-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
Castleman-Like Lymphadenopathy in a Patient with Mixed Connective Tissue Disease: A Case Report and Review of the Literature
Authors: Evelina Rogges 1; Sabrina Pelliccia 2; Gianluca Lopez 3; Roberta Soscia 4; Arianna Di Napoli 3
Authors place of work: PhD School in Translational Medicine and Oncology, Department of Medical and Surgical Sciences and Translational Medicine, Faculty of Medicine and Psychology, Sapienza University of Rome, Rome, Italy 1; Haematology Unit, Department of Clinical and Molecular Medicine, Sant’Andrea University Hospital, Sapienza University, Rome, Italy 2; Pathology Unit, Department of Clinical and Molecular Medicine, Sant’Andrea University Hospital, Sapienza University, Rome, Italy 3; Department of Neurosciences, Mental Health and Sensory Organs, Sapienza University of Rome, Rome, Italy 4
Published in the journal: Čes.-slov. Patol., 62, 2026, No. 1, p. 50-57
Category: Original article
Summary
Differentiating reactive lymphadenopathies in the context of autoimmune disease from Idiopathic Multicentric Castleman Disease (iMCD) poses a significant diagnostic challenge. Castleman-like histological features have been described in various autoimmune disorders, necessitating a strict and comprehensive integration of clinical and laboratory findings to reach the correct diagnosis. Although the Castleman Disease Collaborative Network (CDCN) consensus guidelines list several autoimmune conditions as exclusion criteria for an iMCD diagnosis, mixed connective tissue disease (MCTD) is not currently among them. We report the case of a 77-year-old woman presenting with fatigue, Raynaud’s phenomenon, sclerodactyly, mild generalized lymphadenopathy, in whom the lymph node biopsy revealed a Castleman-like histology. The absence of systemic inflammatory symptoms and the presence of high-titer anti-U1 - RNP antibodies were, however, inconsistent with iMCD, favouring the diagnosis of a reactive Castleman-like lymphadenitis secondary to MCTD. This report highlights that Castleman-like lymphadenopathy can occur in MCTD, closely mimicking iMCD. Therefore, in patients with autoimmune diseases not explicitly listed among the CDCN exclusion criteria, comprehensive clinicopathological integration is essential to avoid misdiagnosis and potentially inappropriate antiIL-6-based therapy.
Keywords:
autoimmune disease – mixed connective tissue disease – Castleman-like lymphadenopathy – Sclerodactyly – Raynaud’s phenomenon - anti-U1-RNP
Castleman disease (CD) represents a spectrum of disorders characterized by distinctive but not specific lymph node histopathology and clinical manifestations, including systemic inflammation, cytopenias, and, in severe cases, life-threatening multiorgan dysfunction (1-4). Significant advances in the understanding, diagnosis, and management of CD have been achieved by the Castleman Disease Collaborative Network (CDCN) consensus guidelines for diagnostic and therapeutic stratification (2, 5-7). According to these criteria, CD is subdivided into unicentric CD (UCD) and multicentric CD (MCD), that can be further categorized into three main subtypes (1, 2). Among these the idiopathic form is further divided into iMCD-TAFRO (defined by thrombocytopenia, acites (anasarca), bone-marrow reticulin fibrosis, renal impairment, and organomegaly), Idiopathic Plasmacytic Lymphadenopathy (IPL) (characterized by thrombocytosis, hypergammaglobulinemia, and a less severe clinical presentation), and iMCD not otherwise specified (8, 9). The etiology of iMCD remains incompletely understood, although excessive production of interleukin-6 (IL-6) and vascular endothelial growth factor (VEGF) are recognized as key pathogenic mediators (1). Patients with iMCD usually present with multiple enlarged lymph nodes and a vari
able constellation of systemic manifestations —fever, fatigue, weight loss, night sweats, serous effusions, and skin lesions (i.e. cherry angiomas) — accompanied by laboratory abnormalities such as anemia, thrombocytopenia or thrombocytosis, elevated ESR (Erythrocyte Sedimentation Rate) and CRP (C-Reactive Protein), polyclonal hypergammaglobulinemia, hypoalbuminemia, and renal dysfunction. The clinical course is often relapsing and remitting, requiring systemic therapy with anti-IL-6 agents (e.g., siltuximab) or anti-CD20 drugs (e.g. rituximab) (7, 10). From a histopathological standpoint, CD is classified into hyaline-vascular (in UCD) or hypervascular (in MCD) type (HVCD), plasma-cell type (P-CD), and mixed (mixed-CD) type, depending on the degree of follicular regression or hyperplasia and the extent of interfollicular vascular proliferation and plasmacytosis (2). Because CD-like histopathological and clinical features may also appear in other diseases, rigorous exclusion of alternative causes is essential before establishing an iMCD diagnosis (11-14). Among these mimicking conditions, the CDCN guidelines listed infections, rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), adult-onset Still’s disease, juvenile idiopathic arthritis, autoimmune lymphoproliferative syndrome (ALPS), HIV-associated lymphadenopathy, and both Hodgkin and non-Hodgkin lymphomas. Other autoimmune diseases that may present with CD-like lymphadenopathy, such as mixed connective tissue disease (MCTD), are less well characterized and do not necessarily exclude a diagnosis of CD (13). MCTD is a rare systemic autoimmune disease characterized by the presence of anti-U1-ribonucleoprotein (RNP) antibodies, Raynaud phenomenon and signs/symptoms of at least 2 over lapping connective tissue diseases, including SLE, systemic sclerosis, polymyositis (15, 16). Given the anti-U1-RNP antibody is the hallmark of MCTD, it is believed that it plays a role in the pathogenesis of MCTD with two main proposed mechanisms. The first involves the direct binding to endothelial cells through U1 RNP peptides or recognition of nucleosome RNP fragments in endothelial cell apoptotic blebs (17, 18). This may lead to the vascular diseases (Raynaud phenomenon), skin sclerosis, and pulmonary hypertension. The second is through immune complex formation, which can activate the complement cascade and lead to tissue inflammation and damage, such as myositis, arthritis, and interstitial lung disease (17, 18). The inflammatory cascade induced by the anti-U1 RNP antibodies is also responsible for the elevated serum levels of IL-6, IL-1, TNF-alpha, and IFN-gamma observed in patients with MCTD (17, 19, 20). Herein we present the case of a patient with MCTD with CDlike lymphadenopathy, for which in accordance with CDCN recommendations, we employed a comprehensive, multidisciplinary evaluation integrating clinical, serological, radiologic, and pathological findings to assess the correct diagnosis (1, 5).
CASE REPORT
A 77-year-old female was referred to our hospital for evaluation of unexplained fatigue and mild generalized lymphadenopathy. She denied fever, weight loss, or night sweats. Clinical examination revealed non-tender lymph nodes in the supraclavicular region. Initial laboratory studies showed normal hematologic, hepatic, and renal parameters. Inflammatory markers (ESR, CRP) were within normal limits. Ultrasound and CT scans revealed multiple small lymph nodes (<1.5 cm)
in both supradiaphragmatic and infradiaphragmatic stations, along with mild splenomegaly (maximal craniocaudal dimension of 13 cm). An excisional biopsy of a supraclavicular lymph node was performed. Histologically, the lymph node architecture was largely preserved, featuring patent sinuses and normal compartmentalization of the B - and T-cell areas (Figure 1). The B-cell zones displayed irregularly shaped lymphoid follicles whose germinal centres (GCs) showed variable degrees of regression (grade 2 according to the CDCN classification), containing a lower number of GC cells (CD10+/-, BCL6+/-, BCL2 - ) and with moderate prominence of CD21+, CD23+ follicular dendritic cells (FDCs) (grade 2 according to the CDCN classification). Characteristically, some germinal centres were penetrated by radially oriented hyalinized capillaries, creating a “lollipop” appearance. These were surrounded by mantle-zone lymphocytes, occasionally concentrically arranged forming an “onion skin” pattern. The interfollicular areas contained a moderate proliferation of small blood vessels (grade 2 according to the CDCN classification), abundant reactive T cells, scattered immunoblasts, and a few polytypic plasma cells (grade 0 according to the CDCN classification) (Figure 1). Immunostaining for human herpes virus 8 (LANA-1) and in situ hybridization for EBER were both negative. The overall lymph node findings were interpreted as a reactive Castleman-like lymphadenitis, necessitating integration with clinical, radiological and laboratory data to differentiate HV-CD from an autoimmune or infectious etiology. Additional clinical investigations revealed sclerodactyly and a history of episodic Raynaud’s phenomenon. The patient did not report other systemic signs. Serologic tests demonstrated the presence of anti-nuclear (ANA) antibodies and high titers of anti-U1 RNP antibodies. Although the excised lymph node exhibited some morphological features reminiscent of Castleman disease—including regressed germinal centers, concentric mantle zones, and mildly increased vascular proliferation—the overall findings did not meet the established diagnostic criteria for iMCD. Specifically, the absence of systemic inflammatory symptoms and serological inflammatory markers, coupled with the presence of high-titer anti-U1 RNP antibodies and signs of connective tissue diseases, was more consistent with an underlying autoimmune process. Therefore, integrating the histopathological, serological, and clinical data, the lymphadenopathy was interpreted as a reactive Castleman-like lymphadenitis secondary to MCTD rather than an iMCD associated with MCTD.
Fig. 1. Histological features of the excised lymph node. 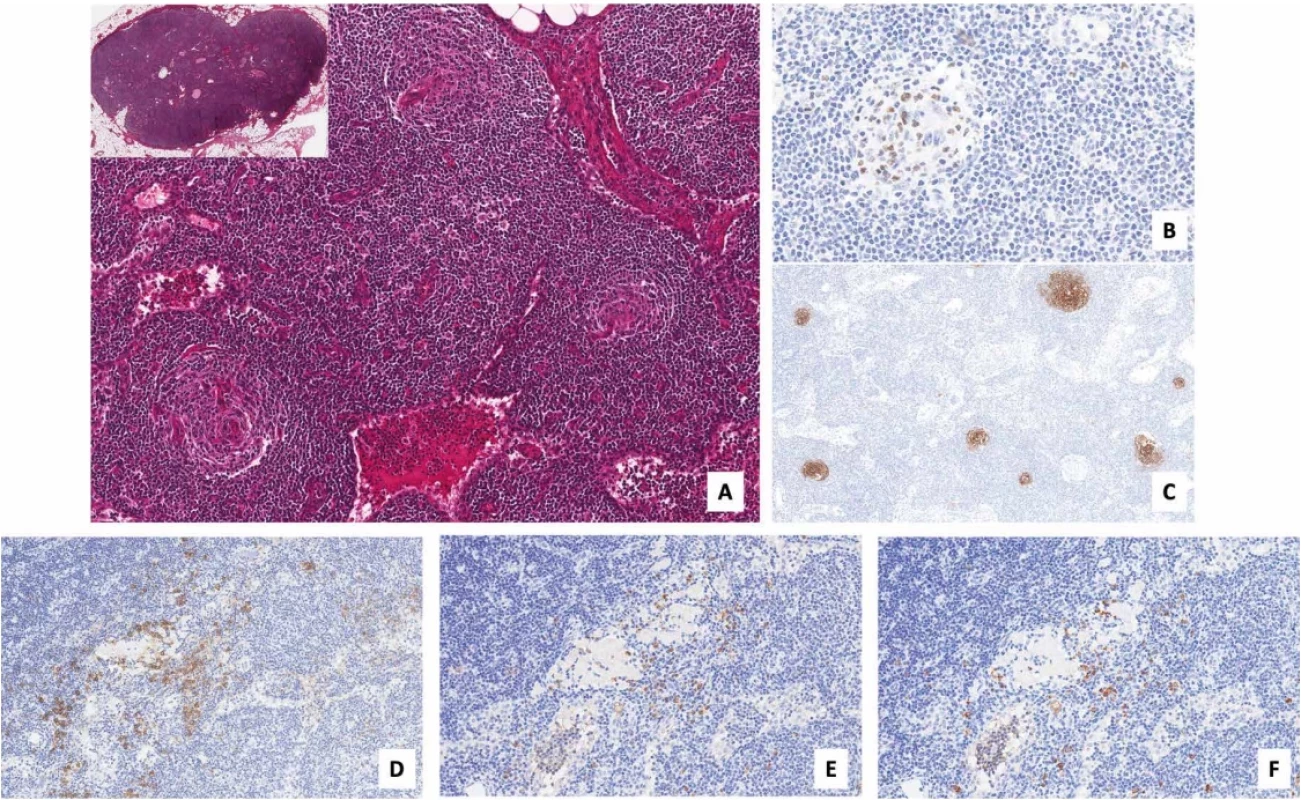
Supraclavicular lymph node with largely preserved architecture (A, upper insert, hematoxylin and eosin (H&E), original magnification (o.m.) x15). The lymphoid follicles showed germinal centers (GCs) with differing degrees of regression, some of which were penetrated by radially oriented hyalinized capillaries and surrounded by concentrically arranged mantle-zone lymphocytes (A, H&E, o.m. x100). The regressed GCs contained a fewer number of GC cells (B: BCL6, o.m. x200) and were centered by a compact meshwork of follicular dendritic cells (C: CD23, o.m. x70). The interfollicular areas were expanded and contained numerous small blood vessels, and a mild polytypic plasma cell infiltrate (D: CD138, E: kappa, F: lambda, o.m. x200). DISCUSSION
The presence of regressed germinal centers, concentric mantle zones, marked vascular proliferation, and prominent interfollicular plasmacytosis can closely resemble the spectrum of histological lesions seen in iMCD. However, Castleman-like morphology alone does not establish a diagnosis of Castleman Disease (CD), as these findings may also occur in reactive conditions secondary to immune dysregulation or infection (11-14). According to the CDCN guidelines, a diagnosis of iMCD necessitates both characteristic histopathology and compatible systemic clinical and laboratory findings, as well as the exclusion of alternative conditions, including selected autoimmune and autoinflammatory diseases when the disease-specific clinical criteria are met (11, 13, 14). These criteria, however, do not specifically address less common overlap syndromes, such as MCTD. Indeed, CD-like histopathological changes have been described in a variety of established autoimmune diseases, such as SLE, RA, and ALPS (11-14), but are also reported—though less frequently—in MCTD (21-26). Several reports, summarized in Table 1, highlight cases of concomitant diagnosis of MCTD and CD (21-26). On the contrary, in our case we were unable to diagnose a MCD due to unmet diagnostic criteria. However, given that MCTD is also a multisystem autoimmune disorder with immune-mediated lymphoid activation, it remains uncertain whether its presence should be regarded as an exclusion criterion for iMCD or whether both conditions may genuinely coexist. This ambiguity has important implications for the interpretation of Castleman-like lymphadenopathy in patients with connective tissue diseases other than SLE or RA.
In MCTD the autoimmune background might provide a plausible mechanism for CD-like histological changes. Immune activation of the downstream inflammatory cascade driven by antiU1-RNP antibodies induces elevated serum levels of IL-6, IL-1alpha, TNF-alpha, and IFN-gamma, (17, 19, 20) which are responsible of the broad histologic heterogeneity observed in MCTD-associated lymphadenopathy. In Table 1 and 2, we summarize reported cases of MCTD with available lymph node histology. The lesions span a wide morphologic spectrum, including sarcoid-like granulomatous reactions (27), Kikuchi-like necrotizing lymphadenitis (28-33), non-specific reactive lymphadenitis (16, 34-39) and lymphadenitis with CD-like changes (21-26).
Tab. 1. Reported cases of CD associated with MCTD. 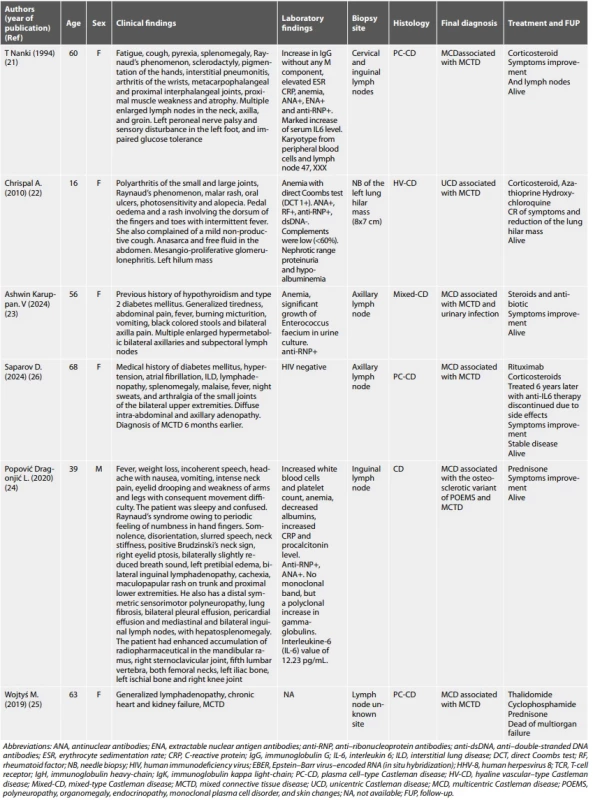
Tab. 2. Other lymph node histopathological patterns reported in association with MCTD. 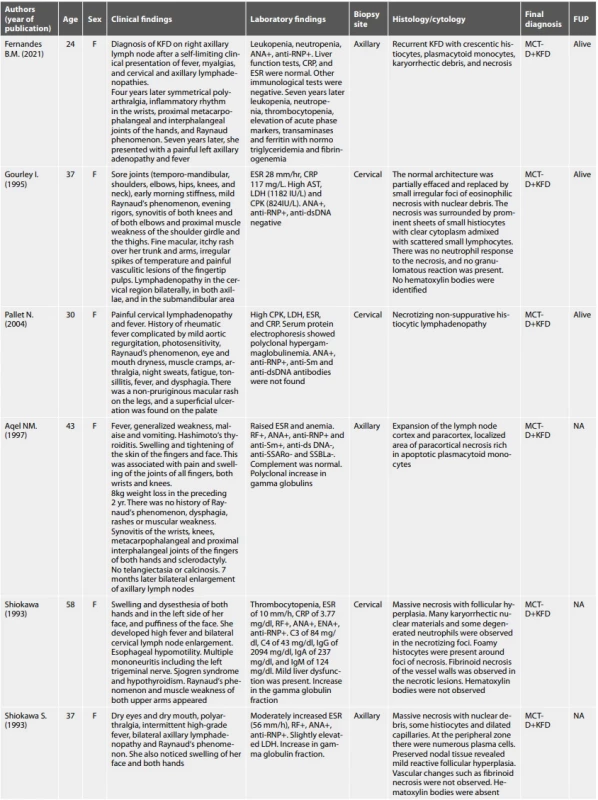
The distinction between iMCD and autoimmune-driven reactive conditions is clinically critical: misclassification as iMCD could lead to unnecessary anti-IL-6–based therapies, while correct recognition of an autoimmune-driven reactive process allows for appropriate immunomodulatory management. Recognition that Castleman-like morphology may represent a reactive pattern rather than a diagnostic entity prevents the overdiagnosis of iMCD and reinforces the need to interpret lymph node histopathology within the broader clinical and immunological context. In our case, the integration of clinical, serological, and pathological data strongly favoured the diagnosis of reactive Castleman-like lymphadenitis associated with MCTD, thus emphasizing the importance of a multidisciplinary, clinicopathological correlation when evaluating lymphadenopathy with Castleman-like features.
In conclusion, with the present case we highlighted that Castleman disease-like lymphadenopathies may occur as a histological pattern in the context of less characterized autoimmune diseases such as MCTD. A careful multidisciplinary assessment integrating clinical, serological, and pathological findings is therefore essential to distinguish reactive Castleman-like lymphadenitis from iMCD to avoid misdiagnosis and inappropriate treatment.
Zdroje
1. Fajgenbaum DC, Uldrick TS, Bagg A, et al. International, evidence-based consensus diagnostic criteria for HHV-8-negative/idiopathic multicentric Castleman disease. Blood 2017; 129(12): 1646-1657.
2. Dispenzieri A, Fajgenbaum DC. Overview of Castleman disease. Blood 2020; 135(16): 1353–1364.
3. Dispenzieri A. POEMS syndrome: 2017 Update on diagnosis, risk stratification, and management. Am J Hematol 2017; 92(8): 814-829.
4. Kawabata H, Takai K, Kojima M, et al. Castleman-Kojima disease (TAFRO syndrome) : a novel systemic inflammatory disease characterized by a constellation of symptoms, namely, thrombocytopenia, ascites (Anasarca), microcytic anemia, myelofibrosis, renal dysfunction, and organomegaly : a status report and summary of Fukushima (6 June, 2012) and Nagoya meetings (22 September, 2012). J Clin Exp Hematop 2013; 53(1): 57–61.
5. van Rhee F, Voorhees P, Dispenzieri A, et al. International, evidence-based consensus treatment guidelines for idiopathic multicentric Castleman disease. Blood 2018; 132(20): 2115-2124.
6. van Rhee F, Oksenhendler E, Srkalovic G, et al. International evidence-based consensus diagnostic and treatment guidelines for unicentric Castleman disease. Blood Adv 2020; 4(23): 6039-6050.
7. Gérard L, Bérezné A, Galicier L, et al. Prospective study of rituximab in chemotherapy-dependent human immunodeficiency virus associated multicentric Castleman’s disease: ANRS 117 CastlemaB Trial. J Clin Oncol 2007; 25(22): 3350-3356.
8. Nijim S, Fajgenbaum DC. Identifying Castleman disease from non-clonal inflammatory causes of generalized lymphadenopathy. Hematology Am Soc Hematol Educ Program 2024; 2024(1): 582-593.
9. Nishikori A, Nishimura MF, Nishimura Y, et al. Idiopathic Plasmacytic Lymphadenopathy Forms an Independent Subtype of Idiopathic Multicentric Castleman Disease. Int J Mol Sci 2022; 23(18): 10301.
10. van Rhee F, Fayad L, Voorhees P, et al. Siltuximab, a novel anti-interleukin-6 monoclonal antibody, for Castleman’s disease. J Clin Oncol 2010; 28(23): 3701-3708.
11. Frizzera G, Peterson BA, Bayrd ED, Goldman A. A systemic lymphoproliferative disorder with morphologic features of Castleman’s disease: clinical findings and clinicopathologic correlations in 15 patients. J Clin Oncol 1985; 3(9): 1202-1216.
12. Deshpande V, Zen Y, Chan JK, et al. Consensus statement on the pathology of IgG4-related disease. Mod Pathol 2012; 25(9): 1181-1192.
13. Pelliccia S, Rogges E, Cardoni A, et al. The application of a multidisciplinary approach in the diagnosis of Castleman disease and Castleman-like lymphadenopathies: A 20-year retrospective analysis of clinical and pathological features. Br J Haematol 2024; 204(2): 534-547.
14. Krishnan J, Danon AD, Frizzera G. Reactive lymphadenopathies and atypical lymphoproliferative disorders. Am J Clin Pathol 1993; 99(4): 385-396.
15. John KJ, Sadiq M, George T, et al. Clinical and Immunological Profile of Mixed Connective Tissue Disease and a Comparison of Four Diagnostic Criteria. Int J Rheumatol 2020; 2020 : 9692030.
16. Bennett RM, O’Connell DJ. Mixed connective tisssue disease: a clinicopathologic study of 20 cases. Semin Arthritis Rheum 1980; 10(1): 25-51.
17. Okawa-Takatsuji M, Aotsuka S, Uwatoko S, et al. Endothelial cell-binding activity of anti-U1-ribonucleoprotein antibodies in patients with connective tissue diseases. Clin Exp Immunol 2001; 126(2): 345-54.
18. Biggiogera M, Bottone MG, Martin TE, Uchiumi T, Pellicciari C. Still immunodetectable nuclear RNPs are extruded from the cytoplasm of spontaneously apoptotic thymocytes. Exp Cell Res 1997; 234(2): 512-520.
19. Okawa-Takatsuji M, Aotsuka S, Uwatoko S, Kinoshita M, Sumiya M. Increase of cytokine production by pulmonary artery endothelial cells induced by supernatants from monocytes stimulated with autoantibodies against U1-ribonucleoprotein. Clin Exp Rheumatol 1999; 17(6): 705-712.
20. Stypińska B, Lewandowska A, Felis-Giemza A, Olesińska M, Paradowska-Gorycka A. Association study between immune-related miRNAs and mixed connective tissue disease. Arthritis Res Ther 2021; 23(1): 19.
21. Nanki T, Tomiyama J, Arai S. Mixed connective tissue disease associated with multicentric Castleman’s disease. Scand J Rheumatol 1994; 23(4): 215-217. 22. Chrispal A, Vasuki Z, Thomas EM, Boorugu HK. Mixed connective tissue disorder and Castleman’s disease. J Assoc Physicians India 2010; 58 : 515-517.
23. Karuppan AV, Ganesh D, Jayaseelya CPS. Case Report on Presentation of Castleman’s Disease in a Patient with Mixed Connective Tissue Disorder. Indian Journal of Pharmacy Practice 2024; 17(1): 75-77.
24. Popović Dragonjić L, Jovanović M, Vrbić M, et al. Castleman’s disease associated with mixed connective tissue disorder and cerebral ischaemia and vasculitis: A rare case and a diagnostic challenge for an infectologist. Vojnosanit Pregl 2020; 77(8): 872–877.
25. Wojtyś M, Piekarska A, Kunc M, et al. Clinicopathological comparison and therapeutic approach to Castleman disease-a case-based review. J Thorac Dis 2019; 11(11): 4859-4874.
26. Saparov D, Gold-Olufadi S, Wasifusddin M, et al. Atypical Combination of Mixed Connective Tissue Disease and Multicentric Castleman Disease. Cureus 2024; 16(9): e70325.
27. Szodoray P, Szollosi Z, Gyimesi E, et al. Sarcoidosis in patients with mixed connective tissue disease: clinical, genetic, serological and histological observations. Rheumatol Int 2008; 28(8): 743-747.
28. Fernandes BM, Bernardes M, Barroca H, Costa L. Kikuchi-Fujimoto Disease Associated With Mixed Connective Tissue Disease: A Late Recurrence Case. J Clin Rheumatol 2021; 27(8S): S779-S780.
29. Gourley I, Bell AL, Biggart D. Kikuchi’s disease as a presenting feature of mixed connective tissue disease. Clin Rheumatol 1995; 14(1): 104-107.
30. Pallet N, Aaron L, Larousserie F, Therby A, Dupont B, Viard JP. Kikuchi-Fujimoto disease associated with mixed connective tissue disease. Scand J Rheumatol 2004; 33(6): 434-436.
31. Aqel NM, Amr SS, Najjar MM, Henry K. Kikuchi’s lymphadenitis developing in a patient with mixed connective tissue disease and Hashimoto’s thyroiditis. Br J Rheumatol 1997; 36(11): 1236-1238.
32. Shiokawa S, Yasuda M, Kikuchi M, Yoshikawa Y, Nobunaga M. Mixed connective tissue disease associated with lupus lymphadenitis. J Rheumatol 1993; 20(1): 147-150.
33. Sharma V, Rankin R. Fatal Kikuchi-like lymphadenitis associated with connective tissue disease: a report of two cases and review of the literature. Springerplus 2015; 4 : 167.
34. Karsulovic C, Hojman LP, Seelmann DL, Wurmann PA. Diffuse Lymphadenopathy Syndrome as a Flare-Up Manifestation in Lupus and Mixed Connective Tissue Disease Following Mild COVID-19. Am J Case Rep 2021; 22: e932751.
35. Erlij D, Cuellar MC, Badilla N, et al. Poliadenopatías de origen reumatológico y las claves del diagnóstico diferencial: Análisis de 19 casos [Lymphadenopathies in patients with rheumatic diseases. Review of 19 cases]. Rev Med Chil 2020; 148(3): 320-326.
36. Guit GL, Shaw PC, Ehrlich J, Kroon HM, Oudkerk M. Mediastinal lymphadenopathy and pulmonary arterial hypertension in mixed connective tissue disease. Radiology 1985; 154(2): 305-306.
37. Frayha RA, Nasr FW, Mufarrij AA. Mixed connective tissue disease, Sjögren’s syndrome, and abdominal pseudolymphoma. Br J Rheumatol 1985; 24(1): 70-73.
38. Yiannopoulos G, Daoussis D, Kourea H, Kalogeropoulou C, Andonopoulos AP. Massive Abdominal Lymphadenopathy as a Manifestation of Connective Tissue Diseases. Report of Two Cases and Review of the Literature. J Orthop Rheumatol 2013; 1 : 1.
39. Bagio MT, Queiroz Guedes L, Takeshi Ishizava R, et al. Lymphadenopathy with high PET-CT uptake: a case of mixed connective tissue disease mimicking lymphoma. Rev Eletr Acervo Saude 2025.
Štítky
Patologie Soudní lékařství Toxikologie
Článek vyšel v časopiseČesko-slovenská patologie

2026 Číslo 1-
Všechny články tohoto čísla
- Testing Claudin 18.2 Expression in Gastric and Gastroesophageal Junction Adenocarcinoma: Current Status and Near‑Future Outlook
- Castleman disease – one name, many faces
- Lymfatická tkáň v souvislostech: od nenápadných změn k zásadním diagnózám
- The advantages and limitations of the new FIGO 2023 staging system for endometrial carcinoma from the perspective of the clinician and pathologist
- Od archeologické brigády k americké rezidentuře: proč je patologie „nejlepší obor medicíny“
- Targeted gene expression profiling as a tool for diagnostic cell-of-origin determination and prognostic stratification in diffuse large B-cell lymphoma
- Castleman-Like Lymphadenopathy in a Patient with Mixed Connective Tissue Disease: A Case Report and Review of the Literature
- EBV-associated plasmacytic variant of Castleman disease: more than a decade-long diagnostic odyssey – a case report
- MONITOR, aneb nemělo by vám uniknout, že…
- Česko-slovenská patologie
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Castleman disease – one name, many faces
- Testing Claudin 18.2 Expression in Gastric and Gastroesophageal Junction Adenocarcinoma: Current Status and Near‑Future Outlook
- The advantages and limitations of the new FIGO 2023 staging system for endometrial carcinoma from the perspective of the clinician and pathologist
- Targeted gene expression profiling as a tool for diagnostic cell-of-origin determination and prognostic stratification in diffuse large B-cell lymphoma
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Autoři: prof. MUDr. Pavel Horák, CSc., doc. MUDr. Ludmila Brunerová, Ph.D., doc. MUDr. Václav Vyskočil, Ph.D., prim. MUDr. Richard Pikner, Ph.D., MUDr. Olga Růžičková, MUDr. Jan Rosa, prof. MUDr. Vladimír Palička, CSc., Dr.h.c.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání





