-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
Top novinky
ReklamaLong-term trends in mortality and AIDS-defining events after combination ART initiation among children and adolescents with perinatal HIV infection in 17 middle- and high-income countries in Europe and Thailand: A cohort study
In a multi-country cohort study, Ali Judd and colleagues study morbidity, mortality, and relevant risk factors in children and adolescents with HIV infection receiving antiretroviral treatment.
Published in the journal: . PLoS Med 15(1): e32767. doi:10.1371/journal.pmed.1002491
Category: Research Article
doi: https://doi.org/10.1371/journal.pmed.1002491Summary
In a multi-country cohort study, Ali Judd and colleagues study morbidity, mortality, and relevant risk factors in children and adolescents with HIV infection receiving antiretroviral treatment.
Introduction
Studies have reported declining mortality since the introduction of combination antiretroviral therapy (cART) about 20 years ago, both in adults and children [1–3]. For example, in the United Kingdom (UK) and Irish nationwide paediatric cohort, among 1,441 children with median age 9 years at last follow-up, mortality rates declined from 8.2 per 100 person-years before 1996 to 0.6 in 2003–2006 [1]. In an Italian multicentre study, survival of children with perinatal HIV significantly improved between 1996 and 1998, following the introduction of cART [4]. Similarly, in a United States cohort of 3,553 children with median age at enrolment of 6 years and median follow-up of 5 years, mortality rates declined from 7.2 to 0.8 per 100 person-years between 1994 and 2000 and remained relatively stable to the end of 2006 [2]. Mortality rates in adults have shown a similar trend, and life expectancy among those with a CD4 count ≥500 cells/mm3 is now thought to be close to that of the general population [5].
However, AIDS is now a top 10 leading cause of death in adolescents globally [6], and evidence suggests worsening outcomes as children with HIV grow up and transition to adult care. European data from individual cohorts suggest that young people with perinatal HIV infection have a higher risk of treatment failure [7–10], care disengagement [11], and death [12] compared to those who acquire HIV in adulthood and children with other routes of transmission. Thus, there is the need for vigilance to ensure that health outcomes in this population who have survived paediatric HIV are maximised as they enter adulthood.
Longitudinal cohort studies are well placed to measure changes in mortality rates over time. However, due to relatively limited numbers of children with perinatal HIV infection in some European countries [13], pooling of data across cohorts is the only practical way of obtaining reliable mortality rates across the European region. The European Pregnancy and Paediatric HIV Cohort Collaboration (EPPICC) is a network of 19 cohorts across 17 countries in Western and Central Europe (W&CE), Eastern Europe (Russia, Ukraine), and Asia (Thailand). In this study, data from these cohorts were used to describe rates and risk factors for mortality and AIDS-defining events in perinatally HIV-infected children and adolescents after initiating cART.
Methods
This study was carried out in accordance with the EPPICC Paediatric merger 2014 SOP and the project-specific Concept Sheet (S1 Text). Nineteen cohorts across 17 countries contributed to individual patient data (see S2 Text: “Writing Group members and collaborating cohorts” for list of collaborators). Children were included in this analysis if they had perinatal HIV infection and initiated cART (defined as a ≥3 drug, ≥2 class regimen [excluding unboosted PIs] or ≥3 non-nucleoside reverse transcriptase inhibitor [NNRTI]-only regimen, including abacavir) after 1996 (with no prior antiretroviral therapy [ART] use), aged <18 years. They were at risk from cART initiation until their censor date, defined as the earliest of 21st birthday, last visit in paediatric care, death, or loss to follow-up (as defined by each cohort), with data available until 31 December 2013. Demographic, clinical, laboratory, and treatment-related data from routine clinic visits (typically every 3–6 months) were pooled electronically using a modified HICDEP protocol (www.hicdep.org). Pooled data were subjected to a battery of consistency checks. Data on all children at participating clinics were included; data are pseudo-anonymised, and therefore individual informed consent was not obtained. All cohorts received approval from local and/or national ethical committees. For example, the UK/Ireland CHIPS cohort had approval from the London Central Research Ethics Committee.
Causes of death, along with CD4 and clinical event data, were reviewed by the Project Team and confirmed by the reporting clinician. Cause of death was coded using the International Classification of Diseases version 10 and categorised into 4 groups: HIV-related infectious causes, other HIV-related causes, deaths not directly related to HIV, and those with unknown cause. AIDS events were considered to be reliably reported, as they are an important clinical indicator of disease progression for children in routine care.
Statistical methods
Rates of death and first AIDS-defining event were calculated per 100,000 person-years. Rates were summarised overall, within 6 months of ART initiation (‘early’; children were at risk from ART initiation to ART initiation plus 6 months), and after 6 months of ART (‘late’; at risk from ART initiation plus 6 months to their censor date), and rates of first AIDS-defining event were additionally summarised by event. Early deaths and AIDS-defining events were summarised separately, as they were likely to be related to late presentation/initiation of cART. AIDS-defining events were classified according to the US Centers for Disease Control and Prevention 2014 surveillance criteria [14]. Cohorts reported date of AIDS diagnosis as the date of the child’s earliest WHO stage 3/4 or US Centers for Disease Control (CDC) stage C event. Continuous variables were compared using Wilcoxon’s rank-sum test and categorical variables using a chi-squared test.
Risk factors for early death, late death, early first AIDS-defining event, and late first AIDS-defining event were assessed using univariable and multivariable proportional hazard models with inverse-probability-of-censoring weights. The probability of being censored was estimated using logistic regression (including all factors listed below) to account for informative censoring in those lost to follow-up. Weights were calculated as the reciprocal of these estimated probabilities. Association with the following factors at cART initiation were considered: age (continuous), sex, ethnicity (black African, Asian, other, unknown), year of birth (continuous), born abroad (versus in country of cohort), country group (Eastern Europe and Thailand [EE&T] [Russia/Ukraine/Thailand]) versus W&CE (all others), previous AIDS diagnosis (yes, no), year of cART initiation (continuous), initial regimen (NNRTI-based, other), World Health Organization (WHO) severe immune suppression for age (defined as a CD4 lymphocyte percentage [CD4%] <25% for children <1 year of age, <20% for children aged 1–3 years, <15% for children aged 3–5 years, and <200 cells/mm3 or <15% for children aged ≥5 years [15]), viral load (≤100,000 c/mL, >100,000 c/mL), and BMI-for-age z-score (>0, 0–−3, <−3, calculated using WHO child growth standards, 2007).
Time-updated factors considered for late death and first AIDS event were age, severe immune suppression for age, HIV viral load, BMI-for-age z-score, and proportion of time suppressed since cART initiation (≥80% versus <80%, as a proxy for adequate adherence). Values for time-updated factors based on clinical/laboratory measurements were considered to occur on the date recorded and were carried forward for up to 6 months (if no subsequent measurement was recorded), after which they were considered to be unknown. Individuals with missing data for any given factor were classified as missing, with a separate level of the variable (the missing indicator method).
Nonlinear effects of continuous variables were explored using natural cubic splines with 5 knots, and in final models, where appropriate, statistically significant nonlinearity was represented by piecewise linear functions. All characteristics (excluding those that were highly correlated) were included in a multivariable model. Covariate interactions between country group, calendar year of ART initiation, and any other statistically significant (p < 0.05) characteristics were considered and the proportional hazards assumption assessed.
BMI-for-age z-score was chosen rather than weight-for-age z-score, as WHO has normative data across all ages for BMI for age (WHO reference data for weight are only available for children aged <10 years), and to minimise differences due to variation in height for age between ethnic groups in the general population. A sensitivity analysis used weight-for-age z-score based on UK normative data for Western, Central, and Eastern European cohorts and Thai normative data for the Thai cohort. Analyses investigating rates and risk factors for early and late AIDS events separately were not prespecified but conducted following discussion of the results from a combined analysis investigating overall rates of AIDS events. Statistical analyses were performed using Stata version 14.2 (Stata Corporation, College Station, Texas). STROBE recommendations were followed (S1 STROBE Checklist).
Results
Of 3,953 HIV-infected, ART-naïve children in EPPICC at the time of initiating cART, 3,526 (89%) acquired HIV perinatally and were eligible for this analysis; 1,124 (32%) were from the UK or Ireland, 700 (20%) from Thailand, 508 (14%) from Ukraine, and 1,194 (34%) from elsewhere across Europe (Table 1). The median age at cART initiation for the 3,526 participants was 5.2 (IQR 1.4–9.3) years, and the median year of cART initiation was 2006 (2003–2009). Median duration of follow-up was 5.6 (2.9–8.7) years, with 20,574 person-years of follow-up overall (of which 4,531 were of participants aged <5 years, 6,836 aged 5–<10 years, 6,577 aged 10–<15 years, 2,139 aged 15–<18 years, and 512 aged ≥18 years).
Tab. 1. Characteristics of children overall, and by timing of death, for those who died. 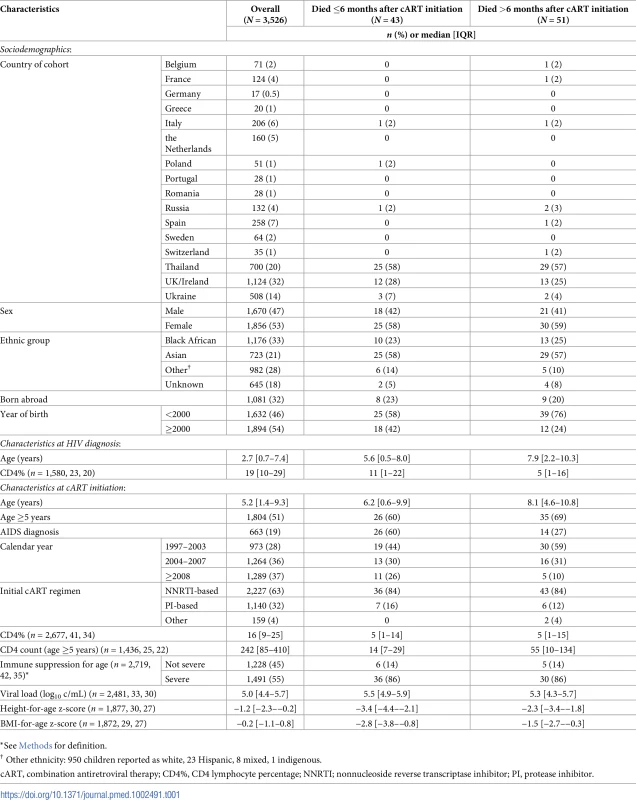
*See Methods for definition. At cART initiation, 1,491/2,719 (55%) children overall had severe immune suppression for their age (466/555 [84%] for Thailand) and 663 (19%) had a previous AIDS diagnosis. The proportion of children aged <5 years at ART initiation who had a CD4% less than 15% declined over the study period, from 35% (134/381) in 1997–2003 to 15% (24/157) from 2011 onwards (p < 0.001) (Fig 1), and similarly, the proportion aged ≥5 years with a CD4 count <200 cells/mm3 at cART initiation declined from 53% (194/365) to 18% (36/196) (p < 0.001).
Fig. 1. CD4% (left panel) and count (right panel) at initiation of cART, by age group and calendar period. 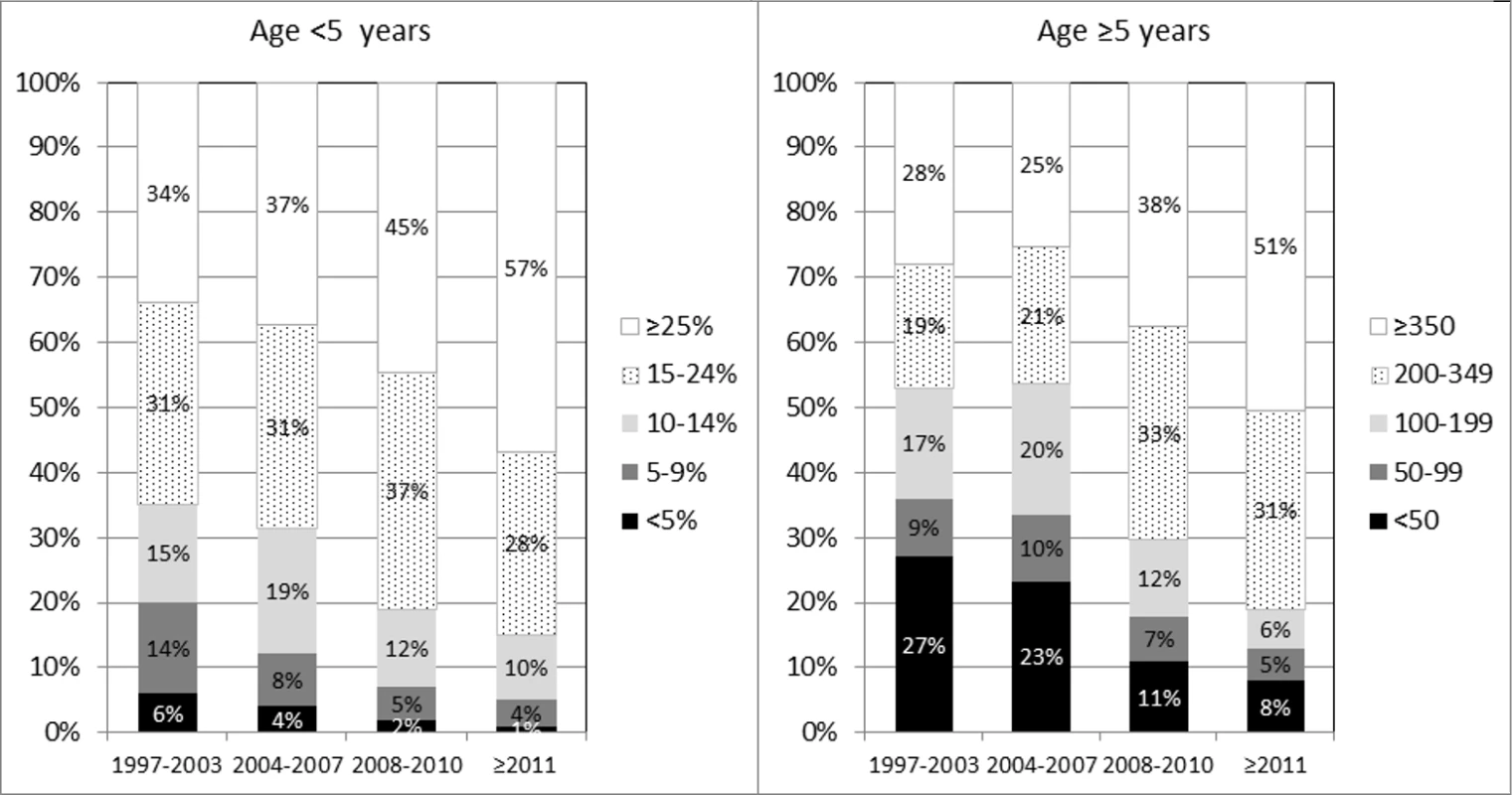
cART, combination antiretroviral therapy; CD4%, CD4 lymphocyte percentage. By the end of the study period, 94 (3%) children had died, 453 (13%) had been lost to follow-up, 305 (9%) had transferred to adult care, 314 (9%) had dropped out for other reasons (including having moved to another clinic or withdrawn consent), and 2,322 (66%) were still in follow-up. Among those still in paediatric care, the median age at last visit was 11.6 (7.6–15.0) years.
Causes of death
There were 94 deaths, of which 43 (46%) were within 6 months of cART initiation. Among the early deaths, 37% (16/43) occurred within 1 month and 79% (34/43) within 3 months of cART start. The crude mortality rate was 2,502 (95% CI 1,856–3,374) per 100,000 person-years in the first 6 months of treatment and 270 (206–356) thereafter. Fig 2 shows the decreasing risk of death after the first 6 months of cART, in all calendar periods and country groups. The probability of survival to 5 years after initiating cART was 97.6% (97.0%–98.1%) overall and improved with calendar period from 96.2% (94.8%–97.2%) in 1997–2004 to 98.4% (97.2%–99.1%) for 2008 onwards. Overall, it was 98.7% (98.0%–99.1%) in W&CE countries and 95.8% (94.4%–96.8%) in EE&T countries. The mortality rate was highest in earlier calendar years, peaking at 1,773 (95% CI 1,030–3,054) per 100,000 person-years in 2003 and decreasing to 360 in 2006, after which it was relatively stable at between 122 and 435 cases per 100,000 person-years (Fig 3), with a similar trend for deaths both before and after 6 months of cART (S1 Fig). The peak in mortality rate in 2003 coincided with the introduction of cART in Thailand, and Thailand accounted for over half of the deaths in the periods for both early and late deaths (Table 1).
Fig. 2. 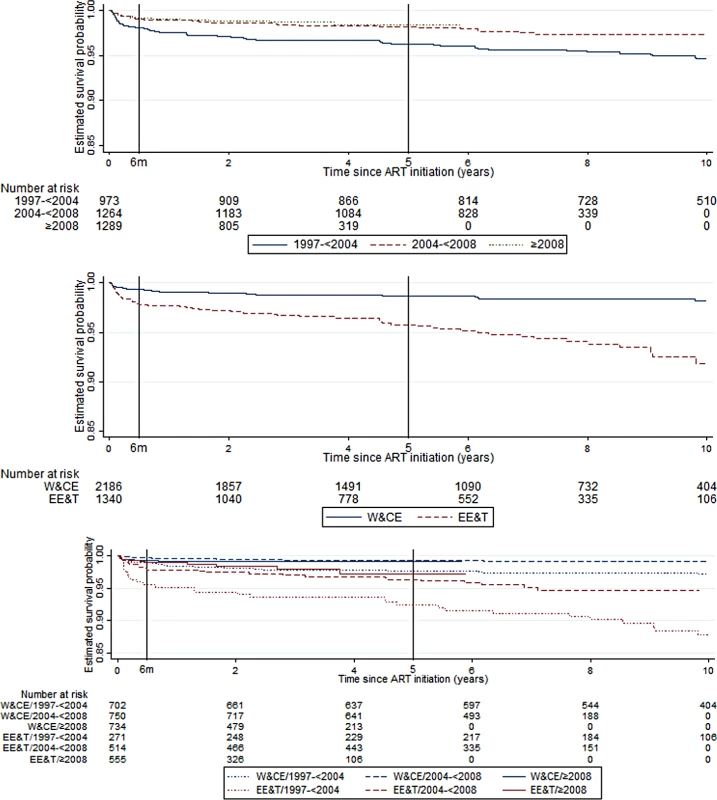
Kaplan-Meier survival curve for time from cART initiation to death (A) by calendar period of cART initiation, (B) by country group*, and (C) by calendar period and country group. ART, antiretroviral therapy; cART, combination antiretroviral therapy; EE&T, Eastern Europe and Thailand; W&CE, Western and Central Europe. Fig. 3. Mortality rates (95% CI) by calendar year of follow-up, 2000–2013. 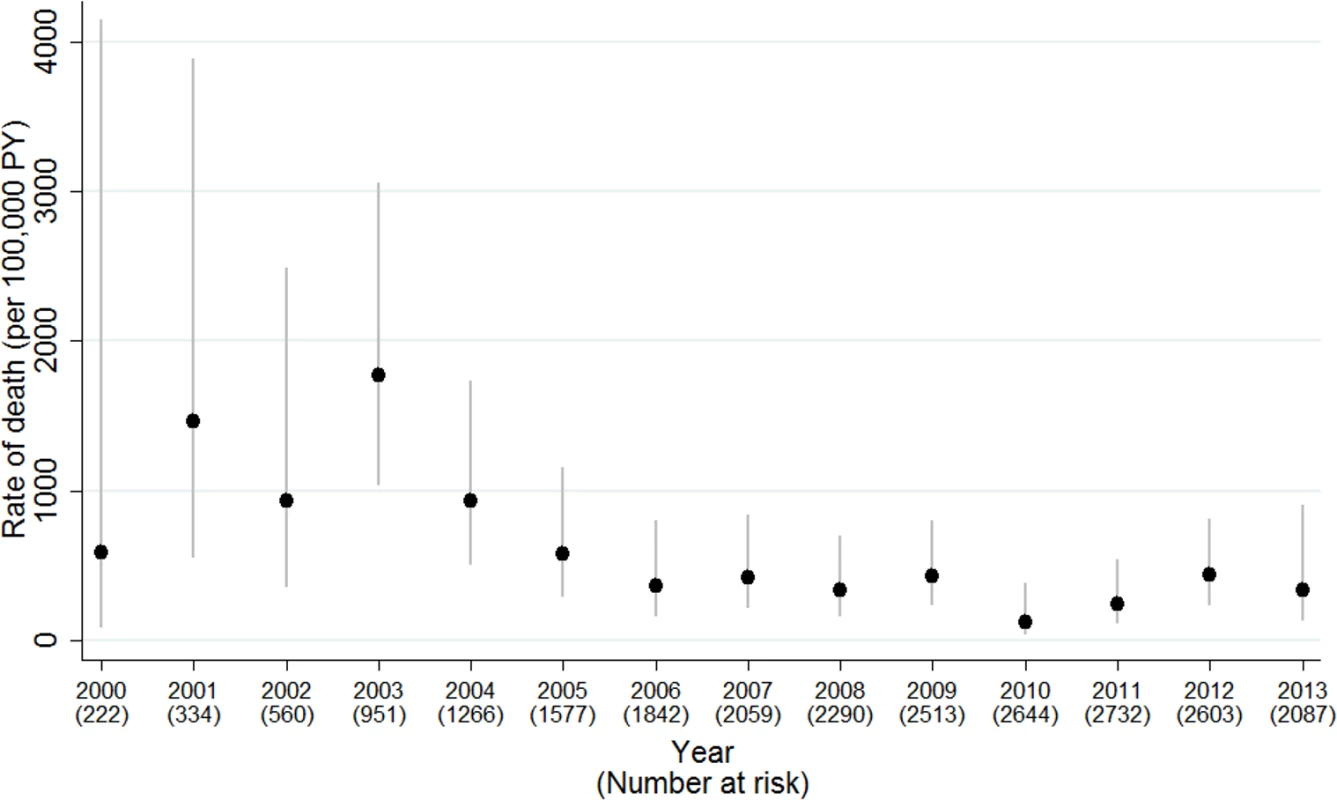
The median age at death was 9.5 (IQR 3.3–13.9) years; those who died within 6 months of cART initiation were younger than those who died after 6 months (median age 6.5 versus 12.8, p < 0.001). Among those who died after 6 months of cART, the median (IQR) viral load was 9,393 (400–107,654), CD4% was 6% (2%–24%), and CD4 count (among children aged ≥5 years, n = 26) was 68 (9–304) cells/mm3. Twelve (24%) children with late deaths were not taking cART at the time of death, and a further 4 (8%) had a treatment interruption of >30 days in the 6 months prior to death.
Overall, 58 (64%) deaths were due to HIV-related infections, predominantly bacterial infection and/or sepsis (Table 2), and these accounted for 76% (31/41 with cause available) of early deaths and 55% (27/49) of late deaths (p = 0.043). Twenty-four deaths (27%) were from other HIV-related causes (22% versus 31% early versus late, p = 0.355), 8 (9%) were not directly related to HIV (2% versus 14%, p = 0.049), and 4 causes were unknown. Over the calendar periods 2000–2004, 2005–2008, and 2009 onwards, the proportions of deaths caused by HIV-related infections (19/30 : 63%, 21/29 : 72%, and 18/31 : 58%, respectively) and other HIV-related causes (10/30 : 33%, 3/29 : 10%, and 11/31 : 35%, respectively) did not significantly change (p = 0.504 and p = 0.053, respectively). Overall, 45 (50%) deaths were AIDS related (59% of early deaths versus 43% later, p = 0.138), with no discernible trend by calendar year period (8/30 : 27%, 15/29 : 52%, 11/31 : 35%) or country group (EE&T: 30/59, 51%; W&CE: 15/31, 48%; p = 0.824). Twenty-five (27%) children who died did not have an AIDS-defining illness at the time of their death; their most common cause of death was bacterial infection/sepsis.
Tab. 2. Causes of death for children who died, overall, and by timing of death. 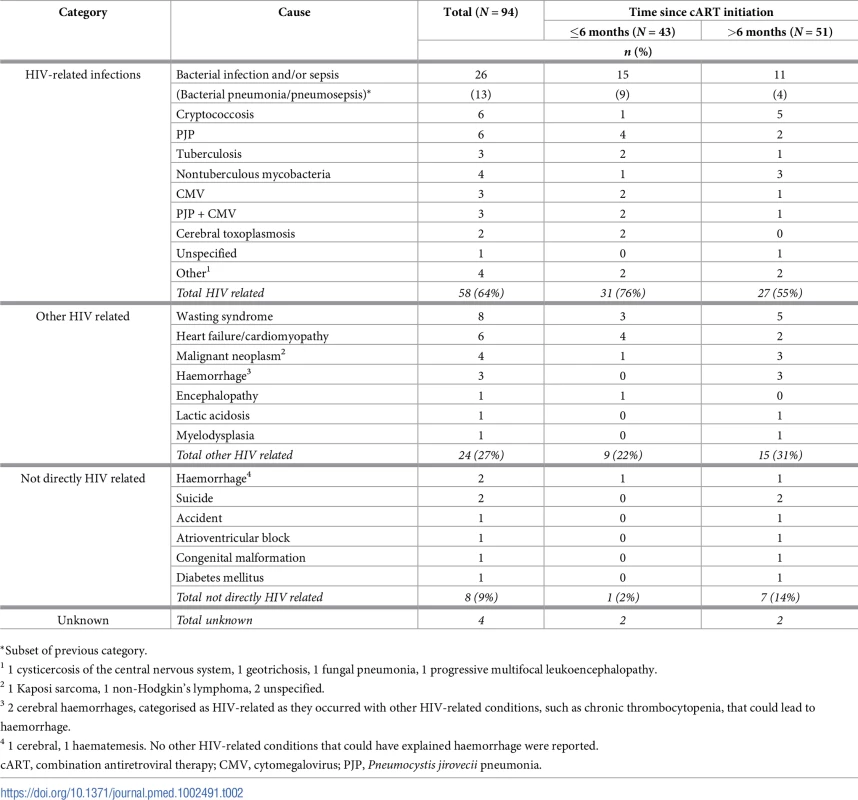
*Subset of previous category. Of the 51 late deaths, 21 (41%) were in adolescents aged ≥14 years. Median ages at HIV diagnosis and cART initiation of these 21 children were 9.9 (IQR 8.5–11.3) and 10.8 (8.7–11.9) years, respectively, and median duration of cART was 6.2 (4.5–7.6) years. Thirteen (62%) of these deaths were due to an HIV-related infection, 6 to other HIV-related causes, and 2 were unrelated to HIV.
Risk factors for death
In multivariable analysis, being in the first year of life, EE&T country group, AIDS diagnosis at cART initiation, initiating cART on an NNRTI-based regimen (versus any other), severe immune suppression for age, and low BMI-for-age z-score were associated with increased risk of early death (Table 3). Sensitivity analysis replacing BMI-for-age z-score with weight-for-age z-score resulted in similar estimates (S1 Table).
Tab. 3. Rates and baseline risk factors for death within 6 months of cART initiation. 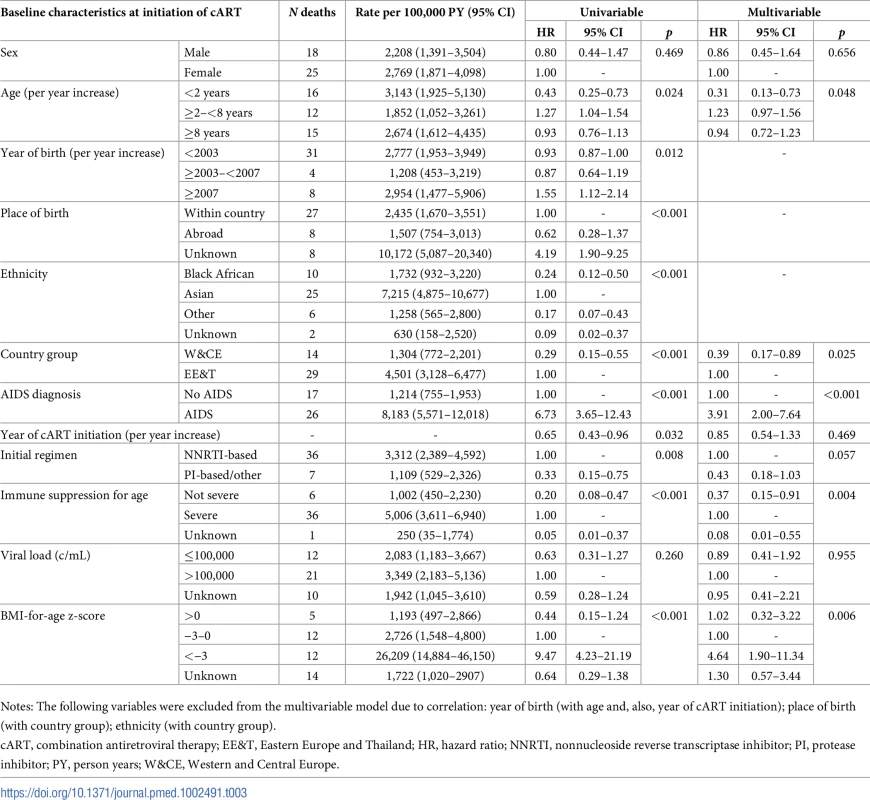
Notes: The following variables were excluded from the multivariable model due to correlation: year of birth (with age and, also, year of cART initiation); place of birth (with country group); ethnicity (with country group). Rates and risk factors for death after 6 months of cART are shown in Table 4. In multivariable analysis, no baseline characteristics were strongly associated with higher risk of late death. Time-updated characteristics associated with higher risk of death were severe immune suppression for age, current viral load >400 c/mL, and low current BMI-for-age z-score. There was a nonsignificantly elevated risk of late death in EE&T (p = 0.073). After adjustment for other factors, year of cART initiation was no longer associated with early or late death.
Tab. 4. Rates and risk factors (baseline and time updated) for death after 6 months of cART. 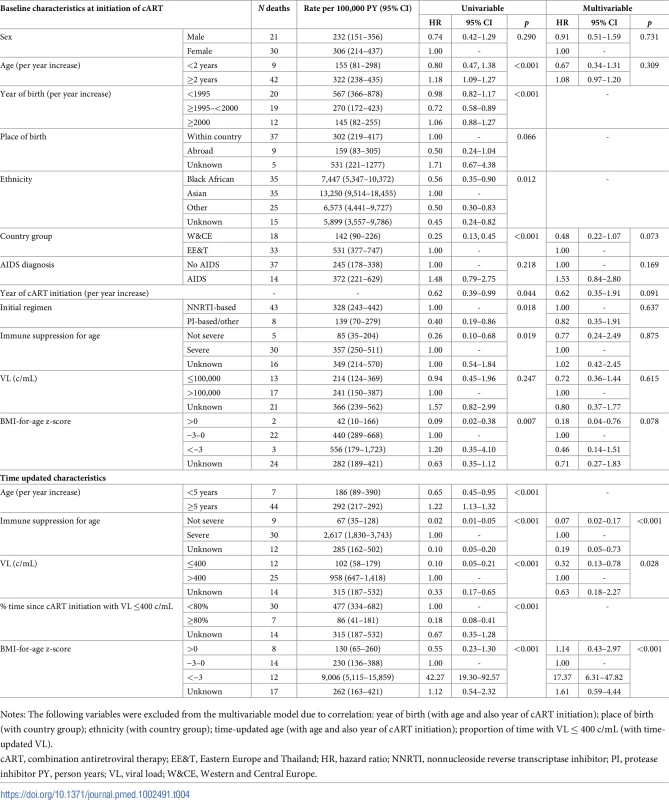
Notes: The following variables were excluded from the multivariable model due to correlation: year of birth (with age and also year of cART initiation); place of birth (with country group); ethnicity (with country group); time-updated age (with age and also year of cART initiation); proportion of time with VL ≤ 400 c/mL (with time-updated VL). In the sensitivity analysis, the model with weight-for-age z-score was broadly similar to the BMI model (S2 Table), but the effect of weight-for-age at ART start was attenuated (>0 adjusted hazard ratio [aHR] 0.57 [0.11–2.88], <−3 aHR 1.07 [0.32–3.63], unknown aHR 1.21 [0.46–3.15] versus −3–0, p = 0.875).
AIDS-defining events
Among those without an AIDS diagnosis by the start of cART (n = 2,863), 278 AIDS-defining events were reported in 237 children: 113 (41%) were from Thailand, 75 (27%) in the UK or Ireland, 48 (17%) in Russia or Ukraine, and 42 (15%) in the rest of W&CE. The overall rate of first AIDS-defining event was 1,549 (95% CI 1,364–1,760) per 100,000 person-years and was higher in the first 6 months of cART (7,305 [6,005–8,887]) compared to later (984 [832–1,163], p < 0.001). The most commonly reported AIDS events were encephalopathy (38 events in 38 children, 14%), tuberculosis (TB)(34 events in 32 children, 12%), and wasting syndrome (31 events in 30 children, 11%) (Table 5).
Tab. 5. Rates of AIDS-defining events among children with no previous AIDS diagnosis at initiation of cART, N = 2,863. 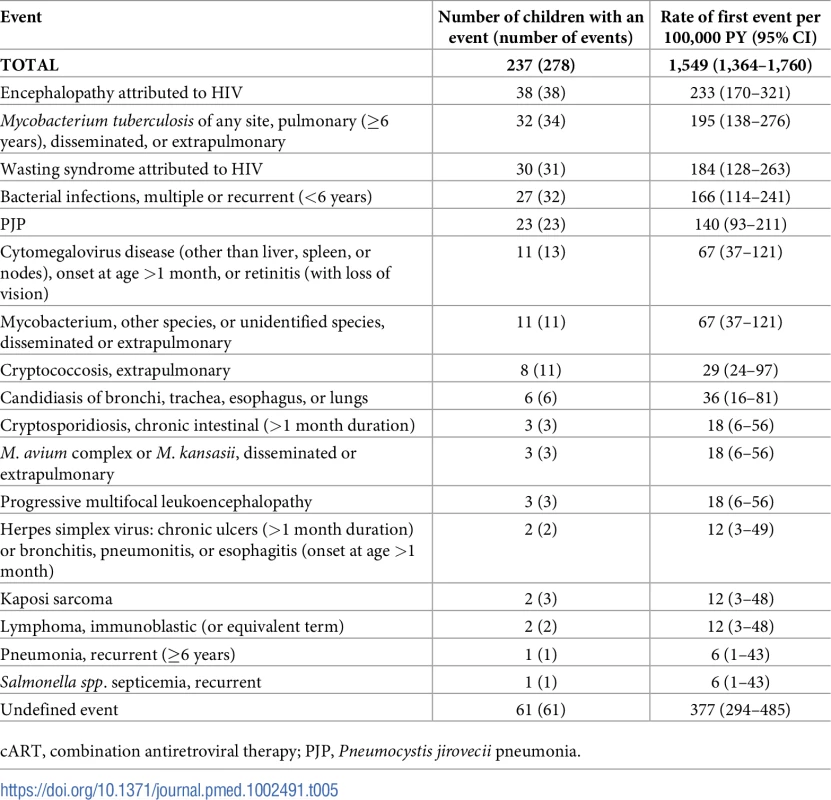
cART, combination antiretroviral therapy; PJP, Pneumocystis jirovecii pneumonia. Among the 663 children with an AIDS diagnosis prior to cART initiation, there were a further 80 AIDS-defining events after initiation of cART in 66 (10%) children. The most common events were encephalopathy (21 events in 21 children, 26%), multiple/recurrent bacterial infections (11 events in 10 children, 14%), and TB (11 events in 8 children, 14%); 61 events had a missing cause.
Risk factors for first AIDS-defining events after cART initiation in those with no previous AIDS diagnosis
In multivariable analysis, factors associated with increased risk of the first AIDS-defining event within 6 months of cART initiation were severe immunosuppression and lower BMI-for-age z-score (<−3 versus 0–−3) at cART initiation (S3 Table). Factors associated with increased risk after 6 months of cART were earlier year of cART initiation, initiating cART on an NNRTI-based regimen (versus any other), higher viral load at cART initiation, severe current immune suppression for age for those in EE&T, current viral load >400 c/mL, and lower current BMI-for-age z-score (S4 Table).
Discussion
Our study included more than 3,500 children and young adults with HIV infection followed across 16 Western, Central, and Eastern European countries and Thailand. Median age at cART initiation was 5 years and median follow-up 5 years, with a quarter of participants being followed for ≥9 years. The median year of birth was 2000, with a quarter being born before 1997; thus, many children were born before the introduction of cART, and outcomes in older children who survived reflect the first cohort of patients with perinatal HIV progressing to adult life [13]. Nearly half of all deaths (46%) and 42% of first AIDS-defining events were within 6 months of cART start in our study. We found that multivariable predictors of early death were very young age, residence in Eastern European countries and Thailand, AIDS diagnosis at cART initiation, initiating cART on an NNRTI-based regimen, severe immune suppression for age, and low BMI-for-age z-score at cART initiation. Time-updated (current) severe immune suppression for age and low BMI-for-age z-score were also associated with late death, as was current viral load >400 c/mL.
Our results confirm that mortality rates in children starting treatment across the European region and Thailand fell markedly since the introduction of cART in 1996 but also suggest that they have remained stable since 2006, at 122 and 435 deaths per 100,000 person-years, respectively. Our 5-year survival probability after initiating cART was 97.6% overall, and was higher in W&CE countries than EE&T. These findings are consistent with results from other studies. For example, a study of HIV-infected youth in the US reported mortality rates of 660 per 100,000 person years (PY) for the period 2008–2014 and a 5-year survival probability of 97.6% [16], and another US study reported survival probabilities of 76% at 6 years for those born in 1991–1996 and exposed to mono or dual therapy, and 91% at 6 years for those born in 1997–2004 and exposed to cART [3].
Interestingly, our results suggest that mortality rates (and also incidence of first AIDS events) in children initiating cART in the European region and Thailand have not declined since 2006. It is encouraging that the proportion of children presenting late decreased over time, but from 2011 onwards, 1 in 7 children aged <5 years and 1 in 5 children aged ≥5 years at cART initiation still presented with CD4% < 15 or CD4 count <200 cells/mm3, despite the CD4 threshold for initiating treatment increasing over time [17]. This highlights the ongoing importance of identifying and testing children most at risk of HIV. Enhanced prophylaxis against infections has been shown to reduce the risk of early death in adults and children with advanced disease in low-income countries [18] and may be relevant in our high-income setting, especially as a third of patients in our study were born abroad, many in sub-Saharan Africa. Further work is needed to ascertain possible contributing factors, such as late HIV diagnosis, suboptimal immune reconstitution despite cART, late access to cART, nonadherence, ART toxicity, and socioeconomic factors. Our results suggest that mortality rates in HIV-infected children initiating cART in the European region are 3–12 times higher than in the general population, as the mortality rate for 0–14-year-olds in 27 countries in the European Union (EU) in 2013 was 35 per 100,000 person-years [19]. Similarly, higher rates of mortality have been found in perinatally HIV-infected older youth compared to the general population in the US [20].
Almost half of all deaths (46%) occurred in the first 6 months of cART, with the majority already having an AIDS diagnosis by the time of cART initiation. Indeed, AIDS may have been an indication for initiating treatment at the time, and with guidelines now recommending universal treatment for HIV [21], it is unlikely that we would observe so many deaths now, with a higher proportion of children being diagnosed earlier (when asymptomatic), through screening. The Therapeutic Research, Education and AIDS Training (TREAT) Asia Pediatric HIV Observational Database (TApHOD) cohort of children in Asia-Pacific found similar results [22], and very young age has been found to predict mortality in other studies [23–26]. In our study, three-quarters of early deaths were due to HIV-related infections, with bacterial/sepsis-related causes being the most common, whilst 22% were due to other HIV-related causes, and only 1 death had a cause not directly related to HIV. Some of these early deaths may have been associated with immune reconstitution inflammatory syndrome [27,28]. The causes of early death contrast with the deaths after 6 months, of which 14% were not directly related to HIV. The trend in causes of death (i.e., HIV-related infections, other HIV-related causes, and not HIV related) did not change over calendar periods, although we had limited power for this. Similarly, we did not detect a decline over the calendar period in the proportion of deaths attributed to AIDS, in contrast to other studies [2,3]. One study of adults with HIV found different trends in the proportion of deaths due to AIDS by region across Europe and Argentina [29], with higher risk of mortality from AIDS-related causes in Eastern Europe and higher non-AIDS mortality in northern Europe.
The strongest predictors of mortality in our analysis were having an AIDS diagnosis at cART initiation, current severe immune suppression for age, and being underweight. These are known risk factors that have been consistently reported in many earlier HIV cohorts [3,20,22]. As expected, children in W&CE countries had a lower risk of death within the first 6 months of cART initiation, perhaps due to better access to medical care, a higher standard of clinical care, higher nutritional status, and better tolerated combinations of cART. One key component of clinical care is vaccination against infectious diseases among children with HIV, and this remains an area in which improvements can be made [30]. Of concern, our findings indicate a raised mortality risk in the first year of life, independent of AIDS diagnosis and immune suppression for age, similar to other study findings [31,32]. This suggests the need for continued vigilance in this group.
Among those who did not have AIDS prior to initiating treatment, 6% of children had a subsequent AIDS-defining event, with the most commonly reported events being encephalopathy, TB, and wasting syndrome. Predictors of first AIDS-defining event were similar to those found for death.
Our study has a number of limitations. We analysed observational data from several cohorts across the European region and Thailand, but events were distributed over a long time period and we lacked statistical power to investigate trends over calendar time of patterns and causes of death in some analyses. Data were not collected in a standardised way across cohorts, but we took time to verify causes of death with reporting clinicians, and each cohort had their own system for data validation and querying. We were unable to analyse data on serious adverse events and CDC disease stage B events, as these were incompletely collected across cohorts. For a small number of children with an AIDS event, information on the type of AIDS event was not available. Around half (52%) of patients came from the UK/Ireland or Thailand, but these countries accounted for 81% of the deaths observed. Conversely, 6 (38%) of the 16 countries that observed no deaths at all (Germany, Greece, the Netherlands, Portugal, Romania, and Sweden) only accounted for 9% of patients.
In conclusion, in our study of more than 3,500 children across the European region and Thailand, mortality rates fell after the introduction of cART in 1997 but have remained stable since 2006, and the prevalence of low CD4 at initiation of cART decreased over the period. Five-year survival probability after initiating cART across the whole period studied was 97.6%. Almost half (46%) of the 94 deaths observed were in the first 6 months of cART, suggesting that close clinical follow-up is recommended over the first 6 months after cART initiation, with enhanced prophylaxis against infections in those presenting with advanced disease. The indication of raised early mortality risk in infants and those in EE&T raises concern and warrants vigilance. It highlights the need to direct additional clinical resources to the care of these groups, as well as further prospective studies evaluating morbidity and mortality in older adolescents.
Supporting Information
Zdroje
1. Judd A, Doerholt K, Tookey PA, Sharland M, Riordan A, Menson E, et al. (2007) Morbidity, mortality, and response to treatment by children in the United Kingdom and Ireland with perinatally acquired HIV infection during 1996–2006: planning for teenage and adult care. Clin Infect Dis 45 : 918–924. doi: 10.1086/521167 17806062
2. Brady MT, Oleske JM, Williams PL, Elgie C, Mofenson LM, Dankner WM, et al. (2010) Declines in mortality rates and changes in causes of death in HIV-1-infected children during the HAART era. J Acquir Immune Defic Syndr 53 : 86–94. doi: 10.1097/QAI.0b013e3181b9869f 20035164
3. Kapogiannis BG, Soe MM, Nesheim SR, Abrams EJ, Carter RJ, Farley J, et al. (2011) Mortality trends in the US Perinatal AIDS Collaborative Transmission Study (1986–2004). Clin Infect Dis 53 : 1024–1034. doi: 10.1093/cid/cir641 22002982
4. de Martino M, Tovo PA, Balducci M, Galli L, Gabiano C, Rezza G, et al. (2000) Reduction in mortality with availability of antiretroviral therapy for children with perinatal HIV-1 infection. Italian Register for HIV Infection in Children and the Italian National AIDS Registry. JAMA 284 : 190–197. 10889592
5. Lewden C, Bouteloup V, De Wit S, Sabin C, Mocroft A, Wasmuth JC, et al. (2012) All-cause mortality in treated HIV-infected adults with CD4 >/ = 500/mm3 compared with the general population: evidence from a large European observational cohort collaboration. Int J Epidemiol 41 : 433–445. doi: 10.1093/ije/dyr164 22493325
6. Mahy M (2016) Explaining global adolescent mortality data: where do the numbers come from and what do they mean? Abstract TUSY0902 AIDS 2016. Durban, South Africa.
7. Judd A, Lodwick R, Noguera-Julian A, Gibb DM, Butler K, Costagliola D, et al. (2017) Higher rates of triple class virologic failure in perinatally HIV-infected teenagers compared to heterosexually infected young adults in Europe. HIV Med 18 : 171–180. doi: 10.1111/hiv.12411 27625109
8. Weijsenfeld AM, Smit C, Cohen S, Wit FW, Mutschelknauss M, van der Knaap LC, et al. (2016) Virological and social outcomes of HIV-infected adolescents and young adults in the Netherlands before and after transition to adult care. Clin Infect Dis 63 : 1105–1112. doi: 10.1093/cid/ciw487 27439528
9. Xia Q, Shah D, Gill B, Torian LV, Braunstein SL (2016) Continuum of Care Among People Living with Perinatally Acquired HIV Infection in New York City, 2014. Public Health Rep 131 : 566–573. doi: 10.1177/0033354916662215 27453601
10. Judd A, Collins IJ, Parrott F, Hill T, Jose S, Ford D, et al. (2017) Growing up with perinatal HIV: changes in clinical outcomes before and after transfer to adult care in the UK. J Int AIDS Soc 20 : 71–80.
11. Rice BD, Delpech VC, Chadborn TR, Elford J (2011) Loss to follow-up among adults attending human immunodeficiency virus services in England, Wales, and Northern Ireland. Sex Transm Dis 38 : 685–690. doi: 10.1097/OLQ.0b013e318214b92e 21844719
12. Fish R, Judd A, Jungmann E, O'Leary C, Foster C (2013) Mortality in perinatally HIV-infected young people in England following transition to adult care: an HIV Young Persons Network (HYPNet) audit. HIV Med 15 : 239–244. doi: 10.1111/hiv.12091 24112550
13. Writing group for the Kids to Adults Working Group and Data Management and Harmonisation Group in EuroCoord (2016) Children and young people with perinatal HIV in Europe: epidemiological situation in 2014 and implications for the future. Euro Surveill 21: ppi = 30162.
14. Centers for Disease Control and Prevention (2014) Revised Surveillance Case Definition for HIV Infection—United States, 2014. MMWR Morb Mortal Wkly Rep 63 : 1–10. 24402465
15. World Health Organization (2007) WHO case definitions of HIV for surveillance and revised clinical staging and immunological classification of HIV-related disease in adults and children. Geneva: World Health Organization.
16. Mirani G, Williams PL, Chernoff M, Abzug MJ, Levin MJ, Seage GR 3rd, et al. (2015) Changing trends in complications and mortality rates among US youth and young adults with HIV infection in the era of combination antiretroviral therapy. Clin Infect Dis 61 : 1850–1861. doi: 10.1093/cid/civ687 26270680
17. Bamford A, Turkova A, Lyall H, Foster C, Klein N, Bastiaans D, et al. (2018) Paediatric European Network for Treatment of AIDS (PENTA) guidelines for treatment of paediatric HIV-1 infection 2015: optimizing health in preparation for adult life. HIV Med 19:e1–e42. doi: 10.1111/hiv.12217 25649230
18. Hakim J, Musiime V, Szubert AJ, Siika A, Mallewa J, Agutu C, et al. (2016) Enhanced infection prophlaxis reduces mortality in severely immunosuppressed HIV-infected adults and older children initiating antiretroviral therapy in Kenya, Malawi, Uganda and Zimbabwe: the REALITY trial. Abstract FRAB0101LB. 21st International AIDS Conference. Durban, South Africa.
19. Eurostat (2016) Population (Demography, Migration and Projections), Death and life expectancy data. Luxembourg: Euripean Commission.
20. Neilan AM, Karalius B, Patel K, Van Dyke RB, Abzug MJ, Agwu AL, et al. (2017) Association of risk of viremia, immunosuppression, serious clinical events, and mortality with increasing age in perinatally human immunodeficiency virus-infected youth. JAMA Pediatr 171 : 450–460. doi: 10.1001/jamapediatrics.2017.0141 28346597
21. World Health Organization (2016) Consolidated guidelines on the use of antiretroviral drugs for treating and preventing HIV infection. Geneva: World Health Organization.
22. Lumbiganon P, Kariminia A, Aurpibul L, Hansudewechakul R, Puthanakit T, Kurniati N, et al. (2011) Survival of HIV-infected children: a cohort study from the Asia-Pacific region. J Acquir Immune Defic Syndr 56 : 365–371. doi: 10.1097/QAI.0b013e318207a55b 21160429
23. Collins IJ, Jourdain G, Hansudewechakul R, Kanjanavanit S, Hongsiriwon S, Ngampiyasakul C, et al. (2010) Long-term survival of HIV-infected children receiving antiretroviral therapy in Thailand: a 5-year observational cohort study. Clin Infect Dis 51 : 1449–1457. doi: 10.1086/657401 21054181
24. Sanjeeva GN, Gujjal Chebbi P, Pavithra HB, Sahana M, Sunil Kumar DR, Hande L (2016) Predictors of Mortality and Mortality Rate in a Cohort of Children Living with HIV from India. Indian J Pediatr 83 : 765–771. doi: 10.1007/s12098-016-2047-9 26916891
25. Zanoni BC, Phungula T, Zanoni HM, France H, Feeney ME (2011) Risk factors associated with increased mortality among HIV infected children initiating antiretroviral therapy (ART) in South Africa. PLoS ONE 6: e22706. doi: 10.1371/journal.pone.0022706 21829487
26. Fenner L, Brinkhof MW, Keiser O, Weigel R, Cornell M, Moultrie H, et al. (2010) Early mortality and loss to follow-up in HIV-infected children starting antiretroviral therapy in Southern Africa. J Acquir Immune Defic Syndr 54 : 524–532. doi: 10.1097/QAI.0b013e3181e0c4cf 20588185
27. Elston JWT, Thaker H (2009) Immune reconstitution inflammatory syndrome. Int J STD AIDS 20 : 221–224. doi: 10.1258/ijsa.2008.008449 19304962
28. Puthanakit T, Aurpibul L, Oberdorfer P, Akarathum N, Kanjananit S, Wannarit P, et al. (2007) Hospitalization and mortality among HIV-infected children after receiving highly active antiretroviral therapy. Clin Infect Dis 44 : 599–604. doi: 10.1086/510489 17243067
29. Reekie J, Kowalska JD, Karpov I, Rockstroh J, Karlsson A, Rakhmanova A, et al. (2012) Regional differences in AIDS and non-AIDS related mortality in HIV-positive individuals across Europe and Argentina: the EuroSIDA study. PLoS ONE 7: e41673. doi: 10.1371/journal.pone.0041673 22911841
30. Bamford A, Manno EC, Mellado MJ, Spoulou V, Marques L, Scherpbier HJ, et al. (2016) Immunisation practices in centres caring for children with perinatally acquired HIV: A call for harmonisation. Vaccine 34 : 5587–5594. doi: 10.1016/j.vaccine.2016.09.035 27727030
31. Violari A, Cotton MF, Gibb DM, Babiker AG, Steyn J, Madhi SA, et al. (2008) Early antiretroviral therapy and mortality among HIV-infected infants. N Engl J Med 359 : 2233–2244. doi: 10.1056/NEJMoa0800971 19020325
32. Tovo PA, de Martino M, Gabiano C, Cappello N, D'Elia R, Loy A, et al. (1992) Prognostic factors and survival in children with perinatal HIV-1 infection. The Italian Register for HIV Infections in Children. Lancet 339 : 1249–1253. 1349667
Štítky
Interní lékařství
Článek vyšel v časopisePLOS Medicine
Nejčtenější tento týden
2018 Číslo 1- Efekt itopridu na motilitu jícnu a funkci dolního jícnového svěrače
- Strava bohatá na arginin a prolin slibuje urychlení hojení ran u diabetických pacientů
- Úloha růstového faktoru TGF-β v procesu hojení ran
- Topicky aplikovaný propolis může urychlovat hojení diabetických bércových vředů
- Itoprid v léčbě funkční dyspepsie
-
Všechny články tohoto čísla
- What about drinking is associated with shorter life in poorer people?
- Life course socioeconomic position, alcohol drinking patterns in midlife, and cardiovascular mortality: Analysis of Norwegian population-based health surveys
- Pelvic inflammatory disease risk following negative results from chlamydia nucleic acid amplification tests (NAATs) versus non-NAATs in Denmark: A retrospective cohort
- Increased risk of ischemic heart disease, hypertension, and type 2 diabetes in women with previous gestational diabetes mellitus, a target group in general practice for preventive interventions: A population-based cohort study
- Sexually transmitted infections in the era of antiretroviral-based HIV prevention: Priorities for discovery research, implementation science, and community involvement
- Traumatic brain injury and the risk of dementia diagnosis: A nationwide cohort study
- Association between intake of less-healthy foods defined by the United Kingdom's nutrient profile model and cardiovascular disease: A population-based cohort study
- Progression of the first stage of spontaneous labour: A prospective cohort study in two sub-Saharan African countries
- The cost-effectiveness of alternative vaccination strategies for polyvalent meningococcal vaccines in Burkina Faso: A transmission dynamic modeling study
- PD-L1 checkpoint inhibition and anti-CTLA-4 whole tumor cell vaccination counter adaptive immune resistance: A mouse neuroblastoma model that mimics human disease
- Safety and pharmacokinetics of the Fc-modified HIV-1 human monoclonal antibody VRC01LS: A Phase 1 open-label clinical trial in healthy adults
- Immune-related genetic enrichment in frontotemporal dementia: An analysis of genome-wide association studies
- Long-term trends in mortality and AIDS-defining events after combination ART initiation among children and adolescents with perinatal HIV infection in 17 middle- and high-income countries in Europe and Thailand: A cohort study
- What’s coming for health science and policy in 2018? Global experts look ahead in their field
- Brain and blood metabolite signatures of pathology and progression in Alzheimer disease: A targeted metabolomics study
- Estimated mortality on HIV treatment among active patients and patients lost to follow-up in 4 provinces of Zambia: Findings from a multistage sampling-based survey
- From macro- to microfactors in health: Social science approaches in research on sexually transmitted infections
- The WHO 2016 verbal autopsy instrument: An international standard suitable for automated analysis by InterVA, InSilicoVA, and Tariff 2.0
- Long-term risks and benefits associated with cesarean delivery for mother, baby, and subsequent pregnancies: Systematic review and meta-analysis
- PLOS Medicine
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Traumatic brain injury and the risk of dementia diagnosis: A nationwide cohort study
- Pelvic inflammatory disease risk following negative results from chlamydia nucleic acid amplification tests (NAATs) versus non-NAATs in Denmark: A retrospective cohort
- PD-L1 checkpoint inhibition and anti-CTLA-4 whole tumor cell vaccination counter adaptive immune resistance: A mouse neuroblastoma model that mimics human disease
- Safety and pharmacokinetics of the Fc-modified HIV-1 human monoclonal antibody VRC01LS: A Phase 1 open-label clinical trial in healthy adults
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Autoři: prof. MUDr. Pavel Horák, CSc., doc. MUDr. Ludmila Brunerová, Ph.D., doc. MUDr. Václav Vyskočil, Ph.D., prim. MUDr. Richard Pikner, Ph.D., MUDr. Olga Růžičková, MUDr. Jan Rosa, prof. MUDr. Vladimír Palička, CSc., Dr.h.c.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



