-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
- Témata
Top novinky
Reklama- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Volná místa
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaWastage of medicines and its financial impact on the healthcare system in the Czech Republic
Nevyužívaná léčiva a jejich finanční dopad na zdravotní systém v České republice
Úvod:
Nevyužívaná léčiva představují významný faktor, který přispívá ke zvyšování nákladů na zdravotní péči. K plýtvání s léčivy (k vzniku farmaceutického odpadu) dochází v důsledku non-compliance pacientů, nadměrného a neracionálního předepisování nebo nedostatečné kontroly výdeje předepsaných léčivých přípravků v lékárně.
Cíl:
Popsat a analyzovat problematiku nevyužívaných léčivých přípravků z kvantitativního, kvalitativního a finančního hlediska, jejich nákladů pro systém veřejné zdravotní péče a chování české populace při reálném využívání léčiv.
Metoda:
Analyzovaný materiál tvořily léčivé přípravky vrácené do 66 lékáren v České republice v období 16. 5. až 29. 6. 2012. Pozornost byla věnována zejména hromadně vyráběným léčivým přípravkům (HVLP).
Hlavní míry výsledků:
Analýza vrácených léčivých přípravků na základě kritérií: z finančního hlediska, nevyužívaného množství, terapeutické skupiny, účinné látky a názvu léčivého přípravku.
Výsledky:
V analyzovaném materiálu bylo zjištěno 18 890 položek. Počet vrácených HVLP činil 16 520 léčivých přípravků. Celková hodnota těchto léčivých přípravků byla vypočtena na cca 2,1 mil. Kč, resp. 82 000 EUR. Úhrada zdravotních pojišťoven činila cca 1,4 mil. Kč. (55 000 EUR). Nejčastějšími vrácenými léčivy byly přípravky působící na kardiovaskulární systém (17,0 %). Nejčastější účinnou látkou byl paracetamol, který se opakoval 405krát.
Závěr:
Výsledky studie o nevyužívaných léčivých přípravcích z kvantitativního, kvalitativního a finančního pohledu popisují situaci nejen pro tvůrce lékové politiky a systémů tvorby cen a politik úhrady, ale také pro bezpečnější a účinnější lékárenskou péči a posílení funkce lékárny.
Klíčová slova:
nevyužitá léčiva – vrácená léčiva – lékový odpad – farmaceutický odpad – Česká republika
Authors: Jozef Kolář Jan Kostřiba Jana Kotlářová Tünde Ambrus Lenka Smejkalová
Published in the journal: Čes. slov. Farm., 2018; 67, 192-199
Category: Původní práce
Summary
Background:
Medication wastage is an important factor that contributes to escalating healthcare costs. Medication wastage may be due to poor compliance of patients, excessive and irrational prescribing, or a lack of control of the dispensing of prescription medications in the community pharmacy.
Objective:
To describe and analyze the situation regarding the unused medicines from the quantitative, qualitative and financial point of view, their cost to the public healthcare system, and behaviours regarding medication wastage of the Czech population.
Method:
The analysed material consisted of medicines returned to 66 community pharmacies in the Czech Republic between May 16 and June 29, 2012. Thorough analysis was aimed only at medicinal products.
Main outcome measure:
The analysis of the returned medicines according to the price, amount, therapeutic group, active substance and brand name.
Results:
In the analysed material there were 18,890 items. The number of returned medicines was 16,520 products. The total value of these products was calculated at about Czech crowns (CZK) 2.1 mil., approx. Euro (EUR) 82,000. Reimbursement of health insurance companies was about CZK 1.4 mil. (approx. EUR 55,000). The most frequently returned drugs were medicines acting on the cardiovascular system (17.0%). The most frequent active substance was paracetamol, which recurred 405 times.
Conclusion:
The results about the unused medicines from the quantitative, qualitative and financial perspectives describe the situation not only for medicines policy makers and adjustment of the systems of pricing and reimbursement policies, but also for safer and more effective pharmaceutical care and strengthening of the function of community pharmacies.
Keywords:
unused medicines – returned medicines – medication waste – medication wastage – Czech Republic
Introduction
Irrational use of medicines is a major problem all around the world today. The World Health Organization (WHO) estimates that half of all medicines are inappropriately prescribed, dispensed or sold and that half of all patients fail to take their medicine properly. The overuse, underuse or misuse of medicines result in wastage of scarce resources and cause a health risks increase1).
The ever-increasing drug use and consequent rise in healthcare costs led to a search for opportunities for cost reduction and savings in most industrialized countries2). The increased costs of medicines, the expenses incurred in the development of new drug products and medication wastage are the most important factors that contribute to escalating healthcare costs all around the world. Medication wastage is defined as any drug product, either dispensed on a prescription order or purchased over-the-counter (OTC) that is never fully consumed. Medication wastage may be due to poor compliance of patients, excessive and irrational prescribing, or a lack of control of the sales of prescription medications in the community pharmacy3).
Reducing medication wastage is possible from the perspective of the physicians, patients, politics and society4). There are many causes of medication waste, including excess supply, treatment changes, and patient non-adherence to therapy5, 6). Investigating medication returns may indicate areas for targeting interventions to reduce waste6). Understanding the behaviour of people regarding medicines wastage and the implementation of this information in practice may help to formulate systematic solutions to reduce the waste of medicines. The most studies focused on the patient’s perspectives rather than any other stakeholder, such as healthcare professionals7).
Wastage of medicines is closely related to the type of healthcare system, its financing, and behaviour of patients in it. The Czech Republic has a pluralistic healthcare system with compulsory health insurance for all citizens8). The total expenditure was slightly rising up to EUR 11.5bn (USD 14.4bn) in 2012, i.e. EUR 1,090 (USD 1,372) per 1 inhabitant9). This total expenditure represents 7.63% of the Czech gross domestic product (GDP) in 20129). The structure of healthcare expenditure in the Czech Republic has been almost constant in the long term. In 2012, as in the preceding years, the predominant part of health expenditure was financed by the public health insurance system, which covered 78.8% of the total expenditures on health. The state and territorial budgets covered 5.3% and private expenditure covered 15.9%10). Direct participation of citizens and private health expenditures thus belong to the lowest among all OECD countries11). The share of private expenditure in total increased mainly after 2008 due to new regulation fees (but by only about 2–3%). Public expenditure, i.e. from public budgets and from public health insurance, was EUR 9.63bn (USD 12.13bn)9).
Rough information about consumption of medicines in the Czech Republic (10.5 mil. inhabitants in 2012) is obtained from the State Institute for Drug Control (SIDC) and the Czech Statistical Office12, 13). They acquired the number from the quantity of packages of medicines delivered to pharmacies, healthcare facilities and to vendors of selected medicinal products. In the Czech Republic in 2012 it was 280.19 mil., 5.6% less than in 2011. Drug consumption in defined daily doses (DDD) in 2012 reached 5.94bn. The financial value of distributed medicines in producers’ prices was EUR 2.29bn (USD 2.88bn). This value remained almost the same as in 2011. It is clearly seen that in 2012 the number of distributed packages decreased, but they contained in total more defined daily doses12).
In the last decades the number of medicines in the market has increased several times which resulted in the medicines being a common part of nearly everyone’s life in industrial countries. In most households there can be found several packages of medicines which will never be used. Most often the medicines are not used due to improper use, patient’s death, occurrence of adverse events, treatment modification or excessive supplies. Wasting medicines has an enormous impact all over the world. Unused medicinal products gathered in people’s households represent a medicinal, professional, financial and environmental issue14). Financial losses caused by non-using medicines burden the healthcare system. A whole range of unused medicines indicates a medicinal issue of a great importance. If medicine is left unused, this could lead to worsening symptoms and extra treatments that could have been avoided. Special examples are oral antibiotics, where the whole package is supposed to be used. If they frequently occur among the discarded products, it means that patients did not use them in the right way. That could have been due to insufficient communication between the patient and the doctor or pharmacist. It is important that pharmacists and doctors always explain to the patients the right use of the medicines, what they got it for, why it is important for them to use it and how to dispose of it properly if they do not use it for some reason.
Czech citizens know about free (and voluntary) return of unused drugs back to the pharmacy by providing many targeted public education campaigns, but a quarter to a third of medicines discarded from households end up in bins together with normal waste15).
The analyses concerning the general public’s behaviour when using medicines and handling them in case that they do not use them any more are very important in establishing medicines policies not only for financial reasons but from the point of view of rational pharmacotherapy as well.
Aim of the study
This work follows on a study “The real use of medicines in the Czech Republic” 15), conducted with the participation of the SIDC – medicines agency and regulatory authority in the Czech Republic. The aim of this research is to describe the situation concerning medication wastage in the Czech Republic from the quantitative, qualitative and financial point of view, and their cost to the public healthcare system.
Method
The material analysed was made up of medicines returned to 66 pharmacies in the Czech Republic (33 pharmacies in Brno and 33 in Hradec Králové). The medicines were collected by Pharmacy Students from the Faculties of Pharmacy of the University of Veterinary and Pharmaceutical Sciences Brno (Brno) and Charles University (Hradec Králové). 18 pharma students took part in the collection of the data in the periods from 16th May to 29th June, 2012. They personally checked the contents of the containers designed for collection of unused medicines in a given pharmacy and made up a list, or, more precisely, a specification of their contents to a given form. When making up the list they focused on the name of the medicine, its type, completeness or remaining contents of the package. A similar process and method were used that had been applied in the past by the SIDC in a similar study15).
The preparations returned were divided into the following six groups: medicinal products (human industrially-produced medicinal products), parapharmaceutical products (food supplements, cosmetics, diet products),
veterinary medicinal products, individually-prepared medicinal products, ingredients for individually-prepared medicinal products, discarded products (products where were not stated the strength of the product, size of the package, remaining amount, as well as empty packages or products that could not be found in the used information systems).
The resulting data were analysed and evaluated. A detailed analysis was only performed in the first group (medicinal products – human industrially-produced medicinal products). The other groups were not (regarding their big heterogeneity and small representation) analysed in detail. In these groups only a small number of products was evaluated.
The first group – medicinal products – were processed according to their names, ATC code, authorisation number, total number returned, availability (on prescription/over-the-counter), health insurance reimbursements (yes/no), market price, the health insurance reimbursement and the main active substance.
As information systems were used AISLP (Computerized Information System of Medicines in the Czech Republic) and database of medicines on the website of the SIDC16).
When assessing prices and health insurance reimbursement the “age” of the medicinal products was not considered, as within the circumstances it was not possible to find out when the product was dispensed. All the products were assigned a uniform price (reimbursement) valid in January 2010.
The following parameters were evaluated:
Representation of medicines past the expiration date and medicines before the expiration date – 31st May 2012 was set as the expiration date.
Classification of the medicines for dispensing – whether the medicine is available on prescription or over-the-counter.
Total financial amount approximately spent on the medicinal product.
The following method was employed to calculate the total prices of the unused medicines: Every group of medicinal products packages was assigned a guide price (if the price was not stated, a reserve – maximum price was used). To assess the number of the whole packages, incomplete samples were recalculated.
For calculation of the financial value corresponding with the quantity of the medicines returned to pharmacies in 2011, the method from the SIDC study was chosen15).
The method is based on a study by the SIDC15). It differs in the recalculation of the whole packages and in the price used. The results obtained by the used modified method are similar.
The extrapolation of the results from our survey for the whole country estimation used the methodology and the results from publications of official institutions – the State Institute for Drug Control15) and the Institute of Health Information and Statistics of the Czech Republic10).
The insurance reimbursement that was approximately spent for the medicines.
Classification according to the ATC group and expression of the financial value and the value of the insurance reimbursement for each of the ATC groups.
Most frequently returned prescription medicines, OTC, active substance.
Results
In the material analysed there were 18,890 items in total. These items were divided into six groups. The first group included the medicinal products of the total number of 16,520 (87.5%). The second group contained parapharmaceuticals, of which 359 (1.9%) were returned. The third group was made up of 11 veterinary products (0.1%). The fourth group included individually-prepared products, the number of which makes up 49 (0.3%). The fifth group included 4 raw materials (0.02%). The last group was made up of discarded products in the number of 1,947 (10.3%).
Medicinal products
There were 84.5% products past the expiration date among the total number of the medicinal products returned to pharmacies and only 15.5% were still usable. The medicines dispensed on prescription made up 66.0%, the rest (34.0%) was made up of OTC drugs.
In total, in 988 returned items of the medicinal products the price and insurance reimbursement of the product could not be found, that is why the products were not included in further processing.
The value of the analysed sample was calculated at EUR 82,060 (USD 103,340) and the insurance reimbursement at the sum from EUR 52,960 to 55,970 (from USD 66,700 to 70,480). This span is due to the fact that some of the products could be both dispensed on prescription and sold over-the-counter. Both variants had been expected. The total value of the medicines returned to pharmacies in 2011 was calculated at more than EUR 39.0 mil. (USD 49.1 mil.), which corresponds with the SIDC calculation. Summary is shown in Table 1.
Tab. 1. The total price of the unused medicines 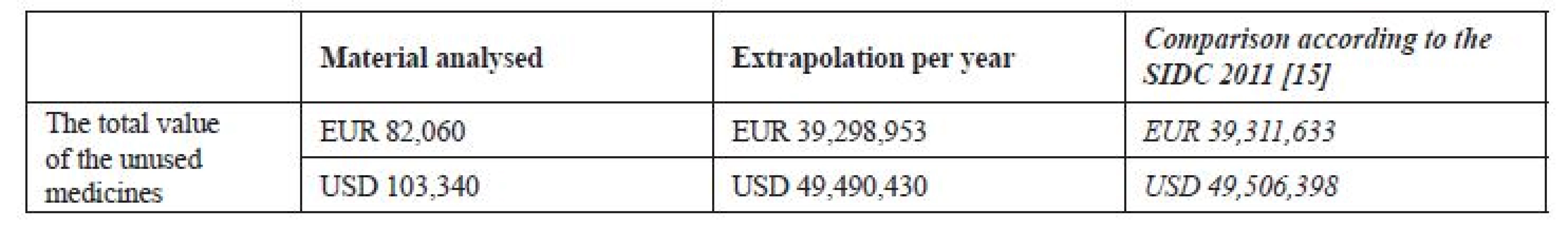
The analysis of the returned medicines according to the therapeutic groups showed that the most often returned products were those acting on the cardiovascular system (16.95%) and the nervous system (15.98%). From the financial point of view, the most resources were spent for unused medicines acting on the nervous system (20.51%). Their value in the analysed group was calculated at EUR 16,828 (USD 21,192), counted per the whole year the sum would reach EUR 8.06 mil. (USD 10.15 mil.). Other costly medicines were those acting on the cardiovascular system (worth EUR 14,950, USD 18,830) and the digestive tract (worth EUR 9,860, USD 12,410). Detailed results and conversion of the financial values of the medicines per the whole year are shown in Table 2.
Tab. 2. The analysis of the returned medicines according to the therapeutic groups (N = 15,532) 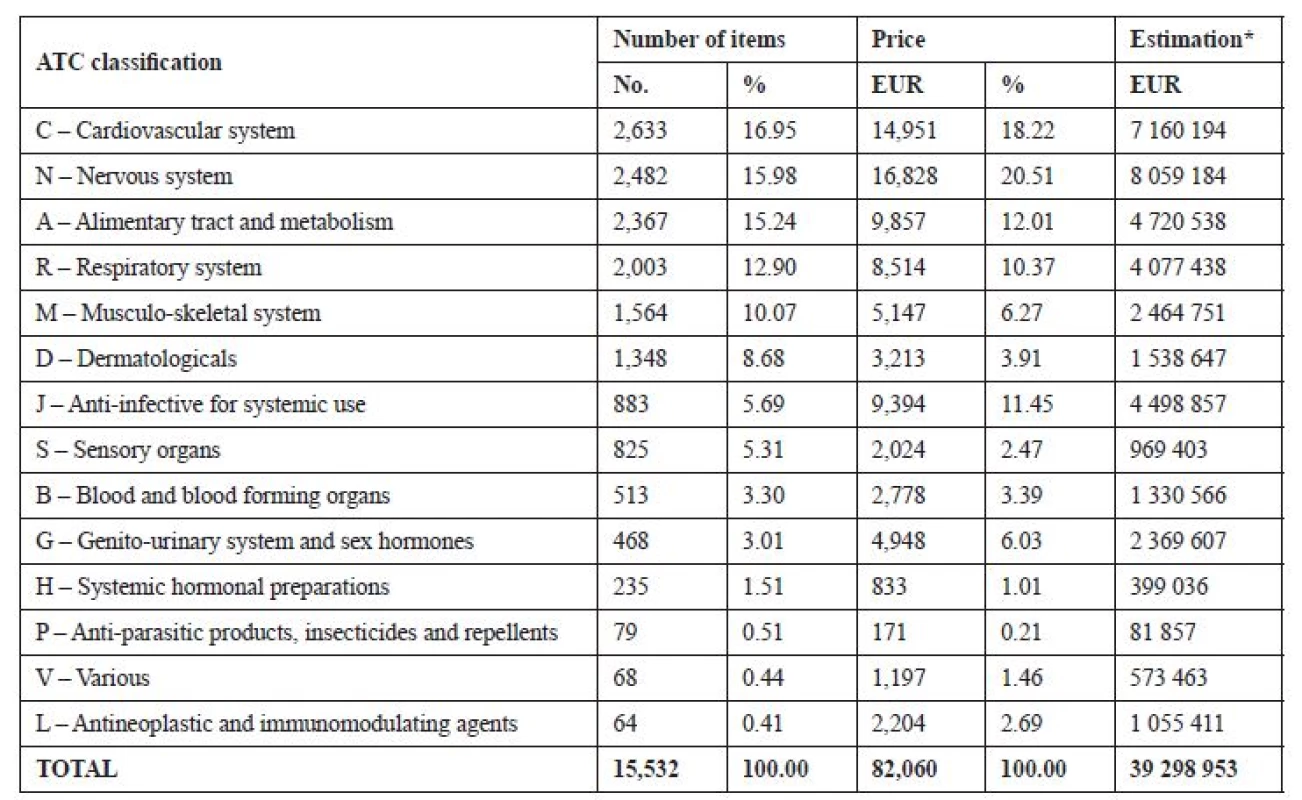
*Extrapolation to the total financial value all returned medicines in the Czech Republic per year. When assessing the frequency of occurrence of a medicinal product, drugs dispensed only on prescription (10,251, i.e. 66%) and OTC medicines (5,281, i.e. 34%) were evaluated separately. The results show that of the prescription drugs the most frequently recurred combined bacitracin-neomycin ointment (brand name Framykoin®), it appeared in total 130 times in the whole group. The detailed results are shown in Table 3.
Tab. 3. Most frequently returned prescription medicines (N = 10,251) 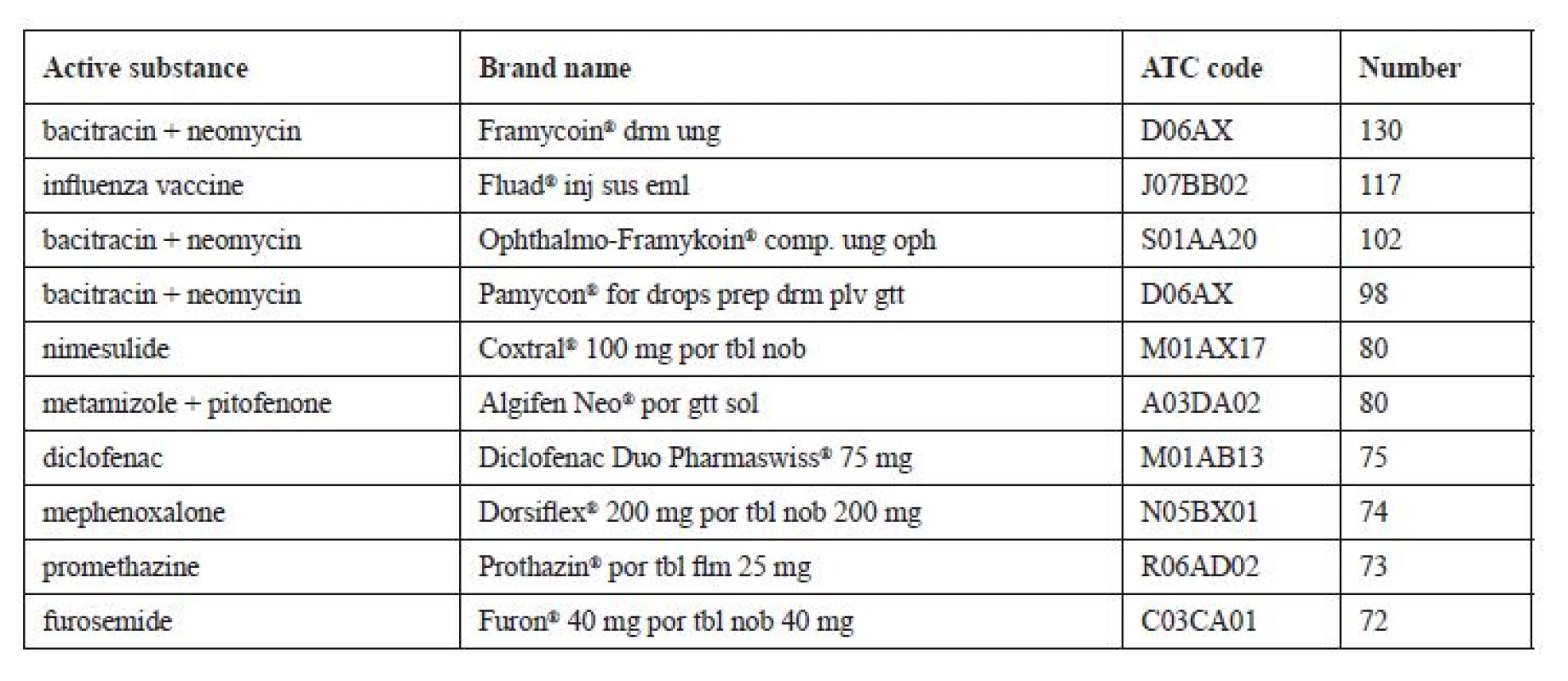
The most often returned OTC medicine was a package of the product with the brand name Paralen® (active substance paracetamol). Altogether it recurred 263 times. Other products are stated in Table 4.
Tab. 4. Most frequently returned OTC medicines (N = 5,281) 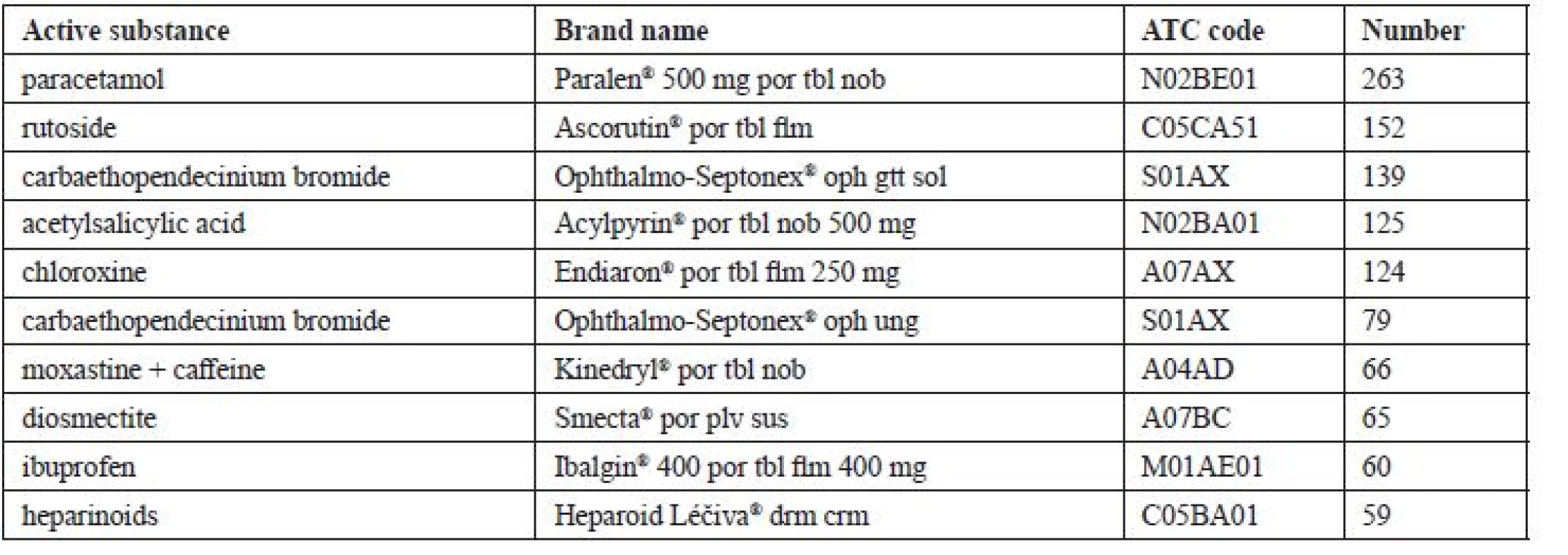
When assessing the most frequently recurring active substances, only products with one main active substance were used to simplify the procedure. Subsequently Table 5 was drawn up, which shows 25 most frequently recurring active substances. Medicinal products that contain as the main active substance one of the substances shown in the Table 5, make up 28% altogether of all the returned products of the analysed group.
Tab. 5. Most frequently unused active substances (N = 15,532) 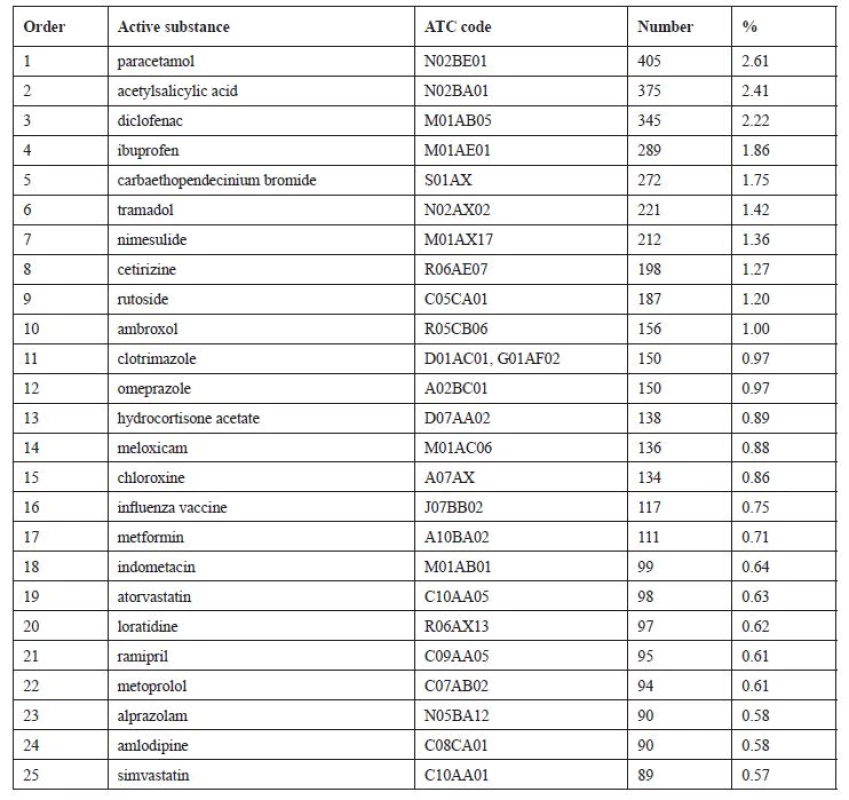
Discussion
For the survey two cities in the Czech Republic were chosen, where there are two pharmaceutical faculties in the country. Hradec Králové (94,000 inhabitants, located in the north-eastern part of Bohemia region), Brno (371,000 inhabitants, located in the southern part of Moravia region). The 33 pharmacies from each city were chosen by random selection.
Besides deliveries to healthcare facilities in the Czech Republic, distributors are also required to provide the SIDC with reports on distribution to foreign countries (to other distributors or healthcare facilities). Data on consumption of medicines from these legal re-exports abroad are adjusted.
For a better understanding and comparison, the values in CZK were converted into EUR and USD at the average exchange rate values at the collection period. According to the Czech National Bank they were 25.64 EUR/CZK and 20.36 USD/CZK. These values correspond only to the reference period17).
The total financial loss for the unused medicines in Czech health care system was estimated by the SIDC at EUR 60.5 mil. (USD 76.1 mil.) in 2011. That means that (according to the financial statement for 2011) in the Czech Republic approx. 2.6% of the total supply of wholesalers to pharmacies was not used (was thrown away or returned)13, 15). According to the assessment, returned to pharmacies were unused drugs worth EUR 39.3 mil. (USD 49.5 mil.) in total, and the estimated amount of pharmaceuticals thrown into the trash along with normal waste or dropped off in collection yards as hazardous waste represented another EUR 15.6 mil. (USD 19.6 mil.)15). Additionally, people handed in to their doctor or nurse medicines worth at least EUR 6.0 mil. (USD 7.6 mil.)15). Besides pharmacies, bins and doctors’ offices, the general public are used to giving medicines to their relatives, or they do not solve the disposal of medicines and gather them at home15).
The results of all the studies on this subject are, in financial expression, always heavily dependent on the current prices of the medicinal products and the chosen methodology of collection and calculation. It is therefore necessary to perceive and compare the analysed values not only in terms of time but also according to the way and methods of collection.
Actual value of unused medicines will probably be even higher. The SIDC used in their calculations just a part of households that throw away medicines into bins or drop them off in collection yards (20.4% of all households). They did not include households that use bins just as one of the ways of unused medicines disposal15).
A significant part of medicinal products (precisely 988 returned items) was not included in the analysis, because it was not possible to find the price of the product and the health insurance reimbursement, the products were too old and not traded for a long time, they were too damaged and illegible, or they were not registered in the Czech Republic at all (they had been bought abroad).
Another study18) from 2001 estimates that the losses for the returned medicines made up 5.4% of the costs on medicines, although in this paper the medicines were not included that had been thrown into the bins (outside pharmacies) or dropped off in collection yards. It can thus be supposed that the comparable quantity in 2001 was even bigger. The two studies lead to the conclusion that the quantity of unused medicines in the Czech Republic is decreasing. It has to be stated, nevertheless, that it is very difficult to assess the exact quantity of the unused medicines and the estimates are always just rough and can be inaccurate. According to various methodologies it can be deduced that the actual value of the unused medicines will always be higher than the analysed and analysable part15).
The most frequently returned products were, in our research, those working on the cardiovascular system (17.0%). These medicines are also the most frequently distributed (62,671,000 were distributed in 2011)19). Most of these products are used over a long period and on a doctor’s recommendation. The reason for not using them can be the death of the patient, adverse events and, according to the results, also probably the wrong use or abuse of these medicines. The cause of non-using could be that some of the cardiovascular diseases like hypertension or hypercholesterolemia do not substantially limit the patient’s life, do not cause any pain, until the consequences occur.
The results of the analysis of the therapeutic groups of the returned medicines can also be compared with the results of studies conducted in New Zealand20) or Spain21). In our study (see Table 2) the most frequently returned were medicines acting on the cardiovascular system (16.95%), the nervous system (15.98%), the digestive tract and metabolism (15.24%) and the respiratory system (12.90%).
In the New Zealand study, it was found that the biggest quantity of the unused medicines is that acting on the nervous system (17%), followed by the digestive system (14%), cardiovascular system (12%), respiratory a musculoskeletal system (11%)20). In a similar study conducted in Spain the top places were occupied by medicines acting on the digestive tract (18.3%), nervous system (18.2%), cardiovascular system (12%) or respiratory system (9%)21). Other results are reported in a Taiwan study. It must be mentioned, however, that they did not use the ATC classification. Here, the biggest part is made up of the medicines acting on the gastrointestinal tract (26%) and digestive system (22.5%). These are followed by anti-inflammatory medicines (12%), antidiabetics (9.5%) and medicines against the common cold (7%)22). In Egypt, then, among the most frequently returned medicines belonged those acting on the cardiovascular system (19.4%) and anti-infective drugs (19.2%)23).
The most frequently returned prescription medicines were very often antibiotic products for local use. Such a high number indicates not evidence-based and unappropriated using and overprescribing. These antibiotics are often inappropriately prescribed for patients with various skin problems not in accordance with evidence-based guideline and recommendations. Another frequent occurrence, influenza vaccines, has caused their short expiration date.
Among seven most frequently unused medicinal substances were six that are used as analgesics (paracetamol, acetylsalicylic acid, diclofenac, ibuprofen, tramadol, nimesulide). This result can partly be considered positive as these substances are not supposed to be used all, given that they are only used to supress symptoms and when the symptoms disappear it is not necessary to take the medicine any longer. In a study conducted in Otago the authors state simvastatin as the most frequently returned substance, which, in our study, is in 25th place. Whereas paracetamol, which is here in the 1st place, in New Zealand takes only the 9th place20).
Prescription medicines made up 66% of the total in our sample, the remaining products (34%) were available over-the-counter. In a study conducted in Vienna, the authors reached relatively similar results. There unused prescription medicines made up 74% and OTC drugs 26%24).
Different states all over the world have different policies regarding household medical waste collection25).
Better communication between the patient and the doctor or pharmacist would surely help to improve the situation in the field of unused medicines. If the patients are not familiar with the right administration, the nature and risk factors of the illness, and the advantages or risks of the treatment, it is more than likely that they will use the medicine in the wrong way or not use it at all. One of the ways is also to dispense some medicines (analgesics) in individual pieces, not in whole packages. Other convenient ways would be to introduce trial prescriptions, especially for medicinal products with a high incidence of adverse events or to give the whole unused and still useable packages of medicines to charitable purposes.
Limitation of the study
There are some limitations of this study. First, the sample size was small (66 pharmacies vs. 2,513 pharmacies and 241 outlets in the Czech Republic in 2012. It makes difficult to extrapolate the results to the general population and to the national level.); second, these findings may not be generalizable to the whole population (this study has been carried out in two university cities); third, data were gathered over an 6-week period from community pharmacies (the return of unused medicines may be subjected to seasonal variations).
Conclusion
Our research analyses the medication wastage in the Czech Republic from the quantitative, qualitative and financial point of view and results of this study confirm the need and importance of collecting data about returned and unused medicines, not only for medicines policy makers and adjustment of the systems of pricing and reimbursement policies, but also for safer and more effective pharmaceutical care and strengthening the function of public pharmacies. The results can help to achieve a better management in the field of drug delivery planning, better compliance, continuous improvement of evidence-based guidelines and their observance, all leading to better practices in the use of medicines.
Moreover, the presence of unused medications in the domiciliary environment may present a public health risk.
Acknowledgements
The authors would like to thank all the students of both faculties of pharmacy involved in data collection.
The study was supported by the Charles University PRVOUK project (P40 – Study of Drug Development), which constitutes a framework for the organizational and institutional funding of research activities at the Faculty of Pharmacy, and by a grant of Charles University (SVV 260 417).
Conflicts of interest: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The grants had no influence on study design, conduction, analysis, interpretation or writing of this article.
doc. RNDr. Jozef Kolář, CSc. (∗)
Department of Applied Pharmacy, Faculty of Pharmacy, University
of Veterinary and Pharmaceutical Sciences Brno, Czech Republic
Palackého tř. 1946/1, 612 42 Brno
e-mail: kolarj@vfu.cz
Received October 12, 2018
Accepted November 8, 2018
Zdroje
1. World Health Organization. Challenges in expanding access to essential medicines. Geneva 2004; Available from: http://apps.who.int/medicinedocs/en/d/Js5571e/2.html#Js5571e.2 (accessed on 12. 09. 2016).
2. Malý J., Doseděl M., Kuběna A., Vlček J. Analysis of pharmacists’ opinions, attitudes and experiences with generic drugs and generic substitution in the Czech Republic. Acta Pol. Pharm. Drug Res. 2013; 70(5), 923–931.
3. Abou-Auda H. S. An economic assessment of the extent of medication use and wastage among families in Saudi Arabia and Arabian Gulf countries. Clin. Ther. 2003; 25(4), 1276–1292.
4. West L. M., Diack L., Cordina M., Stewart D. A focus group based study of the perspectives of the Maltese population and healthcare professionals on medication wastage. Int. J. Clin. Pharm. 2016; 38(5), 1241–1249.
5. Iuga A. O., McGuire M. J. Adherence and health care costs. Risk Manag. Healthc. Policy 2014; 7, 35–44.
6. James T. H., Helms M. L., Braund R. Analysis of medications returned to community pharmacies. Ann. Pharmacother. 2009; 43(10), 1631–1635.
7. West L. M., Diack L., Cordina M., Stewart D. A systematic review of the literature on ‘medication wastage’: an exploration of causative factors and effect of interventions. Int. J. Clin. Pharm. 2014; 36(5), 873–881.
8. Czech Republic. Act on Public Health Insurance. Pub. L. No. 48/1997 Coll. (01. 12. 2011).
9. Institute of Health Information and Statistics of the Czech Republic. Czech health statistics yearbook 2012, Prague 2013; Available from: http://www.uzis.cz/en/system/files/zdrroccz2012.pdf (accessed on 28. 02. 2017).
10. Institute of Health Information and Statistics of the Czech Republic. Economic information on health care 2012, Prague 2013; Available from: http://www.uzis.cz/system/files/ekinf2012.pdf
11. Organisation for Economic Co-operation and Development. Health expenditure and financing, 2017; Available from: http://stats.oecd.org/Index.aspx?DataSetCode=SHA
12. State Institute for Drug Control. Information On Deliveries of Medicinal Products for 2012, Prague 2013; Available from: http://www.sukl.eu/sukl/avizo-k-dodavkam-lecivych-pripravku-za-rok-2012?highlightWords=drug+consumption
13. Czech Statistical Office. Statistical yearbook of the Czech Republic 2013, Prague 2013; Available from: https://www.czso.cz/csu/czso/statistical-yearbook-of-the-czech-republic-2013-653m9thdn1
14. Kolář J., Chybová L., Holečko D. Unused drugs returned to pharmacies I. Ces. slov. Farm. 2001; 50(4), 181–187.
15. State Institute for Drug Control. Real use of medicines in the Czech Republic, Prague 2012; Available from: http://www.sukl.cz/file/73868_1_1 (accessed on 18. 11. 2017).
16. State institute for Drug Control. Medicinal products database, Prague 2015; Available from: http://www.sukl.eu/modules/medication/search.php (accessed on 03. 03. 2017).
17. Czech National Bank. Selected exchange rates, 2012; Available from: http://www.cnb.cz/en/financial_markets/foreign_exchange_market/exchange_rate_fixing/daily.jsp (accessed on 18. 02. 2017).
18. Kolář J., Chybová L., Holečko D. Unused drugs returned to pharmacies II. Čes. slov. Farm. 2001; 50(5), 243–248.
19. Institute of Health Information and Statistics of the Czech Republic. Economic information on health care 2011, Prague 2012; Available from: http://www.uzis.cz/system/files/ekinf2011.pdf
20. Braund R., Chuah F., Gilbert R., Gn G., Soh A., Tan L. Y., Tiong H. S., Yuen Y. C. Identification of the Reasons for Medication Returns. NZ Fam. Physician. 2008; 35(4), 248–252.
21. Coma A., Modamio P., Lastra C. F., Bouvy M. L., Marino E. L. Returned medicines in community pharmacies of Barcelona, Spain. Pharm. World Sci. 2008; 30(3), 272–277.
22. Chien H. Y., Ko J. J., Chen Y. C., Weng S. H., Yang W. C., Chang Y. C., Liuet H. P. Study of Medication Waste in Taiwan. J. Exp. Clin. Med. 2013; 5(2), 69–72.
23. Ibrahim S. Z., Mamdouh H. M., El-Haddad I. Z. Analysis of medications returned to community pharmacies in Alexandria, Egypt. Life Science J. 2012; 9(2), 746–751.
24. Vogler S., Leopold C., Zuidberg C., Habl C. Medicines discarded in household garbage: analysis of a pharmaceutical waste sample in Vienna. J. Pharm. Policy Pract. 2014; 7, 6.
25. Barnett-Itzhaki Z., Berman T., Grotto I., Schwartzberg E. Household medical waste disposal policy in Israel. Israel Journal of Health Policy Research 2016; 5 : 48.
Štítky
Farmacie Farmakologie
Článek Nové knihyČlánek Autorský rejstřík
Článek vyšel v časopiseČeská a slovenská farmacie
Nejčtenější tento týden
2018 Číslo 5-6- Biomarker NT-proBNP má v praxi široké využití. Usnadněte si jeho vyšetření POCT analyzátorem Afias 1
- Psilocybin je v Česku od 1. ledna 2026 schválený. Co to znamená v praxi?
- Ukažte mi, jak kašlete, a já vám řeknu, co vám je
-
Všechny články tohoto čísla
- Co-processed excipients for direct compression of tablets
- Metal complexes in medicine and pharmacy – the past and the present I
- Wastage of medicines and its financial impact on the healthcare system in the Czech Republic
- NMR and IR analysis of natural substances isolated from Cordyceps medicinal mushrooms
- Clinical pharmacist involvement in fall management in a polymorbid geriatric patient with a history of recurrent falls
- Beer with reduced carbohydrates and alcohol content suitable for diabetics
- The beginnings of health libraries of the Czech Brothers Hospitallers in the 18th century
- Emeritní děkan a bývalý předseda ČFS profesor Luděk Jahodář slaví sedmdesátku
- Životné jubileum Márie Kohútovej
- Nové knihy
- Autorský rejstřík
- Česká a slovenská farmacie
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Beer with reduced carbohydrates and alcohol content suitable for diabetics
- Metal complexes in medicine and pharmacy – the past and the present I
- Co-processed excipients for direct compression of tablets
- Clinical pharmacist involvement in fall management in a polymorbid geriatric patient with a history of recurrent falls
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: doc. MUDr. Norbert Pauk, Ph.D.
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MUDr. Irena Krčmová, CSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



