlesimo; G. Bistoni; F. Conte; F. Palumbo; N. Scuderi
Působiště autorů: University of Rome “La Sapienza”, Department of Dermatology and Plastic Surgery, Rome, Italy
Vyšlo v časopise: ACTA CHIRURGIAE PLASTICAE, 50, 2, 2008, pp. 55-58
INTRODUCTION
Glomus tumors are benign hamartomas arising from the glomus apparatus and are located in the subcutaneous tissue. These tumors are caused by proliferation of glomus cells, which make up a portion of the glomus body (1, 5, 8, 10, 11, 14).
The normal glomus body is a contractile neuromyoarterial receptor comprising an afferent arteriole, an anastomotic Sequet-Hoyer canal, an efferent venule, actin-containing glomus cells, the intraglomerular reticulum, and a capsular portion that contributes to the regulation of subcutaneous tissue temperature (1, 8, 10).
Although glomus tumors are thought to arise from glomus cells, they have been observed in extracutaneous locations not known to contain glomus cells. Two variants exist: solitary glomus tumors and multiple glomus tumors, also known as glomangiomas (1, 5, 8, 10, 14).
The initiating event for glomus cell proliferation is still unknown. Some authors have postulated that trauma induces solitary subungual glomus tumors, although this theory has yet to be confirmed (1, 10, 11, 14). Multiple lesions are slightly more common in males. Multiple glomus tumors, particularly those of the disseminated variant, are inherited in an autosomal-dominant pattern with incomplete penetrance (1). Most hereditary glomangiomas are associated with defects in the glomulin gene, located on chromosome 1 (1, 5, 8, 10, 11).
The diagnosis of glomus tumors is generally clinical. However, several imaging techniques are still being used to support the clinical diagnosis before undergoing surgical treatment (1, 11).
The instrumental tests used to confirm the clinical evaluation may include: X-ray, MRI, arteriography and ultrasound, with or without Doppler analysis (2, 3, 4, 6, 13).
Clinically, the glomus tumor is common and includes pain, pinpoint pain, hypersensitivity to cold, a bluish or purple color to the finger, and papules or nodules that blanch on pressure.
The therapy for glomus tumors is surgical excision, with a high rate of success (2, 3, 4, 6).
Upon review of our treated cases, here we report our assessment scale that simplifies the diagnosis of this neoplasm and may, in our opinion, avoid unnecessary expensive radiological examinations.
MATERIALS AND METHODS
Patients
From 1997 to 2007, 21 patients were diagnosed and surgically treated for solitary glomus tumor of the digit at the Department of Plastic, Reconstructive and Aesthetic Surgery of the University of Rome “La Sapienza”.
The patient population comprised of 19 females and 2 males. The mean age at diagnosis was 35.5 years (range 19 to 53 years). All patients had a single glomus tumor located in the distal phalanx of the right hand. Patients were divided in two groups.
Group I included nine most recent cases treated surgically after clinical diagnosis alone using our method (Table 1).
Tab. 1. Major and minor symptoms in the diagnosis of digital glomus tumors 
Group II included twelve cases previously treated and retrospectively analyzed and compared to Group I. The analysis of Group II was done using recording charts (Table 2).
Tab. 2. Association of symptoms in the two groups of patients 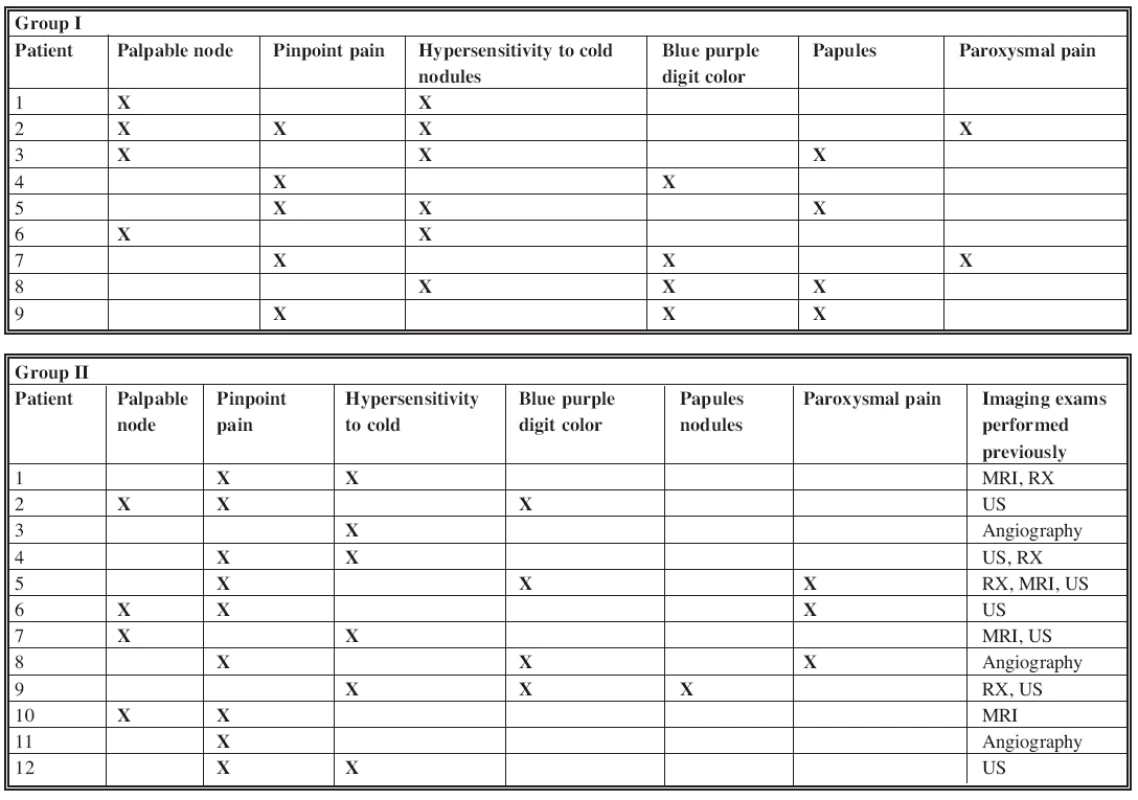
All but two cases resolved with a single operation. Two patients had undergone two operations for incomplete excision of the lesion before being admitted to our Department. Before our clinical examination, the patients in Group II had undergone numerous invasive and extensive instrumentaltests. On ten patients, 18 imaging procedures were performed: 4 X-rays, 7 ultrasound, 4 MRI and 3 angiography (see Table 2).
Surgical excision of the nodule was performed under local anesthesia with mepivacaine 1% digital block on digital ischemia by finger tourniquet. The excision of the sensitive afferent nervous branch was performed under loupe magnification (2.5 X to 4 X) in order to visualize the afferent nervous branch.
Pathological features
The histological appearance of the glomus tumor is a solid neoplasm composed of groups of monomorphic glomus cells with large circular nuclei and a small amount of cytoplasm.
In the center of the neoplasm are well-circumscribed vascular spaces delimited by endothelium surrounded by a vallum of fibrous tissue that is compressed and dislocated by the mass. There are three distinct histologic types of glomus tumor, a mucoid hyaline, a solid, and an angiomatous type.
Among the different types of glomus tumors, immunohistochemistry revealed a positive presence to alfa-actin and alfa-caldesin and negativity to CD-34, S-100, and CK tests.
RESULTS
After review of our treated cases, the symptoms in the two groups were classified according to frequency and clinical relevance to diagnosis. Major symptoms were the most frequent and the most specific, whereas minor symptoms were less frequent and less specific (see Table 1).
Major symptoms included paroxysmal pain caused by glomus-cell contraction, pinpoint pain and hypersensitivity to cold. Minor symptoms included blue or purple color of the digit, papules and nodules that blanched upon pressure (see Table 1). On review of the case histories, minor symptoms appeared to be related to the time of tumor onset, appearing years after the start of the first symptoms that were usually paroxysmal pain and hypersensitivity to cold.
All treated patients had complete regression of pain after surgical excision of the tumor, except for one patient who had hypoesthesia of the treated area for three months after surgery. All patients were examined at follow-up at 6 months and 1 year by cold-sensitivity test and pinpoint test (Love test) and had a negative response.
Three patients had a hypertrophic scar that was treated with silicone gel for 2 to 6 months.
DISCUSSION
Glomus tumors, also known as glomic tumors, glomangiomas, or hemangiopericytomas are benign neoplasms smaller than one centimeter in diameter and commonly located in the distal extremities, particularly in subungual areas. This tumor represents from 1 to 5% of all soft tissue tumors of the hand (1, 5, 7, 8, 10, 11).
Clinically there are several symptoms and signs that have been reported to be related to this neoplasm. Three clinical tests are used to identify the glomus tumor. The Love test consists of eliciting pain by applying pressure to a precise area with the tip of a pencil. The Hildreth sign eradicates pain after application of a tourniquet proximally on the arm. The cold-sensitivity test should result in increased pain when the affected region is exposed to cold temperature (7, 11). However, none of these tests were considered specific to accurately diagnose the tumor. For this reason patients undergo imaging studies for the possible occurrence of glomus tumor prior to surgery.
In the physical examination the differential diagnosis considered other tumors that appeared to be glomus tumors. Schwannoma or neurilemmoma are tumors that most frequently mimic the clinical presentation of a glomus tumor. However, they are not usually painful and do not cause hypersensitivity to cold. Other conditions that must be considered in differential diagnosis for hand tumors may include hemangiomas, leiomyomas, angiolipomas, mucous cysts, neurofibromas, Raynaud phenomenon, and osteogenic tumors (1, 5, 11).
Several imaging studies have been used to define the radiological aspect of the lesion and facilitate differential diagnosis. In the past we used these studies to confirm the diagnosis before surgical excision, particularly when clinical evaluation was not adequate to achieve a correct diagnosis.
Roentgenograms showed bone erosion in 14-60% of patients and an increased distance between the dorsum of the phalanx and the underside of the nail. However, X-rays are not highly repeatable, due to the ionizing power, and more importantly are not very accurate (11).
Ultrasound scanning images may show a hypoechoic-nodular lesion compared to the surrounding fat, which is usually marked off by a regular, thin hyperechoic vallum. This test is accurate, low-cost, non-invasive, non-ionizing and highly repeatable. Ultrasound can also be performed with the power-Doppler mode to assess the blood-flow study (2, 3, 4, 6). The ultrasound power-Doppler has been used to study the artero-venous shunt, a typical characteristic of the glomus tumor, and has now replaced arteriography in the study of vascular morphology. The vascular study is effective in confirming the diagnosis but does not add information that can facilitate surgical excision (2, 3, 4, 6). In fact, arteriography is used also for an accurate study of the vascular composition of the glomus tumor, but this method is invasive, poorly tolerated by patients, and has a low repeatability level, due to its invasiveness and ionizing power (2, 3, 4, 6).
High-resolution magnetic-resonance imaging (MRI) has proved to be a valuable method for imaging glomus tumors and can be useful in efficiently identifying preoperative tumors, particularly those with non-specific symptoms. However, MRI has poor sensitivity to diagnose this type of tumor and is an expensive diagnostic test (1, 11).
In our opinion clinical examination and diagnosis should never replace an instrumental test. A negative clinical examination may influence the surgical approach while the opposite condition will not.
The preferred treatment for solitary glomus tumors is surgical excision that is effective and curative. The prognosis is excellent with complete regression of pain after surgery (9, 11).
Regarding our experience, we believe that clinical examination of the patient is usually sufficient to diagnose glomus tumor. With respect to the nine cases we recently treated, and after review of previously treated cases, it is our opinion that two major symptoms, or one major plus two minor symptoms, are adequate to achieve a high accuracy rate in diagnosing glomus tumor. However, in selected cases with uncertain symptoms or clinical signs or multi-operated patients for recurrence due to inadequate excision of the glomus tumor, ultrasound completed by Doppler ultrasonography may support the diagnosis by the surgeon of subungual glomus tumors. As a result, we achieved a total accuracy of 90% (19/21 patients), 100% on the most recent cases (9/9 patients), and of 82% (9/11 patients) on previously operated cases even though the negative cases were analyzed in a retrospective study of the case histories.
Address for correspondence:
Emanuele Cigna, M.D.
Via Federico Barocci, 3
00147 Rome, Italy
E-mail: emanuelecigna@msn.com
Zdroje
1. Erin M., McDermott BA., Arnold-Peter C., Weiss, MD. Glomus tumours. J. Hand Surg., 31A, 2006, p. 1397-1400.
2. Bruno D., Fornage MD. Glomus tumours in the fingers: diagnosis with ultrasound. Radiology, 167, 1988, p. 183-185.
3. Ogino T., Ohnishi N. Ultrasonography of a subungual glomus tumour. J. Hand Surg., 18B, 1993, p. 746-747.
4. Matsunaga A., Ochiai T., Abe I., Kawamura A., Muto R., Tomita Y., Ogawa M. Subungual glomus tumour: evaluation of ultrasound imaging in preoperative assessment. Eur. J. Dermatol., 17, 2007, p. 67-69.
5. Murthy PS., Rajagopal R., Kar PK., Grover S. Two cases of subungual glomus tumour. Indian J. Dermatol. Venereol. Leprol., 72, 2006, p. 47-49.
6. Trignano M., Campus GV., Soro P., Scuderi N.: Semeiologia strumentale nei tumori gnomici. Rivista Italiana di Chirurgia Plastica, 1988; 14; Fasc. 2.
7. Giele H. Hildreth’s test is a reliable clinical sign for the diagnosis of glomus tumour. J. Hand Surg., 27, 2002, p. 157-158.
8. Bhaskaranand K.,Navadgi BC. Glomus tumour of the hand. J. Hand Surg., 27, 2002, p. 229-231.
9. Takata H. Ikuta Y., Ishida O., Kimori K. Treatment of subungual glomus tumour: J. Hand Surg., 6, 2001, p. 25-27.
10. Carroll RE., Berman AT. Glomus tumours of the hand: review of the literature and report on 28 cases. J. Bone Joint Surg., 4, 1972, p. 691-703.
11. Abou Jaoude JF., Roula Farah A., Sargi Z., Khairallah S., Fakih C. Glomus tumours: report on eleven cases and review of the literature. Chir. Main, 19, 2000, p. 243-252.
12. Dailiana ZH., Drape JL., Le Viet D. A glomus tumour with four recurrences. J. Hand Surg. [Br], 24, 1999, p. 131-132.
13. Opdenakker G., Gelin G., Palmers Y. MR imaging of a subungual glomus tumour. Am. J. Roentgenol., 172, 1999, p. 250-251.
14. Van Geertruyden J., Lorea P., Goldschmidt D., de Fontaine S., Schuind F., Kinnen L., Ledoux P., Moermans JP. Glomus tumours of the hand. A retrospective study of 51 cases. J. Hand Surg. [Br], 21, 1996, p. 257-260.
Štítky
Chirurgie plastická Ortopedie Popáleninová medicína Traumatologie
Článek vyšel v časopiseActa chirurgiae plasticae
Nejčtenější tento týden
2008 Číslo 2- Trombóza portální žíly jako komplikace infekce COVID-19 – kazuistika
- Bezpečnostní profil metamizolu – systematický přehled
- Spasmolytický účinek metamizolu
-
Všechny články tohoto čísla
- ARTERIALIZED VENOUS FREE FLAPS – A RECONSTRUCTIVE ALTERNATIVE FOR LARGE DORSAL DIGITAL DEFECTS
- RECONSTRUCTION OF LARGE UPPER LIP DEFECTS BY FREE TISSUE TRANSFER
- THE VALUE OF CLINICAL DIAGNOSIS OF DIGITAL GLOMUS TUMORS
- GIANT CELL REPARATIVE GRANULOMA IN THE MANDIBLE – CASE REPORT AND REVIEW OF THE LITERATURE
- IPSILATERAL FIBULAR TRANSFER: A VALUABLE OPTION FOR TREATMENT OF LARGE TIBIAL DEFECTS IN CHILDREN
- ČESKÉ A SLOVENSKÉ SOUHRNY
- Acta chirurgiae plasticae
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- ARTERIALIZED VENOUS FREE FLAPS – A RECONSTRUCTIVE ALTERNATIVE FOR LARGE DORSAL DIGITAL DEFECTS
- GIANT CELL REPARATIVE GRANULOMA IN THE MANDIBLE – CASE REPORT AND REVIEW OF THE LITERATURE
- RECONSTRUCTION OF LARGE UPPER LIP DEFECTS BY FREE TISSUE TRANSFER
- THE VALUE OF CLINICAL DIAGNOSIS OF DIGITAL GLOMUS TUMORS
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Autoři: prof. MUDr. Pavel Horák, CSc., doc. MUDr. Ludmila Brunerová, Ph.D., doc. MUDr. Václav Vyskočil, Ph.D., prim. MUDr. Richard Pikner, Ph.D., MUDr. Olga Růžičková, MUDr. Jan Rosa, prof. MUDr. Vladimír Palička, CSc., Dr.h.c.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



