-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
HEMOCOAGULATION DISORDERS IN EXTENSIVELY BURNED PATIENTS: PILOT STUDY FOR SCORING OF THE DIC
Autoři: B. Lipový 1; Y. Kaloudová 1; M. Matýšková 2; M. Penka 2; H. Řihová 1; P. Brychta 1
Působiště autorů: Department of Burns and Reconstructive Surgery, Medical Faculty, Masaryk University Brno, and 1; Department of Clinical Hematology, Medical Faculty, Masaryk University Brno, Czech Republic 2
Vyšlo v časopise: ACTA CHIRURGIAE PLASTICAE, 50, 4, 2008, pp. 115-118
INTRODUCTION
Disseminated intravascular coagulation (DIC) is a syndrome accompanying serious conditions and in 82.9% it is part of systemic inflammatory response syndrome (SIRS). Changes in the system of blood coagulation play an important role in the development of multi-organ dysfunction syndrome – MODS (1). Disseminated intravascular coagulation can be caused by various clinical conditions, for example sepsis, trauma, organ dysfunction, malignant changes, pregnancy complications, vascular abnormality, severe liver failure, severe poisoning, fat embolism and many others.
In 2001 the Scientic and Standardisation Committee (SSC), International Society on Thrombosis and Haemostasis (ISTH) divided its Disseminated intravascular coagulation into: non-overt DIC (indolent, compensated, “low grade” form), which is characterized by mild hemostatic dysfunction with mainly hypocoagulation trend, and overt DIC (acute, fulminant, decompensated, “high-grade”) with dominating hypercoagulation status (2).
Homeostasis of the hemocoagulation system is during the burn trauma more or less impaired practically always. Extensive burn trauma leads to activation of coagulation, decrease of fibrinolytic activity and decrease of natural anticoagulation activity (3).
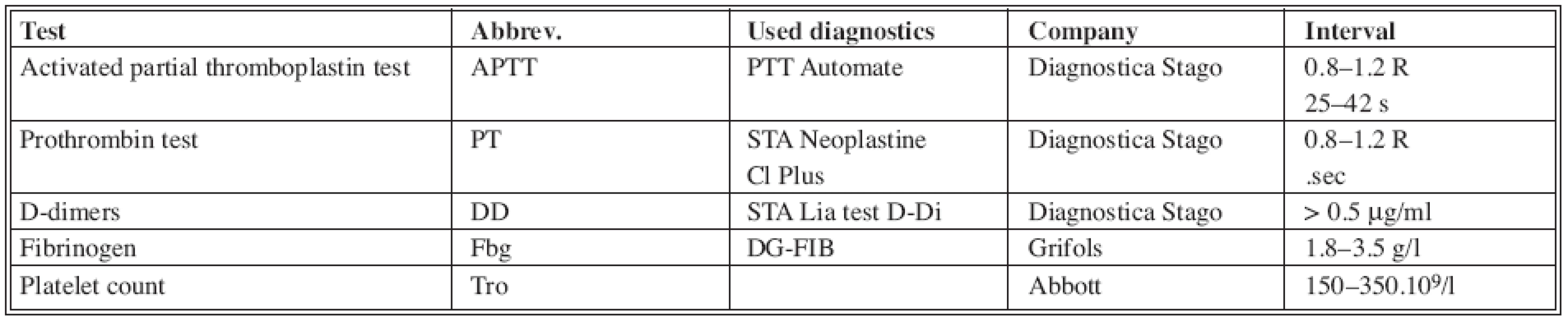
With such rapid development of more exact and specific laboratory methods, evaluating separate factors of hemocoagulation, we can better explain impending, incipient or already ongoing hemocoagulation disorders (in means of increased bleeding or thrombosis). That allows a renaissance of evaluation and foremost dynamics of growth or decrease of the D-dimers (soluble fibrin monomers) values in circulation. Laboratory evaluation at the Faculty Hospital in Brno uses the methods ELISA and LIA. For our patients we have used the method LIA. D-dimers are not specific enough for prediction of the TED, because values of D-dimers can also be elevated during increased physical activity, insignificant infections, viral infections, SIRS, sepsis or physiologically with aging (4).
MATERIAL AND METHODS
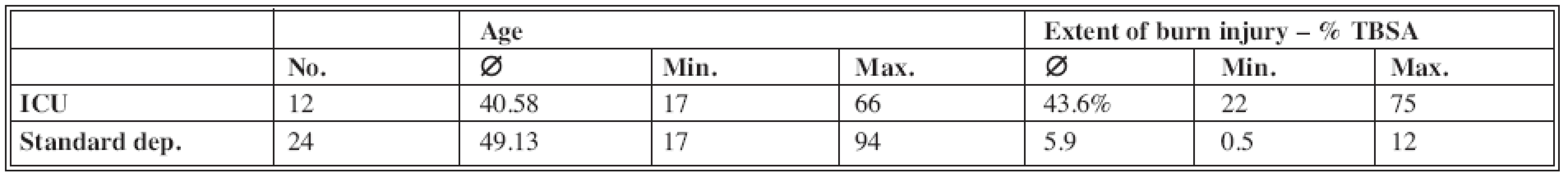
In our pilot study we have included total of 36 patients with various extent of burn injury. They were hospitalized at the Intensive Care Unit (ICU) as well as at Standard Unit (Tab. 1). See the blood tests we used in Tab. 2 (5). In these chosen patients we have established decisive days: the day of injury, the 5th day after the injury, the day after the autotransplantation, (or 8th day of hospitalization if the autotransplantation was not indicated in deep 2nd degree burns – deep partial-thickness skin destruction)) and at the day of discharge. In these days, series of hemocoagulation blood tests were done which were eventually evaluated according to the scheme recommended by ISTH for overt and non-overt DIC (Tab. 3, 4). Value of the overt DIC score above 5 meant positivity i.e. the patient would have acute form of DIC.
Tab. 3. Scoring system for overt disseminated intravascular coagulation (DIC) (6) 
Tab. 4. Scoring system for non-overt disseminated intravascular coagulation (DIC) (6) 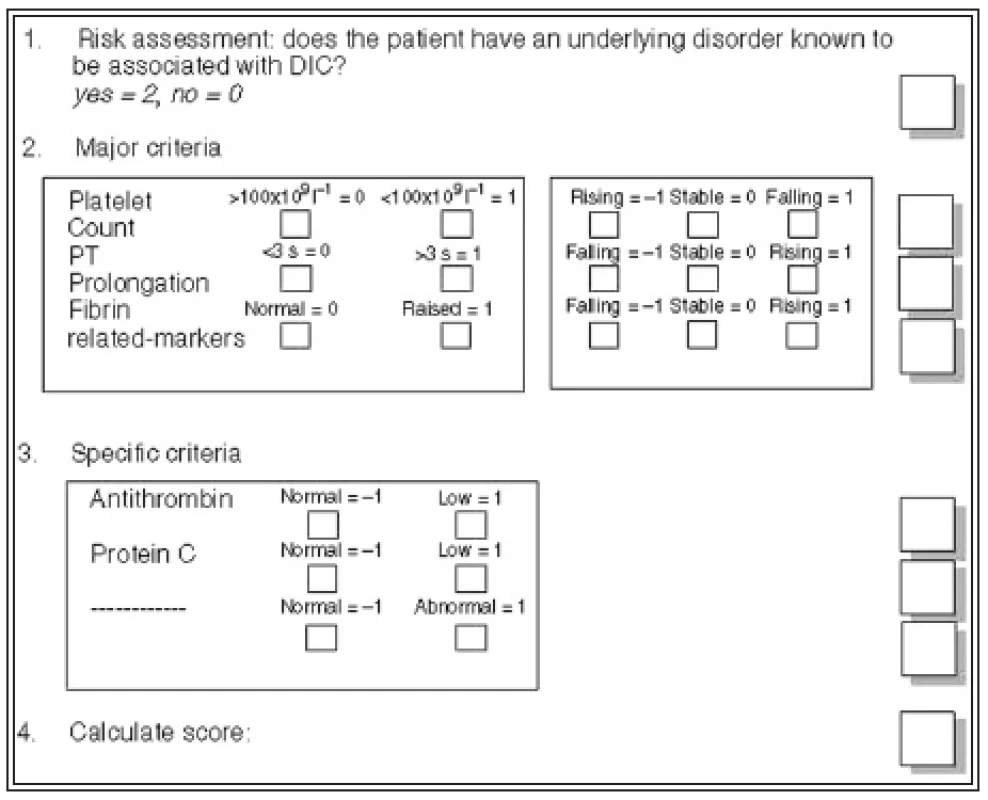
RESULTS
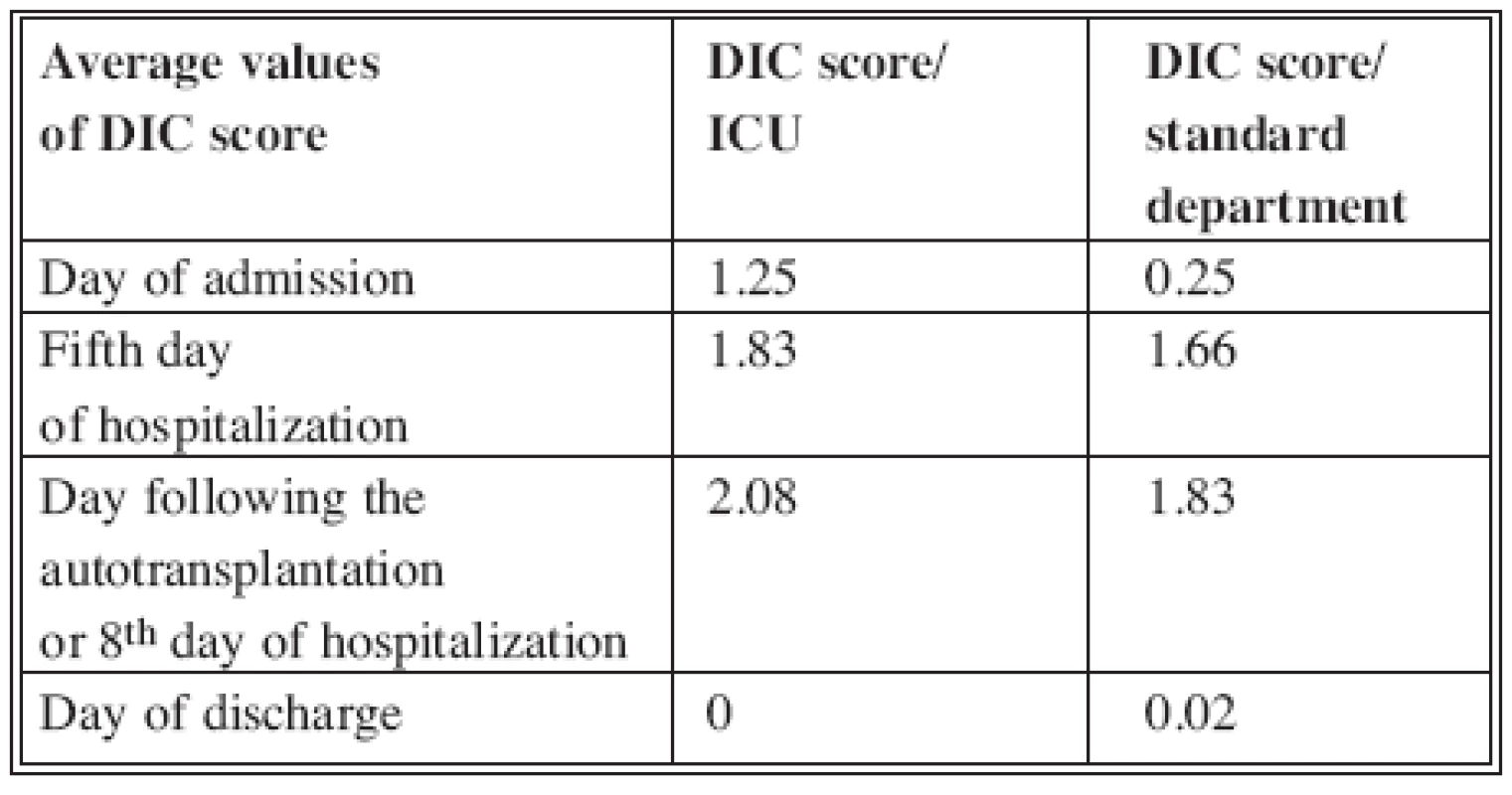
First part of the study included 12 patients hospitalized at our ICU. The average value of D-dimers on the day of admission was 2.06 μg/ml (0.27–7.49), on the 5th day of the hospitalization was the average value of D-dimers 2.32 μg/ml (0.45–5.27), the day after the autotransplantation was the value of D-dimers 4.18 μg/ml (0.78–6.97) and at the day of discharge 0.64 μg/ml (0.22–0.96) (Graph 1). Only one patient of 12 in this group died following a septic shock. On the day of injury the value of overt DIC score reached 1.25 (0–3). Fifth day after the injury the average value of DIC score was 1.83 (0–3). Day after the autotransplantation it was 2.08 (0–3) and on the day of discharge it was 0 (Tab. 5). The DIC score reached 2 in the deceased patient.
Second part of the study included 24 patients hospitalized at the Standard Unit of our Department. The average extent of burn injury in these patients was 5.9% TBSA (0.5–12%) and the average age was 49.13 years (17–94 years). At the day of admission the average value of D-dimers was 0.51 μg/ml (0.07–1.93). On the 5th day of hospitalization the average value of the D-dimers was 1.37 μg/ml (0.88–1.77), on the day of autotransplantation or the 8th day of hospitalization was the value of D-dimers 1.72 μg/ml (0.35–4.31) and at the day of discharge it was 0.92 μg/ml (0.65–1.08) (Graph 2). On the day of the injury the value of overt DIC score reached 0.25 (0–2), the day after the autotransplantation it was 1.83 (0–3) and on the day of discharge 0.02 (0–2) (see Tab. 5).
Graph 1. D-dimers values in patients hospitalized at the Intensive Care Unit 
Graph 2. D-dimers values in patients hospitalized at the standard department 
DISCUSSION
For the scoring of DIC it is important to establish values of D-dimers in μg/ml, which would be considered as mid or high growth of the D-dimers level. For these purposes, Dempfle et al. defined stratification, which we also used for our scoring. Values of D-dimers 0–1 μg/ml are evaluated by 0 points, values 1–3 μg/ml are evaluated by 2 points and growth above 3 μg/ml then by 3 points (7). In 2001 the ISTH issued a recommendation for the frequency of blood taking and also evaluation of the DIC score, which in patients with high risk should be completed daily. Therefore the basic principle of DIC scoring is the evaluation of dynamics of growth or decrease of the obtained values.
CONCLUSION
The ISTH overt DIC score system can assist in more accurate diagnostics in patient with severe sepsis and multi-organ failure. According to the literature results of the scoring clearly correlate with mortality, particularly in septic patients. Growth of the overt DIC score during sepsis means high probability of other organ involvement within the multi-organ failure and decreases the probability of survival. We have not found any elevated overt DIC score (over 5) in any of our patients, which means that we did not confirm ongoing acute DIC. DIC score can be an interesting promise for objectification of impending and ongoing disorders of hemocoagulation; can lead to a purposeful prevention and faster therapeutic intervention, which can reverse the unfavorable prognosis of the patient. Of course, it does not mean that burned patients are out of the life-threatening situation due to DIC, but more likely, it can assist in the therapy management of burn trauma patient by the adequate administration of antithrombotic doses of heparin and, in the indicated cases, substitution of hemocoagulation factors; non-specifically (by administration of fresh frozen plasma) or specifically (for example substitution of Antithrombin). The care of these patients must always be a result of multidiscipline cooperation of a specialist in burns, hematologist, microbiologist and anesthesiologist.
Address for correspondence:
B. Lipový, M.D.
Department of Burns and Reconstructive Surgery,
Medical Faculty,
Masaryk University Brno
Komenského nám. 2
662 43 Brno
Czech Republic
E-mail: B.Lipovy@seznam.cz
Zdroje
1. Penka M., Buliková A., Matýšková M., Zavřelová J. Diseminovaná intravaskulární koagulace. Brno: Avicenum GRADA, 2003, p. 17–71.
2. Taylor FB. Jr., Toh CH., Hoots WK., Wada H., Levi M. Towards denition, clinical and laboratory criteria, and a scoring system for disseminated intravascular coagulation on behalf of the Scientic Subcommittee on Disseminated Intravascular Coagulation of the International Society on Thrombosis and Haemostasis. Thromb. Haemost., 86, 2001, p. 1327–1330.
3. Lavrentieva A., Kontakiotis T., Bitzani M., Parlapani A., Thomareis O., Scourtis H., Tsotsolis N., Lazaridis L., Giala M-A. The efficacy of antithrombin administration in the acute phase of burn injury. Tromb. Haemost., 100, 2008, p. 286–290.
4. Herndon DN. Total Burn Care.: Saunders ELSEVIER, 2007, p. 325–342.
5. Lehman CM., Wilson LW., Rodgers GM. Analytic validation and clinical evaluation of the STA LIA TEST Immunoturbidimetric D-dimer assay for the diagnosis of disseminated intravascular coagulation. Am. J. Clin. Pathol., 122(2), 2004, p. 217–221.
6. Toh CH., Hoots WK., on behalf of the SSC on Disseminated Intravascular Coagulation of the ISTH. The scoring system of the Scientific and Standardisation Committee on Disseminated Intravascular Coagulation of the International Society on Thrombosis and Haemostasis: a 5-year overview. J. Thromb. Haemost., 5, 2007, p. 604–606.
7. Dempfle CE., Wurst M., Smolinski M., Lorenz S., Osika A., Olenik D., Fiedler F., Borggrefe M. Use of soluble fibrin antigen instead of D-dimer as fibrin-related marker may enhance the prognostic power of the ISTH overt DIC score. Thromb. Haemost., 91, 2004, p. 812–818.
8. Königová R. et al. Komplexní léčba popálenin. Praha: Grada, 1999, p. 153–200, 205–215, 240–245.
Štítky
Chirurgie plastická Ortopedie Popáleninová medicína Traumatologie
Článek vyšel v časopiseActa chirurgiae plasticae
Nejčtenější tento týden
2008 Číslo 4- Trombóza portální žíly jako komplikace infekce COVID-19 – kazuistika
- Bezpečnostní profil metamizolu – systematický přehled
- Spasmolytický účinek metamizolu
-
Všechny články tohoto čísla
- CONGRESS HISTORY OF THE INTERNATIONAL SOCIETY OF BURN INJURIES
- LATE COMPLICATION OF RESPIRATORY THERMAL INJURIES
- ENZYMATIC NECROLYSIS OF ACUTE DEEP BURNS – REPORT OF PRELIMINARY RESULTS WITH 22 PATIENTS
- HEMOCOAGULATION DISORDERS IN EXTENSIVELY BURNED PATIENTS: PILOT STUDY FOR SCORING OF THE DIC
- ČESKÉ SOUHRNY
- Contens
- INDEX
- ETHICAL PROBLEMS IN BURN MEDICINE
- Acta chirurgiae plasticae
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- ENZYMATIC NECROLYSIS OF ACUTE DEEP BURNS – REPORT OF PRELIMINARY RESULTS WITH 22 PATIENTS
- CONGRESS HISTORY OF THE INTERNATIONAL SOCIETY OF BURN INJURIES
- HEMOCOAGULATION DISORDERS IN EXTENSIVELY BURNED PATIENTS: PILOT STUDY FOR SCORING OF THE DIC
- ETHICAL PROBLEMS IN BURN MEDICINE
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Autoři: prof. MUDr. Pavel Horák, CSc., doc. MUDr. Ludmila Brunerová, Ph.D., doc. MUDr. Václav Vyskočil, Ph.D., prim. MUDr. Richard Pikner, Ph.D., MUDr. Olga Růžičková, MUDr. Jan Rosa, prof. MUDr. Vladimír Palička, CSc., Dr.h.c.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání