-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Kariéra
Doporučené pozice
Reklama- Praxe
Estimating the health and economic effects of the proposed US Food and Drug Administration voluntary sodium reformulation: Microsimulation cost-effectiveness analysis
Jonathan Pearson-Stuttard and colleagues quantify the potential health and economic impacts of the United States Food & Drug Administration's proposed voluntary sodium reduction goals for processed and commercially prepared foods.
Published in the journal: . PLoS Med 15(4): e32767. doi:10.1371/journal.pmed.1002551
Category: Research Article
doi: https://doi.org/10.1371/journal.pmed.1002551Summary
Jonathan Pearson-Stuttard and colleagues quantify the potential health and economic impacts of the United States Food & Drug Administration's proposed voluntary sodium reduction goals for processed and commercially prepared foods.
Introduction
Sodium consumption is a leading modifiable risk factor for higher blood pressure (BP) and cardiovascular disease (CVD) [1]. The excess risk associated with sodium consumption appears to be mainly mediated through the deleterious effect of excess sodium consumption on BP [2]. CVD remains the leading cause of mortality and morbidity in the US, generating approximately 800,000 deaths and 6 million hospital admissions annually [3]. These CVD burdens cost $318 billion annually in healthcare costs and an additional $237 billion in lost productivity, with further costs of informal care [4]. Average sodium intake in the US is approximately 3,400 milligrams per person per day, or 8.6 grams of salt, approximately 50% above the recommended upper bound consumption level of 2,300 mg/day [5]. About 75% of sodium intake comes from processed and commercially produced foods, making industry reformulation a major priority for reducing population sodium intake [6,7].
Consistent with World Health Organization (WHO) recommendations and other voluntary reformulation policies that have effectively lowered sodium intake in Finland, Turkey, and the United Kingdom [8], the US Food and Drug Administration (FDA) in 2016 proposed short-term (2 year) and long-term (10 year) voluntary, category-specific sodium reformulation targets for commercially processed, packaged, and prepared foods across 155 food categories [9]. This proposal was designed to support the 2015–2020 US dietary guidelines by encouraging food reformulation and new product development [5].
However, the potential health and economic effects of these proposed targets have not been quantified. In addition, in both the 2017 congressional budget and the current proposed 2018 House of Representatives agriculture appropriations bill, the US Congress has instructed the FDA not “to develop, issue, promote or advance final guidance applicable to food manufacturers for long term population-wide sodium reduction,” at least in part because of uncertainty about potential health effects. Recent studies have estimated the potential health gains of general sodium reduction in the US population, but without mapping the effects of the specific FDA-proposed policies for industry or taking a wider societal perspective [10]. In this study, we quantified the potential reductions in CVD and the economic impact of different levels of compliance with the 2016 proposal over a 20-year period. This investigation was performed as part of the Food-PRICE (Food Policy Review and Intervention Cost-Effectiveness) project.
Methods
We used and extended the previously validated US IMPACT Food Policy Model [11,12] to assess the potential health and economic effects of the proposed FDA voluntary sodium reformulation policy over a 20-year period (2017 to 2036). We simulated 3 scenarios: (1) optimal, with optimal implementation of the proposed FDA policy, assuming all processed foods will be reformulated to the FDA proposed 2 - and 10-year sodium targets; (2) modest, assuming 50% compliance with the proposed upper bound of the 2 - and 10-year targets and 50% compliance with the main 2 - and 10-year targets; and (3) pessimistic, assuming all processed foods will be reformulated to the 2-year target but with no further reformulation.
We compared these 3 scenarios with a counterfactual “no intervention” (baseline) scenario. For this, we assumed that the recent observed slow declining trends in sodium consumption [13] will continue in the future. In addition, we modeled a very low compliance scenario, with 7.5% of applicable foods compliant with the 10-year reformulation targets, and present these results in S1 Appendix (Extra Scenario; p. 28).
The US IMPACT Food Policy Model
Our extended US IMPACT Food Policy Model is a stochastic dynamic microsimulation model that simulates the life course of synthetic individuals in a close-to-reality synthetic population under different policy scenarios. Compared to previous versions of the model, it allows for more detailed and flexible simulation of food policies in a competing risk framework, taking into account population heterogeneity and lag times between exposures and outcomes.
Specifically, the model first simulates the life courses of synthetic individuals aged 30 to 84 years under the “no intervention” scenario and records their sodium consumption, systolic BP (SBP), first episode of CVD (coronary heart disease [CHD] or stroke), quality-adjusted life years (QALYs), costs, and death from CVD or any other cause. Then it calculates the life courses of the same synthetic individuals under each of the 3 modeled sodium reformulation scenarios (optimistic, modest, and pessimistic) and records the differences in the aforementioned outcomes (Fig 1). Model data sources are outlined in Table 1. We further describe the model inputs, structure, key assumptions, and outputs below. Detailed description of the model, input sources, and key assumptions are detailed in S1 Appendix.
Fig. 1. Simplified model structure. 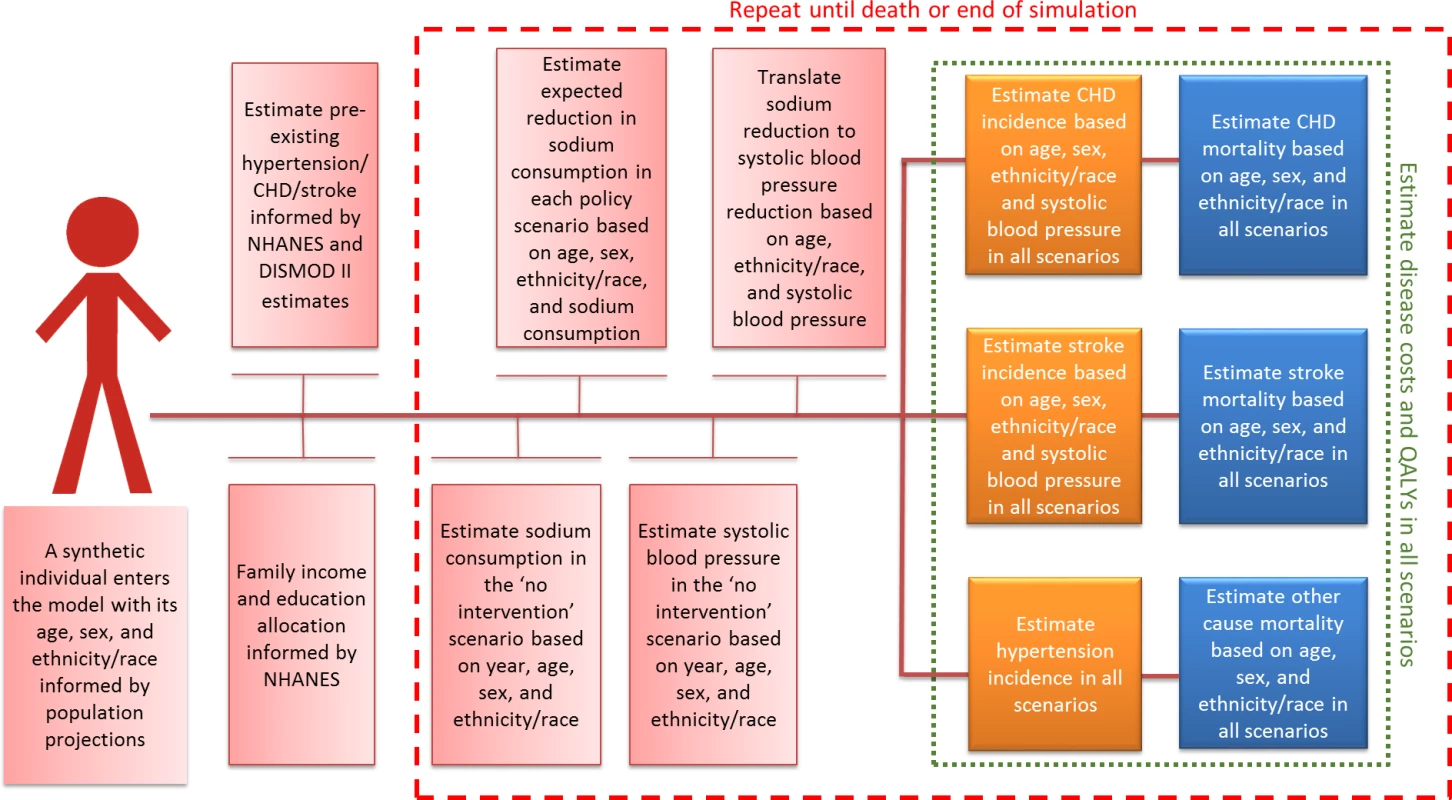
CHD, coronary heart disease; NHANES, National Health and Nutrition Examination Survey; QALY, quality-adjusted life year. Tab. 1. The US IMPACT Food Policy Model data sources. 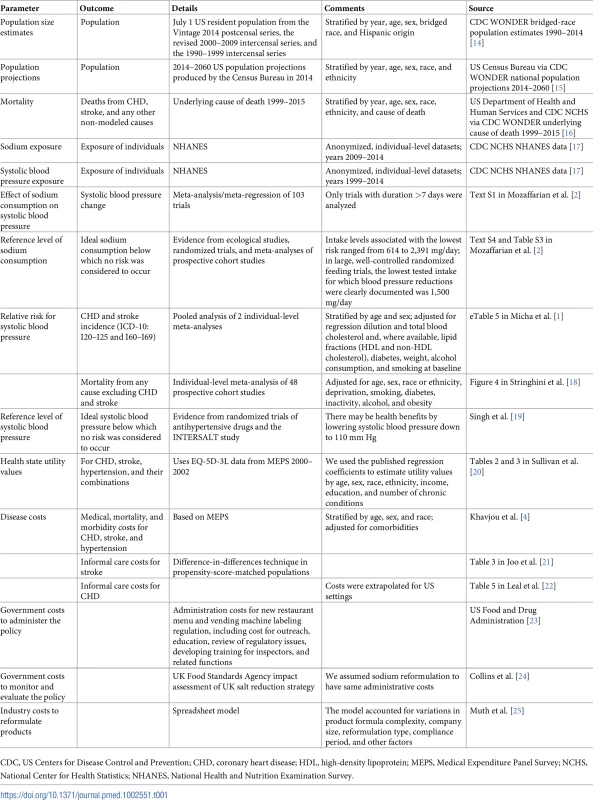
CDC, US Centers for Disease Control and Prevention; CHD, coronary heart disease; HDL, high-density lipoprotein; MEPS, Medical Expenditure Panel Survey; NCHS, National Center for Health Statistics; NHANES, National Health and Nutrition Examination Survey. Model inputs and structure
Demographics, sodium intake, and BP
The model synthesizes information regarding population structure by age, sex, and race/ethnicity [14] and data from the 2 most recent National Health and Nutrition Examination Survey (NHANES) cycles (2011–2014) [13] regarding sodium and SBP exposure, to prime a close-to-reality synthetic population. For this, the model draws the traits of the synthetic individuals from conditional distributions that were estimated from multinomial models fitted in the original survey data. The statistical framework of this method and its extension to modeling have been described elsewhere [26,27], and a detailed description and validation can be found in S1 Appendix. Then, the model projects the recent observed trends in SBP and sodium intake into the future, and uses the projections to evolve the traits of the synthetic individuals over time. We used NHANES 1999–2014 for the SBP projections and NHANES 2009–2014 for the sodium intake projections [13]. The inclusion of exposure trends in our analysis ensures more conservative estimates for the potential impact of the proposed FDA targets compared to an analysis assuming no trends.
CVD endpoints
We used the CDC WONDER database [14] to extract mortality rates for CHD (ICD-10: I20–I25), stroke (ICD-10: I60–I69), and any other cause for the years 1999–2015, stratified by age, sex, and race/ethnicity. We forecasted these trends to 2036, again providing a more appropriate and conservative estimate of the potential impact of the proposed FDA targets. Then, we used WHO DISMOD II to model the incidence and prevalence rates for CHD and stroke for 2014 [28]. To account for future trends in CHD and stroke incidence rates that are not attributable to SBP trends, we assumed that half of the forecasted annual change in CHD and stroke mortality rates is due to changes in incidence rates. We based this assumption on observational evidence from England and modeling studies in England and the US [29–32], and we included this assumption in our probabilistic sensitivity analysis (see below). Using a population attributable risk approach, the model calculates the annual risk of the synthetic individuals developing CHD and stroke based on their SBP and incidence rate forecasts using published relative risks. Finally, the model calibrates the annual case fatality for CHD, stroke, and any other cause to the forecasted mortality rates in a competing risk framework. Specifically, for “any other cause” mortality we assumed that hypertensive synthetic individuals had higher mortality rates to account for diseases other than CHD and stroke that we did not explicitly model but that are causally related to hypertension [18].
Summary of evidence regarding the risks of excess sodium consumption
Excess dietary sodium consumption has been linked to an increased risk of CVD [33]. For CVD, the excess risk appears to be mainly mediated through the deleterious effect of excess sodium consumption on BP [2]. Our methods for evaluating the causality of effects of sodium reduction on BP and of BP reduction on CVD have been previously described [2].
There is some controversy regarding the optimal level of sodium consumption [34]. Some researchers claim that sodium consumption lower than 3,000 mg/day can actually increase the risk of CVD and overall mortality [35,36]. However, it appears that this argument is based on biased measurement methodology [37]. In a recent discussion on the subject, Mozaffarian et al. concluded that the optimal level of sodium consumption, below which further sodium reduction has no further health gains, is somewhere in the range of 614 mg/day to 2,391 mg/day [2]. In our study we incorporated the uncertainty around the ideal sodium consumption in our probabilistic sensitivity analysis.
Evidence that directly links sodium risk reversibility to CVD mortality or morbidity outcomes is lacking. A meta-analysis of several randomized control trials that tested low-sodium diets was underpowered and therefore inconclusive [38]. In comparison, a plethora of evidence exists supporting the effect of a low-sodium diet on BP, which appears to happen within weeks [2,39]. Finally, the cardiovascular risk reversibility of BP has been evident in several randomized control trials and appears to occur within a 5-year period [40].
Policy effects
The FDA-proposed sodium reformulation policy included specific mean and upper bound sodium concentration targets at 2 and 10 years for 155 food categories [9]. In addition, the FDA also provided data to map these 155 food categories to the NHANES 2009–2010 24-hour recall dietary questionnaire [41]. These data enabled the model to estimate the potential impact of the modeled policies on every synthetic individual based on their age, sex, race/ethnicity, and sodium consumption in the “no intervention” scenario. The model then used the estimated reduction in sodium consumption of the synthetic individuals to calculate the effect upon their SBP using a published meta-regression equation [2]. We assumed a gradual reformulation of food products to targets, and immediate change in sodium intake in synthetic individuals according to reformulation. We also assumed that the reformulated products would sustain their sodium concentration thereafter. Although changes in sodium intake influence SBP within weeks [2,39], we conservatively assumed a median duration of 5 years from change in SBP to health outcomes.
Model outputs
For each scenario, the model generated the total numbers of relevant events and reported cases and deaths prevented or postponed (CHD or stroke [CVD] or other), QALYs, life years gained, and disaggregated disease costs. We present the results for US adults aged 30 to 84 years from 2017 to 2036 (simulation horizon of 20 years), rounded to 2 significant digits.
Medical costs and health state utility analysis
We calculated the health state utility values (preference weights) using published equations [20] that used EQ-5D-3L data from the Medical Expenditure Panel Survey (MEPS) 2000–2002 [20]. The disease medical, mortality, and morbidity costs per person-year were derived from an RTI International report that was based on MEPS [4]. We estimated informal care costs using published data [21,22]. All costs were stratified by age, sex, and race/ethnicity except informal care costs. The health state utility values were additionally stratified by income and education.
Policy costs
Policy costs included government costs to administer and monitor the policy as well as industry costs incurred through reformulating products. By taking this societal perspective, we aimed to understand the impact of sodium reduction on the entire US economy. Specifically, for industry costs we used a reformulation cost model developed by RTI International under contract with the FDA [25]. The model accounts for variations in product formula complexity, company size, reformulation type, compliance period, and other factors, which produces a more accurate cost estimate compared to a standard per-product cost approach. Administrative costs were assumed to occur every year, with monitoring and evaluation costs occurring every year after full policy implementation at year 3. We assumed the industry costs were equal in the 2 rounds of reformulation (2 - and 10-year targets) except for the pessimistic scenario (which had no costs for the second round), and divided the costs over the policy implementation years (intervention years 1–3 for the first round, and intervention years 4–10 for the second round). We assumed no policy costs after intervention year 10.
Cost-effectiveness analysis
To inform cost-effectiveness from different relevant perspectives, we evaluated both societal and healthcare cost perspectives, closely adhering to the recommendations from the Second Panel on Cost-Effectiveness in Health and Medicine [42]. All costs were inflated to 2017 US dollars using the Bureau of Labor Statistics Consumer Price Index and discounted at a 3% annual rate. We also discounted QALYs at the same rate. We assumed a willingness to pay of $100,000 per QALY [43].
Sensitivity and uncertainty analyses
We performed probabilistic sensitivity analysis via a second-order Monte Carlo approach that allowed the estimated uncertainty of different model parameters and population heterogeneity to be propagated to the outputs [44]. The sources of uncertainty we considered were the sampling errors of baseline sodium intake, baseline SBP, and the relative risk of CHD and stroke based on SBP; the uncertainties around the lowest sodium and SBP exposures below which no risk is observed; the uncertainty around the effect of sodium on SBP; the uncertainty around the true incidence of CHD and stroke; the uncertainty of mortality forecasts; the uncertainty around which foods will be reformulated; the uncertainty around the quality of life decrements used to calculate QALYs; and the uncertainty of all the costs. We summarize the output distributions by reporting the medians and 95% uncertainty intervals (UIs). We also plotted the annual probability that a scenario was cost-effective or cost-saving over the simulation period. Finally, both discount rate and willingness to pay values were included in a 1-way sensitivity analysis and allowed to vary in steps between 0% and 9% and between $50,000 and $150,000, respectively. Please refer to S1 Appendix (specifically Tables D–F) for more information.
Results
Health-related outcomes
In the baseline scenario, median sodium consumption modestly declined from 3,150 mg/day in 2017 to 2,974 mg/day in 2036. In the optimistic, modest, and pessimistic scenarios, sodium consumption was projected to fall to 2,224 mg/day, 2,524 mg/day, and 2,789 mg/day, respectively, in 2036 (Table 2; Fig 2). The resulting difference in median sodium consumption between the optimal and pessimistic scenarios resulted in a 1.0-mm Hg difference in median SBP. These differences were larger in specific subgroups, for example older adults, those with hypertension, and black individuals.
Fig. 2. Median US sodium consumption among adults aged 30–84 years under the baseline projection and 3 modeled scenarios. 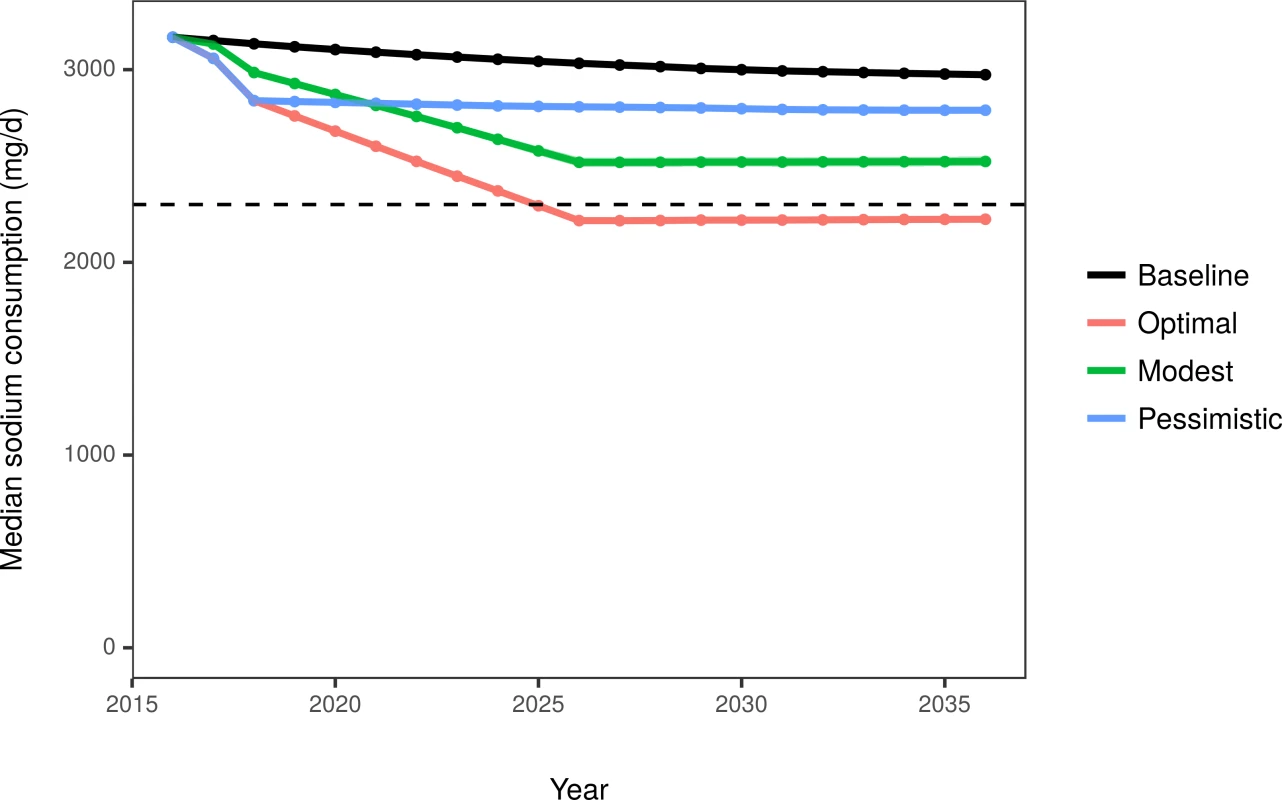
The dashed horizontal line depicts the 2015–2020 Dietary Guidelines for Americans recommended upper bound of 2,300 mg/day [5]. Tab. 2. Health-related model estimates over the 20-year simulation period from 2017 to 2036, for US adults aged 30 to 84 years. 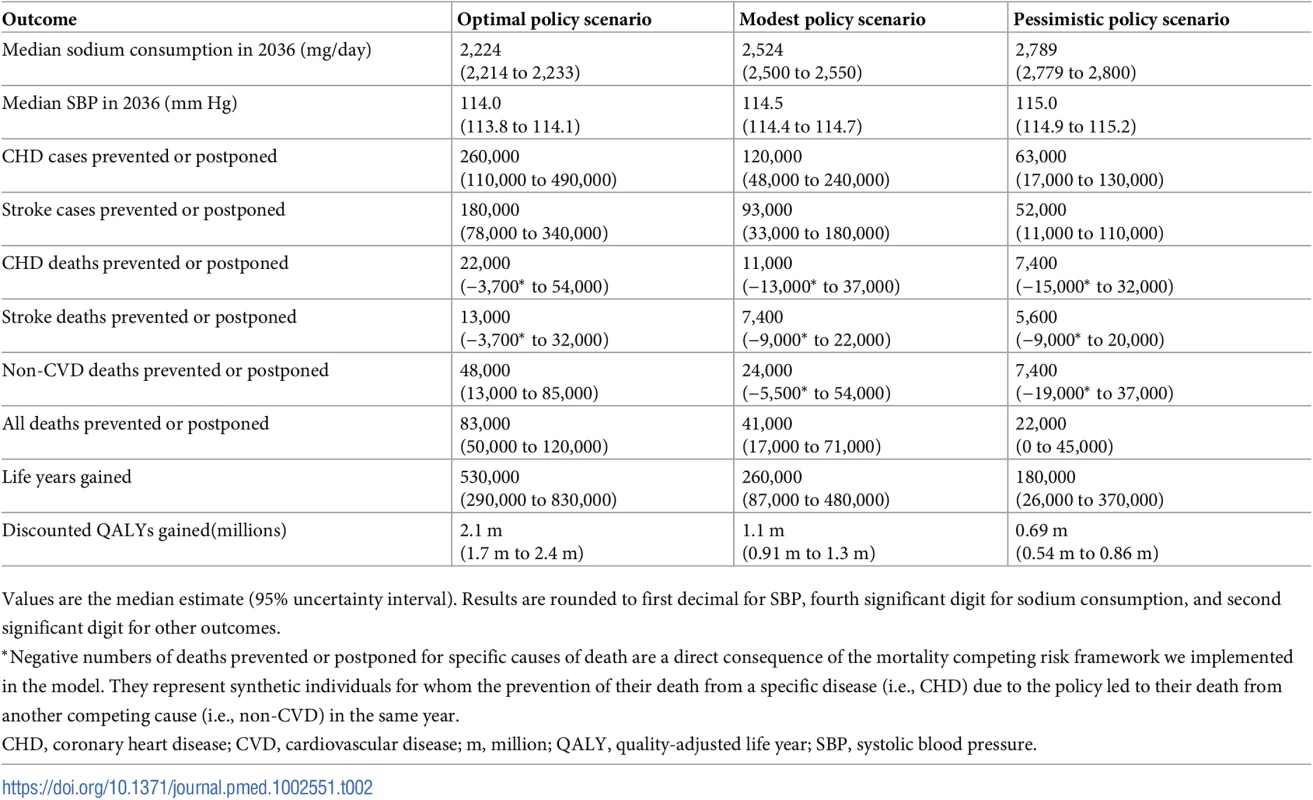
Values are the median estimate (95% uncertainty interval). Results are rounded to first decimal for SBP, fourth significant digit for sodium consumption, and second significant digit for other outcomes. Optimal implementation, achieving the 2015–2020 US dietary guidelines national target of <2,300 mg/day sodium consumption, could potentially prevent or postpone approximately 35,000 CVD deaths (95% UI: 3,700 to 78,000) and 450,000 cases of CVD from 2017 to 2036 (95% UI: 240,000 to 740,000), and potentially generate 2.1 million (95% UI: 1.7 million to 2.4 million) discounted QALYs between 2017 and 2036 (61 QALYs per 100,000 person-years) (Table 3). The modest and pessimistic scenarios might potentially prevent or postpone approximately half as many (220,000) and a quarter as many (120,000) total cases, respectively. Both could still substantially improve the health of the US population, with proportional findings for CVD deaths and QALYs.
Tab. 3. QALYs gained and costs per 100,000 person-years. 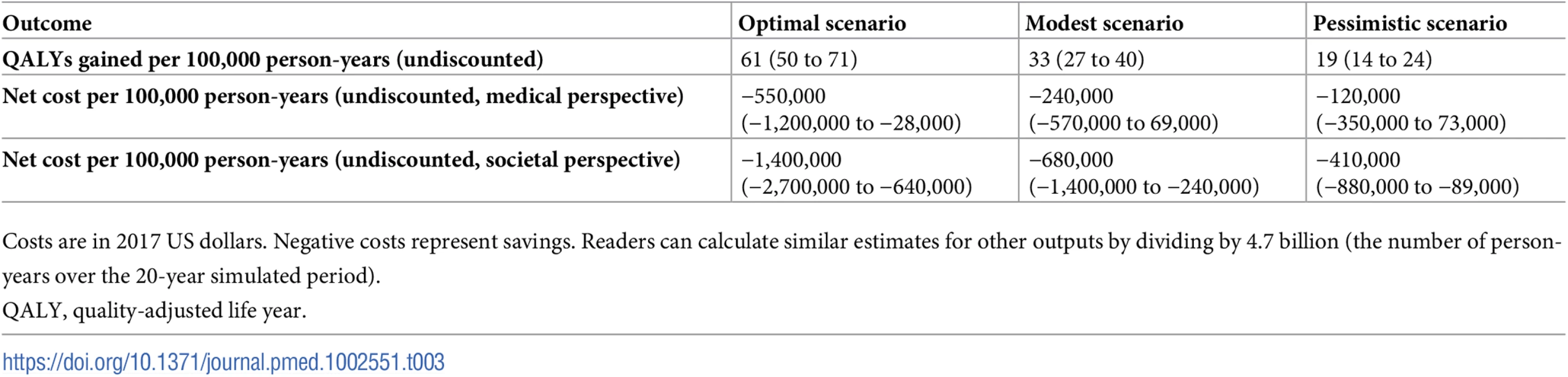
Costs are in 2017 US dollars. Negative costs represent savings. Readers can calculate similar estimates for other outputs by dividing by 4.7 billion (the number of person-years over the 20-year simulated period). The absolute health benefits from the optimistic scenario would be approximately 50% greater among men than among women, reflecting men’s higher sodium intake and higher CVD burden. The benefit would also be greater among non-Hispanic black individuals than among non-Hispanic white individuals, reflecting the higher SBP, higher CVD burden, and greater sensitivity to sodium changes of black individuals [18]. Finally, the largest number of CVD cases would be prevented in the oldest age group (70–84 years), while middle-aged individuals (50–69 years) would gain the most QALYs (please see S1 Appendix, Tables G–I).
Costs and cost-effectiveness
From a healthcare perspective (government and private payers), the optimal scenario would result in an approximately $31 billion (95% UI: $20 billion to $48 billion) reduction in total net costs, a substantial saving over the 20-year period (Table 4; Fig 3). The pessimistic scenario would still yield one-third of the savings, some $9.7 billion (95% UI: $5.9 billion to $16 billion). From the societal perspective, the net savings from 2017 to 2036 would be even larger: an approximately $41 billion (95% UI: $14 billion to $81 billion) reduction in net costs in the optimal scenario. More than 95% of policy costs would be attributable to industry costs of reformulation, with less than 5% attributable to government costs. The largest health-related cost savings for all scenarios would be generated from hypertension medical and productivity costs. The optimal scenario would yield more than 3 times more healthcare and societal savings per 100,000 person-years than the pessimistic scenario (Table 3).
Fig. 3. Estimated disaggregated discounted cumulative costs for the simulated period 2017 to 2036. 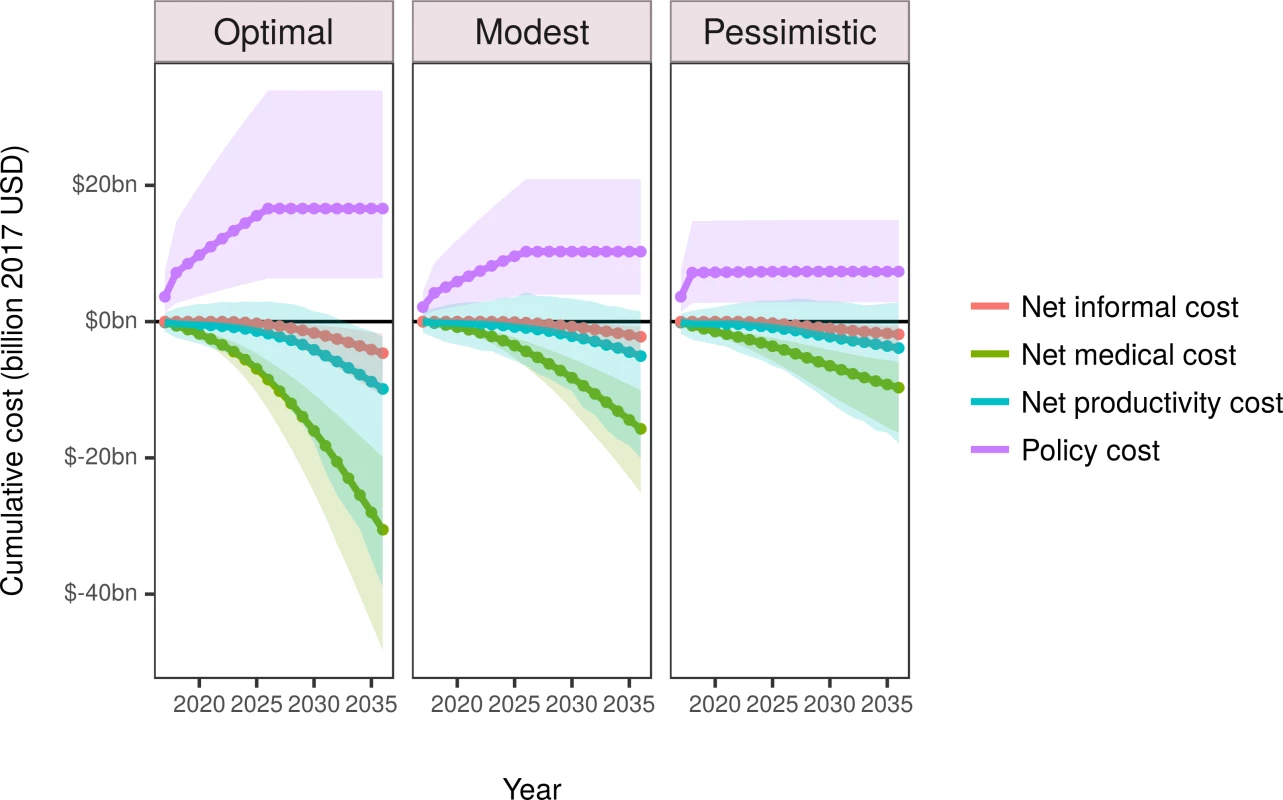
Negative costs represent savings. The shaded areas depict 95% uncertainty intervals. USD, US dollars. Tab. 4. Impact inventory and cost-effectiveness analysis of model outputs for individuals aged 30 to 84 years, assessed cumulatively over the 20-year simulation period from 2017 to 2036. 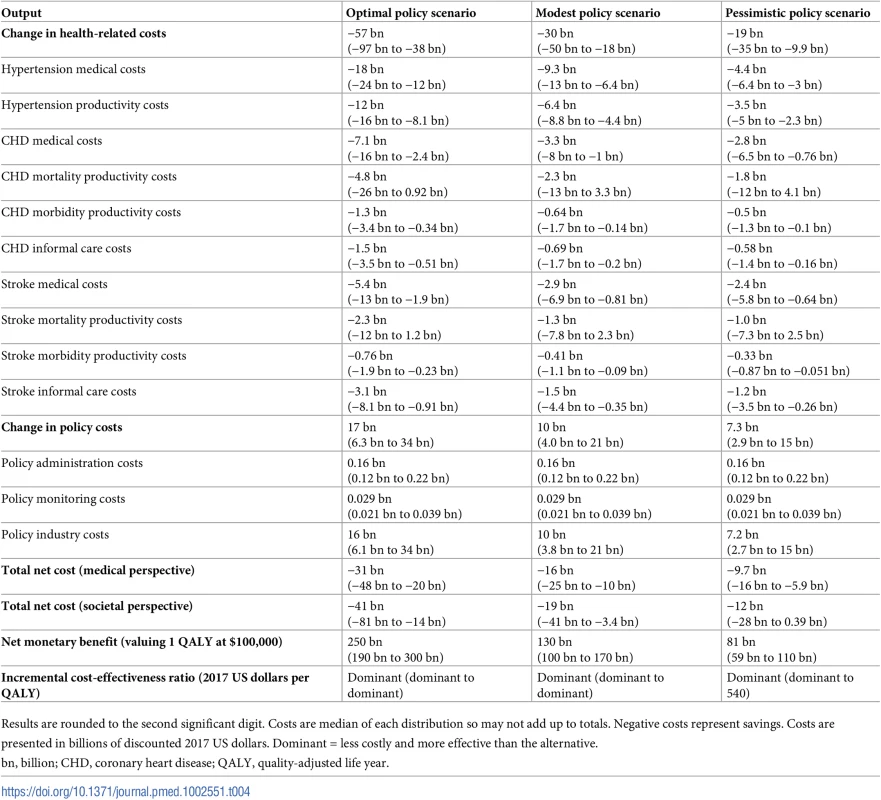
Results are rounded to the second significant digit. Costs are median of each distribution so may not add up to totals. Negative costs represent savings. Costs are presented in billions of discounted 2017 US dollars. Dominant = less costly and more effective than the alternative. All reformulation scenarios would be cost-effective, with the optimal and modest scenarios being dominant, i.e., cost-saving and producing more health than the baseline case. The optimal scenario would be approximately twice and 3 times as cost-effective as the modest and pessimistic scenarios, respectively (Fig 4; S1 Animation), generating a net monetary benefit of approximately $250 billion (95% UI: $190 billion to $300 billion), with each QALY gained valued at $100,000.
Fig. 4. Cost-effectiveness plane by the end of simulation (year 2036). 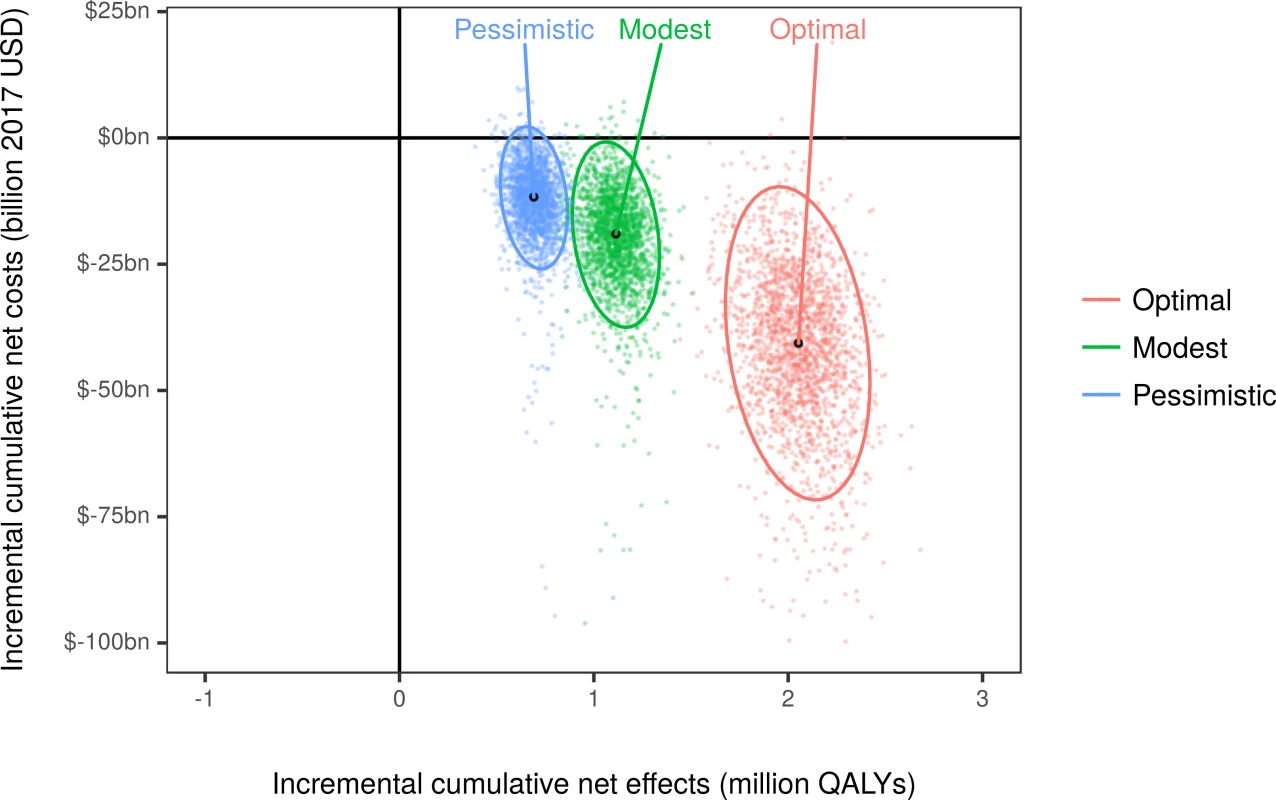
Each colored dot is the result of a stochastic Monte Carlo iteration. The black dots are the median combinations of cumulative discounted net costs (2017 US dollars) and discounted net QALYs for each simulated scenario, and the ellipses depict the 95% uncertainty interval. Negative costs represent savings. QALY, quality-adjusted life year; USD, US dollars. Probability of cost-effectiveness and sensitivity analysis
Including costs, we estimated a probability of near 100% that the optimal and pessimistic scenarios would become cost-effective by 2021, and the modest scenario by 2023. All scenarios were likely to be cost-saving by 2036 (99.9%, 99.0%, and 97.1% probability for optimal, modest, and pessimistic scenarios, respectively). The optimal and pessimistic scenarios would have more than 80% probability of becoming cost-saving by 2029, and the modest scenario by 2031 (Fig 5). In a set of 1-way sensitivity analyses, net monetary benefit remained positive when willingness to pay for a QALY was varied down from $100,000 to $50,000, and when annual discount rates were varied up from 3% to 9% (see S1 Appendix, Tables E and F).
Fig. 5. Estimated probability of cost-effective and cost-saving policy over the 20-year simulated period. 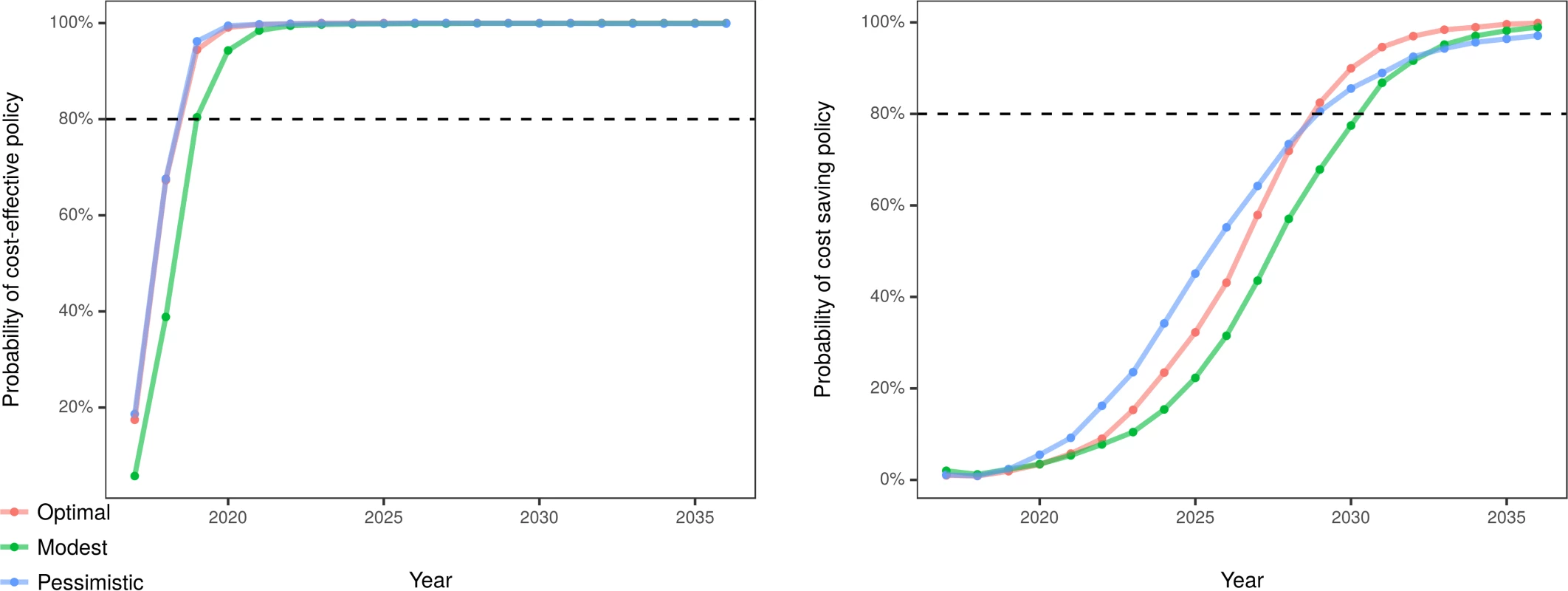
Cost-effectiveness at the willingness to pay value of 0,000 per quality-adjusted life year. Discussion
We used a microsimulation approach of a close-to-reality synthetic population (US IMPACT Food Policy Model) to estimate the potential health and economic effects, over a 20-year period, of the FDA’s proposed voluntary sodium reformulation targets under 3 scenarios of differing compliance. Our study suggests that implementation of and full compliance with the FDA voluntary sodium reformulation targets would result in substantial decreases in CVD incidence and mortality whilst also offering impressive cost savings to the healthcare payers and the wider economy. The optimal scenario saved the most lives and generated the most QALYs and economic savings. However, even lower compliance, i.e., with just the 2-year targets or 50% of the 10-year targets, yielded health and cost savings. This finding highlights the substantial health and economic opportunity costs of inaction or poorly sustained efforts to reduce sodium consumption.
Suboptimal diet is a leading cause of mortality and morbidity in the US and worldwide, with excess sodium being a significant contributor [1]. The burden of hypertension continues to grow despite advances in screening and evidence-based medications [45], emphasizing the importance of population initiatives to reduce BP. Due to their well-documented effects on BP, sodium reduction policies have been characterized as a “best buy” government intervention by WHO. Despite this and many efforts, sodium remains overconsumed in the US, highlighting the challenging nature of dietary behavior change. To date, the largest population-wide reductions in sodium consumption have been achieved in Finland, Japan, and the UK via comprehensive “upstream” strategies involving population-wide, multicomponent policies. In contrast, more “downstream” approaches such as individual approaches and worksite or community interventions are much weaker [8], again demonstrating the effectiveness hierarchy of public health interventions [46]. These declines in population sodium consumption in Finland, Japan, and the UK have corresponded with expected reductions in population BP, supporting our findings. In addition, long-term follow-up from the largest randomized control trials of sodium reduction demonstrated expected reductions in risk of CVD events [47]. Gradual sodium reduction achieves mean population sodium intake reductions without noticeable changes to consumers and their palates [48–50]. This is unlikely to trigger compensatory behaviors resulting in additional sodium used by the consumer at the table or in cooking [51–53]. Together with prior results, our investigation therefore supports the potential health and economic benefits of implementing the proposed FDA voluntary sodium reformulation targets.
When stratified by population subgroup, our results suggest the largest beneficial effects in non-Hispanic black individuals, based on higher BP responsiveness and CVD mortality rates [54]. These findings suggest additional benefits of the sodium reformulation targets in reducing disparities, consistent with previous evidence demonstrating that upstream population interventions are more equitable than downstream, individual-focused strategies [55].
Our findings are consistent with previous analyses quantifying potential benefits of general population reduction of sodium consumption [10,56–59] and build upon them significantly. A previous modeling study estimated 194,000 to 392,000 QALYS gained annually in the US with a reduction in salt consumption of 3 g/day [10], whilst another simulation study found that 312,000 QALYs could be generated annually by reducing sodium consumption to the recommended upper bound of 2,300 mg/day [57]. These previous findings are reassuringly consistent with our study, which projected an average 305,000 QALYs generated each year of the study. However, our analysis includes several notable advances. For example, we evaluated and incorporated background trends in sodium intake, SBP, and CVD, which reduces the estimated potential benefits of our policy scenarios, providing the most conservative estimation of benefits. In addition, we specifically modeled the 2016 FDA proposal, providing direct relevance to current policy considerations in the US. Furthermore, we evaluated cost-effectiveness from distinct relevant perspectives, including societal and healthcare perspectives. We are thus able to provide policy makers and health advocacy groups with more accurate and timely real-world estimations of the likely effects of the proposed policy, and the foregone opportunity costs if the desired reformulations are not achieved. By including industry costs, the present study aimed to include all relevant costs and provide objective results for all stakeholders.
This study has potential limitations. The effect estimates in the model are based on interventional and prospective observational studies. They are therefore subject to biases and confounding that may have influenced our model estimates. However, the etiological effects of dietary changes were estimated from meta-analyses with confirmatory validity analyses, including from randomized clinical trials. Our estimates may be conservative and underestimate the full health and economic benefits of sodium reformulation, as (1) our baseline scenario assumed that recent observed declines in sodium intake would continue into the future, moderating the benefit of all reformulation scenarios; (2) we only evaluated diseases mediated through BP, while decreasing sodium consumption could have beneficial effects upon other health burdens such as gastric cancer [60,61]; and (3) reductions in sodium consumption achieved through the proposed policy might additionally increase potassium intake though substitution of NaCl with KCl [62], but we did not include this potential beneficial effect in our model. We did not include unrelated medical costs in the main analysis or a sensitivity analysis as this study focused only on costs for CVD. Our modeling results cannot replace evidence from evaluating the actual policy intervention over time in the US, indicating that any implementation of the FDA policy should be accompanied by robust independent assessment.
In conclusion, our findings suggest that the proposed FDA voluntary sodium reformulation targets could result in substantial health benefits and cost savings across the US population. However, suboptimal compliance or a delay in reaching these targets could result in a significant number of preventable CVD cases, CVD deaths, and costs to the wider economy.
Supporting Information
Zdroje
1. Micha R, Penalvo JL, Cudhea F, Imamura F, Rehm CD, Mozaffarian D. association between dietary factors and mortality from heart disease, stroke, and type 2 diabetes in the United States. JAMA. 2017;317(9):912–24. doi: 10.1001/jama.2017.0947 28267855
2. Mozaffarian D, Fahimi S, Singh GM, Micha R, Khatibzadeh S, Engell RE, et al. Global sodium consumption and death from cardiovascular causes. N Engl J Med. 2014;371(7):624–34. doi: 10.1056/NEJMoa1304127 25119608
3. Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M, et al. Heart disease and stroke statistics—2016 update: a report from the American Heart Association. Circulation. 2016;133(4):e38–360. doi: 10.1161/CIR.0000000000000350 26673558
4. Khavjou O, Phelps D, Leib A. Projections of cardiovascular disease prevalence and costs: 2015–2035. Dallas: American Heart Association; 2016 [cited 2018 Mar 12]. Available from: http://www.heart.org/idc/groups/ahaecc-public/@wcm/@global/documents/downloadable/ucm_491130.pdf.
5. Dietary Guidelines Advisory Committee. Scientific report of the 2015 Dietary Guidelines Advisory Committee. Washington (DC): US Department of Health and Human Services; 2015 [cited 2018 Mar 12]. Available from: http://health.gov/dietaryguidelines/2015-scientific-report/pdfs/scientific-report-of-the-2015-dietary-guidelines-advisory-committee.pdf.
6. Harnack LJ, Cogswell ME, Shikany JM, Gardner CD, Gillespie C, Loria CM, et al. Sources of sodium in US adults from 3 geographic regions. Circulation. 2017;135(19):1775–83. doi: 10.1161/CIRCULATIONAHA.116.024446 28483828
7. Mattes RD, Donnelly D. Relative contributions of dietary sodium sources J Am Coll Nutr. 1991;10(4):383–93. 1910064
8. Hyseni L, Elliot-Green A, Lloyd-Williams F, Kypridemos C, O’Flaherty M, McGill R, et al. Systematic review of dietary salt reduction policies: evidence for an effectiveness hierarchy? PLoS ONE. 2017;12(5):e0177535. doi: 10.1371/journal.pone.0177535 28542317
9. Food and Drug Administration. Draft guidance for industry: voluntary sodium reduction goals: target mean and upper bound concentrations for sodium in commercially processed, packaged, and prepared foods. Silver Spring: Food and Drug Administration; 2016.
10. Bibbins-Domingo K, Chertow GM, Coxson PG, Moran A, Lightwood JM, Pletcher MJ, et al. Projected effect of dietary salt reductions on future cardiovascular disease. N Engl J Med. 2010;362(7):590–9. doi: 10.1056/NEJMoa0907355 20089957
11. Pearson-Stuttard J, Bandosz P, Rehm CD, Penalvo J, Whitsel L, Gaziano T, et al. Reducing US cardiovascular disease burden and disparities through national and targeted dietary policies: a modelling study. PLoS Med. 2017;14(6):e1002311. doi: 10.1371/journal.pmed.1002311 28586351
12. Pearson-Stuttard J, Bandosz P, Rehm CD, Afshin A, Penalvo JL, Whitsel L, et al. Comparing effectiveness of mass media campaigns with price reductions targeting fruit and vegetable intake on US cardiovascular disease mortality and race disparities. Am J Clin Nutr. 2017;106(1):199–206. doi: 10.3945/ajcn.116.143925 28566311
13. National Center for Health Statistics. National Health and Nutrition Examination Survey data 2011–2014. Atlanta: Centers for Disease Control and Prevention; 2014.
14. Centers for Disease Control and Prevention. CDC WONDER: bridged-race population estimates 1990–2014 request. Atlanta: Centers for Disease Control and Prevention; 2017 [cited 2017 Feb 11]. Available from: https://wonder.cdc.gov/Bridged-Race-v2014.HTML.
15. Centers for Disease Control and Prevention. CDC WONDER: national population projections 2014–2060 request. Atlanta: Centers for Disease Control and Prevention; 2017 [cited 2017 Feb 11]. Available from: https://wonder.cdc.gov/population-projections-2014-2060.html.
16. Centers for Disease Control and Prevention. CDC WONDER: about underlying cause of death, 1999–2016. Atlanta: Centers for Disease Control and Prevention; 2017 [cited 2017 Apr 18]. Available from: https://wonder.cdc.gov/ucd-icd10.html.
17. Centers for Disease Control and Prevention National Center for Health Statistics. National Health and Nutrition Examination Survey: continuous NHANES. Atlanta: Centers for Disease Control and Prevention; 2016 [cited 2016 Nov 15]. Available from: https://wwwn.cdc.gov/nchs/nhanes/ContinuousNhanes/.
18. Stringhini S, Carmeli C, Jokela M, Avendano M, Muennig P, Guida F, et al. Socioeconomic status and the 25 x 25 risk factors as determinants of premature mortality: a multicohort study and meta-analysis of 1.7 million men and women. Lancet. 2017;389(10075):1229–37. doi: 10.1016/S0140-6736(16)32380-7 28159391
19. Singh GM, Danaei G, Farzadfar F, Stevens GA, Woodward M, Wormser D, et al. The age-specific quantitative effects of metabolic risk factors on cardiovascular diseases and diabetes: a pooled analysis. PLoS ONE. 2013;8(7):e65174. doi: 10.1371/journal.pone.0065174 23935815
20. Sullivan PW, Ghushchyan V. Preference-based EQ-5D index scores for chronic conditions in the United States. Med Decis Making. 2006;26(4):410–20. doi: 10.1177/0272989X06290495 16855129
21. Joo H, Dunet DO, Fang J, Wang G. Cost of informal caregiving associated with stroke among the elderly in the United States. Neurology. 2014;83(20):1831–7. doi: 10.1212/WNL.0000000000000986 25305152
22. Leal J, Luengo-Fernandez R, Gray A, Petersen S, Rayner M. Economic burden of cardiovascular diseases in the enlarged European Union. Eur Heart J. 2006;27(13):1610–9. doi: 10.1093/eurheartj/ehi733 16495286
23. Department of Health and Human Services, Food and Drug Administration. Justification of estimates for appropriations committees. Fiscal year 2012. Silver Spring: Food and Drug Administration; 2012 [cited 2018 Mar 14]. Available from: http://wayback.archive-it.org/7993/20180126110704/https://www.fda.gov/downloads/AboutFDA/ReportsManualsForms/Reports/BudgetReports/UCM243370.pdf.
24. Collins M, Mason H, O’Flaherty M, Guzman-Castillo M, Critchley J, Capewell S. An economic evaluation of salt reduction policies to reduce coronary heart disease in England: a policy modeling study. Value Health. 2014;17(5):517–24. doi: 10.1016/j.jval.2014.03.1722 25128044
25. Muth MK, Bradley S, Brophy J, Capogrossi K, Karns S, Viator C. Reformulation cost model. Contract No. HHSF-223-2011-10005B, Task Order 20. Final report. Research Triangle Park (NC): RTI International; 2015.
26. Kypridemos C, Guzman-Castillo M, Hyseni L, Hickey GL, Bandosz P, Buchan I, et al. Estimated reductions in cardiovascular and gastric cancer disease burden through salt policies in England: an IMPACTNCD microsimulation study. BMJ Open. 2017;7(1):e013791. doi: 10.1136/bmjopen-2016-013791 28119387
27. Alfons A, Kraft S, Templ M, Filzmoser P. Simulation of close-to-reality population data for household surveys with application to EU-SILC. Stat Methods Appl. 2011;20(3):383–407.
28. Barendregt JJ, Van Oortmarssen GJ, Vos T, Murray CJ. A generic model for the assessment of disease epidemiology: the computational basis of DisMod II. Popul Health Metr. 2003;1(1):4. doi: 10.1186/1478-7954-1-4 12773212
29. Smolina K, Wright FL, Rayner M, Goldacre MJ. Determinants of the decline in mortality from acute myocardial infarction in England between 2002 and 2010: linked national database study. BMJ. 2012;344:d8059. doi: 10.1136/bmj.d8059 22279113
30. Young F, Capewell S, Ford ES, Critchley JA. Coronary mortality declines in the U.S. between 1980 and 2000 quantifying the contributions from primary and secondary prevention. Am J Prev Med. 2010;39(3):228–34. doi: 10.1016/j.amepre.2010.05.009 20709254
31. Unal B, Critchley JA, Capewell S. Explaining the decline in coronary heart disease mortality in England and Wales between 1981 and 2000. Circulation. 2004;109(9):1101–7. doi: 10.1161/01.CIR.0000118498.35499.B2 14993137
32. Ford ES, Ajani UA, Croft JB, Critchley JA, Labarthe DR, Kottke TE, et al. Explaining the decrease in U.S. deaths from coronary disease, 1980–2000. N Engl J Med. 2007;356(23):2388–98. doi: 10.1056/NEJMsa053935 17554120
33. Strazzullo, D’Elia L, Kandala NB, Cappuccio FP. Salt intake, stroke, and cardiovascular disease: meta-analysis of prospective studies. BMJ. 2009;339:b4567. doi: 10.1136/bmj.b4567 19934192
34. Ioannidis JP. Commentary: salt and the assault of opinion on evidence. Int J Epidemiol. 2016;45(1):264–5. doi: 10.1093/ije/dyw015 26888871
35. Mente A, O’Donnell M, Rangarajan S, Dagenais G, Lear S, McQueen M, et al. Associations of urinary sodium excretion with cardiovascular events in individuals with and without hypertension: a pooled analysis of data from four studies. Lancet. 2016;388(10043):465–75. doi: 10.1016/S0140-6736(16)30467-6 27216139
36. O’Donnell M, Mente A, Rangarajan S, McQueen MJ, Wang X, Liu L, et al. Urinary sodium and potassium excretion, mortality, and cardiovascular events. N Engl J Med. 2014;371(7):612–23. doi: 10.1056/NEJMoa1311889 25119607
37. Cogswell ME, Mugavero K, Bowman BA, Frieden TR. Dietary sodium and cardiovascular disease risk—measurement matters. N Engl J Med. 2016;375(6):580–6. doi: 10.1056/NEJMsb1607161 27248297
38. Taylor R, Najafi F, Dobson A. Meta-analysis of studies of passive smoking and lung cancer: effects of study type and continent. Int J Epidemiol. 2007;36(5):1048–59. doi: 10.1093/ije/dym158 17690135
39. Aburto NJ, Ziolkovska A, Hooper L, Elliott P, Cappuccio FP, Meerpohl JJ. Effect of lower sodium intake on health: systematic review and meta-analyses. BMJ. 2013;346:f1326. doi: 10.1136/bmj.f1326 23558163
40. Lawes CMM, Vander Hoorn S, Law MR, Elliott P, MacMahon S, Rodgers A. Chapter 6: high blood pressure. In: Ezzati M, Lopez AD, Rodgers A, Murray CJL, editors. Comparative quantification of health risks. Geneva: World Health Organization; 2004 [cited 2018 Mar 12]. Available from: http://www.who.int/publications/cra/en/.
41. Food and Drug Administration. FNDDS mapping file request 082516. Silver Spring: Food and Drug Administration; 2016 [cited 2018 Mar 20]. Available from: https://www.regulations.gov/document?D=FDA-2014-D-0055-0410.
42. Neumann P, Sanders G, Russell L, Siegel J, Ganiats T. Cost-effectiveness in health and medicine. Oxford: Oxford University Press; 2016.
43. Neumann PJ, Cohen JT, Weinstein MC. Updating cost-effectiveness—the curious resilience of the $50,000-per-QALY threshold. N Engl J Med. 2014;371(9):796–7. doi: 10.1056/NEJMp1405158 25162885
44. Groot Koerkamp B, Stijnen T, Weinstein MC, Hunink MG. The combined analysis of uncertainty and patient heterogeneity in medical decision models. Med Decis Making. 2011;31(4):650–61. doi: 10.1177/0272989X10381282 20974904
45. Chobanian AV. The hypertension paradox—more uncontrolled disease despite improved therapy. N Engl J Med. 2009;361(9):878–87. doi: 10.1056/NEJMsa0903829 19710486
46. Capewell S, Capewell A. An effectiveness hierarchy of preventive interventions: neglected paradigm or self-evident truth? J Public Health (Oxf). 2017. doi: 10.1093/pubmed/fdx055 28525612
47. Cook NR, Appel LJ, Whelton PK. Sodium intake and all-cause mortality over 20 years in the trials of hypertension prevention. J Am Coll Cardiol. 2016;68(15):1609–17. doi: 10.1016/j.jacc.2016.07.745 27712772
48. He FJ, MacGregor GA. Reducing population salt intake worldwide: from evidence to implementation. Prog in Cardiovasc Dis. 2010;52(5):363–82. doi: 10.1016/j.pcad.2009.12.006 20226955
49. Levings JL, Cogswell ME, Gunn JP. Are reductions in population sodium intake achievable? Nutrients. 2014;6(10):4354–61. doi: 10.3390/nu6104354 25325254
50. Dotsch M, Busch J, Batenburg M, Liem G, Tareilus E, Mueller R, et al. Strategies to reduce sodium consumption: a food industry perspective. Crit Rev Food Sci Nutr. 2009;49(10):841–51. doi: 10.1080/10408390903044297 19960392
51. Beauchamp GK, Bertino M, Engelman K. Failure to compensate decreased dietary sodium with increased table salt usage. JAMA. 1987;258(22):3275–8. 3682116
52. Janssen AM, Kremer S, van Stipriaan WL, Noort MW, de Vries JH, Temme EH. Reduced-sodium lunches are well-accepted by uninformed consumers over a 3-week period and result in decreased daily dietary sodium intakes: a randomized controlled trial. J Acad Nutr Diet. 2015;115(10):1614–25. doi: 10.1016/j.jand.2015.01.008 25769746
53. Shepherd R, Farleigh CA, Wharf SG. Limited compensation by table salt for reduced salt within a meal. Appetite. 1989;13(3):193–200. 2596842
54. Pearson-Stuttard J, Guzman Castillo M, Penalvo JL, Rehm CD, Afshin A, Danaei G, et al. Modelling future cardiovascular disease mortality in the United States: national trends and racial and ethnic disparities. Circulation. 2016;133(10):967–78. doi: 10.1161/CIRCULATIONAHA.115.019904 26846769
55. Capewell S, Kypridemos C. Socioeconomic inequalities in dietary sodium intake: upstream versus downstream interventions. Am J Public Health. 2017;107(4):499–500. doi: 10.2105/AJPH.2017.303673 28272941
56. Danaei G, Ding EL, Mozaffarian D, Taylor B, Rehm J, Murray CJ, et al. The preventable causes of death in the United States: comparative risk assessment of dietary, lifestyle, and metabolic risk factors. PLoS Med. 2009;6(4):e1000058. doi: 10.1371/journal.pmed.1000058 19399161
57. Palar K, Sturm R. Potential societal savings from reduced sodium consumption in the U.S. adult population. Am J Health Promot. 2009;24(1):49–57. doi: 10.4278/ajhp.080826-QUAN-164 19750962
58. Smith-Spangler CM, Juusola JL, Enns EA, Owens DK, Garber AM. Population strategies to decrease sodium intake and the burden of cardiovascular disease: a cost-effectiveness analysis. Ann Intern Med. 2010;152(8):481–7,W170–3. doi: 10.7326/0003-4819-152-8-201004200-00212 20194225
59. Dall TM, Fulgoni VL 3rd, Zhang Y, Reimers KJ, Packard PT, Astwood JD. Potential health benefits and medical cost savings from calorie, sodium, and saturated fat reductions in the American diet. Am J Health Promot. 2009;23(6):412–22. doi: 10.4278/ajhp.080930-QUAN-226 19601481
60. D’Elia L, Rossi G, Ippolito R, Cappuccio FP, Strazzullo P. Habitual salt intake and risk of gastric cancer: a meta-analysis of prospective studies. Clin Nutr. 2012;31(4):489–98. doi: 10.1016/j.clnu.2012.01.003 22296873
61. World Cancer Research Fund International, Continuous Update Project. Diet, nutrition, physical activity and stomach cancer. London: World Cancer Research Fund International; 2016 [cited 2018 Mar 13]. Available from: https://wcrf.org/sites/default/files/Stomach-Cancer-2016-Report.pdf.
62. Micha R, Shulkin ML, Penalvo JL, Khatibzadeh S, Singh GM, Rao M, et al. Etiologic effects and optimal intakes of foods and nutrients for risk of cardiovascular diseases and diabetes: systematic reviews and meta-analyses from the Nutrition and Chronic Diseases Expert Group (NutriCoDE). PLoS ONE. 2017;12(4):e0175149. doi: 10.1371/journal.pone.0175149 28448503
Štítky
Interní lékařství
Článek vyšel v časopisePLOS Medicine
Nejčtenější tento týden
2018 Číslo 4- Není statin jako statin aneb praktický přehled rozdílů jednotlivých molekul
- Magnosolv a jeho využití v neurologii
- Moje zkušenosti s Magnosolvem podávaným pacientům jako profylaxe migrény a u pacientů s diagnostikovanou spazmofilní tetanií i při normomagnezémii - MUDr. Dana Pecharová, neurolog
- Biomarker NT-proBNP má v praxi široké využití. Usnadněte si jeho vyšetření POCT analyzátorem Afias 1
- Antikoagulační léčba u pacientů před operačními výkony
-
Všechny články tohoto čísla
- Preprints in medical research: Progress and principles
- From surviving to thriving: What evidence is needed to move early child-development interventions to scale?
- Preprints: An underutilized mechanism to accelerate outbreak science
- Attacks on medical workers in Syria: Implications for conflict research
- Determining the scope of attacks on health in four governorates of Syria in 2016: Results of a field surveillance program
- Estimating the health and economic effects of the proposed US Food and Drug Administration voluntary sodium reformulation: Microsimulation cost-effectiveness analysis
- Maternal age and offspring developmental vulnerability at age five: A population-based cohort study of Australian children
- Two-year impact of community-based health screening and parenting groups on child development in Zambia: Follow-up to a cluster-randomized controlled trial
- Universal versus conditional day 3 follow-up for children with non-severe unclassified fever at the community level in Ethiopia: A cluster-randomised non-inferiority trial
- Universal versus conditional day 3 follow-up for children with non-severe unclassified fever at the community level in the Democratic Republic of the Congo: A cluster-randomized, community-based non-inferiority trial
- Breastfeeding during infancy and neurocognitive function in adolescence: 16-year follow-up of the PROBIT cluster-randomized trial
- Impacts 2 years after a scalable early childhood development intervention to increase psychosocial stimulation in the home: A follow-up of a cluster randomised controlled trial in Colombia
- Genetic scores to stratify risk of developing multiple islet autoantibodies and type 1 diabetes: A prospective study in children
- PLOS Medicine
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Breastfeeding during infancy and neurocognitive function in adolescence: 16-year follow-up of the PROBIT cluster-randomized trial
- Preprints in medical research: Progress and principles
- Determining the scope of attacks on health in four governorates of Syria in 2016: Results of a field surveillance program
- Estimating the health and economic effects of the proposed US Food and Drug Administration voluntary sodium reformulation: Microsimulation cost-effectiveness analysis
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MUDr. Irena Krčmová, CSc.
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



