-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
The Difference between Ganglion Cell Complex and Nerve Fiber Layer in the Same Altitudinal Halves of the Retina in Hyper-tension and Normal-tension Glaucomas
Authors: J. Lešták 1,2; Š. Pitrová 1,2; M. Žáková 2
Authors place of work: Oční klinika JL FBMI ČVUT Praha, vedoucí lékař doc. MUDr. Ján Lešták, CSc., FEBO, MBA 1; FBMI ČVUT v Praze, Katedra zdravotnických oborů a ochrany obyvatelstva, Kladno, vedoucí katedry: prof. MUDr. Leoš Navrátil, CSc. 2
Published in the journal: Čes. a slov. Oftal., 73, 2017, No. 5-6, p. 218-221
Category: Původní práce
Summary
Nerve Fiber Layer (RNFL) measurements in the same altitudinal half of the eye in hyper-tension (HTG) and normal-tension glaucomas (NTG).
Materials and Methods:
We examined both eyes in altogether 50 patients (100 eyes). The cohort was divided into two groups, in each of them; there were 25 patients with average age of 55.4 in the NTG group and 54.6 years in the HTG group. The NTG group consisted of 17 women of the average age of 55.35 (range, 25–75) years, and 8 men of the average age of 55.5 (range, 32–69) years. In the HTG group, there were 12 women of the average age of 53.25 (range, 34–69) years, and 13 men of the average age of 60.38 (range, 37–74) years. The criteria for including the patients into the groups were: the visual acuity at least 1.0 (20/20) with possible correction smaller than ± 3 diopters, and approximately identical changes in the visual fields in all patients with initial stage of the glaucoma disease; no other ophthalmic or neurological diseases. In patients in the NTG group, the diagnosis was confirmed by means of electrophysiological examination. The thickness of the GCC was measured by means of SD-OCT RTvue – 100, similarly as the RNFL.Results:
In the statistical survey of the GCC values in HTG and NTG in the same altitudinal halves of the retina we did not establish statistical significant difference. We obtained the same result in statistical evaluation of the RNFL in HTG and NTG groups in the same altitudinal halves of the retinas. Similarly, even after the subtraction the GCC from RNFL values in the same altitudinal halves of the retinas we did not establish statistically significant difference between both groups.Conclusion:
The GCC and the RNFL examination values in the same altitudinal halves of the retinas did not establish statistically significant difference that would differ the patients with HTG from patients with NTG. Although, using the OCT, we did not confirm the differences in both diagnostic groups, it does not mean, that these differences do not exist and they may have several causes.Key words:
Ganglion Cell Complex (GCC), Retinal Nerve Fiber Layer (RNFL), Hyper-Tension Glaucoma (HTG), Normal-Tension Glaucoma (NTG)INTRODUCTION
Glaucomas are currently indicated as a chronic progressive neuropathy with excavation and atrophy of the disc of the optic nerve and subsequent changes in the visual field. This definition not only fails to embrace current knowledge, but at the same time does not differentiate between high-tension (HTG) and normal-tension glaucoma (NTG). Furthermore it overlooks the pathogenesis of excavation, which was summarised by Hayreh (3) in 1974 into three factors which are probably responsible for this abnormality:
1. destruction of nerve tissue in prelaminar region,
2. distortion of the lamina cribriformis in a backward direction, caused by retrolaminar fibrosis, and a lack of support of the posterior part of the lamina due to its loss,
3. weakening of the lamina cribriformis.
However, these changes are not characteristic only of glaucoma atrophy of the disc of the optic nerve, but also other (mainly vascular) causes. We summarised the differences between HTG and NTG in our previous study (8). On this occasion, it is again necessary to point out that in the case of HTG damage to ganglion cells occurs diffusely throughout the entire retina with subsequent decay of their axons. In the case of NTG, in contrast with HTG, the electrical response of the ganglion cells is relatively good. On the basis of this information we can schematically illustrate the layer of the ganglion cells and their fibres as follows (fig. 1).
Fig. 1. Schematic illustration of inner layers of retina (RNFL – retinal nerve fibre layer, GCL – ganglion cell layer, IPL – inner plexiform layer) a) normal finding, b) finding in HTG – reduction of GCL and slightly also RNFL, c) finding in NTG – reduction of RNFL and slightly also GCL 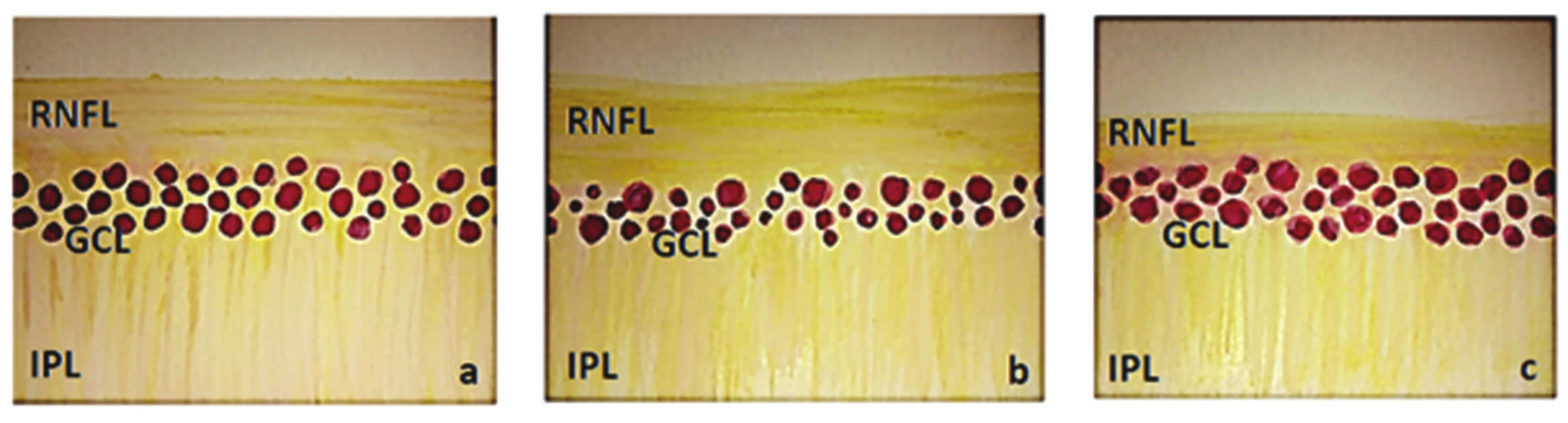
In the case of NTG, the ganglion cell layer (GCL) should be less damaged in comparison with the RNFL, mainly in the initial stages. In our last study on this topic (8) we demonstrated a medium-strong correlation between sensitivity in the altitudinal hemipoles in comparison with the bilateral value of the GCC only in the case of NTG. By contrast, in the case of HTG we did not determine any statistically significant correlation (8).
On the basis of these hypotheses we set the goal of our study, which is to determine whether a statistically significant difference exists between the GCC and RNFL in the same altitudinal half of the retina in HTG and NTG.
COHORT AND METHOD
We examined both eyes in a total of 50 patients (100 eyes). The cohort was divided into two groups, in which each group contained 25 patients with an average age of 55.4 years in the NTG group and 54.6 years in the HTG group. The NTG group comprised 17 women with an average age of 55.35 (25-75) years and 8 men with an average age of 55.5 (32-69) years. The HTG group comprised 12 women with an average age of 53.25 (34-69) years and 13 men with an average age of 60.38 (37-74) years. The criteria serving for classification of the patients into groups were as follows: visual acuity of at least 1.0 after applicable correction of less than ± 3 dioptres (11). Approximately identical changes within the visual field in all patients with incipient glaucoma pathology (7). No other ocular or neurological pathology. In the NTG patients the diagnosis was confirmed by electro-physiological examination. The thickness of the GCC layer was measured with the aid of SD-OCT RTvue -100, similarly in the case of the RNFL.
RESULTS
The measured and calculated values are presented in summary in tables 1 and 2.
Tab. 1. Summary table of measured values in NTG (F/33 = female/33 years, M/47 = male/47 years, GCC = ganglion cell complex, RE = right eye, LE = left eye, UHR = upper half of retina, LHR = lower half of retina, RNFL = retinal nerve fibre layer, Difference = RNFL-GCC 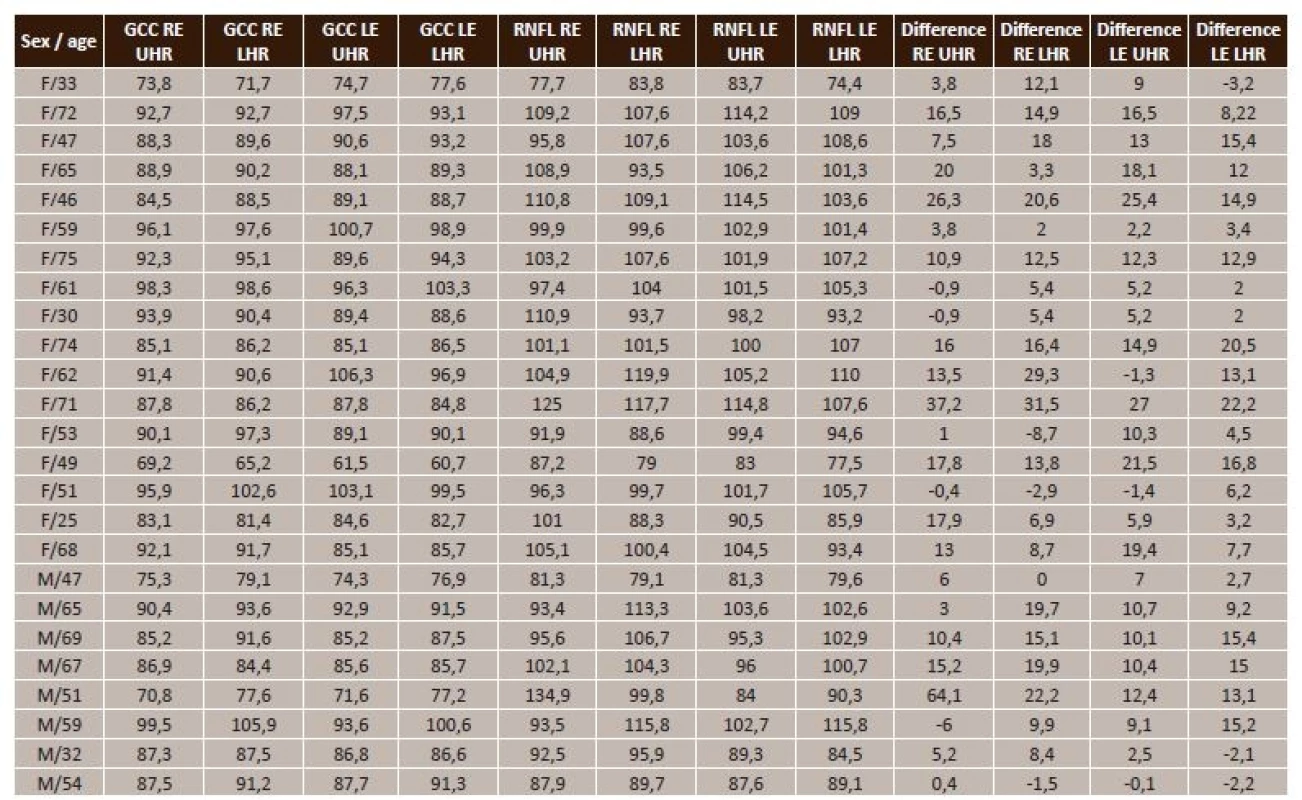
Tab. 2. Summary table of measured values in NTG (F/33 = female/33 years, M/47 = male/47 years, GCC = ganglion cell complex, RE = right eye, LE = left eye, UHR = upper half of retina, LHR = lower half of retina, RNFL = retinal nerve fibre layer, Difference = RNFL-GCC 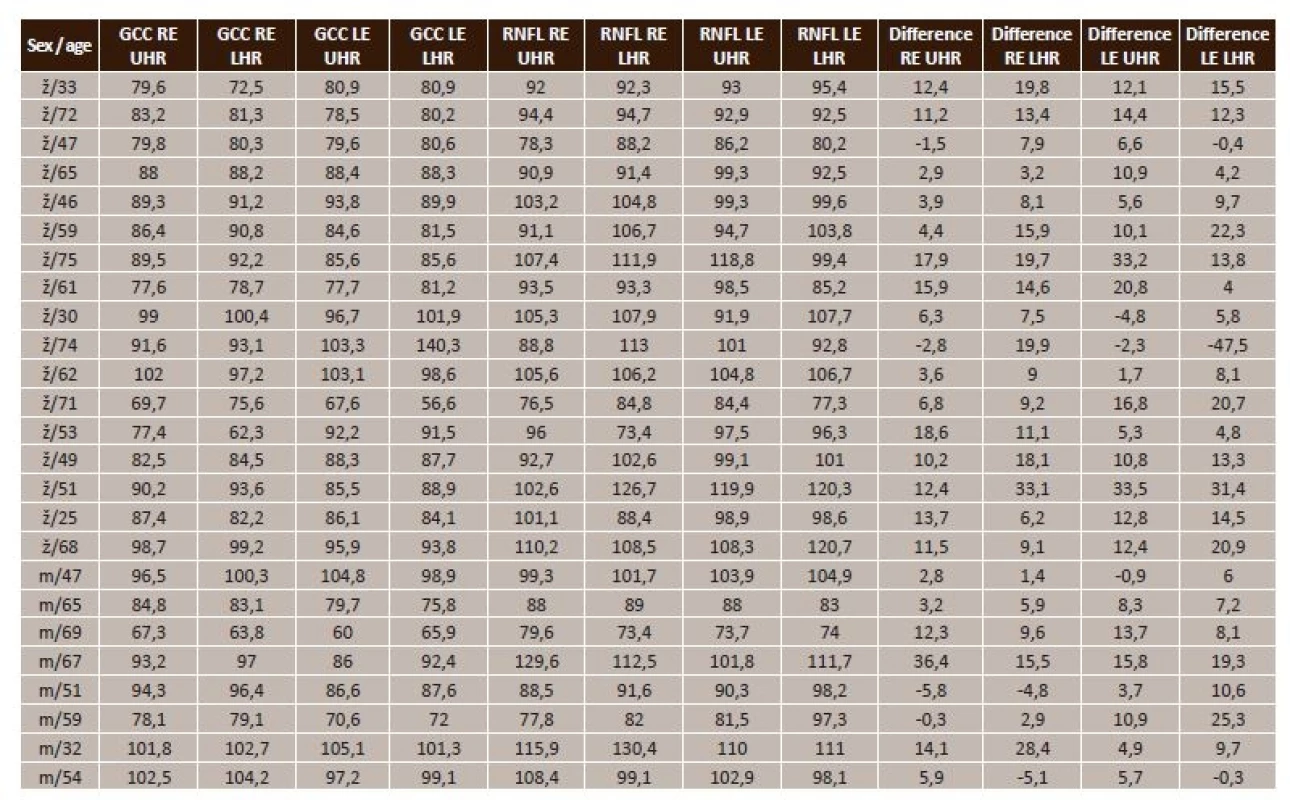
Statistical processing of the GCC and RNFL values in HTG and NTG in the same altitudinal halves of the retina is presented in table 3.
Tab. 3. Values of GCC and RNFL in HTG and NTG (GCC = ganglion cell complex, RNFL = retinal nerve fibre layer) 
Through statistical processing of the values of GCC in HTG and NTG in the same altitudinal halves of the retinas we did not determine any statistically significant difference. We arrived at the same result also upon statistical evaluation of the RNFL in HTG and NTG in the same altitudinal halves of the retinas.
With regard to these results, we subsequently compared the differences between RNFL and GCC in the same altitudinal halves of the retina and conducted statistical processing (table 4).
Tab. 4. Difference between RNFL and GCC in HTG and NTG in same altitudinal halves of retina (GCC = ganglion cell complex, RNFL = retinal nerve fibre layer) 
Similarly also after deduction of the GCC from the RNFL in the same altitudinal halves, we did not determine any statistically significant difference between both groups.
DISCUSSION
The ganglion cell complex (GCC) is composed of three layers. The dendrites of the ganglion cells (IPL) form the internal plexiform layer, and there follows the actual ganglion cell layer (GCL), formed by the bodies of the cells, and finally the axons of the ganglion cells form the retinal nerve fibre layer (RNFL). The GCC, in particular the macular region containing 50% of all ganglion cells of the retina, is an ideal location for detecting early changes in glaucoma and also for monitoring the progression of this pathology (4, 9, 10).
There is a relatively sizeable number of studies in the literature dealing with the issue of GCC and RNFL in high-tension glaucomas, less so on the topic of HTG and NTG. Gracitelli et al. determined that patients with NTG have a thinner RNFL than patients with HTG (2). By comparison, these authors did not record this difference in GCC. Our results did not demonstrate this difference. Kim et al. determined that the average value of RNFL in HTG does not differ from NTG. However, the average value of GCC differed (5). Similar results were attained by Konstantakoupoulou et al. (6), who did not determine any difference in the RNFL in HTG and NTG.
All our patients in the observed cohort had glaucoma pathology in the initial stages. We assumed, entirely logically, that we would find a statistically significant difference at least in the RNFL values. However, we did not demonstrate this hypothesis.
The functional condition of the ganglion cells of the retina and their axons cannot be determined by an examination of the GCC and RNFL, since this concerns techniques serving for structural imaging. It is generally known that first of all a collapse of the ganglion cells takes place, and when the fundamental cause generating this condition exceeds the given time frame, their necrosis takes place. For this reason it is possible to show a decline by PERG (pattern electroretinogram) before structural changes occur. This fact was demonstrated also by Bowd et al. (1). In NTG the electrical responses from the ganglion cells are relatively within the norm.
We did not demonstrate differences in the two diagnostic groups by means of an examination with the aid of OCT. In our opinion, however, this does not mean that these differences do not exist morphologically. The result may be due to the low resolution capacity of the OCT technology, or the variant of the non-existence of differences in the initial stages of the pathology, or a combination thereof.
CONCLUSION
Examination of the GCC and RNFL in the same altitudinal halves of the retina did not demonstrate a statistically significant difference distinguishing patients with HTG from patients with NTG. Although we did not demonstrate differences in the two diagnostic groups with the aid of OCT, this does not mean that these differences do not exist morphologically. The result may be due to the low resolution capacity of the OCT technology, or the variant of the non-existence of differences in the initial stages of the pathology, or a combination thereof.
The authors of the study declare that no conflict of interest exists in the compilation, theme and subsequent publication of this professional communication, and that it is not supported by any pharmaceuticals company.
Doc. MUDr. Ján Lešták, CSc., FEBO, MBA
Oční klinika JL FBMI ČVUT Praha,
V Hůrkách 1296/10,
158 00 Praha 5 – Nové Butovice
E mail: lestak@seznam.cz
Zdroje
1. Bowd C, Tafreshi A, Zangwill LM, Medeiros FA, Sample PA, Weinreb RN: Pattern electroretinogram association with spectral domain OCT structural measurements in glaucoma. Eye (Lond), 25; 2011 : 224–232.
2. Graciletti CP, Moreno PA, Leite MT, Prata TS: Identification of the most accurate spectral-domain optical coherence tomography parameters in eyes with early high-tension and low-tension glaucoma. J. Glaucoma 25; 2016 : 854–859.
3. Hayreh SS: Pathogenesis of cupping of the optic disc. Brit. J. Ophthal. 58; 1974 : 863-876.
4. Jeoung JW, Choi YJ, Park KH, Kim DM: Macular ganglion cell imaging study: glaucoma diagnostic accuracy of spectraldomain optical coherence tomography. Invest Ophthalmol Vis Sci, 54; 2013 : 4422–4429.
5. Kim NR, Hong S, Kim JH, Rho SS, Seong GJ, Kim CY: Comparison of macular ganglion cell complex thickness by Fourier-domain OCT in normal tension glaucoma and primary open-angle glaucoma. J Glaucoma, 22; 2013 : 133–139.
6. Konstatakopoulou E, Reeves BC, Fenerty C, Harper RA: Retinal nerve fiber layer measures in high - and normal-tension glaucoma. Optom Vis Sci. 85; 2008 : 538–542.
7. Lestak J, Nutterova E, Jiraskova N, Navratil L: Ganglion cell complex and nerve fibre layer in hypertension and normal-tension glaucoma. Wulfenia Journal 23; 2016 : 2–12.
8. Lešták J, Pitrová Š: „Ganglion cells complex“ a vrstva nervových vláken u hypertenzních a normotenzních glaukomů. Čs a Slov Oftalmol, 72; 2016 : 199–203.
9. Na JH, Lee K, Lee JR, Baek S, Yoo SJ, Kook MS: Detection of macular ganglion cell loss in preperimetric glaucoma patients with localized retinal nerve fiber defects by spectraldomain optical coherence tomography. Clin Experiment Ophthalmol, 41; 2013 : 870–880.
10. NouriMahdavi K, Nowroozizadeh S, Nassiri N, Cirineo N, Knipping S, Giaconi J, Caprioli J: Macular ganglion cell/inner plexiform layer measurements by spectral domain optical coherence tomography for detection of early glaucoma and comparison to retinal nerve fiber layer measurements. Am J Ophthalmol, 156; v2013 : 1297–1307.
11. Takeyama A, Kita Y, Kita R, Tomita G: Influence of axial length on ganglion cell complex (GCC) thickness and on GCC thickness to retinal thickness ratios in young adults. Jpn J Ophthalmol, 58; 2014 : 86–93.
Štítky
Oftalmologie
Článek Autorský rejstřík
Článek vyšel v časopiseČeská a slovenská oftalmologie
Nejčtenější tento týden
2017 Číslo 5-6- Selektivní laserová trabekuloplastika nesnižuje nitroční tlak více než argonová laserová trabekuloplastika
- Progresi glaukomu je třeba hodnotit strukturálními i funkčními parametry
- Ztráta centrálního vidění po filtrujících operacích glaukomu
- Od PGF-2 alfa-isopropyl esteru k latanoprostu: přehled vývoje Xalatanu
- Compliance u pacientů s glaukomem
-
Všechny články tohoto čísla
- OCT Findings and Long-term Follow-up Results of Vitrectomy in Patients with Optic Disc Pit and Associated Maculopathy
- PERSISTENT HYALOID ARTERY – PERFORM A SURGERY OR NOT?
- Combination of Intravitreal Corticosteroid with Anti-vegf in Macular Edema Secondary to Retinal Vein Occlusion
- Jubileum doc. MUDr. Šárky Pitrové, CSc., FEBO
- Sarkoidosis and its Eye Ocular Manifestation (an Analysis of Six Case Report)
- Results of Treatment of Diabetic Retinopathy by the Laser System PASCAL
- Ciliary body melanoma treatment by stereotactic radiosurgery
- The treatment results in patients with lmphoma disease in the orbit
- The Difference between Ganglion Cell Complex and Nerve Fiber Layer in the Same Altitudinal Halves of the Retina in Hyper-tension and Normal-tension Glaucomas
- Xerosis in Patient with Vitamin A Deficiency – a Case Report
- Recommended procedure for eye examination for infants and children of pre-school age in regular outpatient practice
- Autorský rejstřík
- Česká a slovenská oftalmologie
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Sarkoidosis and its Eye Ocular Manifestation (an Analysis of Six Case Report)
- Recommended procedure for eye examination for infants and children of pre-school age in regular outpatient practice
- PERSISTENT HYALOID ARTERY – PERFORM A SURGERY OR NOT?
- The treatment results in patients with lmphoma disease in the orbit
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Autoři: prof. MUDr. Pavel Horák, CSc., doc. MUDr. Ludmila Brunerová, Ph.D., doc. MUDr. Václav Vyskočil, Ph.D., prim. MUDr. Richard Pikner, Ph.D., MUDr. Olga Růžičková, MUDr. Jan Rosa, prof. MUDr. Vladimír Palička, CSc., Dr.h.c.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání




