-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
Top novinky
ReklamaThe Rac GTP Exchange Factor TIAM-1 Acts with CDC-42 and the Guidance Receptor UNC-40/DCC in Neuronal Protrusion and Axon Guidance
The mechanisms linking guidance receptors to cytoskeletal dynamics in the growth cone during axon extension remain mysterious. The Rho-family GTPases Rac and CDC-42 are key regulators of growth cone lamellipodia and filopodia formation, yet little is understood about how these molecules interact in growth cone outgrowth or how the activities of these molecules are regulated in distinct contexts. UNC-73/Trio is a well-characterized Rac GTP exchange factor in Caenorhabditis elegans axon pathfinding, yet UNC-73 does not control CED-10/Rac downstream of UNC-6/Netrin in attractive axon guidance. Here we show that C. elegans TIAM-1 is a Rac-specific GEF that links CDC-42 and Rac signaling in lamellipodia and filopodia formation downstream of UNC-40/DCC. We also show that TIAM-1 acts with UNC-40/DCC in axon guidance. Our results indicate that a CDC-42/TIAM-1/Rac GTPase signaling pathway drives lamellipodia and filopodia formation downstream of the UNC-40/DCC guidance receptor, a novel set of interactions between these molecules. Furthermore, we show that TIAM-1 acts with UNC-40/DCC in axon guidance, suggesting that TIAM-1 might regulate growth cone protrusion via Rac GTPases in response to UNC-40/DCC. Our results also suggest that Rac GTPase activity is controlled by different GEFs in distinct axon guidance contexts, explaining how Rac GTPases can specifically control multiple cellular functions.
Published in the journal: . PLoS Genet 8(4): e32767. doi:10.1371/journal.pgen.1002665
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1002665Summary
The mechanisms linking guidance receptors to cytoskeletal dynamics in the growth cone during axon extension remain mysterious. The Rho-family GTPases Rac and CDC-42 are key regulators of growth cone lamellipodia and filopodia formation, yet little is understood about how these molecules interact in growth cone outgrowth or how the activities of these molecules are regulated in distinct contexts. UNC-73/Trio is a well-characterized Rac GTP exchange factor in Caenorhabditis elegans axon pathfinding, yet UNC-73 does not control CED-10/Rac downstream of UNC-6/Netrin in attractive axon guidance. Here we show that C. elegans TIAM-1 is a Rac-specific GEF that links CDC-42 and Rac signaling in lamellipodia and filopodia formation downstream of UNC-40/DCC. We also show that TIAM-1 acts with UNC-40/DCC in axon guidance. Our results indicate that a CDC-42/TIAM-1/Rac GTPase signaling pathway drives lamellipodia and filopodia formation downstream of the UNC-40/DCC guidance receptor, a novel set of interactions between these molecules. Furthermore, we show that TIAM-1 acts with UNC-40/DCC in axon guidance, suggesting that TIAM-1 might regulate growth cone protrusion via Rac GTPases in response to UNC-40/DCC. Our results also suggest that Rac GTPase activity is controlled by different GEFs in distinct axon guidance contexts, explaining how Rac GTPases can specifically control multiple cellular functions.
Introduction
In a developing nervous system, axonal growth happens via extension and migration of the growth cone, an actin-rich neuronal structure from which filopodial and lamellipodial structures emanate in order to sense the surrounding environment for appropriate guidance cues [1]–[5]. Guidance receptors such as Deleted in Colorectal Carcinoma (DCC) and Roundabout (ROBO) are present in the leading edge of the growth cone and are activated upon ligand binding (such as Netrin and Slit, respectively) [6]–[10]. Attractive and repellent guidance systems guide the growth cone toward its final target, presumably by affecting the protrusiveness of filopodial and lamellipodial structures in the growth cone [8], [11]. The proper control of the actin cytoskeleton is crucial for the development, maintenance and dynamics of these filopodia and lamellipodia. Failure in the proper extension of axons during development is associated with several neurological disorders, including autism spectrum disorders and dyslexia [12], [13].
The intracellular signaling mechanisms linking guidance receptors to cytoskeletal alterations are an intense area of study, and many remain to be elucidated. Rac GTPases, members of the Rho subfamily of small GTPases (which also includes Rho and Cdc-42), have been well established as modulators of the actin cytoskeleton [14] and axon guidance [15]. Rac GTPases and their downstream effectors modulate filopodia and lamellipodia morphology and dynamics during axon pathfinding in vitro in mammalian cell culture, as well as in vivo in the fruit fly Drosophila melanogaster and the nematode Caenorhabditis elegans [15]–[18]. Rac GTPases act on the actin cytoskeleton via diverse downstream effectors such as the actin-filament nucleator Arp2/3 complex, the severing protein Cofilin, the Pak and LIM kinases, and the actin binding protein UNC-115/abLIM [19]–[23]. In C. elegans growth cone development and axon pathfinding, the two redundant Rac GTPases CED-10/Rac1 and MIG-2/RhoG act via two distinct pathways to activate the Arp2/3 complex: CED-10/Rac1 acts with WVE-1/Wave, while MIG-2/RhoG acts with WSP-1/Wasp [24]. CED-10/Rac1 is also known to act with the actin-binding protein UNC-115/abLIM via the Receptor for Activated C Kinase (RACK-1) [25].
Although much is known about the downstream effectors of the Rac GTPases in axon development, less is known about pathways upstream of Rac GTPases that control their activity in vivo. Rac GTPases switch between an inactive, GDP-bound state, and an active, GTP-bound state. Rac GTPase activation can be reached with the aid of a Guanine-nucleotide Exchange Factor (GEF), which catalyzes the exchange of GTP for GDP [26]. The C. elegans genome encodes 19 predicted Dbl-homology (DH) GEF proteins (Wormbase). UNC-73/Trio is a well-characterized GEF for MIG-2/RhoG and CED-10/Rac1 in C. elegans axon pathfinding [27]–[30]. A mutation that specifically disrupts specifically the RacGEF activity of UNC-73/Trio (rh40) disrupts axon pathfinding [30]. However, pathfinding defects of mig-2; ced-10 rac double mutants are more severe than unc-73(rh40) [28], indicating that additional GEFs act with Rac GTPases to control axon pathfinding.
Here we show that TIAM-1 is a Rac GEF similar to mammalian T-cell lymphoma Invasion and Metastasis Factor 1 (Tiam1) [31] and Drosophila Still Life (Sif) [32]. Previous results suggest that the UNC-73/Trio Rac GEF does not act with CED-10/Rac1 downstream of UNC-40/DCC in neuronal protrusion [9]. Here we show that the TIAM-1 Rac GEF acts downstream of the UNC-40/DCC netrin receptor in neuronal protrusion, demonstrating the modular use of Rac GEFs in the control of distinct Rac-mediated events. We report that TIAM-1 functions mechanistically as a Rac-specific GEF and that TIAM-1 controls Rac GTPases in lamellipodia and filopodia formation. The roles of Rac and CDC-42 GTPases in lamellipodia and filopodia formation have been documented extensively, yet it is unclear how these molecules interact in this process. Here we show that TIAM-1 acts downstream of the CDC-42 GTPase, and might link CDC-42 and Rac signaling in a linear pathway in lamellipodia and filopodia formation downstream of UNC-40/DCC. Sif and Tiam1 have been previously implicated in axon development [33], [34]. We present evidence that TIAM-1 acts in the UNC-40/DCC pathway in axon guidance. Together, these data lead us to speculate that TIAM-1 controls growth cone lamellipodia and filopodia formation during axon guidance downstream of the UNC-40/DCC guidance receptor.
Results
C11D9.1 and T21E12.2 encode a GEF similar to mammalian Tiam1 and Drosophila Still Life
The C11D9.1 gene is predicted to encode a molecule with DH and Pleckstrin homology (PH) domains most similar to mammalian T-cell lymphoma Invasion and Metastasis Factor 1 (Tiam1) and Drosophila Still Life (Sif), but lacks an apparent EVH1 domain and PDZ domain found in Tiam1 and Sif [33], [35]. Three cDNAs representing C11D9.1 were obtained and sequenced (courtesy of Yuji Kohara). One of the cDNAs contained sequences complementary not only to C11D9.1, but also to the predicted upstream gene T21E12.2 (Figures S1 and S2), indicating that C11D9.1 and T21E12.2 are a single gene.
A search of the additional predicted sequence encoded by T21E12.2 using BLASTP and CCD [36], [37] did not identify regions similar to the EVH1 domain and PDZ domain of Tiam1 and Sif. However, two regions in the C. elegans molecule were conserved in the related nematodes Ascaris Suum and Brugia malayi, and these regions from A. suum and B. Malayi were recognized by BLASTP as being similar to the EVH1 and PDZ domain regions of Tiam1 and Sif (Figure S3). While these might not represent bona fide EVH1 and PDZ domains, this evolutionary comparison revealed regions in T21E12.2/C11D9.1 similar to the EVH1 domains and PDZ domains of Tiam1 and Sif. Mammalian Tiam1 contains an N-terminal myristoylation site (Myr) [38], [39], and a potential Myr site is found at the N-terminus of the predicted TIAM-1 molecule (Figure S2). We refer to the T21E12.2/C11D9.1 locus collectively as tiam-1.
The predicted molecules encoded by the tiam-1 cDNAs are shown in Figure S1. In other organisms, the DH-PH domain of Tiam1/Sif is a Rac-specific GEF [34], [40]. Both alleles of tiam-1 used here (ok772 and tm1556) remove at least part of the DH domain (Figures S1 and S2), and are predicted to cause frame shifts. Thus, ok772 and tm1556 are likely to be strong loss of function mutations.
TIAM-1 interacts with Rac GTPases in axon pathfinding
The C. elegans Rac-like GTPases MIG-2/RhoG and CED-10/Rac1 act redundantly in axon pathfinding [28]. Loss-of-function mutations in either mig-2 or ced-10 alone display few defects in pathfinding of the PDE axons (1% and 0%, respectively), but when in a double mutant combination (mig-2(mu28); ced-10(n1993M+); M+ indicates that the genotype has wild-type maternally-supplied gene function), most PDE neurons were affected (84%, Figure 1A and [41]). UNC-73/Trio is a well characterized GEF for MIG-2/RhoG and CED-10/Rac1 in axon pathfinding [30]. A mutation that specifically abolishes the Rac GEF activity of UNC-73 (rh40, a Serine-1216-Phenylalanine (S1216F) substitution in the Rac-specific DH1 domain) [30] displayed severe axon pathfinding defects (42%, Figure 1A), but not as strong as the double mutant of mig-2(mu28); ced-10(n1993M+) (p<0.001). mig-2(mu28); unc-73(rh40) and ced-10(n1993); unc-73(rh40) doubles display pathfinding defects equivalent to mig-2(mu28); ced-10(n1993M+) [41]. Thus, another GEF might regulate Rac activity in axon pathfinding.
Fig. 1. TIAM-1 acts with the Rac GTPases in axon pathfinding. 
A) A graph representing PDE axon pathfinding defects (X axis) in different genotypes (Y axis). At least 100 PDE neurons were scored, and p value significance was determined by Fisher's exact analysis. [tiam-1(+)] indicates a transgene that harbors a wild-type genomic copy of the tiam-1 locus on fosmid clone WRM0633ch01. The error bars represent 2× standard error of the proportion in each direction. M+ indicates that the genotype had wild-type maternal gene function. The inset is a model of the predicted TIAM-1 isoform A molecule (856 amino acid residues) showing the regions of the molecule missing in two deletion alleles. Unless otherwise noted, all backgrounds are wild-type. p value significance as determined by Fisher Exact Analysis is shown. Dashed lines indicate comparisons between genotypes not marked with a p value to those marked with p values. (B–D) Fluorescent micrographs of osm-6::gfp in the PDE neurons of young adults. In all micrographs, dorsal is up and anterior is to the left. The arrows point to axons, which are misguided in mutants in (B) and (D). The ventral nerve cord is represented by a dashed line. The scale bar in (B) represents 5 µm for (B–D). The C. elegans genome encodes 19 predicted DH-containing GEFs including UNC-73/Trio (Wormbase, and data not shown). We screened mutant alleles or RNAi of the other 18 DH-GEFs alone or in combination with ced-10(n1993) and mig-2(mu28) for PDE axon pathfinding defects (EAL, unpublished). The two deletion alleles of tiam-1 described above, ok772 and tm1556, caused low-penetrance defects in PDE axon pathfinding (1% and 3%, respectively) (Figure 1A). In double mutant combination with mig-2(mu28) and ced-10(n1993), these defects were synergistically enhanced (Figure 1A–1C). For example, tiam-1(tm1556); mig-2(mu28) displayed 15% misrouted PDEs (p = 0.001), and tiam-1(tm1556); ced-10(n1993) 12% (p = 0.008). tiam-1 mutations slightly but significantly enhanced unc-73(rh40) (Figure 1A; p = 0.022 and 0.015), indicating that TIAM-1 and UNC-73 might act in parallel in PDE axon guidance. Misguided axons often grew distances that were greater than the wild-type PDE axon, suggesting a defect in guidance rather than a general outgrowth defect. The fosmid clone WRM0633ch01 (Source Bioscience) carrying a wild-type copy of the tiam-1 gene rescued the PDE axon pathfinding defects of tiam-1(tm1556); ced-10(n1993) (12% to 5%, p = 0.028) (Figure 1A). We noted that tiam-1(+) expression in a wild-type background caused a low level of PDE axon pathfinding defects (5%; Figure 1A), equivalent to the level in tiam-1(tm1556); ced-10(n1993) rescued animals. Thus, TIAM-1 transgenic expression caused defects in axon pathfinding.
The phenotype of tiam-1(ok772); mig-2(mu28) was significantly weaker than that of tiam-1(tm1556); mig-2(mu28) (p = 0.02) and other tiam-1 doubles with mig-2 and ced-10 (Figure 1A). This might indicate that ok772 retains some gene function and is a hypomorph. Alternatively, this might be the effect of genetic background. In any case, enhancement in tiam-1(ok772); mig-2(mu28) is still significant compared to tiam-1(ok772) alone (p = 0.046).
tiam-1 mutants alone also displayed weak defects in other neurons. In the AVM neurons, tiam-1(ok772) displayed ventral axon pathfinding defects and 6% ectopic neurite formation, defects not seen in wild-type (Figure S4 and as discussed later). tiam-1(ok772) and tiam-1(tm1556) also displayed weak defects in VD/DD commissural motor neuron pathfinding (0.69% and 1.62%, respectively; p = 0.0051 for tiam-1(tm1556) but not significant for tiam-1(ok772)) (Figure S4).
These studies indicate that TIAM-1 displays a genetic interaction with the Rac GTPases CED-10 and MIG-2 and the UNC-73/Trio GEF in PDE axon pathfinding. That tiam-1 enhances mig-2 and ced-10 could indicate that TIAM-1 acts in parallel to these GTPases or that TIAM-1 acts in both of the redundant MIG-2 and CED-10 pathways in pathfinding. Studies below indicate that TIAM-1 acts with both MIG-2 and CED-10. However, the mig-2; ced-10 double mutant phenotype is much stronger than tiam-1, suggesting that another molecule might regulate MIG-2 and CED-1 in parallel to TIAM-1, possibly another GEF.
A tiam-1promoter::cfp transgene is expressed in neurons
To determine where and when the tiam-1 gene might by expressed, we studied expression of a tiam-1 transcriptional reporter transgene. We generated by PCR a 4793-bp region upstream of T21E12.2 (the entire upstream region to the next predicted gene, T21E12.3) (Figure 2A), and this putative tiam-1 promoter region was placed upstream of the cyan fluorescent protein (cfp) coding region. Expression of the tiam-1promoter::cfp construct was primarily in neurons (Figure 2B–2D). Most if not all neurons expressed the construct, including neurons in the head and tail, the ventral cord commissural motorneurons, the mechanosensory neurons (ALMs, PLMs, AVM, PVM) and the CAN, PDE, and PVD neurons. tiam-1promoter::cfp expression in neurons began in embryos and lasted throughout adulthood. No obvious expression outside of the nervous system was noted. However, tiam-1 might be endogenously expressed in other tissues, as some transcriptional elements might not be present in this transgene.
Fig. 2. The tiam-1 promoter drives expression in the nervous system. 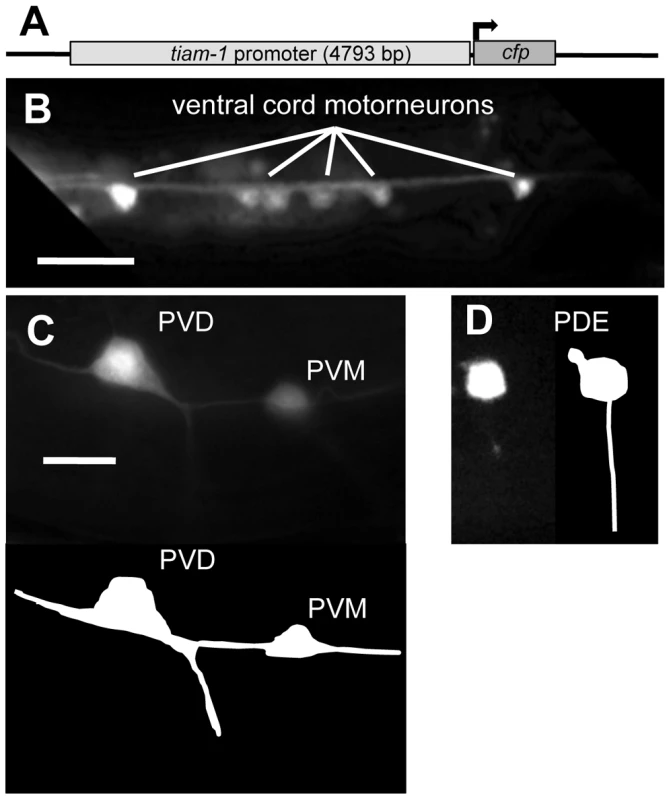
A) A diagram of the tiam-1 promoter::cfp transgene. The tiam-1 promoter included 4793 bp upstream of the tiam-1 A cDNA initiator codon. (B–D) Fluorescent micrographs of young adults harboring tiam-1p::cfp. In all micrographs, dorsal is up and anterior is to the left. B) tiam-1p::cfp expression in ventral cord motor neurons (ventral aspect). C) Expression in the post-deirid ganglion neurons PVD and PVM (lateral aspect) The diagram below depicts the positions of the neurons in the micrograph. D) Expression in the PDE neuron (lateral aspect), with a depiction to the right. The scale bar in (B) represents 10 µm, and the scale bar in (C) represents 2 µm for (C–D). TIAM-1 is a Rac1-specific GTP exchange factor
C. elegans TIAM-1 is similar to the Rac GEFs Tiam1 and Sif. We tested if TIAM-1 from C. elegans acts as a GTP exchange factor in vitro using the mant-GTP RhoGEF exchange assay (Cytoskeleton, Inc; see Methods) (Figure 3). The DH-PH region of TIAM-1 was purified from bacterial expression and tested for exchange activity against human Rac1, Cdc42, and RhoA. In the absence of TIAM-1[DHPH], none of the GTPases incorporated mant-GTP (fluorescence did not change over 30 minutes). Addition of TIAM-1[DHPH] increased fluorescence with Rac1 (Figure 3A), but not Cdc-42 nor RhoA (Figure 3B–3C). This result suggests that TIAM-1[DHPH] has Rac1-specific GTP exchange activity.
Fig. 3. The TIAM-1 DHPH domain is a Rac1-specific GTP exchange factor. 
Graphs plotting arbitrary fluorescence units (A.U.) over time in a mant-GTP exchange assay (Cytoskeleton, Inc.) (see Methods). Dark, cross-hatched points represent exchange activity of the GTPase after the addition of vehicle without the DHPH domain of TIAM-1. Light, diamond-shaped points represent exchange after the addition of the DHPH domain of TIAM-1 in vehicle. The dashed vertical line represents the time of addition of vehicle or vehicle plus TIAM-1 DHPH. A) Exchange activity on human Rac1. B) Exchange activity on human RhoA. C) Exchange activity of human Cdc-42. D) Exchange activity on human Rac1 of a TIAM-1 DHPH domain with the T516F point mutation predicted to eliminate Rac GEF activity (TIAM-1(DEAD)). Error bars represent standard deviations of three independent experiments which each had consistent results. The unc-73(rh40) S1216F missense mutation in the Rac-specific DH GEF domain of UNC-73 specifically abolishes Rac GEF activity [30]. We made an analogous mutation in TIAM-1[DHPH] (threonine 516 changed to phenylalanine (T516F) relative to the A isoform of TIAM-1 (Figure S2)). TIAM-1[DHPH] with the T516F mutation (TIAM-1(DEAD)) no longer showed exchange activity to Rac1 (Figure 3D), suggesting the T516F mutation abolished Rac1 GEF activity of TIAM-1[DHPH], similar to the unc-73(rh40) mutation S1216F. This mutation also abolished the ability of TIAM-1[DHPH] to induce ectopic protrusions in neurons as described below.
The TIAM-1 and UNC-73 Rac GEF domains induce ectopic protrusion from neurons
To test the roles of TIAM-1 and UNC-73 as potential Rac GEFs in vivo, we constructed transgenes that express constitutively-activated forms of the TIAM-1[DHPH] and UNC-73[DHPH1] Rac GEF domains in the PDE neurons. We based these experiments on previous results with Drosophila Trio (UNC-73 in C. elegans), in which a myristoylation sequence was added to the Rac-specific DHPH domain of Trio (Figure 4A). This construct produced a DHPH domain that was localized to cellular membranes via myristoylation [42]. Flies expressing this construct had retinal axon pathfinding defects due to unregulated activity of the DHPH domain. Mammalian Tiam1 has an N-terminal myristoylation site and is normally localized to membranes [38], [39], and TIAM-1 in C. elegans has a potential N-terminal myristoylation site (Figure S2). Furthermore, an N-terminal truncation of mammalian Tiam1, which does not affect the DHPH domains, activates Rac more strongly than full length Tiam1 [31], [43], suggesting that a deleted portion of Tiam1 might normally inhibit GEF activity of the molecule.
Fig. 4. Ectopic protrusions induced by overactive TIAM-1 and UNC-73 were suppressed by loss-of-function of Rac GTPases. 
(A) Diagram of membrane-tethered, Rac-specific MYR::DHPH domains of UNC-73 and TIAM-1 inducing ectopic protrusions. (B–C) Fluorescent micrographs of PDE neurons with ectopic lamellipodial and filopodial protrusions (arrows) induced by MYR::DHPH constructs. Fluorescence is from soluble GFP expressed from an osm-6::gfp co-injection marker for cell shape. Compare to the wild-type PDE neurons in Figure 1. In all micrographs, dorsal is up and anterior is to the left. The scale bar in (B) represents 5 µm for (B–C). D) A graph plotting percentage of ectopic lamellipodial protrusions from the PDE neurons of young adult animals harboring transgenes driving the expression of MYR::DHPH domains of UNC-73 and TIAM-1. MYR::TIAM-1[DHPH] contains the TIAM-1 DHPH region (lqIs165). MYR::UNC-73[DHPH1] (lqIs123) contains the Rac specific GEF domain of UNC-73. A second DH GEF domain of UNC-73 is Rho specific (MYR::UNC-73[DHPH2]) (lqIs125). UNC-73[DH(DEAD)PH1] (lqIs134) and TIAM-1[DH(DEAD)PH] (lqIs193) are transgenes that harbor the point mutations that eliminate Rac GEF activity of the DH domains of UNC-73 DH1 and TIAM-1 (S1216F and T516F, respectively). Unless otherwise noted, all backgrounds are wild-type. At least 100 PDE neurons for each genotype were scored, and p value significance was determined using Fisher's exact analysis. Error bars represent 2× standard error of the proportion in both directions. The asterisk “*” indicates transgenes that have been crossed away from the ced-10 or mig-2 mutations and re-scored in a wild-type background. Dashed lines indicate comparisons between genotypes not marked with a p value to those marked with p values. Based on these results, we created transgenes consisting of the TIAM-1 DHPH domain and both UNC-73 DHPH domains (Figure 4A) fused in frame to an N-terminal myristoylation sequence and to a C-terminal GFP (see Methods). Expression of these molecules in the PDE neurons was driven by the osm-6 promoter and was confirmed by DHPH::GFP fluorescence in the PDE and other osm-6-expressing neurons (data not shown). The MYR::DHPH::GFP fusion proteins displayed a punctate, peripheral accumulation pattern consistent with localization to cellular membranes as seen in other MYR::GFP fusion proteins [44].
In a wild-type background, the Rac-specific MYR::UNC-73[DHPH1] induced ectopic protrusions that emanated from the cell body and axons of PDEs (Figure 4B). MYR::TIAM-1[DHPH] also promoted ectopic lamellipodial and filopodial protrusions (Figure 4C). Axon branching and defects in pathfinding were also observed in each case (Figure 4B and 4C). We generated multiple independent extrachromosomal transgenic lines with each construct, and in every case saw robust lamellipodial protrusions (Figure S5). In Drosophila, the Rho GEF domain of Trio did not cause retinal axon pathfinding defects [42]. We found that the Rho-specific MYR::UNC-73[DHPH2] of UNC-73 caused a low level of ectopic PDE protrusions in multiple transgenic extrachromosomal lines generated (Figure S5 and Figure 4D), significantly lower than the UNC-73 Rac-specific domain and MYR::TIAM-1DHPH (p≤0.001), consistent with the Drosophila studies.
To test the requirement of Rac GEF activity in ectopic protrusion, we introduced point mutations into the transgenes that abolish the Rac GEF activities of UNC-73 and TIAM-1 (the unc-73(rh40) S1216F mutation [30] and the T516F mutation in TIAM-1 used in Figure 3). The “GEF-dead” constructs were expressed as determined by GFP fluorescence. Multiple transgenic extrachromosomal lines were produced for each, and none promoted high levels of ectopic lamellipodial protrusions (Figure S5 and Figure 4D) (p≤0.001 compared to non-mutant transgenes). These results suggest that Rac GEF activity was required for robust ectopic lamellipodial and filopodial induction. These results also indicate that the MYR::[DHPH] transgenes likely produced constitutively activated GEF molecules rather than dominant negative molecules. We assayed the effects of MYR::TIAM-1[DHPH] in the tiam-1(tm1556) background and observed ectopic protrusions similar to the wild-type background (data not shown), consistent with the idea that MYR::TIAM-1DHPH represents a constitutively-activated rather than a dominant negative molecule.
CED-10/Rac and MIG-2/RhoG are required for ectopic protrusions induced by activated TIAM-1 and UNC-73 GEF domains
To determine if Rac function was required for the effects of MYR::[DHPH] constructs, we crossed loss-of-function mig-2 and ced-10 with these overactive MYR::[DHPH] constructs to test for suppression of the ectopic protrusion phenotype. For these studies, we created integrated lines of the extrachromosomal transgenes described in Figure S5 (see Methods). In these experiments, we outcrossed the integrated transgene away from the loss-of-function mutation and re-scored the transgene to ensure that it retained the ability to induce ectopic protrusions (asterisks in Figure 4D). Both mig-2(mu28) and ced-10(n1993) significantly suppressed the ectopic projections caused by MYR::TIAM-1[DHPH] and MYR::UNC-73[DHPH1] (Figure 4D). mig-2(mu28) suppressed the ectopic lamellipodia and filopodia caused by MYR::UNC-73[DHPH1] (96% to 44%, p<0.001) and MYR::TIAM-1[DHPH] (76% to 23%, p<0.001). ced-10(n1993) suppressed MYR::TIAM-1[DHPH] strongly (48% to 13%, p<0.001) and MYR::UNC-73[DHPH1] more weakly but significantly (93% to 79%, p = 0.015). The same integrated MYR::TIAM-1[DHPH] transgene (lqIs165) was used in both the mig-2 and ced-10 double mutants, yet upon outcrossing, the transgene showed significantly different effects (p<0.01). We do not know the nature of this variability, but it might be due to effects of different genetic backgrounds after outcrossing. Regardless, in both cases the effects increased significantly upon outcrossing from ced-10 and mig-2. We also scored integrated lines of the GEF dead constructs and the Rho-specific UNC-73 DHPH2 domain (Figure 4D), and found significantly reduced protrusions, consistent with results with extrachromosomal transgenes in Figure S5. These data indicate that unregulated Rac GEF activity of UNC-73 and TIAM-1 in the PDE neurons led to ectopic lamellipodia and filopodia that were dependent on CED-10/Rac1 and MIG-2/RhoG function. These results are consistent with the GTP exchange assays in Figure 3 and indicate that TIAM-1 interacts with CED-10/Rac1 and MIG-2/RhoG in vivo.
TIAM-1 and Rac GTPases co-localize in C. elegans neurons
To determine the subcellular localization of TIAM-1 in neurons, we fused the longest isoform A tiam-1 cDNA (yk730h9 in Figure S1 and S2) with mCherry at the C-terminus, and placed this construct under the control of the unc-25 promoter (for VD/DD motor neurons) and the osm-6 promoter (for PDE expression). The yk730h9 cDNA contained two errors relative to the reference gene models that led to frame shifts. These were repaired before proceeding (see Methods and Figure S2). This construct likely produced a functional TIAM-1::mCherry molecule, because it was required for ectopic protrusion induced by activated CDC-42(G12V) in the PDE neurons (see below and Figure 6), and it induced ectopic protrusions on its own (Figure 5A and 5B).
Fig. 5. TIAM-1::mCherry is present in ectopic protrusions and growth cones and co-localizes with GFP::MIG-2 and GFP::CED-10. 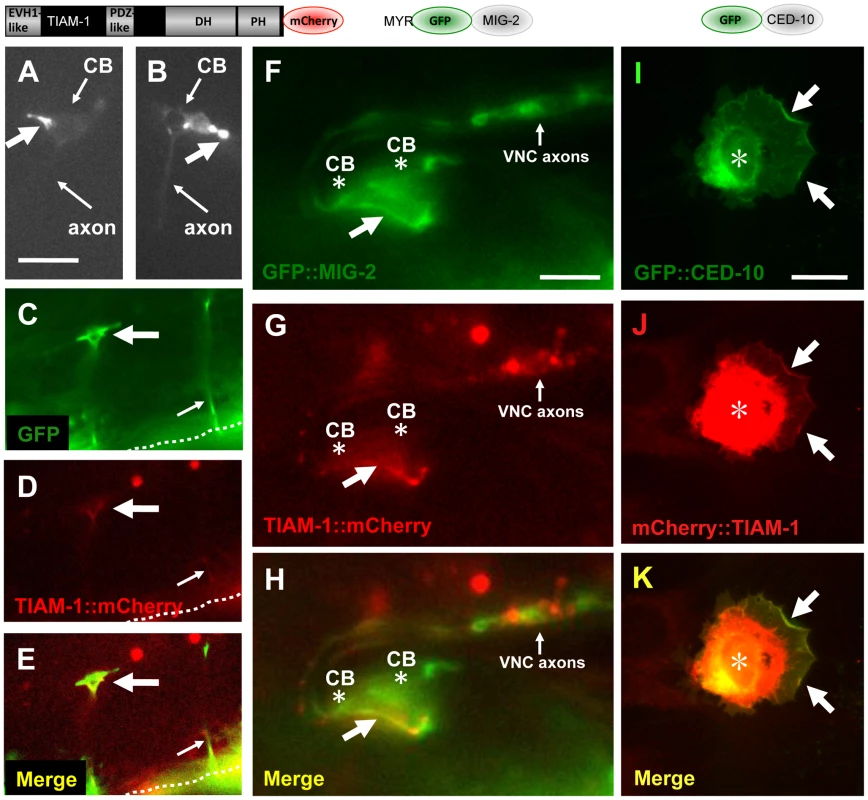
The diagrams above the micrographs represent the fusion proteins produced by the transgenes used: TIAM-1 C-terminally tagged with mCherry; MIG-2 N-terminally tagged with GFP after the predicted myristoylation site; and CED-10 N-terminally tagged with GFP. For the NIH 3T3 studies (I–K), an N-terminally-tagged TIAM-1 was assayed. Panels are fluorescent micrographs of animals harboring transgenes that express TIAM-1::mCherry, GFP, or GFP::MIG-2. In (A–H), dorsal is up and anterior is to the left. (A and B) Micrographs of PDE neurons of animals carrying an osm-6p::tiam-1::mCherry transgene expressed in the PDE neurons. TIAM-1::mCherry accumulated in ectopic protrusions that occurred at a low frequency (large arrows). Small arrows point to the cell bodies (CB) and the axons, which are faint. (C–E) A VD growth cone (large arrow) in an early L2 animal 18–20 hours post-hatching. The growth cone was visualized using cytoplasmic unc-25p::gfp, and also showed TIAM-1::mCherry accumulation driven from the unc-25 promoter. The dashed line indicates the ventral nerve cord, and the small arrow points to a commissural motor axon that has completed commissural extension. The scale bar in (A) represents 5 µm for (A–E). (F–H) TIAM-1::mCherry and GFP::MIG-2 colocalized in ectopic protrusions. A ventral aspect of the ventral nerve cord is shown. (F) unc-25p::gfp::mig-2 expression in an ectopic protrusion from VD or DD motor neurons (arrow). Cell bodies are indicated (CB, asterisk), as is the ventral cord neuropil (VNC axons). (G) unc-25p::tiam-1::mCherry expression in the ectopic protrusion of the same VD/DD neuron. (H) Co-localization of GFP::MIG-2 and TIAM-1::mCherry in the ectopic protrusion from the VD/DD neuron. The scale bar in (F) represents 3 µm for (F–H). (I–K) CED-10::GFP and TIAM-1::mCherry expression in NIH 3T3 cells. (I) GFP::CED-10 accumulated in membranous regions around the nucleus (asterisk), as well as at the periphery of lamellipodial ruffles induced by GFP::CED-10 (arrows). (J–K) TIAM-1::mCherry co-localized with GFP::CED-10 at the periphery of CED-10::GFP-induced lamellipodial ruffles (arrows). mCherry::TIAM-1 also accumulated to a perinuclear region that was more widespread than GFP::CED-10 accumulation (asterisk). The scale bar in (I) represents 10 µm for (I–K). In the PDE and VD/DD neurons, TIAM-1::mCherry localized to puncta primarily at the periphery of the cell bodies, consistent with plasma membrane localization, as well as in the axons (Figure S6). Consistent with this observation, mammalian Tiam1 associates with membranes [38]. We noted that full-length TIAM-1::mCherry produced a low percentage (5%) of PDE and VD/DD neurons with ectopic neurites and protrusions (Figure 5A and 5B), similar to the TIAM-1 MYR::DHPH construct, suggesting that this molecule is active and drives ectopic protrusions. This is also consistent with the low level of PDE axon pathfinding defects observed upon tiam-1(+) expression in Figure 1A. TIAM-1::mCherry accumulated strongly in these ectopic protrusions (Figure 5A and 5B). TIAM-1::mCherry also accumulated in the growth cones of developing VD axons (Figure 5C–5E), consistent with a role of TIAM-1 in the growth cone during outgrowth.
We constructed mig-2/RhoG and ced-10/Rac1 transgenes tagged with gfp driven by the unc-25 promoter. MIG-2 contains an N-terminal myristoylation site and a C-terminal CAAX prenylation site. We placed gfp immediately after the Myr sequence in frame with mig-2, such that both myristoylated and prenylated forms of MIG-2 could be tagged with GFP. CED-10 was N-terminally tagged with GFP. GFP::CED-10 and GFP::MIG-2 accumulated at the cell periphery as previously described [28], [45] (data not shown). GFP::MIG-2 and TIAM-1::mCherry co-localized to the periphery of the ectopic protrusions of VD/DD neurons induced by expression of these molecules (Figure 5F–5H). GFP::CED-10 also co-localized with TIAM-1::mCherry (data not shown). Similar to TIAM-1::mCherry, GFP::CED-10 and GFP::MIG-2 were also found in the VD growth cones (data not shown). While TIAM-1::mCherry and GFP::MIG-2 and GFP::CED-10 showed co-localization, it was not completely overlapping co-localization (compare GFP in Figure 5F to mCherry in Figure 5G). This suggests that TIAM-1 and the GTPases MIG-2 and CED-10 might interact in specific regions in the cell (e.g. ectopic protrusions at the cell periphery).
TIAM-1::mCherry and GFP::CED-10 co-localized to the plasma membrane in NIH-3T3 cultured mammalian fibroblasts
We analyzed the localization of N-terminally tagged mCherry::TIAM-1 and GFP::CED-10 in cultured NIH-3T3 fibroblasts. As expected, expression of GFP::CED-10 in fibroblasts caused marginal lamellipodial ruffling, a phenotype canonically associated with expression of vertebrate Rac1 in these cells (Figure 5I and Figure S7). GFP::CED-10 localized to intracellular regions around the nucleus that might represent membranous organelles, as well as to the plasma membrane, particularly in lamellipodial regions (Figure 5I and Figure S7A). Expression of mCherry::TIAM-1 did not affect cell shape as did GFP::CED-10, but did accumulate at the cell margins and intracellular membranous organelles (Figure S7B). These patterns are likely to represent CED-10 and TIAM-1 specific localization, as mCherry and GFP expression alone did not show these patterns (Figure S7C and S7D).
When expressed together, mCherry::TIAM-1 and GFP::CED-10 co-localized to the lamellipodial regions of the plasma membrane induced by GFP::CED-10 (Figure 5I–5K). Both molecules also localized to a perinuclear region, but overlap was not complete (TIAM-1::mCherry was more widely distributed in these perinuclear regions than was GFP::CED-10; compare Figure 5I and 5J). Similar to the results in C. elegans, these data indicate that TIAM-1 and CED-10/Rac1 might interact in specific regions of the cell. In 3T3 cells, they co-localize where CED-10/Rac1 induces lamellipodial ruffles at the cell periphery (Figure 5K). Possibly, TIAM-1 and the GTPases MIG-2 and CED-10 co-localize only where TIAM-1 is activating them (i.e. acting as a Rac GEF).
TIAM-1 acts cell-autonomously downstream of CDC-42
Evidence from cell and neuronal culture studies [46], [47] suggests mammalian Tiam1 acts downstream of Cdc-42 in the activation of Rac1 in axon development. We tested if TIAM-1 acts downstream of CDC-42 in C. elegans. A glycine-12-valine substitution in the GTPase binding pocket of small GTPases has been shown to favor the GTP-bound state of these molecules, constitutively activating them [41]. Previous studies have shown that Rac(G12V) activity (CED-10/Rac and MIG-2/RhoG) in C. elegans neurons induces ectopic lamellipodia and filopodia, similar to those described for MYR::TIAM-1(DHPH) (Figure 4) [41]. We constructed a CDC-42(G12V) transgene driven by the osm-6 promoter in the PDE neurons. Transgenic animals harboring cdc-42(G12V) displayed ectopic lamellipodial and filopodial protrusions (44%) (Figure 6A), as well as disruption of proper axon pathfinding (50%; n = 100) in the PDE neurons, similar to Rac(G12V) and MYR::TIAM-1(DHPH). Thus, unregulated CDC-42 activity promotes lamellipodial and filopodial formation in neurons. We generated multiple cdc-42(G12V) transgenic lines, and each induced ectopic protrusions. We used one representative line for further studies.
Fig. 6. TIAM-1 acts cell autonomously downstream of CDC-42. 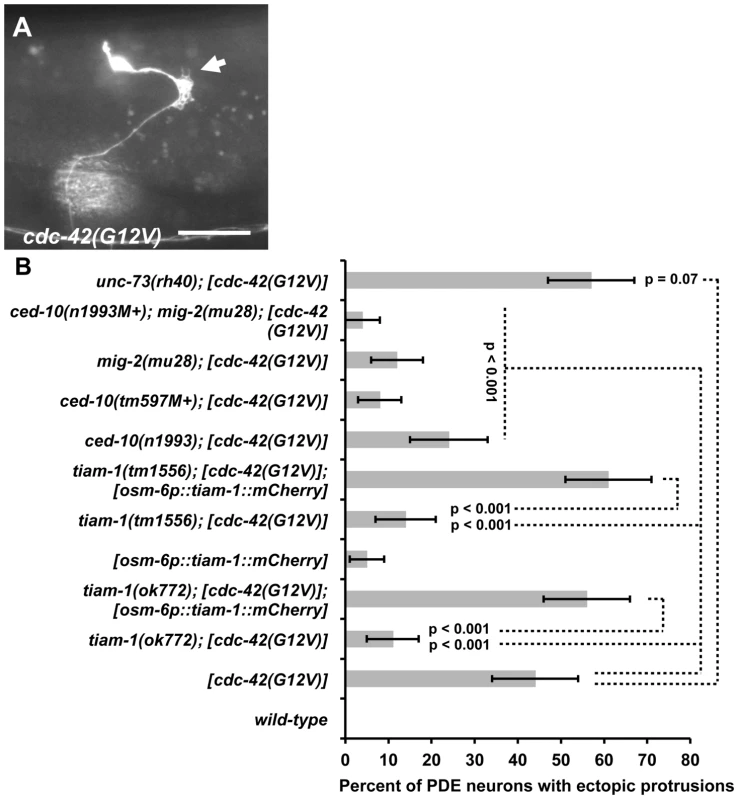
(A) Fluorescent micrograph of a PDE neuron from an animal with an integrated cdc-42(G12V) transgene expressed with the osm-6 promoter. An osm-6::gfp marker transgene was included to label the PDE cell body and processes. Dorsal is up and anterior is to the left. An arrow points to an ectopic lamellipodial protrusion. The scale bar in represents 5 µm. (B) A graph charting the percentage of PDE axons with ectopic lamellipodial and filopodial protrusions (X axis) in different genotypes (Y axis). Unless otherwise noted, all backgrounds are wild type. M+ indicates that the genotype had wild-type maternal gene function. [cdc-42(G12V)] indicates a transgene that harbors activated cdc-42(G12V) driven by the osm-6 promoter in the PDEs. [osm-6p::tiam-1(+)::mCherry] indicates a transgene that harbors the corrected tiam-1 cDNA yk730h9 fused in frame to mCherry and driven by the osm-6 promoter in the PDE neurons (see Methods and Figure S2). At least 100 PDE neurons were scored for each genotype, and p value significance was determined using Fisher's Exact analysis. Error bars represent 2× standard error of the proportion in both directions. Dashed lines indicate comparisons between genotypes not marked with a p value to those marked with p values. To test if TIAM-1 was required for the effects of CDC-42(G12V), we assayed the effects of cdc-42(G12V) in tiam-1(ok772) and tiam-1(tm1556) mutants. In both cases, the ectopic lamellipodia and filopodia induced by CDC-42(G12V) were significantly suppressed (12% for ok772 and 15% for tm1556, p<0.0001) (Figure 6B). These data suggest that TIAM-1 is required for the activity of CDC-42(G12V). In contrast, unc-73(rh40) did not suppress CDC-42(G12V) (47% ectopic lamellipodia and filopodia in unc-73(rh40); cdc-42(G12V) compared to 44% in cdc-42(G12V) alone; p = 0.07)). These data suggest that TIAM-1, but not UNC-73, is required for the effects of CDC-42(G12V).
osm-6p::tiam-1::mCherry, which produces full-length TIAM-1 tagged with mCherry in the PDE neurons but not surrounding cells, restored the effects of CDC-42(G12V) in tiam-1(ok772) and tiam-1(tm1556) mutants (Figure 6B) (p<0.001 in each case). These results indicate that TIAM-1 is required cell autonomously for the ectopic lamellipodia and filopodia induced by CDC-42(G12V) in the PDE neurons. A genomic fosmid containing tiam-1 with its endogenous promoter also restored CDC-42(G12V)-induced ectopic lamellipodia and filopodia (Figure S8).
With osm-6p::tiam-1::mCherry rescue, the effects of CDC-42(G12V) were consistently enhanced compared to CDC-42(G12V) alone (p = 0.079 for ok772 rescue, and p = 0.014 for tm1556 rescue). This was also the case with fosmid rescue (p = 0.106 for ok772 rescue, and p = 0.02 for tm1556 rescue). Possibly, the excess TIAM-1 expressed from the transgene is activated by CDC-42(G12V), leading to increased defects. However, TIAM-1 expression alone causes ectopic protrusions (5%; Figure 6B), possibly explaining the increase.
These findings were corroborated with loss-of-function studies. The cdc-42(gk388) deletion removes the initiator codon and a large portion of the coding region and is likely a null allele (Wormbase). cdc-42(gk388M+) homozygotes, with wild-type maternal gene function, survived until late larval/early adulthood and were sterile. Alone, cdc-42(gk388M+) animals display 15% PDE axon pathfinding defects (Figure 7). tiam-1(ok772); cdc-42(gk388M+) animals showed no increase in pathfinding defects and resembled cdc-42(gk388M+) alone. However, unc-73(rh40); cdc-42(gk388M+) mutants displayed a synergistic increase in the number of PDE pathfinding defects (90%) compared to cdc-42(gk388M+) and unc-73(rh40) alone (p<0.001) (Figure 7). These loss-of function studies are consistent with the idea that TIAM-1 and CDC-42 act in the same pathway in PDE axon guidance, possibly in parallel to UNC-73/Trio.
Fig. 7. tiam-1 and cdc-42 interactions with axon guidance mutations. 
A graph representing PDE axon pathfinding defects (X axis) in different genotypes (Y axis). At least 100 PDE neurons were scored, and p value significance was determined by Fisher's exact analysis. The error bars represent 2× standard error of the proportion in each direction. M+ indicates that the genotype had wild-type maternal gene function. Dashed lines indicate comparisons between genotypes not marked with a p value to those marked with p values. CED-10/Rac1 and MIG-2/RhoG are required for the effects of CDC-42(G12V)
Our results suggest that TIAM-1 acts downstream of CDC-42 in ectopic lamellipodial protrusion. Given that TIAM-1 can act as a Rac GEF, we expect that MIG-2/RhoG and CED-10/Rac1 might also be required for the effects of CDC-42(G12V). Indeed, mig-2(mu28) and ced-10(n1993) suppressed the effects of CDC-42(G12V) (Figure 6B; p<0.001). The ced-10(tm597M+) deletion allele also suppressed CDC-42(G12V) (p<0.001). Suppression by ced-10(n1993) was significantly weaker than that of mig-2(mu28) (24% compared to 12%; p = 0.022) and ced-10(tm597M+) (8%; p = 0.001). ced-10(n1993) is a mutation in the C-terminal prenylation sequence and is a hypomorph, consistent with weaker suppression [48]. The ced-10(n1993M+); mig-2(mu28) double mutant suppressed (4%, p<0.001) slightly better than mig-2(mu28) or ced-10(tm597M+) alone (p = 0.029 and 0.0194, respectively). These data indicate that CED-10/Rac1 and MIG-2/RhoG are required for the ectopic protrusions caused by CDC-42(G12V).
TIAM-1 might act in the UNC-6/Netrin pathway in ventral axon guidance
We have shown evidence that the Rac GEF TIAM-1 regulates the Rac GTPases CED-10 and MIG-2 in response to CDC-42 signaling. We next sought to determine which dorsal-ventral axon guidance ligand/receptor pathway might be upstream of TIAM-1. Distinct parallel ventral guidance pathways control dorsal ventral guidance in C. elegans. UNC-6/Netrin is a secreted ligand expressed in the ventral side of the animal that attracts axons that express its receptor UNC-40/DCC [49]. SLT-1/Slit is expressed primarily in the dorsal side and is thought to act as a repellent for ventrally-guided axons that express its receptor SAX-3/Robo [50]. Ephrins have also been involved in directing proper axon pathfinding [51]. VAB-1/EphR is the only C. elegans Ephrin receptor [52] and has been shown to work in parallel with UNC-6/Netrin and SLT-1/Slit in axon ventral guidance [53].
We constructed double mutants of tiam-1 with the loss-of-function alleles unc-6(ev400), slt-1(eh15), and vab-1(dx31), and scored defects in PDE axon ventral guidance (Figure 7). tiam-1(ok772) significantly increased the defects of vab-1(dx31) (4% to 46%, p<0.001), and tiam-1(tm1556) enhanced slt-1(eh15) (11% to 22%, p = 0.032), suggesting that TIAM-1 might act in parallel to VAB-1/Eph and SLT-1/Slit. In contrast, neither tiam-1(ok772) nor tiam-1(tm1556) increased the amount of PDE pathfinding defects of unc-6(ev400) (from 59% to 56% and 50%; p = 0.622 and 0.256). slt-1(eh15) is a small deletion and is likely a null [50], an vab-1(dx31) is a complex rearrangement that deletes exons 1–4 and is also likely a null [52]. That tiam-1 enhanced slt-1 and vab-1 but did not enhance unc-6 is consistent with TIAM-1 acting in the UNC-6/Netrin pathway in parallel to VAB-1 and SLT-1 in PDE ventral guidance.
TIAM-1 might act downstream of UNC-40/DCC
Previous studies indicated that the Rac GTPase CED-10 acts downstream of the UNC-6/Netrin receptor UNC-40/DCC in the AVM neuron [9]. A myristoylated form of the cytoplasmic tail of the Netrin receptor UNC-40 missing the extracellular and transmembrane domains induced axon defects and ectopic lamellipodia and filopodia in the AVM neurons due to constitutive activation of UNC-40 signaling (see diagram in Figure 8A) [9]. Loss of ced-10/Rac1 function suppressed myr::unc-40, but loss of unc-73/Trio did not [9], suggesting a distinct GEF might act downstream of UNC-40.
Fig. 8. TIAM-1 acts with UNC-6/netrin and UNC-40/DCC in axon guidance and protrusion. 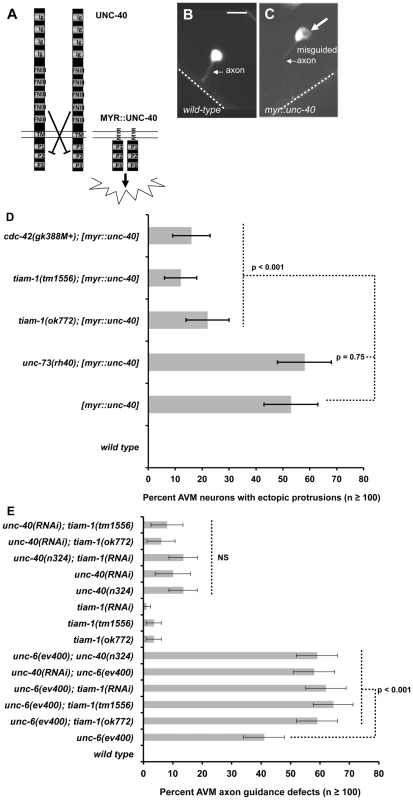
(A) A diagram illustrating attractive axon guidance via the UNC-40/DCC receptor. In the absence of UNC-6/Netrin ligand, the extracellular domains of UNC-40 prevent dimerization of the cytoplasmic regions and activation of receptor signal transduction. A myristoylated version of the cytoplasmic domain lacking the transmembrane domain and extracellular domains is believed to constitutively dimerize and is constitutively active, resulting in ectopic protrusion from the AVM neuron. Ig = immunoglobulin domain; FNIII = fibronectin type II domain; TM = transmembrane domain; P1–P3 = conserved proline-rich domains; MYR = covalent myristoyl group. (B) A wild-type AVM neuron visualized with soluble GFP from a mec-4::gfp transgene, with an arrow pointing to the unbranched ventrally-directed axon. The dashed line indicates the ventral nerve cord. (C) An AVM neuron with expression of unc-86p::myr::unc-40, a transgene that drives a myristoylated version of the cytoplasmic domain of UNC-40 in AVM. Fluorescence is soluble GFP expressed from mec-4::gfp to highlight cell shape. A large arrow points to an ectopic lamellipodium, and a smaller arrow points to a misguided and branched AVM axon. In all micrographs, dorsal is up and anterior is to the left. The scale bar represents 5 µm for B and C. (D) A graph charting the percentage of AVM axons with ectopic lamellipodial protrusions (X axis) in different genotypes (Y axis). Unless otherwise noted, all backgrounds are wild type. [myr::unc-40] represents animals harboring a transgene that expresses the myristoylated UNC-40 cytoplasmic domain under the mec-7 promoter (mec-7p::myr::unc-40). M+ indicates wild-type maternal gene function. Alone, none of the single mutants tiam-1(ok772), cdc-42(gk388M+), or unc-73(rh40) displayed ectopic protrusions. (E) A graph plotting the percentage of animals with AVM ventral axon guidance defects in different genotypes. Reported here are defects in AVM guidance that include failure of the axon tom reach the ventral nerve cord and axon wandering. If we just consider failure to reach the ventral nerve cord, similar significant results are found (unc-40 and tiam-1 enhance unc-6(ev400), and tiam-1 does not enhance unc-40). At least 100 AVM neurons were scored for each genotype, and p value significance was determined using Fisher's Exact analysis. Error bars represent 2× standard error of the proportion in both directions. Dashed lines indicate comparisons between genotypes not marked with a p value to those marked with p values. NS = not significantly different. We tested if TIAM-1 might act downstream of MYR::UNC-40 in AVM. When expressed in the PDE neurons, MYR::UNC-40 did not result in ectopic lamellipodial protrusions. We do not know why this is the case, but possibly the PDE neuron is less sensitive to the effects of MYR::UNC-40 than is AVM, or the osm-6 promoter is not expressed at the correct time or level. We expressed MYR::UNC-40 in the AVMs using the mec-7 promoter as previously described [9]. In our hands, MYR::UNC-40 caused 53% of AVMs to form ectopic lamellipodial protrusions (Figure 8B, 8C, and 8D). As expected, unc-73(rh40) did not suppress (Figure 8D; 58%), confirming the results described previously [9]. However, tiam-1(ok772) significantly suppressed the ectopic lamellipodia incidence in these neurons (22%, p<0.001). Furthermore, cdc-42(gk388M+) suppressed MYR::UNC-40 (16%, p<0.001), suggesting that CDC-42 is also downstream of UNC-40. Consistent with this idea, cdc-42(gk388M+) did not enhance unc-6(ev400) in PDE ventral axon guidance (Figure 7). In fact, cdc-42(gk388M+) slightly suppressed unc-6(ev400) (p = 0.027). The nature of this possible suppression is unclear, but it is possible that cdc-42(gk388M+) might also normally attenuate UNC-6/Netrin signaling. While tiam-1 loss of function suppressed the ectopic protrusions driven by MYR::UNC-40, the axon guidance and branching defects were not suppressed significantly (data not shown), suggesting that MYR::UNC-40 perturbation of axon guidance and branching are not dependent upon TIAM-1 and might be due to effects distinct from ectopic lamellipodial protrusion.
We assayed AVM axon guidance in single and double loss-of-function mutants involving unc-6, unc-40, and tiam-1 (Figure 8E). unc-6(ev400) and unc-40(n324) both displayed defective AVM ventral guidance as expected, but unc-6(ev400) was significantly stronger than unc-40(n324) (41% versus 14%; p<0.001). Both are predicted null mutations, suggesting that UNC-40 might not be the only receptor for UNC-6 in AVM ventral guidance. AVM defects in unc-6(ev400) mutants were scored previously by another group [54], and fewer defects were noted in this study (∼25% compared to 41% in our hands). We scored AVM guidance as defective if the axon failed to reach the ventral cord or if the axon wandered more than 45 degrees away from straight ventrally. When we consider only axons that failed to reach the ventral nerve cord, we see 27% defects in unc-6(ev400) (data not shown), similar to those previously described [54].
We found that tiam-1(tm1556), tiam-1(ok772), and tiam-1(RNAi) significantly enhanced the AVM ventral guidance defects of unc-6(ev400) (Figure 8E). This result is in contrast to the PDE, where tiam-1 did not enhance unc-6 (Figure 8D), suggesting cell-specific differences in TIAM-1 function with UNC-6. While tiam-1 enhanced unc-6, we found that tiam-1 did not enhance unc-40. For these studies we used RNAi, as tiam-1 and unc-40 are closely linked and we were unable to construct a double mutant. unc-40(RNAi) caused defects in AVM guidance similar to unc-40(n324) that were not enhanced by tiam-1 mutations; and tiam-1(RNAi), which enhanced unc-6(ev400), did not enhance unc-40(n324) (Figure 8E). These data are consistent with TIAM-1 acting in the UNC-40/DCC pathway. In further support of this notion, we found that unc-40(n324) and unc-40(RNAi) enhanced unc-6(ev400) in a manner similar to tiam-1. Enhancement of unc-6(ev400) by tiam-1 and unc-40 was also significant when only failure to reach the ventral nerve cord was considered (data not shown). Previous results showed that unc-40(e1430); unc-6(ev400) doubles had only slightly increased AVM guidance defects (∼25% to ∼33%) [54]. We used unc-40(n324) and unc-40(RNAi) and the previous study used unc-40(e1430) [54], raising the possibility that the stronger enhancement of unc-6(ev400) that we report is due to different unc-40 alleles used.
In sum, these genetic interactions suggest that UNC-6 and UNC-40 act partially in parallel and also suggest that UNC-6 might not be the only ligand for UNC-40/DCC in AVM ventral guidance. They are also consistent with TIAM-1 acting in the UNC-40 pathway in AVM ventral axon guidance. That unc-40 has a more severe phenotype than tiam-1 suggests that additional molecules might act in parallel to TIAM-1 downstream of UNC-40.
Discussion
In recent years, attention has focused on how guidance receptors engage cytoplasmic signaling mechanisms to control growth cone outgrowth and turning during axon guidance. In this report we present evidence of a signaling pathway (Figure 9A) involving the previously uncharacterized C. elegans molecule TIAM-1, which acts specifically as a GEF for Rac GTPases and interacts with them in axon guidance and neuronal protrusion. We show that TIAM-1 regulates neuronal protrusion downstream of the CDC-42 GTPase and upstream of the two redundant Rac GTPases MIG-2/RhoG and CED-10/Rac1. Genetic interactions of these molecules suggest that that they act in a common pathway in axon pathfinding. Furthermore, we provide evidence that TIAM-1 acts with the UNC-6/Netrin receptor UNC-40/DCC in axon guidance and downstream of UNC-40/DCC in neuronal protrusion. Previous studies have implicated Rac GTPases and the Rac GEF Trio as players in Netrin signaling and interaction with the Netrin receptor DCC [55]–[57]. Here we present evidence that the Rac GEF TIAM-1, along with CDC-42, CED-10/Rac1, and MIG-2/RhoG, acts downstream of UNC-40/DCC in the formation of neuronal protrusions, which might reflect a role in protrusive activity in the growth cone during outgrowth.
Fig. 9. A model of the distinct roles of the Rac GEFs TIAM-1 and UNC-73/Trio in axon guidance. 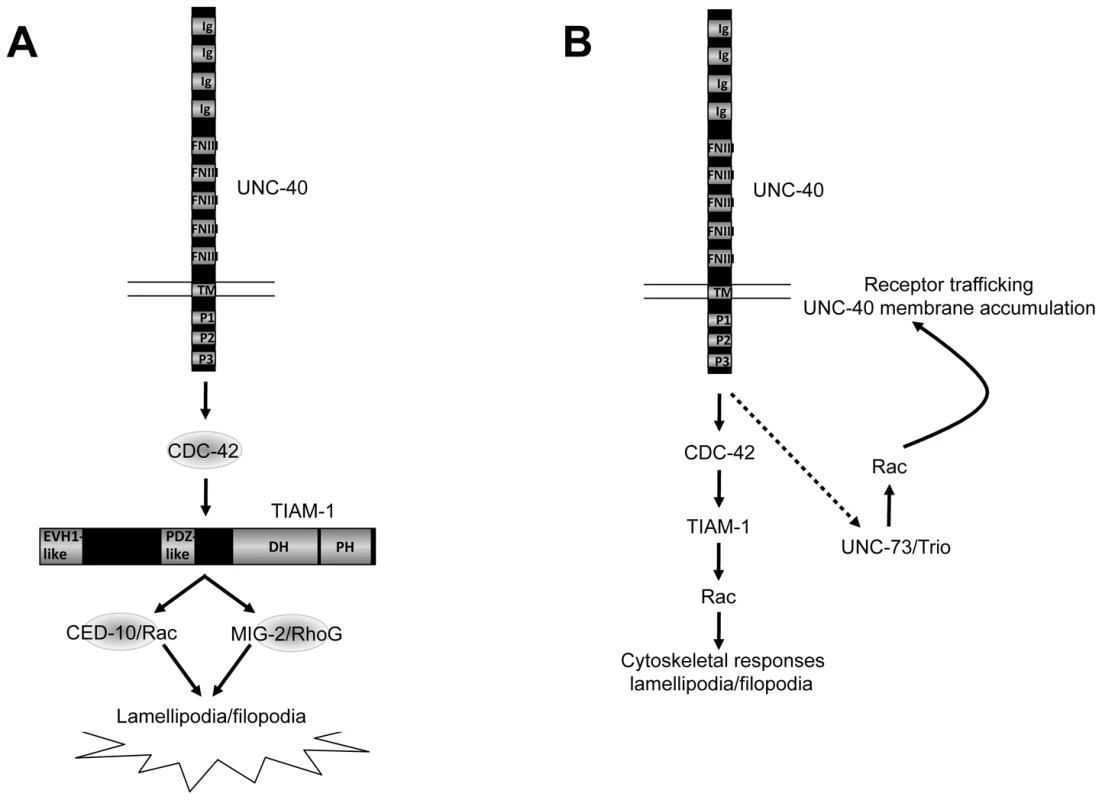
Arrows represent functional or genetic interactions between molecules and do not imply physical interactions. A) TIAM-1 controls protrusive events downstream of UNC-40/DCC. Results presented here suggest that TIAM-1 acts downstream of UNC-40/DCC and CDC-42, and upstream of the Rac GTPases CED-10 and MIG-2, in ectopic lamellipodial and filopodia formation and attractive axon guidance. B) TIAM-1 and UNC-73/Trio might regulate distinct aspects of Rac function in UNC-40/DCC-mediated axon guidance. TIAM-1 might regulate Rac GTPases downstream of UNC-40/DCC involving cytoskeletal protrusive events. UNC-73/Trio might control Rac GTPases upstream of UNC-40/DCC in trafficking of UNC-40/DCC to the membrane [55], [62]. The dashed arrow indicates a potential feedback loop where by UNC-40/DCC might control its own membrane trafficking via UNC-73. Ig = immunoglobulin I domain; FNIII = fibronectin type III domain; TM = transmembrane domain; P1–P3 = conserved proline-rich domains; EVH1 = EVH1 domain; PDZ = PDZ domain; DH = Dbl-homology domain; PH = Pleckstrin homology domain. TIAM-1 links CDC-42 and Rac GTPases in neuronal protrusion downstream of UNC-40/DCC
GTP exchange assays indicated that TIAM-1 is a Rac-specific GEF (Figure 3). The activated Rac-specific MYR::DHPH domains of TIAM-1 and UNC-73 induced ectopic protrusions from PDE neurons that resembled lamellipodia and filopodia. Protrusions were dependent upon a functional DH GEF domain in these molecules as well as on the presence of MIG-2/RhoG and CED-10/Rac1. TIAM-1 was required for ectopic protrusions induced by CDC-42 and activated MYR::UNC-40; and CDC-42 was required for MYR::UNC-40-induced protrusions. Together, these data suggest a pathway of neuronal lamellipodial and filopodial protrusion wherein TIAM-1 links CDC-42 and Rac GTPase signaling downstream of UNC-40/DCC.
A caveat with the studies using DHPH Rac GEF domains of TIAM-1 and UNC-73 is that the GEF domains were not in the context of the full-length molecules, and thus these MYR::DHPH constructs might not reflect the normal cellular roles of these molecules in neurons. These studies do indicate, however, that TIAM-1 and UNC-73 have the ability to act as Rac GEFs in vivo, whether or not their normal role is in protrusion. However, we found that transgenic expression of full-length TIAM-1::mCherry also induced ectopic protrusions in the PDE neurons similar to but at a much lower level than the MYR::DHPH construct, suggesting that regulation of protrusion is a normal cellular role of TIAM-1. This is consistent with tiam-1 suppression of protrusion caused by CDC-42(G12V) and MYR::UNC-40. In contrast, unc-73 suppressed neither of these, suggesting that UNC-73/Trio might not normally regulate protrusive events downstream of UNC-40. We speculate that UNC-73 might control Rac activity upstream of guidance receptors to control their cell surface trafficking (Figure 9B and see below).
TIAM-1::mCherry co-localized with GFP::MIG-2 in ectopic protrusions from C. elegans neurons, and with GFP::CED-10 in lamellipodial ruffles induced by GFP::CED-10 in cultured NIH 3T3 fibroblasts. That these molecules did not co-localize in other regions of the cell suggests that TIAM-1 associates with CED-10/Rac1 and MIG-2/RhoG only where they are actively inducing protrusion.
TIAM-1 displayed genetic interactions with the Rac GTPases in PDE axon guidance. tiam-1 mutations enhanced mutations in mig-2 and ced-10, which have redundant roles in axon guidance [15], [24], [28], [41]. Thus, TIAM-1 might act with both molecules. This idea is supported by gain-of-function studies showing that both MIG-2 and CED-10 are required for the ectopic protrusions induced by the activated MYR::DHPH domain of TIAM-1. TIAM-1 might act in parallel to another molecule, possibly another GEF, that regulates Rac GTPases in axon guidance, as the mig-2; ced-10 double mutant phenotype is more severe than tiam-1.
Genetic interactions suggest that TIAM-1 acts in the UNC-40/DCC axon guidance pathway
The UNC-6/Netrin ligand is expressed in ventral cells, and growth cones are attracted to or repelled from the ventral source [58]. A series of loss-of-function genetic interactions suggest that CDC-42, TIAM-1, MIG-2/RhoG, and CED-10/Rac1 act in the UNC-40/DCC axon guidance pathway. In PDE axon guidance (Figure 8D), tiam-1 enhanced slt-1/Slit and vab-1/EphR but not unc-6/Netrin. In AVM axon guidance (Figure 8E), tiam-1 enhanced unc-6, suggesting cell specific differences in the roles of these molecules in axon guidance. However, tiam-1 did not enhance unc-40 in AVM axon guidance, suggesting that TIAM-1 might act with UNC-40. Consistent with this idea, unc-40 also enhanced unc-6 in AVM guidance, similar to tiam-1. However, tiam-1 mutations were less severe than unc-40, suggesting that other molecules act in parallel to TIAM-1 in UNC-40 signaling. Indeed, the TRIM molecule MADD-2 acts with UNC-6 and UNC-40 signaling in AVM ventral guidance, but is not required for the protrusive effects of MYR::UNC-40 [54]. We find that TIAM-1 is required for MYR::UNC-40 protrusive effects but not axon guidance defects caused by MYR::UNC-40, suggesting that distinct pathways might act downstream of UNC-40 to mediate distinct roles of UNC-40 in axon guidance (i.e. protrusive versus non-protrusive events).
These results have implications about UNC-6/Netrin signaling in axon guidance. First, unc-6 AVM axon guidance defects were more severe than unc-40, suggesting that UNC-6 signals through other receptors in addition to UNC-40/DCC. Second, that unc-40 enhances unc-6 suggests that these molecules act at least in part in parallel, and suggest that UNC-6 might not be the only ligand for UNC-40/DCC in AVM ventral guidance. Possible candidates for such interactions might be the SLT-1/Slit ligand and its receptor SAX-3/Robo, which have been shown to functionally interact with Netrin and DCC [59]. In any case, genetic interactions suggest that TIAM-1 acts in the UNC-40 pathway in AVM axon guidance.
The relationship of lamellipodial and filopodial protrusion to axon guidance
Our results demonstrate that TIAM-1 is required for axon guidance and that TIAM-1 regulates lamellipodial and filopodial protrusions in neurons. Growth cones, including those in C. elegans, display dynamic lamellipodial and filopodial protrusions [19], [60]. Our recent findings indicate that the Netrin receptors UNC-40/DCC and UNC-5 modulate growth cone lamellipodial and filopodial protrusion that correlates with attractive versus repulsive responses to UNC-6/Netrin (i.e. UNC-6/Netrin inhibits protrusion in repelled growth cones via UNC-5, and stimulates protrusion in attracted growth cones via UNC-40/DCC) [61]. We speculate that TIAM-1 normally stimulates growth cone lamellipodial and filopodial protrusion in response to UNC-40/DCC in UNC-6/Netrin attractive axon guidance (Figure 9). While it is possible that TIAM-1 controls axon guidance by a mechanism distinct from lamellipodia and filopodia formation, this model is consistent with our data presented here.
Distinct roles of TIAM-1 and UNC-73/Trio in axon guidance
Our results and those of others [9] suggest that UNC-73/Trio acts in parallel to UNC-6/Netrin signaling in PDE axon guidance, and might not act downstream of UNC-40/DCC in AVM neuronal protrusion. In contrast to these results, Trio has been shown in multiple systems to act with Rac GTPases in Netrin signaling. In C. elegans, UNC-73/Trio and the kinesin-like VAB-8L molecule interact and control accumulation of the UNC-6/Netrin receptor UNC-40/DCC and the SLT-1/Slit receptor SAX-3/Robo to the cell surface (i.e. UNC-73/Trio acts upstream of UNC-40/DCC to control its accumulation) [55], [62]. In Drosophila, Trio and the Abelson kinase Abl interact with the Netrin receptor Frazzled (the fly equivalent of UNC-40 and DCC) to control guidance events involving Netrin [56]. Trio was required for the ectopic midline crossing of axons caused by a hybrid chimeric Frazzled-Robo receptor composed of the extracellular and transmembrane domains of Robo and the cytoplasmic domain of Frazzled. In vertebrates, Trio is required for Netrin mediated activation of Rac1 in neurons, and is required for neurite extension in response to Netrin [57].
How might our results fit with these known roles of Trio in Netrin signaling? Cell-specific and context-specific differences in Trio function downstream of guidance receptors are possible explanations. In other words, Trio might act with UNC-40/DCC in some neurons and not in others, or might display distinct, cell-specific interactions with guidance receptors.
An alternative but not mutually exclusive explanation, consistent with previous results, is that Trio and TIAM-1 affect distinct aspects of Rac function in axon guidance. It is becoming apparent that the role of Rac GTPases in axon pathfinding is complex. Rac GTPases clearly regulate cytoskeletal events in the growth cone in response to guidance cues. For example, in C. elegans, Rac GTPases engage the actin-nucleating Arp2/3 complex and the actin-binding protein UNC-115/abLIM to promote filopodial extension [19], [24], [41]. It is also clear that Rac GTPases (e.g. MIG-2/RhoG) control the accumulation of guidance receptors (UNC-40/DCC) at the cell surface via trafficking [55], [62]. Thus, Rac GTPases might act both upstream and downstream of guidance receptors (Figure 9B). This might also explain in part why the axon guidance defects of mig-2; ced-10 doubles are more severe than tiam-1 alone. Rac GTPases might affect multiple aspects of axon guidance, whereas TIAM-1 might specifically regulate them in downstream cytoskeletal effects.
Trio might be a GEF that controls Rac activity in receptor trafficking upstream of UNC-40/DCC, possibly in a feedback mechanism (Figure 9B). In other words, UNC-6/Netrin activation of UNC-40/DCC might alter UNC-40/DCC trafficking to the cell surface, mediated by UNC-73/Trio and Rac. While the studies of Trio and Rac function in flies and vertebrates discussed above [56], [57] did not specifically address UNC-40/DCC trafficking, each can be explained by Trio and Rac regulating cell surface accumulation of Frazzled/DCC and the chimeric Frazzled-Robo receptor, similar to UNC-73/Trio and VAB-8L in C. elegans [55], [62]. In our studies, the myristoylated UNC-40 cytoplasmic domain is likely trafficked to the cell surface by a mechanism that is distinct from the endogenous UNC-40 molecule and might be independent of UNC-73/Trio and Rac-regulated cell surface accumulation, explaining why unc-73 did not suppress MYR::UNC-40. Thus, suppression of MYR::UNC-40 might target those molecules that act downstream of UNC-40/DCC but not those involved in it's trafficking (Figure 9B). Our studies showing that tiam-1 suppressed MYR::UNC-40 indicate that TIAM-1 might regulate Rac GTPases specifically downstream of UNC-40/DCC to mediate cytoskeletal responses.
Rho GTPases and Rho GEFs in axon guidance
Many studies have shown that Rac and CDC-42 GTPases control lamellipodia and filopodia formation, yet it has been unclear how these molecules interact to regulate protrusion. Our results indicate that they act in a linear pathway, with CDC-42 controlling Rac activity mediated by TIAM-1. However, the ced-10; mig-2 double mutant is much more severe than tiam-1, so it is likely that another molecule regulates MIG-2 and CED-10 in parallel to TIAM-1 in axon pathfinding, possibly another GEF.
Rho-family GTPases control many cellular functions, and it has been assumed that upstream regulators confer specificity to Rho GTPase activity. Indeed, genomes encode many GTP exchange factors for Rho GTPases (e.g. the C. elegans genome encodes 19 DH GEF proteins). Our results provide experimental evidence for this idea, as we have shown that the TIAM-1 GEF but not the UNC-73/Trio GEF controls Rac function downstream of UNC-6/Netrin signaling in protrusion. UNC-73/Trio might act upstream of UNC-40/DCC and regulate its trafficking [55], [62] (Figure 9B). Future studies will be aimed at understanding how UNC-40/DCC links to CDC-42 to regulate TIAM-1, as well as understanding the roles of Rho GTPases and GEFs in repulsive axon guidance in response to UNC-6/Netrin.
Methods
C. elegans genetics and culture
C. elegans handling and culture were performed using standard techniques [63]. All experiments were performed at 20°C. The tiam-1(tm1556) allele was provided by the National Bioresource Project for the Experimental Animal “Nematode C. elegans” (S. Mitani), and the tiam-1(ok772) allele was provided to us by the C. elegans Gene Knockout Consortium via the C. elegans Genetic Center (CGC). Polymerase chain reaction (PCR) was used to verify the homozygosity of tiam-1(ok772) and (tm1556) in strains. The following mutations and genetic constructs were used: LGI: unc-73(rh40), tiam-1(ok772) and (tm1556), lqIs37[osm-6::cdc-42(G12V)]; LGII: cdc-42(gk388), mIn1,vab-1(dx31), juIs76[unc-25::gfp]; LGIV: ced-10(n1993), zdIs4[mec-4::gfp], lqIs3[osm-6::gfp]; LGV: lqIs123[osm-6::myr::unc-73(DHPH1)]; LGX: mig-2(mu28), unc-6(ev400), slt-1(eh15), lqIs2[osm-6::gfp]; LG?: lqIs125[osm-6::myr::unc-73(DHPH2)], lqIs131[mec-7::myr::unc-40(cyto)::gfp], lqIs134[osm-6::myr::unc-73(DHPH1 S1216F)], lqIs165[osm-6::myr::tiam-1(DHPH)], lqIs216[osm-6::myr::tiam-1(DHPH T516F)]. Extrachromosomal arrays: lqEx504-506, osm-6::myr::unc-73(DHPH1); lqEx509-510, osm-6::myr::unc-73(DHPH)2; lqEx517-518, osm-6::myr::unc-73(DHPH1 S1216F); lqEx559-561, osm-6::myr::tiam-1(DHPH); lqEx571-572, lqEx584, osm-6::myr::tiam-1(DHPH T516F).
C. elegans transformation was performed with standard methods by microinjection of DNA into the hermaphroditic syncytial germline [64]. Transgenes were integrated into the genome using trimethylpsoralen and standard techniques. RNAi was performed using standard feeding RNAi protocols and clones from the Ahringer RNAi library that were confirmed by PCR [65].
All micrographs were obtained on a Leica DMRE microscope with a Qimaging Rolera MGi EMCCD camera or a Qimaging Retiga CCD camera. Openlab and IPlab software were used to obtain images.
Scoring of PDE, VD/DD, and AVM axon defects
Fluorescence microscopy was used to score axon pathfinding defects of fourth larval stage (L4) or young adult hermaphrodite animals expressing a green fluorescent protein transgene for specific cells. To score and visualize PDE axons, the integrated transgenes lqIs2 X and lqIs3 IV [osm-6 promoter::gfp] were used [41], [66]. To score and visualize VDs and DDs axons, the integrated transgene juIs76 II [unc-25 promoter::gfp] was used [67]. To score and visualize AVM axons, the integrated transgene zdIs4 IV [mec-4 promoter::gfp] was used.
PDE axon pathfinding
The cell bodies of the PDE neurons (PDEL and PDER) are situated in the posterior lateral post-deirid ganglion. PDEs extend an axon ventrally to the ventral nerve cord, which then bifurcates and extends anteriorly and posteriorly in the ventral nerve cord. If the axon failed to reach the ventral nerve cord or wandered beyond a 45° angle from a straight line ventrally from the cell body, it was considered mutant.
VD/DD axon pathfinding
A VD/DD commissural axon that failed to reach the dorsal nerve cord or that wandered laterally before reaching the dorsal nerve cord was considered mutant. The percent of animals with pathfinding defects was noted, and the percentage of defective axons was noted.
AVM axon pathfinding
The AVM cell body is situated laterally on the right side, in the anterior half of the body of the animal. The axon extends ventrally towards the ventral nerve cord, which then extends anteriorly until the first pharyngeal bulb. Similarly to PDE axons, if the axon failed to reach the ventral nerve cord or wandered beyond a 45° angle from a straight line ventrally, it was considered mutant. p-value significance was determined using Fisher Exact Analysis.
Ectopic PDE and AVM protrusions
Ectopic lamellipodial and filopodial protrusions were identified as any thin ruffle-like or finger-like protrusion from the cell body, which is normally smooth and round, or axon of the neurons.
Transgenes for expression in C. elegans
All C. elegans expression plasmids utilized the standard Fire expression vectors with the unc-54 3′ UTR (A. Fire, Stanford University). Primer sequences used to amplify C. elegans genomic and cDNA sequences to construct these plasmids are shown in Figure S9. For each transgene, at least three independent transgenic lines were generated, each with similar results. A tiam-1 transcriptional promoter fusion plasmid was generated by amplifying by PCR the tiam-1 upstream promoter region, a 4793-bp region upstream of T21E12.2 (the entire upstream region to the next predicted gene, T21E12.3) (Figure 2), and placing it upstream of the cyan fluorescent protein (cfp) coding region. For cell specific expression of full length TIAM-1, the open reading frame of the yk370h9 tiam-1 cDNA was amplified by PCR and fused to mCherry at the 3′ end to create a C-terminally tagged TIAM-1::mCherry molecule (Figure 5). tiam-1::mCherry expression plasmids contained the osm-6 promoter (to drive expression in the PDEs) [66] and the unc-25 promoter (VD/DDs) [67] in separate constructs. The yk730h9 cDNA contained errors relative to the reference gene models that resulted in frame shifts. These were repaired in the yk730h9 cDNA by site-directed mutagenesis. A G residue after base 1039 relative to the start codon in Figure S2 was removed, and the A residue at position 2478 was added (see Figure S2).
A gfp::mig-2 fusion plasmid was constructed by placing gfp in frame after the N-terminal myristoylation sequence of mig-2 and before the remaining genomic coding region, allowing for GFP tagging of both myristoylated and prenylated forms of MIG-2 (Figure 5). gfp::ced-10 was constructed by placing gfp at the N-terminus as previously described [48] (Figure 5). gfp::mig-2 and gfp::ced-10 expression was driven by the osm-6 or unc-25 promoter.
A plasmid containing an activated cdc-42(G12V) genomic coding region under the control of the osm-6 promoter was constructed using site directed mutagenesis as described previously for ced-10(G12V) [41]. A myr::unc-40(cyto) plasmid under the control of the mec-7 promoter was constructed as described in [9] (Figure 8A). The myr::GEF(DHPH) plasmids with the osm-6 promoter were used as means of overactivating the GEFs as previously described [42] (Figure 4A). Three distinct DHPH GEF domains were fused in frame to an N-terminal myristoylation sequence as described in [44] and to a C-terminal GFP: the DHPH region of TIAM-1 (residues 488–856 relative to the A isoform); the Rac-specific UNC-73 DHPH domain (DHPH1) (residues 1193–1527 relative to the F55C7.7A isoform); and the Rho-specific UNC-73 DHPH domain (DHPH2) (residues 1795–2129). The coding regions of transgenes were sequenced after PCR to ensure that no mutations had been introduced.
Cell culture, transfections, and co-localization assays in NIH3T3 fibroblasts
For expression in NIH 3T3 cells, a cmv::mCherry::tiam-1 N-terminal fusion construct was generated by placing the yk37h9 tiam-1 cDNA open reading frame, fused N-terminally to mCherry, behind the CMV promoter in the pTriEx-mCherry vector (K. Hahn, University of North Carolina). cmv::gfp::ced-10 was constructed using the ced-10 cDNA open reading frame [48] and the pEGFP-C1 vector (Clontech).
NIH 3T3 mouse fibroblasts were grown in DMEM (Cellgro) supplemented with 10% Cosmic Calf serum (HyClone) and 1% penicillin-streptomycin (P/S) and maintained in 5% CO2 at 37°C. Cells were plated onto coverslips the day before transfection at a density of 200,000 cells per well in a 6-well plate. For co-localization assays, pEGFP-C1 (vector only, “GFP”), pEGFP-C1 expressing CED-10 (“GFP::CED-10”), pTriEx-mCherry (vector only, “mCherry”) and pTriEx-mCherry expressing TIAM-1 (“mCherry::TIAM-1”) were transiently transfected into NIH3T3 cells using GeneExpresso Transfection reagent (Lab Supply Mall) according to the manufacturer's specifications. To visualize subcellular protein localizations at high magnification, the NIH 3T3 cells grown on coverslips and transiently transfected were fixed in 3.7% formaldehyde for 1 h at room temperature. Coverslips were then washed three times in 1× Phosphate Buffered Saline (PBS), one time in ddH2O, and then mounted onto a glass slide using ProLong Gold antifade (Invitrogen/Molecular Probes) mounting medium. The cells were then visualized using compound fluorescence microscopy (a Leica DMRE microscope with a Qimaging Rolera MGi EMCCD camera was used along with the IPLab and ImageJ software).
Mant-GTP based exchange assay
We amplified the DHPH domains of TIAM-1 (residues 488–856 relative to isoform A), placed it into the pET-28b vector (Novagen), which has a T7 IPTG-inducible promoter and a C-terminal 6HIS-tag, and transformed this construct into BL21(DE3) Gold competent cells (Agilent Technologies). For the TIAM-1[DH(T516F)PH] construct, we used site directed mutagenesis to make a point mutation in the wild-type construct using the Quikchange kit (Stratagene). We purified TIAM-1[DHPH]::6HIS using Ni-column based chromatography (Qiagen). For the mant-GTP assay, we used the RhoGEF Exchange Assay Biochem Kit (Cytoskeleton, Inc) and followed instructions in the kit. We used 12.5 µM of TIAM-1[DHPH] in these experiments.
N - methylanthraniloyl-GTP (mant-GTP) is a fluorescently labeled nucleotide analog that can be incorporated by small GTPases. Using a Varian Cary Eclipse fluorescent spectrophotometer, we measured the amount of mant-GTP incorporated by the GTPase due to the spectroscopic difference between bound and free mant-GTP (when in the GTPase binding pocket, mant-GTP increases its fluorescence). The GTPase (human Rac1, Cdc-42 or RhoA) was incubated in buffer and mant-GTP for five minutes, after which time solvent (control) or the GEF (TIAM-1[DHPH]) was added. Experiments were recorded for one half hour.
Supporting Information
Zdroje
1. GalloGLetourneauPC 2004 Regulation of growth cone actin filaments by guidance cues. J Neurobiol 58 92 102
2. WelchMDMullinsRD 2002 Cellular control of actin nucleation. Annu Rev Cell Dev Biol 18 247 288
3. SvitkinaTMBulanovaEAChagaOYVignjevicDMKojimaS 2003 Mechanism of filopodia initiation by reorganization of a dendritic network. J Cell Biol 160 409 421
4. KorobovaFSvitkinaT 2008 Arp2/3 complex is important for filopodia formation, growth cone motility, and neuritogenesis in neuronal cells. Mol Biol Cell 19 1561 1574
5. PakCWFlynnKCBamburgJR 2008 Actin-binding proteins take the reins in growth cones. Nat Rev Neurosci 9 136 147
6. BroseKBlandKSWangKHArnottDHenzelW 1999 Slit proteins bind Robo receptors and have an evolutionarily conserved role in repulsive axon guidance. Cell 96 795 806
7. ZallenJAYiBABargmannCI 1998 The conserved immunoglobulin superfamily member SAX-3/Robo directs multiple aspects of axon guidance in C. elegans. Cell 92 217 227
8. KilleenMTSybingcoSS 2008 Netrin, Slit and Wnt receptors allow axons to choose the axis of migration. Dev Biol 323 143 151
9. GitaiZYuTWLundquistEATessier-LavigneMBargmannCI 2003 The netrin receptor UNC-40/DCC stimulates axon attraction and outgrowth through enabled and, in parallel, Rac and UNC-115/AbLIM. Neuron 37 53 65
10. ShekarabiMKennedyTE 2002 The netrin-1 receptor DCC promotes filopodia formation and cell spreading by activating Cdc42 and Rac1. Mol Cell Neurosci 19 1 17
11. AdlerCEFetterRDBargmannCI 2006 UNC-6/Netrin induces neuronal asymmetry and defines the site of axon formation. Nat Neurosci 9 511 518
12. AnithaANakamuraKYamadaKSudaSThanseemI 2008 Genetic analyses of roundabout (ROBO) axon guidance receptors in autism. Am J Med Genet B Neuropsychiatr Genet 147B 1019 1027
13. Hannula-JouppiKKaminen-AholaNTaipaleMEklundRNopola-HemmiJ 2005 The axon guidance receptor gene ROBO1 is a candidate gene for developmental dyslexia. PLoS Genet 1 e50 doi:10.1371/journal.pgen.0010050
14. BishopALHallA 2000 Rho GTPases and their effector proteins. Biochem J 348 Pt 2 241 255
15. LundquistEA 2003 Rac proteins and the control of axon development. Curr Opin Neurobiol 13 384 390
16. AzumaTWitkeWStosselTPHartwigJHKwiatkowskiDJ 1998 Gelsolin is a downstream effector of rac for fibroblast motility. Embo J 17 1362 1370
17. BiyashevaASvitkinaTKundaPBaumBBorisyG 2004 Cascade pathway of filopodia formation downstream of SCAR. J Cell Sci 117 837 848
18. NobesCDHallA 1995 Rho, rac and cdc42 GTPases regulate the assembly of multimolecular focal complexes associated with actin stress fibers, lamalliposia and filopodia. Cell 81 53 62
19. NorrisADDyerJOLundquistEA 2009 The Arp2/3 complex, UNC-115/abLIM, and UNC-34/Enabled regulate axon guidance and growth cone filopodia formation in Caenorhabditis elegans. Neural Dev 4 38
20. PollardTDBorisyGG 2003 Cellular motility driven by assembly and disassembly of actin filaments. Cell 112 453 465
21. AizawaHWakatsukiSIshiiAMoriyamaKSasakiY 2001 Phosphorylation of cofilin by LIM-kinase is necessary for semaphorin 3A-induced growth cone collapse. Nat Neurosci 4 367 373
22. DanCKellyABernardOMindenA 2001 Cytoskeletal changes regulated by the PAK4 serine/threonine kinase are mediated by LIM kinase 1 and cofilin. J Biol Chem 276 32115 32121
23. SymonsM 2000 Adhesion signaling: PAK meets Rac on solid ground. Curr Biol 10 R535 537
24. ShakirMAJiangKStruckhoffECDemarcoRSPatelFB 2008 The Arp2/3 Activators WAVE and WASP Have Distinct Genetic Interactions With Rac GTPases in Caenorhabditis elegans Axon Guidance. Genetics 179 1957 1971
25. DemarcoRSLundquistEA 2010 RACK-1 acts with Rac GTPase signaling and UNC-115/abLIM in Caenorhabditis elegans axon pathfinding and cell migration. PLoS Genet 6 e1001215 doi:10.1371/journal.pgen.1001215
26. HallA 1992 Ras-related GTPases and the cytoskeleton. Molecular biology of the cell 3 475 479
27. KubiseskiTJCulottiJPawsonT 2003 Functional analysis of the Caenorhabditis elegans UNC-73B PH domain demonstrates a role in activation of the Rac GTPase in vitro and axon guidance in vivo. Mol Cell Biol 23 6823 6835
28. LundquistEAReddienPWHartwiegEHorvitzHRBargmannCI 2001 Three C. elegans Rac proteins and several alternative Rac regulators control axon guidance, cell migration and apoptotic cell phagocytosis. Development 128 4475 4488
29. WuYCChengTWLeeMCWengNY 2002 Distinct rac activation pathways control Caenorhabditis elegans cell migration and axon outgrowth. Dev Biol 250 145 155
30. StevenRKubiseskiTJZhengHKulkarniSMancillasJ 1998 UNC-73 activates the Rac GTPase and is required for cell and growth cone migrations in C. elegans. Cell 92 785 795
31. HabetsGGScholtesEHZuydgeestDvan der KammenRAStamJC 1994 Identification of an invasion-inducing gene, Tiam-1, that encodes a protein with homology to GDP-GTP exchangers for Rho-like proteins. Cell 77 537 549
32. SoneMHoshinoMSuzukiEKurodaSKaibuchiK 1997 Still life, a protein in synaptic terminals of Drosophila homologous to GDP-GTP exchangers. Science 275 543 547
33. MatsuoNHoshinoMYoshizawaMNabeshimaY 2002 Characterization of STEF, a guanine nucleotide exchange factor for Rac1, required for neurite growth. J Biol Chem 277 2860 2868
34. MatsuoNTeraoMNabeshimaYHoshinoM 2003 Roles of STEF/Tiam1, guanine nucleotide exchange factors for Rac1, in regulation of growth cone morphology. Mol Cell Neurosci 24 69 81
35. HoshinoMSoneMFukataMKurodaSKaibuchiK 1999 Identification of the stef gene that encodes a novel guanine nucleotide exchange factor specific for Rac1. J Biol Chem 274 17837 17844
36. Marchler-BauerALuSAndersonJBChitsazFDerbyshireMK CDD: a Conserved Domain Database for the functional annotation of proteins. Nucleic Acids Res 39 D225 229
37. AltschulSFGishWMillerWMyersEWLipmanDJ 1990 Basic local alignment search tool. J Mol Biol 215 403 410
38. BaumeisterMAMartinuLRossmanKLSondekJLemmonMA 2003 Loss of phosphatidylinositol 3-phosphate binding by the C-terminal Tiam-1 pleckstrin homology domain prevents in vivo Rac1 activation without affecting membrane targeting. J Biol Chem 278 11457 11464
39. MichielsFStamJCHordijkPLvan der KammenRARuuls-Van StalleL 1997 Regulated membrane localization of Tiam1, mediated by the NH2-terminal pleckstrin homology domain, is required for Rac-dependent membrane ruffling and C-Jun NH2-terminal kinase activation. J Cell Biol 137 387 398
40. WorthylakeDKRossmanKLSondekJ 2000 Crystal structure of Rac1 in complex with the guanine nucleotide exchange region of Tiam1. Nature 408 682 688
41. StruckhoffECLundquistEA 2003 The actin-binding protein UNC-115 is an effector of Rac signaling during axon pathfinding in C. elegans. Development 130 693 704
42. NewsomeTPSchmidtSDietzlGKelemanKAslingB 2000 Trio combines with dock to regulate Pak activity during photoreceptor axon pathfinding in Drosophila. Cell 101 283 294
43. LeeuwenFNKainHEKammenRAMichielsFKranenburgOW 1997 The guanine nucleotide exchange factor Tiam1 affects neuronal morphology; opposing roles for the small GTPases Rac and Rho. J Cell Biol 139 797 807
44. YangYLundquistEA 2005 The actin-binding protein UNC-115/abLIM controls formation of lamellipodia and filopodia and neuronal morphogenesis in Caenorhabditis elegans. Mol Cell Biol 25 5158 5170
45. ZipkinIDKindtRMKenyonCJ 1997 Role of a new Rho family member in cell migration and axon guidance in C. elegans. Cell 90 883 894
46. NishimuraTYamaguchiTKatoKYoshizawaMNabeshimaY 2005 PAR-6-PAR-3 mediates Cdc42-induced Rac activation through the Rac GEFs STEF/Tiam1. Nat Cell Biol 7 270 277
47. Montenegro-VenegasCTortosaERossoSPerettiDBollatiF MAP1B regulates axonal development by modulating Rho-GTPase Rac1 activity. Mol Biol Cell 21 3518 3528
48. ReddienPWHorvitzHR 2000 CED-2/CrkII and CED-10/Rac control phagocytosis and cell migration in Caenorhabditis elegans. Nat Cell Biol 2 131 136
49. HedgecockEMCulottiJGHallDH 1990 The unc-5, unc-6, and unc-40 genes guide circumferential migrations of pioneer axons and mesodermal cells on the epidermis in C. elegans. Neuron 4 61 85
50. HaoJCYuTWFujisawaKCulottiJGGengyo-AndoK 2001 C. elegans slit acts in midline, dorsal-ventral, and anterior-posterior guidance via the SAX-3/Robo receptor. Neuron 32 25 38
51. FlanaganJGVanderhaeghenP 1998 The ephrins and Eph receptors in neural development. Annu Rev Neurosci 21 309 345
52. GeorgeSESimokatKHardinJChisholmAD 1998 The VAB-1 Eph receptor tyrosine kinase functions in neural and epithelial morphogenesis in C. elegans. Cell 92 633 643
53. ZallenJAKirchSABargmannCI 1999 Genes required for axon pathfinding and extension in the C. elegans nerve ring. Development 126 3679 3692
54. HaoJCAdlerCEMebaneLGertlerFBBargmannCI 2010 The tripartite motif protein MADD-2 functions with the receptor UNC-40 (DCC) in Netrin-mediated axon attraction and branching. Dev Cell 18 950 960
55. Watari-GoshimaNOguraKWolfFWGoshimaYGarrigaG 2007 C. elegans VAB-8 and UNC-73 regulate the SAX-3 receptor to direct cell and growth-cone migrations. Nat Neurosci 10 169 176
56. ForsthoefelDJLieblECKolodziejPASeegerMA 2005 The Abelson tyrosine kinase, the Trio GEF and Enabled interact with the Netrin receptor Frazzled in Drosophila. Development 132 1983 1994
57. Briancon-MarjolletAGhoghaANawabiHTrikiIAuziolC 2008 Trio mediates netrin-1-induced Rac1 activation in axon outgrowth and guidance. Mol Cell Biol 28 2314 2323
58. IshiiNWadsworthWGSternBDCulottiJGHedgecockEM 1992 UNC-6, a laminin-related protein, guides cell and pioneer axon migrations in C. elegans. Neuron 9 873 881
59. YuTWHaoJCLimWTessier-LavigneMBargmannCI 2002 Shared receptors in axon guidance: SAX-3/Robo signals via UNC-34/Enabled and a Netrin-independent UNC-40/DCC function. Nat Neurosci 5 1147 1154
60. KnobelKMJorgensenEMBastianiMJ 1999 Growth cones stall and collapse during axon outgrowth in Caenorhabditis elegans. Development 126 4489 4498
61. NorrisADLundquistEA 2011 UNC-6/netrin and its receptors UNC-5 and UNC-40/DCC modulate growth cone protrusion in vivo in C. elegans. Development 138 4433 4442
62. Levy-StrumpfNCulottiJG 2007 VAB-8, UNC-73 and MIG-2 regulate axon polarity and cell migration functions of UNC-40 in C. elegans. Nat Neurosci 10 161 168
63. BrennerS 1974 The genetics of Caenorhabditis elegans. Genetics 77 71 94
64. MelloCCKramerJMStinchcombDAmbrosV 1991 Efficient gene transfer in C. elegans: extrachromosomal maintenance and intregration of transforming sequences. EMBO J 10 3959 3970
65. KamathRSFraserAGDongYPoulinGDurbinR 2003 Systematic functional analysis of the Caenorhabditis elegans genome using RNAi. Nature 421 231 237
66. ColletJSpikeCALundquistEAShawJEHermanRK 1998 Analysis of osm-6, a gene that affects sensory cilium structure and sensory neuron function in Caenorhabditis elegans. Genetics 148 187 200
67. JinYJorgensenEHartwiegEHorvitzHR 1999 The Caenorhabditis elegans gene unc-25 encodes glutamic acid decarboxylase and is required for synaptic transmission but not synaptic development. J Neurosci 19 539 548
Štítky
Genetika Reprodukční medicína
Článek A Genome-Wide Screen for Genetic Variants That Modify the Recruitment of REST to Its Target GenesČlánek Population Structure of Hispanics in the United States: The Multi-Ethnic Study of AtherosclerosisČlánek Differing Requirements for RAD51 and DMC1 in Meiotic Pairing of Centromeres and Chromosome Arms inČlánek Transcriptional Regulation of Rod Photoreceptor Homeostasis Revealed by NRL Targetome AnalysisČlánek Cell Contact–Dependent Outer Membrane Exchange in Myxobacteria: Genetic Determinants and MechanismČlánek Formation of Rigid, Non-Flight Forewings (Elytra) of a Beetle Requires Two Major Cuticular Proteins
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2012 Číslo 4- Kazuistika – Perspektivy využití precizované medicíny v rámci personalizované specifické terapie onkologických pacientů
- Nobelova cena za chemii pro genetické nůžky: Objev, který změní naši budoucnost?
- Technologie na bázi RNA v klinické praxi: od přebarvených petúnií k terapii vzácných a dosud jen obtížně léčitelných chorob u lidí
- „Nepředstavovali jsme si, že náš výzkum povede přímo ke vzniku nových léků, dokonce ještě za našeho života“
- Bezplatné služby pro diagnostiku ATTRv amyloidózy pro kardiology
-
Všechny články tohoto čísla
- Runs of Homozygosity Implicate Autozygosity as a Schizophrenia Risk Factor
- Modifier Genes and the Plasticity of Genetic Networks in Mice
- The DSIF Subunits Spt4 and Spt5 Have Distinct Roles at Various Phases of Immunoglobulin Class Switch Recombination
- A Genome-Wide Screen for Genetic Variants That Modify the Recruitment of REST to Its Target Genes
- Population Structure of Hispanics in the United States: The Multi-Ethnic Study of Atherosclerosis
- Deep Sequencing of Plant and Animal DNA Contained within Traditional Chinese Medicines Reveals Legality Issues and Health Safety Concerns
- Differing Requirements for RAD51 and DMC1 in Meiotic Pairing of Centromeres and Chromosome Arms in
- Insulin Signaling Mediates Sexual Attractiveness in
- Progressive Telomere Dysfunction Causes Cytokinesis Failure and Leads to the Accumulation of Polyploid Cells
- Long-Range Chromosome Organization in : A Site-Specific System Isolates the Ter Macrodomain
- Regulation of Budding Yeast Mating-Type Switching Donor Preference by the FHA Domain of Fkh1
- Polyglutamine Toxicity Is Controlled by Prion Composition and Gene Dosage in Yeast
- Patterns of Regulatory Variation in Diverse Human Populations
- Sequence-Specific Targeting of Dosage Compensation in Favors an Active Chromatin Context
- Whole-Exome Sequencing and Homozygosity Analysis Implicate Depolarization-Regulated Neuronal Genes in Autism
- Replication Fork Reversal after Replication–Transcription Collision
- Common Variants at 9p21 and 8q22 Are Associated with Increased Susceptibility to Optic Nerve Degeneration in Glaucoma
- Coordinate Regulation of Lipid Metabolism by Novel Nuclear Receptor Partnerships
- Epigenome-Wide Scans Identify Differentially Methylated Regions for Age and Age-Related Phenotypes in a Healthy Ageing Population
- A Coordinated Interdependent Protein Circuitry Stabilizes the Kinetochore Ensemble to Protect CENP-A in the Human Pathogenic Yeast
- Budding Yeast Dma Proteins Control Septin Dynamics and the Spindle Position Checkpoint by Promoting the Recruitment of the Elm1 Kinase to the Bud Neck
- , a Homolog of a Deaf-Blindness Gene, Regulates Circadian Output and Slowpoke Channels
- Transcriptional Regulation of Rod Photoreceptor Homeostasis Revealed by NRL Targetome Analysis
- Cell Contact–Dependent Outer Membrane Exchange in Myxobacteria: Genetic Determinants and Mechanism
- Defective Membrane Remodeling in Neuromuscular Diseases: Insights from Animal Models
- Formation of Rigid, Non-Flight Forewings (Elytra) of a Beetle Requires Two Major Cuticular Proteins
- SPE-44 Implements Sperm Cell Fate
- A Shared Role for RBF1 and dCAP-D3 in the Regulation of Transcription with Consequences for Innate Immunity
- A Companion Cell–Dominant and Developmentally Regulated H3K4 Demethylase Controls Flowering Time in via the Repression of Expression
- The HEN1 Ortholog, HENN-1, Methylates and Stabilizes Select Subclasses of Germline Small RNAs
- Improved Statistics for Genome-Wide Interaction Analysis
- The Probability of a Gene Tree Topology within a Phylogenetic Network with Applications to Hybridization Detection
- Context-Dependent Dual Role of SKI8 Homologs in mRNA Synthesis and Turnover
- Mu Insertions Are Repaired by the Double-Strand Break Repair Pathway of
- Competition between Replicative and Translesion Polymerases during Homologous Recombination Repair in Drosophila
- An Unbiased Assessment of the Role of Imprinted Genes in an Intergenerational Model of Developmental Programming
- Type 2 Diabetes Risk Alleles Demonstrate Extreme Directional Differentiation among Human Populations, Compared to Other Diseases
- Mutations in and Cause “Splashed White” and Other White Spotting Phenotypes in Horses
- Fine-Scale Mapping of Natural Variation in Fly Fecundity Identifies Neuronal Domain of Expression and Function of an Aquaporin
- Dynamics of Brassinosteroid Response Modulated by Negative Regulator LIC in Rice
- Genetic Inhibition of Solute-Linked Carrier 39 Family Transporter 1 Ameliorates Aβ Pathology in a Model of Alzheimer's Disease
- The Functions of Mediator in Support a Role in Shaping Species-Specific Gene Expression
- Patterns of Ancestry, Signatures of Natural Selection, and Genetic Association with Stature in Western African Pygmies
- Dissection of Pol II Trigger Loop Function and Pol II Activity–Dependent Control of Start Site Selection
- PIWI Associated siRNAs and piRNAs Specifically Require the HEN1 Ortholog
- Genome-Wide Patterns of Gene Expression in Nature
- Hypoxia Disruption of Vertebrate CNS Pathfinding through EphrinB2 Is Rescued by Magnesium
- A New Role for Translation Initiation Factor 2 in Maintaining Genome Integrity
- Sex Reversal in C57BL/6J XY Mice Caused by Increased Expression of Ovarian Genes and Insufficient Activation of the Testis Determining Pathway
- The Rac GTP Exchange Factor TIAM-1 Acts with CDC-42 and the Guidance Receptor UNC-40/DCC in Neuronal Protrusion and Axon Guidance
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- A Coordinated Interdependent Protein Circuitry Stabilizes the Kinetochore Ensemble to Protect CENP-A in the Human Pathogenic Yeast
- Coordinate Regulation of Lipid Metabolism by Novel Nuclear Receptor Partnerships
- Defective Membrane Remodeling in Neuromuscular Diseases: Insights from Animal Models
- Formation of Rigid, Non-Flight Forewings (Elytra) of a Beetle Requires Two Major Cuticular Proteins
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Autoři: prof. MUDr. Pavel Horák, CSc., doc. MUDr. Ludmila Brunerová, Ph.D., doc. MUDr. Václav Vyskočil, Ph.D., prim. MUDr. Richard Pikner, Ph.D., MUDr. Olga Růžičková, MUDr. Jan Rosa, prof. MUDr. Vladimír Palička, CSc., Dr.h.c.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



