-
Články
- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
- Kongresy
- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
Use of ex-press® implant in glaucoma surgery - retrospective study
Authors: V. Fialová 1; M. Váša 2
Authors place of work: Oční klinika Fakultní nemocnice Hradec Králové, Sokolská tř. 581, 500 05 Hradec Králové přednosta kliniky: Prof. MUDr. Naďa Jirásková, Ph. D., FEBO 1; VISUS spol. s. r. o., Němcové 738, 547 01 Náchod primář: MUDr. Karel Havlíček, MBA 2
Published in the journal: Čes. a slov. Oftal., 74, 2018, No. 1, p. 9-14
Category: Původní práce
Summary
Purpose:
The objective of the study is to evaluate of primary open-angle glaucoma (POAG) with the use of EX-PRESS drainage implant. Evaluated was the decrease of intraocular pressure (IOP), visus stabilization, perimeter Humphrey - related finding (T 30-2), Heidelberg Retina Tomograph (HRT) and possibility of reducing the local drug therapy.
Patients and methods:
Retrospective data analysis was performed in 40 eyes with POAG in 28 subjects (14 female - and 14 male patients) average-aged 69.5 years. In all 40 eyes, surgery was performed by one surgeon within the years 2011-2017. Indications for EX-PRESS implantation in our study were the POAG with decompensated IOP, decompensated chronic secondary glaucoma or failure previously anti-glaucoma surgical operations. Within the preoperative period, in all cases the progression was observed on the perimeter T 30-2 or on the HRT. Before and after surgery, all 40 eyes were evaluated for the following factors: the IOP, visus, pachymetry, therapy by anti-glaucomatic drugs and regular inspections of the perimeter and HRT. The average post-surgery following-up time in our total of patients was 3 years and 8 months. The identified data of our total of patients were statistically processed using the Wilcoxon Signed-Rank Test.
Results:
The average pre-surgery IOP was 21.4 mm Hg, 6 months post-surgery 11.2 mm Hg, and 13.2 mm Hg at the last inspection. Pre-surgery anti-glaucoma therapy: monotherapy in 2 eyes, dual therapy in 25 eyes, triple therapy in 13 eyes. Post-operatively without the need of therapy were 19 eyes, the need for monotherapy in 7 eyes, dual therapy in 13-eyes and triple therapy in 1 eye. During the last inspection of the perimeter or HRT within the postoperative period, we identified stationary finding in 39 eyes and only a mild progression in 1 eye. Peroperatively, we have not identified any serious complications. Within the postoperative period, we observed choroid ablation in 10 eyes as recovered within 6-7 days. As a more serious complication, we noted endophthalmitis in 1 eye on the background of generalized lichen planus complicating skin disease. In 5 eyes within the late postoperative period we found occlusion of lateral orifices in the EX-PRESS implant by the fibrotic tissue, the 4 eyes developed cataracts, and the eyeball hypotonia persists in 1 eye at the level of 5 torr without affecting the visus of the operated eye.
Conclusion:
It outflows from the above results that the use of EX-PRESS implant in the surgery of glaucoma is an effective and safe method with a minimal number of complications.
Keywords:
primary open-angle glaucoma, filtering surgery, EX-PRESS implant.
INTRODUCTION
Glaucoma is a multifactorial pathology, defined as a progressive loss of retinal ganglion cells leading to the appearance of a defect in the visual field [9]. At present we are capable of influencing increased intraocular pressure as the most significant risk factor using conservative treatment, as well as laser or surgical procedures [7]
Trabeculectomy is still considered to be the traditional surgical solution [1, 9, 12]. In recent years an alternative to trabeculectomy has been offered not only by the EX-PRESS implant, but also by other types of implants (Aquaflow, Healaflow, T flux, Xen Gel stent, Starflow, Ahmed glaucoma valve, iStent, CyPass, …) [5].
Indications for surgical intervention are insufficient compensation of glaucoma pathology by anti-glaucoma medication, laser treatment with insufficient reduction of intraocular pressure (IOP) or unsuccessful prior anti-glaucoma surgery.
The aim of surgical treatment of glaucoma is to achieve a reduction or stabilisation of intraocular pressure by creating an alternative pathway for the drainage of the intraocular fluid, and thus to attain a retardation or complete halting of the progression of the glaucoma pathology [13].
An EX-PRESS implant is a 2-3 mm long tube with a diameter of 0.4 mm, connecting the anterior chamber with the subscleral space (see fig. no. 1). It is manufactured from 316 L stainless steel. It is not subject to temperature changes, up to a magnetic resonance (MR) strength of 3 Tesla it does not change its position or location. There are 2 alternative lumen sizes: P50 and P200. The use depends on the state of the ocular finding, decompensation of intraocular pressure and the need for attaining target intraocular pressure [4]. Approximately 2 weeks after implantation the patient may undergo examination by magnetic resonance imaging.
Fig. 1. Description of the EX-PRESS implant (Source: Image provided by the manufacturer) 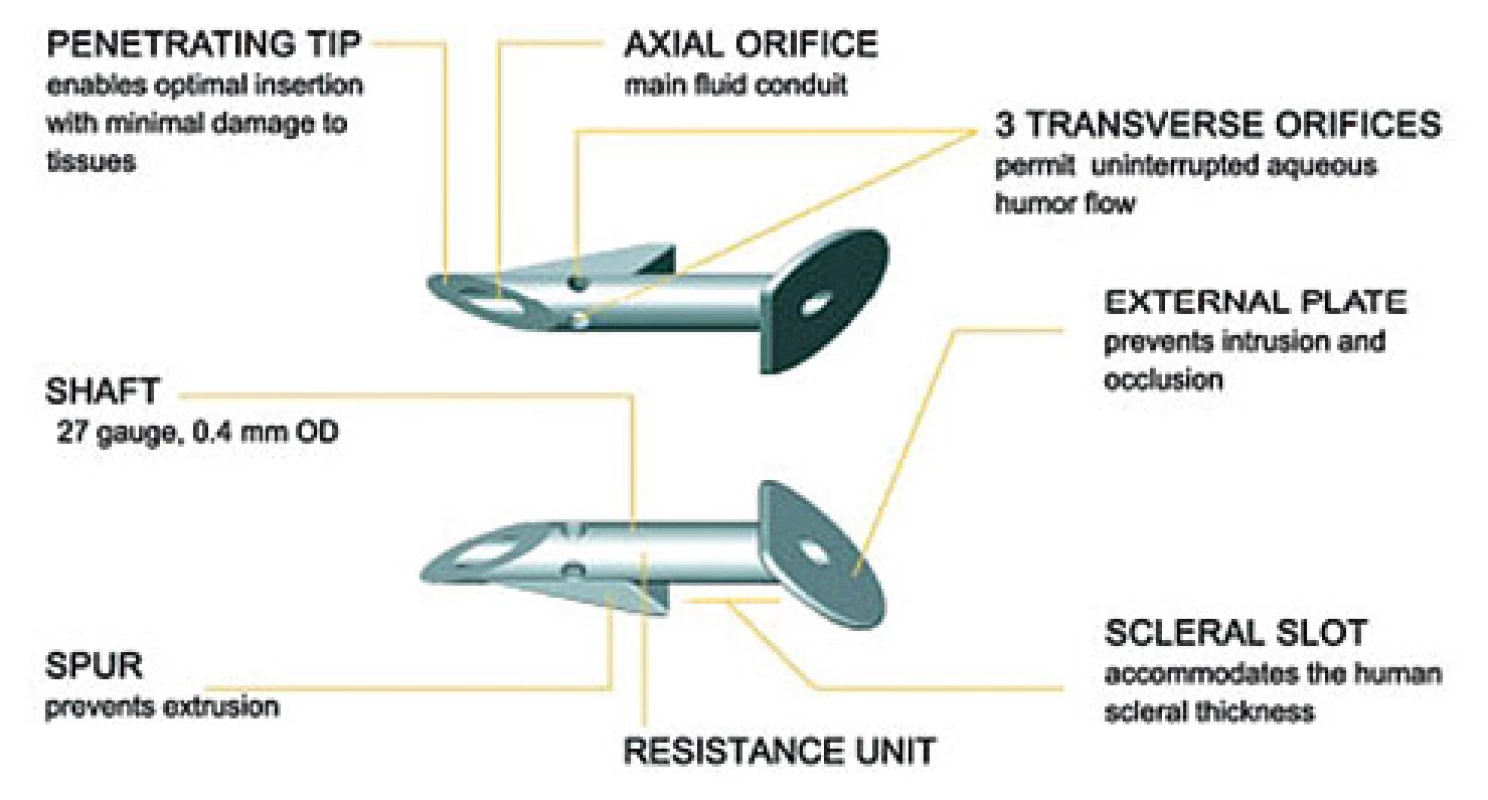
It is supplied inserted in a specially designed single-use EX-PRESS™ Delivery System (see fig. no. 2), which maintains the correct orientation of the implant throughout the entire implantation time, and is controlled by one finger. It is designed only for one use [4].
Fig. 2. Single Deployment Implant Tool - EX-PRESS ™ Delivery System (Source: Image provided by the manufacturer) 
METHOD AND STUDY COHORT
The retrospective study incorporated a total of 40 eyes of 28 patients, of whom 14 were women and 14 men, with an average age of 69.5 years. The average time of therapy undergone before implantation of an EX-PRESS implant was 10.63 years. The operation was performed on all 40 eyes by a single surgeon during the period of 2011-2017. A model EX-PRESS P50 was implanted in 24 eyes, and a P200 in 16 eyes. Indications for the implantation of EX-PRESS were primary open-angle glaucoma (POAG) with decompensated IOP, failure of maximally conservative therapy with progression to the perimeter or unsuccessful prior anti-glaucoma surgery. Within the cohort 15 eyes had undergone previous anti-glaucoma surgery (trabeculectomy, deep sclerectomy, cyclocryocoagulation). Implantation was performed on 23 artephakic (1 eye with anterior chamber intraocular pressure, 22 eyes with posterior chamber intraocular lens) and 17 phakic eyes. The average observation period of the cohort of operated patients was 3 years and 8 months.
Success was defined as full if intraocular pressure was within the range of 5.21 mm Hg and partial in the case that intraocular pressure was within the same range, but with anti-glaucoma therapy.
Examination of patient
For all patients, the average frequency of examinations before the implantation of an EX-PRESS implant was at minimum once per 3 months. After surgery the follow-up examinations took place according to the clinical finding and the condition of IOP, and were progressively extended until they were at least once every 3 months. Before and after surgery, the following factors were recorded and evaluated in every patient: IOP (measured by applanation on a slit lamp CSO SL-980, manufacturer: CSO Italy), vision (examined on Snellen's optotypes), examination of pachymetry (on instrument Auto Kerato-Refracto-Tonometer TRK-1P, manufacturer: TOPCON, Japan), fundus including condition of optic nerve (examined on slit lamp CSO SL-980, manufacturer: CSO Italy), therapy using anti-glaucomatous agents and regular examinations on a computer perimeter (T 30-2) HFA 740i (manufacturer: Carl Zeiss Meditec AG, Germany) and Heidelberg Retina Tomograph III (HRT) (manufacturer: Heidelberg Engineering).
In all cases, in the preoperative period, upon examination progression was recorded on a perimeter T 30-2 or on HRT.
Surgical technique
The surgical procedure covers the creation of a conjunctival flap, the cleansing of the sclera and the creation of a scleral flap, which should be of 50% of the thickness of the sclera and a size of 4x3 mm to 5x5 mm (depending on the affiliated visual defect, anatomical proportions of the eye, and last but not least on the experience of the surgeon in evaluating the degree of risk). We then apply mitomycin 0.2 mg/l for 1-2 minutes subconjunctivally and beneath the scleral flap in order to reduce the risk of failure of filtration. There follows thorough rinsing with a physiological solution. We then create a puncture through the remaining scleral tissue using a 25 G needle in the lower part of the grey zone, parallel to the iris. This is followed by the actual implantation of the EX-PRESS implant, and thorough checking of the position and then also the function subsequently via paracentesis in the cornea. We perform suturing of the scleral flap using individual stitches, and suturing of the conjunctiva on the limbus or fornix according to the place of creation of the conjunctival flap. At the end of the operation we apply Diprophos subconjunctivally, Tobradex unguent and monoculus.
In exceptional cases it is possible to use 1 or more EX-PRESS implants into further quadrants. The placing is determined by the surgeon on the basis of the anatomical proportions and condition of the chamber angle. In the majority of cases, placing into the upper quadrants is recommended. In all cases we implanted one EX-PRESS implant into the upper quadrants (see Fig. 3 and 4).
Fig. 3. EX-PRESS implant located in the upper outer quadrant of the right eye (Source: own processing, year: 2015) 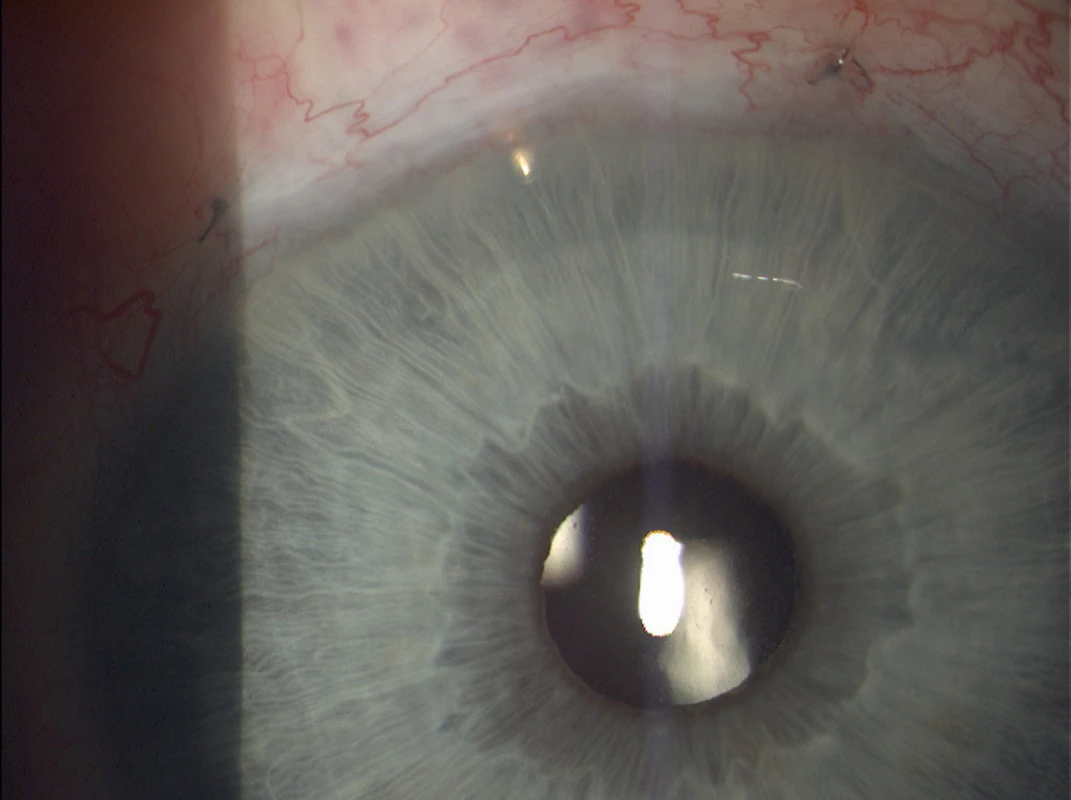
Fig. 4. EX-PRESS implant located in the upper outer quadrant of the left eye (Source: own processing, year: 2015) 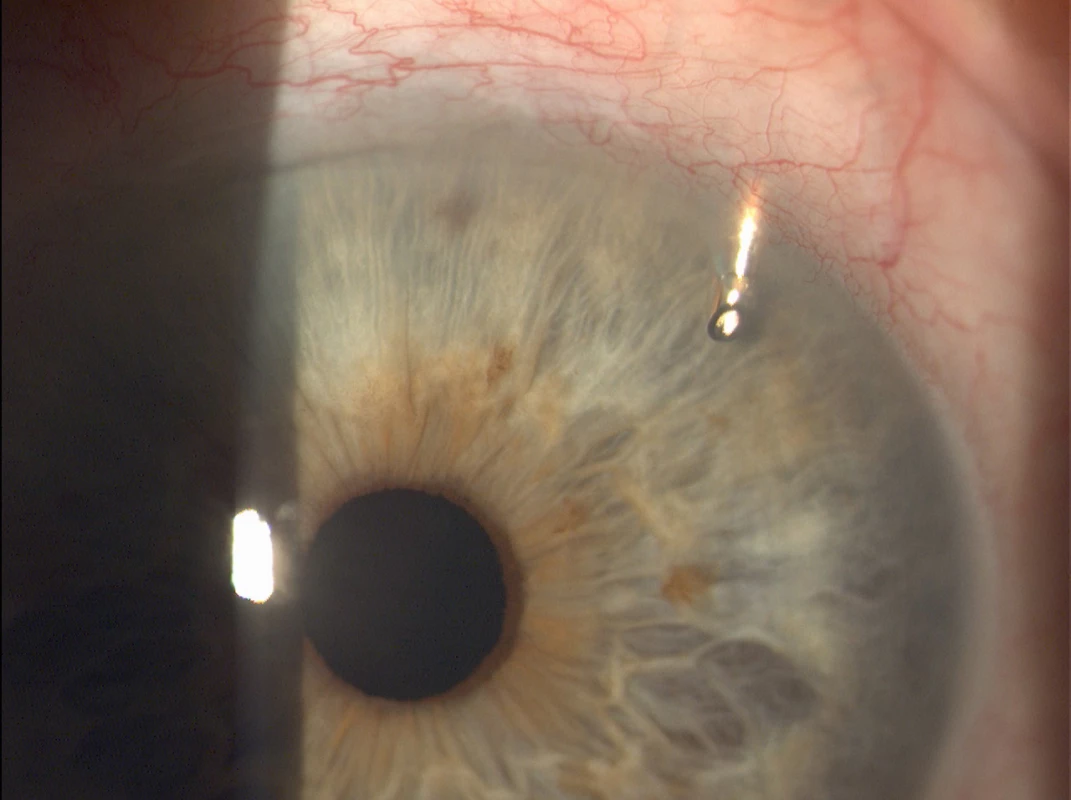
RESULTS
Average IOP before surgery was 21.14 mm Hg, 6 months after surgery 11.2 mm Hg and at the last follow-up examination at least 8 months after surgery 13.2 mm Hg. The average reduction of IOP at the last follow-up examination is 8.2 mm Hg, therefore, there was a reduction by 38.3 %. We recorded compensation of intraocular pressure within the range of 5 - 21 mm Hg without anti-glaucomatous medication in 47.5 %, with the application of anti-glaucomatous medication within the range of 5-21 mm Hg we recorded in 52.5 %.
Graph 1. Intraocular average pressure (IOP) in mm Hg vs. tracking time 
We statistically evaluated the measured values of intraocular pressure with the aid of an unpaired Wilcoxon signed-rank test. The Wilcoxon test that was used refused a zero hypothesis of a concordance of both medians, and in its result inclined toward the alternative hypothesis that the median before surgery is statistically significantly higher, on a level of significance of p <0.0000005. Table no. 1 presents the limits of reliability which here indicate the interval at which the actual median lies with a high, 95% probability. From the table it is evident that the actual values of the medians do not in any way “meet” here, and are clearly different.
Tab. 1. The confidence limit indicating the interval in which the actual interval lies with 95 % confidence 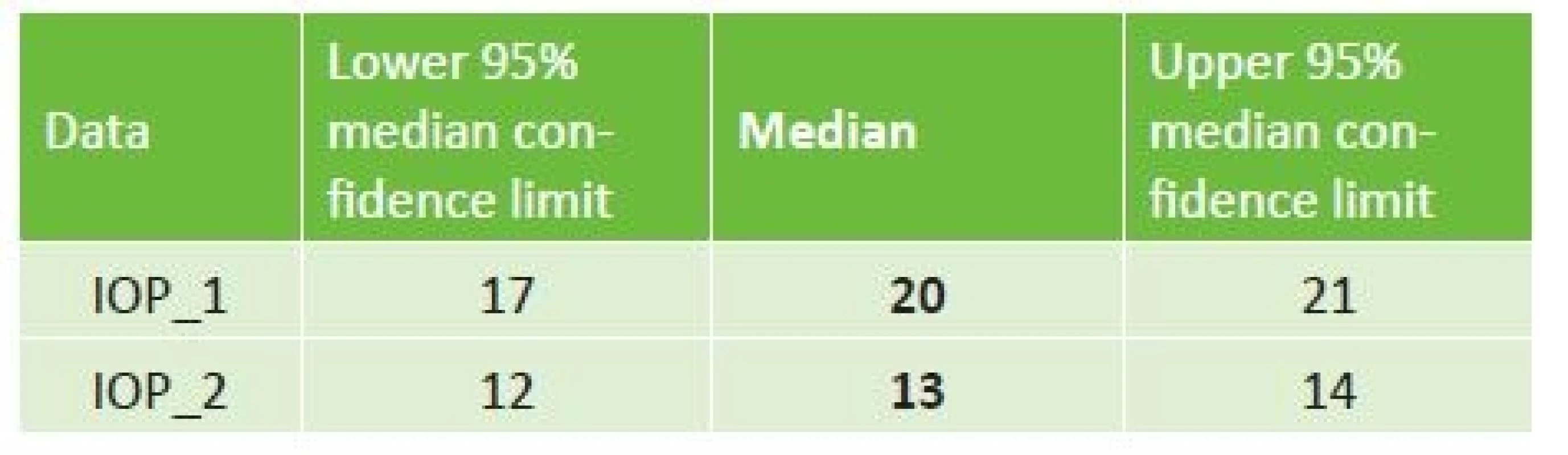
On the basis of the conducted test, we therefore demonstrated a statistically significant reduction of IOP following the implantation of an EX-PRESS implant.
Anti-glaucoma therapy before surgery: monotherapy in 2 eyes, double combination in 25 eyes, triple combination in 13 eyes. Postoperatively 19 eyes were without the need for therapy, there was a need for monotherapy in 7 eyes, double combination in 13 eyes and triple combination in 1 eye. The average number of applied anti-glaucomatous agents was reduced from 2.27 before surgery to 0.9 at the last follow-up examination. The percentage reduction of applied anti-glaucomatous agents before surgery and at the last follow-up is by 60.4 %.
Graph 2. Antiglaucoma therapy before surgery vs. after surgery 
Visual acuity before and after surgery remained the same in 27 eyes, in 6 eyes there was an improvement by 1-2 rows and in 7 eyes there was a deterioration by 1-2 rows, which was caused in 4 cases by cataract and in the remaining 3 by progression of age related macular degeneration and progression of glaucoma changes.
At the last follow-up examination of the perimeter in the postoperative period we recorded a stationary finding in 37 eyes and slight progression in only 3 eyes.
COMPLICATIONS
We did not record any more serious complications perioperatively. In the early postoperative period we recorded choroid ablation in 10 eyes, with recovery within 6-7 days due to excessive hypotonia. As a more serious complication we recorded endophthalmitis in 1 eye upon a background of general complicating lichen planus skin disease, with subsequent explantation of the EX-PRESS implant. In the later postoperative period we found occlusion of lateral orifices in the EX-PRESS implant by fibrous tissue in 5 eyes, 4 eyes developed cataracts, and hypotonia of the eyeball persists in 1 eye at the level of 5 torr, but without the formation of hypotonic maculopathy and with vision of 5/5.
DISCUSSION
Surgical therapeutic procedures for decompensated glaucomas are evaluated mainly on the basis of efficacy, safety and practicability of the method.
In recent years there has been ever increasing use of the method of implantation of an EX-PRESS implant in comparison with trabeculectomy, even if the latter is the method still considered to be the gold standard [1, 9, 12].
EX-PRESS is a miniature drainage implant designated for the regulation of intraocular pressure in eyes with refractory glaucoma, where conservative, laser or other prior anti-glaucoma therapy has failed. Indications for implantation of an EX-PRESS implant are advanced form POAG, decompensated or poorly compensated glaucoma, failure of pharmaceutical therapy, prior anti-glaucoma surgery, aphakic or pseudophakic glaucoma indicated for filtration operation, secondary glaucoma indicated for filtration operation (uveitis, neovascular glaucoma) [1, 9, 13, 14, 15].
Contraindications for the performance of implantation are: acute uveitis, ocular infection, severe sicca syndrome, blepharitis, existing ocular or systemic pathology (cicatrical pathology, autoimmune disorder, vascular diseases etc.), which the surgeon considers could cause the complications described below after implantation, as well as a patient with diagnosed glaucoma with narrow or closed angle. Potential complications and side effects: shallow anterior chamber, short-term or long-term choroid ablation, significant reduction of visual acuity, appearance of scar tissue in place of operation, erosion of conjunctiva, hyphaema [4].
Positive aspects in comparison with trabeculectomy are as follows: higher percentage of success, safer performance, less invasive method, faster and easier performance, repeatability of method, better healing, precisely calibrated drainage (due to the shape and size of the lumen of the implant and the suture of the scleral flap), reduction of complications (hypotonia, haemorrhage from surgical wound or iridectomy, shallow anterior chamber, choroid ablation, postoperative inflammations (uveitis, endophthalmitis)) [1, 9, 12, 15].
In our study we recorded choroid ablation in 10 eyes, with recovery within 6-7 days, due to excessive postoperative hypotonia. As a more serious complication we identified endophthalmitis in 1 eye upon a background of of general complicating lichen planus skin disease. In the later postoperative period we found occlusion of lateral orifices in the EX-PRESS implant by fibrous tissue in 5 eyes, 4 eyes developed cataracts, and hypotonia of the eyeball persists in 1 eye at the level of 4 torr, but without the formation of hypotonic maculopathy and with vision of 5/5.
In our retrospective study we attained full success in 42.5 % and partial success in 52.5 %. Average IOP before surgery was 21.14 mm Hg, 6 months after surgery 11.2 mm Hg, at the last follow-up examination at least 8 months after surgery 13.2 mm Hg. In the study conducted by Dahan et al. [2] with a cohort of 24 eyes with POAG, in which conservative therapy had failed, IOP was reduced from an average of 27.2 ± 7.1 mm Hg before surgery to 14.5 ± 5.0 mm Hg 12 months after surgery, and 14.21 ± 4.2 24 months after surgery. According to the study by Lankaranian et al. [8] on a cohort of 100 eyes, full and partial success was defined in the same manner as in our study, full success was attained in 60 % of patients and partial success in 24 %, with a reduction of intraocular pressure from average preoperative values of 27.7 mm Hg ± 9.2 to 14.02 ± 5.1 mm Hg.
Mariotti et al. [11] conducted a study in which an EX-PRESS implant was implanted independently in 136 eyes (55 %), whereas in 112 eyes (45 %) implantation of an EX-PRESS implant was performed simultaneously, together with cataract surgery, in which the results of both groups were grouped together. Average preoperative intraocular pressure was reduced from 27.63 ± 8.26 mm Hg (n = 248) to 13.95 ± 2.70 mm Hg (n = 95) 5 years after implantation. With set criteria for full success, i.e. without glaucoma pathology, postoperative intraocular pressure is 5-18 mm Hg and partial success, i.e. same intraocular pressure but with or without pharmaceutical medication, full and partial success was 83% and 85% in the given order after 1 year, and 57% and 63% respectively after 5 years of observation. In the study by Samková et al. (15), a cohort of 22 eyes with decompensated secondary glaucoma was evaluated, in which an EX-PRESS implant was implanted. After surgery the authors again demonstrated a significant reduction of intraocular pressure (p <0.005), a reduction of local anti-glaucomatous therapy and a complete discontinuation of general therapy.
On the basis of the studies listed above, it is evident that as soon as 6 months, and also 5 years after surgery, there is not only a significant reduction in IOP, but that according to the results it is possible to document that an EX-PRESS implant is a very reliable method in the surgical solution of glaucoma.
Since the EX-PRESS implant is produced from stainless steel, it presents a potential problem upon MR imaging. Geffen et al. [6] studied the influence of a magnetic field 1.5 and 3 T on an EX-PRESS implant under various conditions, including human eyes (post mortem), and came to the conclusion that undergoing MR with an implanted EX-PRESS implant up to 3 T is safe as a result of the ocular tissue which prevents the movement of the device. Seibold et al. [17] examined the movement of an EX-PRESS implant in various magnetic fields, and also determined that the implant is safe for MR of as much as 3 T. Mabray et al. [10] also demonstrated that the presence of an EX-PRESS implant in the eyes of patients did not cause any significant artefacts influencing the diagnostic interpretation of their MR. In our study none of the patients has yet had do undergo MR examination.
As standard we performed an examination of the position and function of the EX-PRESS implant in all cases, both biomicroscopically and gonioscopically. Detorakis et al. [3] performed imaging of an EX-PRESS implant with the aid of an ultrasound B scan and on optical coherence tomography (OCT). It ensues from their results that imaging can be performed by both methods, since both enable visualisation of the position of the implant in relation to the iris and cornea, and delineate the internal structure of the filtration cicatrix. It would therefore be appropriate to use these methods in cases with a large filtration cicatrix exceeding beyond the cornea or in the case of the opacity of the anterior segment.
In our study we did not examine the differences between the implant sizes of P50 and P200, nevertheless Samsudin et al. [15] investigated this quantity and came to the conclusion that the luminary diameters are very similar, and that both diameters have low resistance values, and therefore do not themselves prevent the occurrence of postoperative hypotonia.
CONCLUSION
Before the anti-glaucoma operation with implantation of an EX-PRESS implant, 6% of eyes had undergone a different anti-glaucoma procedure (trabeculectomy, deep sclerectomy, cyclocryocoagulation). All the patients in our selected cohort had decompensated POAG or secondary glaucoma, and without treatment would have suffered an irreversible deterioration of vision or total blindness.
In our cohort we demonstrated a statistically significant reduction of IOP following surgery, on average by 38.3 %, and a reduction of the number of applied anti-glaucomatous agents by 60.4 %.
Despite the fact that trabeculectomy is still considered to be the gold standard in the surgery of POAG, and the majority of ocular surgeons still incline toward this procedure, it ensues from our results that the use of an EX-PRESS implant in the surgery of decompensated primary open angle glaucoma and secondary glaucoma is a safer and more effective method with a minimal number of complications.
This study was presented in abbreviated form at the 12th European Glaucoma Society Congress, 2016, Prague, Czech Republic, and at Futurum Ophthalmologicum 2016, Lázně Bělohrad.
The authors of the study declare that no conflict of interest exists in the compilation, theme and subsequent publication of this professional communication, and that it is not supported by any pharmaceuticals company.
Delivered to the editorial board 8. 11. 2017
Received for printing 26. 2. 2018
Dr. Veronika Fialová
Department of Ophthalmology, University Hospital Hradec Králové
Sokolská tř. 581
500 05 Hradec Králové
Zdroje
1. Bing, L., Da-Dong, G., Xiu-Juan, D., et al.: Evaluation of EX-PRESS implantation combined with phacoemuldification in primary angle-closure glaucoma. Medicine (Baltimore). [online]. 2016, Sep, 96(36): e4613. [cit. 2001-01-03]. Dostupné na https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5023874/.
2. Dahan, E., Carmichael, TR.: Implantation of a miniature glaucoma device under a scleral flap. J Glaucoma. 2005, April, 14(2) : 98-102.
3. Detorakis, E., Stojanovic, N., Chalkia, A. , et al.: EX-PRESS® Implant Position and Function: Comparative Evaluation with Ultrasound Biomicroscopy and Optical Coherence Tomography. J Ophthalmol. 2016, Jan-Mar, 23(1): 110-4.
4. EX-PRESS. Návod k obsluze zdravotnického prostředku.
5. Francis, B. A., Sarkisian, S. R. Jr., Tan, J. C.: Minimally invasive glaucoma surgery. A practical guide. New York/Stuttgart, Thieme Publishers. 2016, 236 p. ISBN 978-1-62,623-156-6.
6. Geffen, N., Trope, GE., Alasbali, T., et al.: Is the Ex-PRESS glaucoma shunt magnetic resonance imaging safe? J Glaucoma. 2010, Feb, 19(2): 116-8.
7. Kuchynka, P. a kol.: Oční lékařství. 2. Doplněné a přepracované vydání. Praha: Grada Publishing a.s. 2016. 650s.
8. Lankaranian, D., Razeghinejad, MR., Prasad, A., et al.: Intermediate-term results of the Ex-PRESS miniature glaucoma implant under a scleral flap in previously operated eyes. Clin Exp Ophthalmol. 2011, Jul, 39(5): 421-8.
9. Leo, J., Antoine, L., Anne-Sophie, A., et al.: Five-year extension of a clinical trial comparing the EX-PRESS glaucoma filtration device and trabeculectomy in primary open-angle glaucoma. Clin Ophthalmol. 2011, 5 : 527–533.
10. Mabray, M., Uzelac, A., Talbott, JE., Lin, SC., et al: Ex-PRESS glaucoma filter: an MRI compatible metallic orbital foreign body imaged at 1.5 and 3 T. Clin Radiol. 2015, May, 70(5): e28-34.
11. Mariotti, C., Dahan, E., Nicolai, M., et al.: Response to: RE: Long-term outcomes and risk factors for failure with the EX-press glaucoma drainage device. Eye (Lond). 2014, 28 : 1-8.
12. Marzette, L., Herndon, LW.: A comparison of the Ex-PRESS™ mini glaucoma shunt with standard trabeculectomy in the surgical treatment of glaucoma. Ophthalmic Surg Lasers Imaging. 2011, 42(6): 453-9.
13. Mlčák, P., Karhanová, M., Marešová, K.: Chirurgická léčba glaukomu. Praktické lékárenství. 2009, 5(3): 118 s.
14. Salim, S.: The role of the Ex-PRESS glaucoma filtration device in glaucoma surgery. Semin Ophthalmol. 2013, 28 : 180–184.
15. Samková K., Rejmont L.: Řešení dekompenzovaného sekundárního glaukomu pomocí antiglaukomového implantátu EXPRESS. Čes a slov. Oftal. 69, 2013, 5, s.198-200.
16. Samsudin, A., Eames, I., Brocchini, S., et al.: Evaluation of Dimensional and Flow Properties of ExPress Glaucoma Drainage Devices. Journal of Glaucoma. 2016, January, Volume 25, Issue 1, pp 39–45.
17. Seibold, LK. , Rorrer, RA. , Kahook, M.: MRI of the Ex-PRESS stainless steel glaucoma drainage device. Br J Ophthalmol. 2011, Feb, 95(2): 251-4.
Štítky
Oftalmologie
Článek vyšel v časopiseČeská a slovenská oftalmologie
Nejčtenější tento týden
2018 Číslo 1- Selektivní laserová trabekuloplastika nesnižuje nitroční tlak více než argonová laserová trabekuloplastika
- Progresi glaukomu je třeba hodnotit strukturálními i funkčními parametry
- Ztráta centrálního vidění po filtrujících operacích glaukomu
- Od PGF-2 alfa-isopropyl esteru k latanoprostu: přehled vývoje Xalatanu
- Compliance u pacientů s glaukomem
-
Všechny články tohoto čísla
- Change of tear osmolarity after refractive surgery
- Optic nerve orbital meningioma
- Pachychoroid disease of the macula
- Femtosecond Laser - assisted intrastromal corneal segment implantation - our experience
- Merkel cell carcinoma of the eyelid and orbit
- Use of ex-press® implant in glaucoma surgery - retrospective study
- Česká a slovenská oftalmologie
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Pachychoroid disease of the macula
- Optic nerve orbital meningioma
- Use of ex-press® implant in glaucoma surgery - retrospective study
- Merkel cell carcinoma of the eyelid and orbit
Kurzy
Zvyšte si kvalifikaci online z pohodlí domova
Revma Focus: Spondyloartritidy
nový kurz
Autoři: prof. MUDr. Vladimír Palička, CSc., Dr.h.c., doc. MUDr. Václav Vyskočil, Ph.D., MUDr. Petr Kasalický, CSc., MUDr. Jan Rosa, Ing. Pavel Havlík, Ing. Jan Adam, Hana Hejnová, DiS., Jana Křenková
Autoři: MDDr. Eleonóra Ivančová, PhD., MHA
Autoři: prof. MUDr. Eva Kubala Havrdová, DrSc.
Autoři: prof. MUDr. Pavel Horák, CSc., doc. MUDr. Ludmila Brunerová, Ph.D., doc. MUDr. Václav Vyskočil, Ph.D., prim. MUDr. Richard Pikner, Ph.D., MUDr. Olga Růžičková, MUDr. Jan Rosa, prof. MUDr. Vladimír Palička, CSc., Dr.h.c.
Všechny kurzyPřihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



