-
Medical journals
- Career
Taylor’s flap reconstruction following abdominoperineal resection for advanced hidradenitis suppurativa – a case series and literature review
Authors: I. Ben Ayad 1; F. Château 2
Authors‘ workplace: Department of Surgery, Cliniques Universitaires Saint Luc, Brussels, Belgium 1; Department of Plastic and Reconstructive Surgery, CHU de Reims, Reims, France 2
Published in: ACTA CHIRURGIAE PLASTICAE, 67, 4, 2025, pp. 245-251
doi: https://doi.org/10.48095/ccachp2025245Introduction
Hidradenitis suppurativa (HS), also known as acne inversa or Verneuil’s disease, is a chronic inflammatory condition primarily affecting intertriginous skin regions rich in apocrine sebaceous glands and hair follicles [1]. The prevalence is estimated to range between < 1% and 4% in the overall population [2]. The pathogenesis of HS remains incompletely understood. Follicular occlusion is considered the initiating event leading to lesion formation [3]. Clinically, HS manifests along a spectrum from recurrent inflamed nodules and abscesses to draining sinus tracts and extensive cicatricial bands. The associated symptoms – including pain, malodor, chronic drainage, and disfigurement – may contribute to significant psychosocial distress in affected individuals [4].
Management strategies for HS can be broadly classified into medical and surgical approaches, with treatment selection guided by disease severity at presentation. Early-stage disease may respond to exclusive medical therapy and advanced cases often necessitate surgical intervention aiming at the complete excision of involved tissue [5,6].
This report presents two cases of extensive perineal HS in which abdominoperineal amputation, followed by reconstruction with an oblique rectus abdominis flap, or Taylor’s flap, was performed as an elective salvage procedure after all other therapeutic options had been exhausted. Additionally, an updated review of the literature on the disease is provided.
Case description
Patient 1
A 49-year-old male, clinically diagnosed with hidradenitis suppurativa 5 years prior, presented with recurrent and progressively worsening lesions affecting the perineal, perianal, and gluteal regions. His past medical history included active smoking. His disease history had been characterized by multiple episodes of abscess formation, necessitating repeated surgical drainage and local excisions. Despite prolonged conservative management, including prolonged (> 12 weeks) period of doxycycline therapy, extensive local wound care, and repeated courses of topical antibiotics, he continued to experience recurrent relapses and progressive perineal disease. The patient also received adalimumab for severe HS at standard dosing (160 mg initially, followed by 80 mg at week 2, and then 40 mg weekly thereafter). He did not receive other biologic agents such as secukinumab or bimekizumab, as these treatments were not available in our institution nor reimbursed for this indication at the time of management. A multidisciplinary consultation was conducted to explore definitive surgical options. After thorough discussion, a radical surgical approach was proposed to these patients, consisting of an abdominoperineal resection with a definitive colostomy, followed by reconstruction using a pedicled Taylor’s flap. Surgical management was scheduled as a single-stage procedure performed under general anesthesia in the absence of active lesions. He underwent preoperative consultations with a psychologist and a stomatherapist to ensure informed decision-making and optimize postoperative compliance.
Skin markings delineating the perineal resection and the flap design were made preoperatively (Fig. 1, 2).
1. Patient 1 – preoperative skin markings, donor-site. 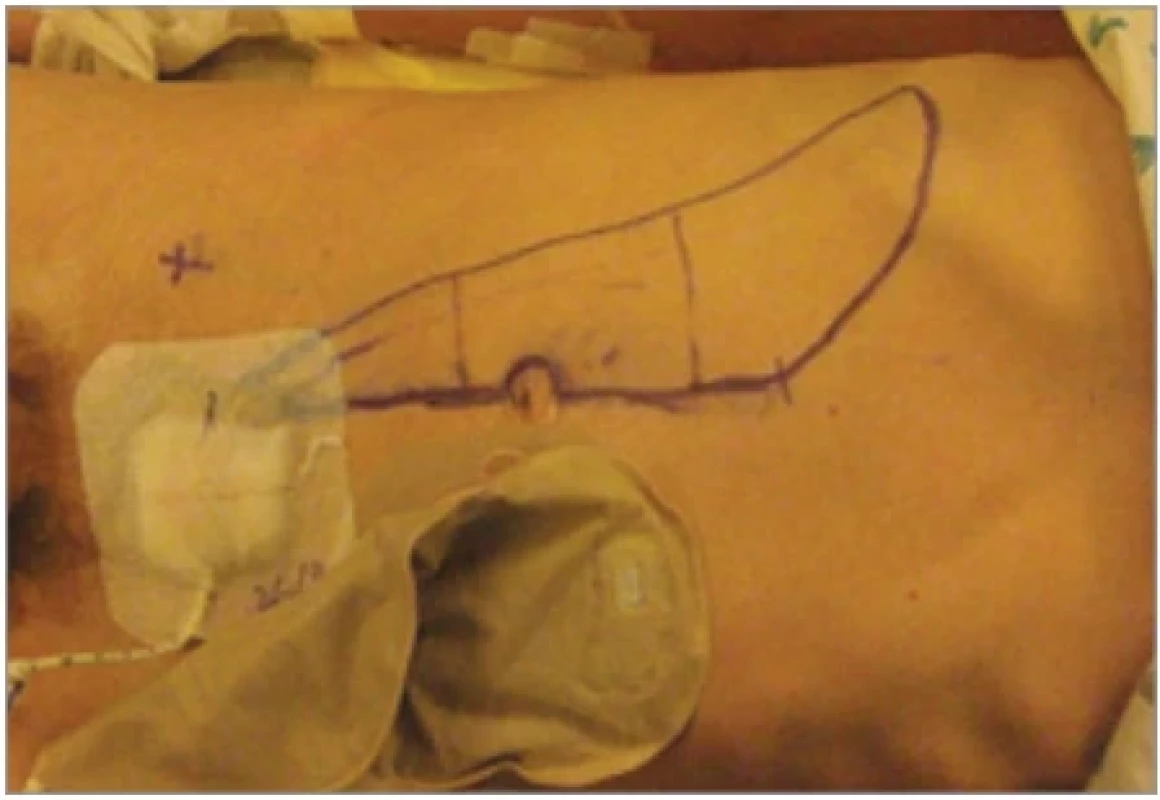
2. Patient – 1 perineal Hurley III–HS lesions. 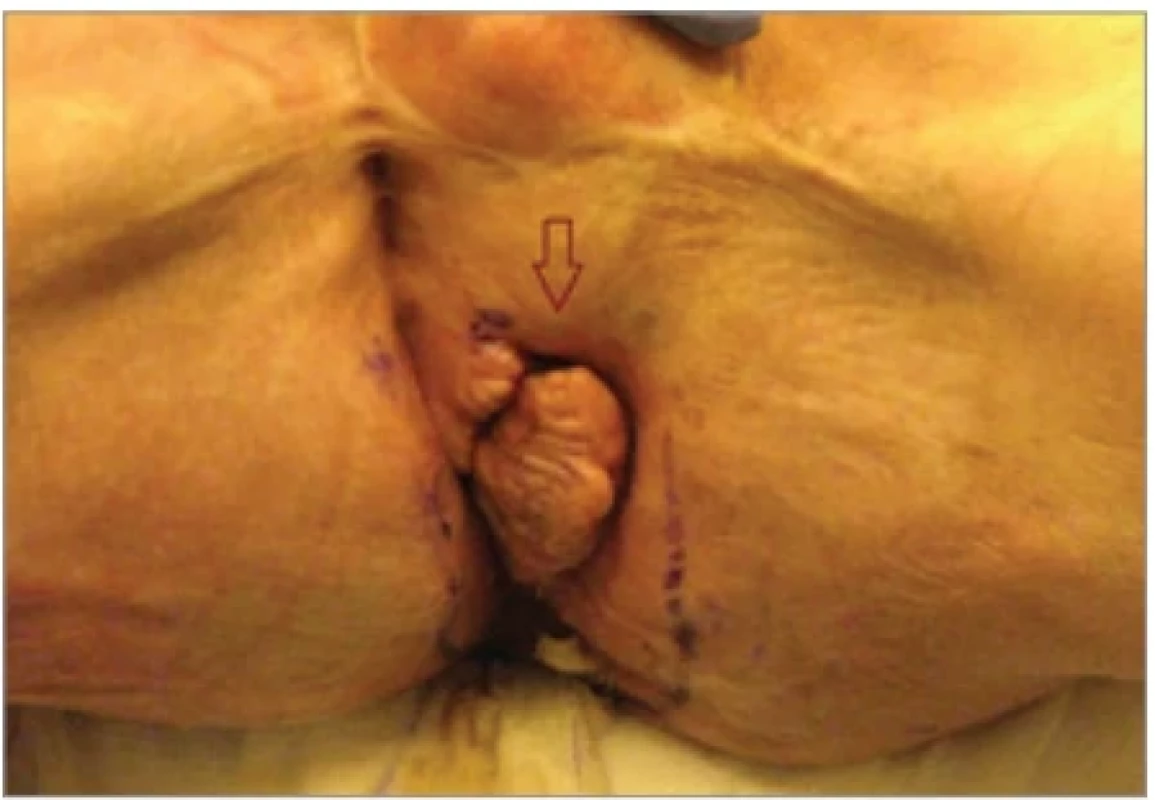
The musculocutaneous flap was harvested in a retrograde fashion, incorporating a segment of anterior fascia at the periumbilical region to preserve the emergence of perforating vessels. The flap was then tunneled to the perineum, posterior to the bladder, with careful intraoperative assessment of the deep inferior epigastric vascular pedicle to ensure optimal perfusion (Fig. 3). Two drains were placed to prevent fluid accumulation, which could compromise the flap’s vascularization (Fig. 4). Due to an initial mismatch between the defect and the skin paddle, the flap was loosely secured before definitive fixation. The abdominal wall was then closed with reinforcement of the aponeurosis using a mesh of polyglactin. At the end of the procedure, the skin paddle appeared well-perfused and norm colored. Additionally, two gluteal fistulae were explored, with methylene blue instillation confirming no communication with the resected area. These fistulae were subsequently incised and managed accordingly.
3. Flap’s position after tunneling to the perineal region. 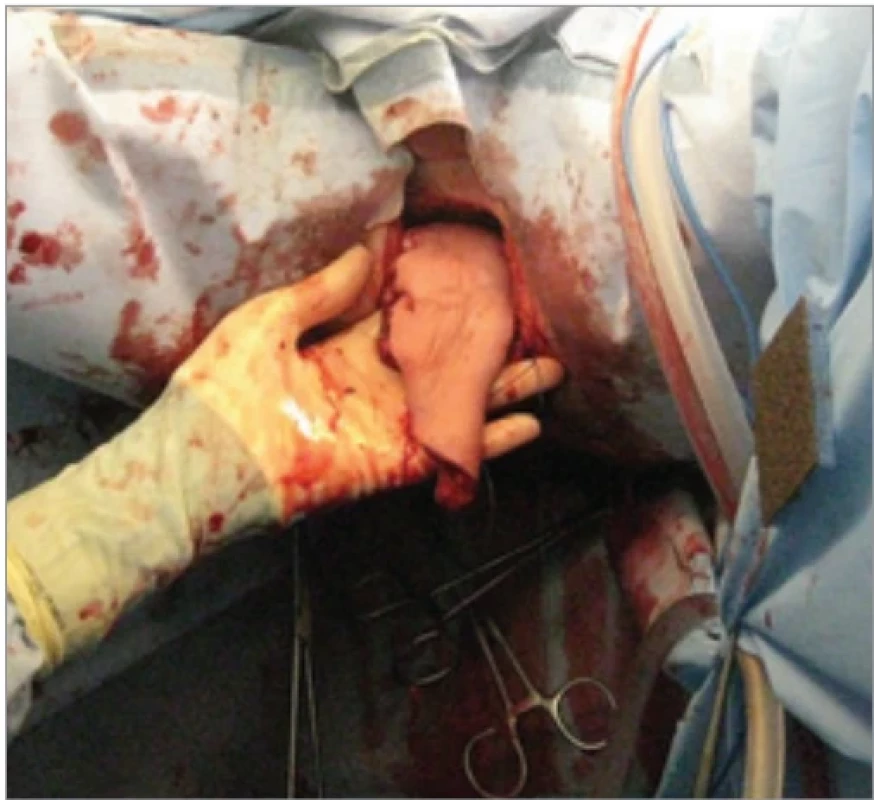
4. Flap’s position after its defi nitive fi xation; note that two drains were placed to prevent fl uid accumulation. 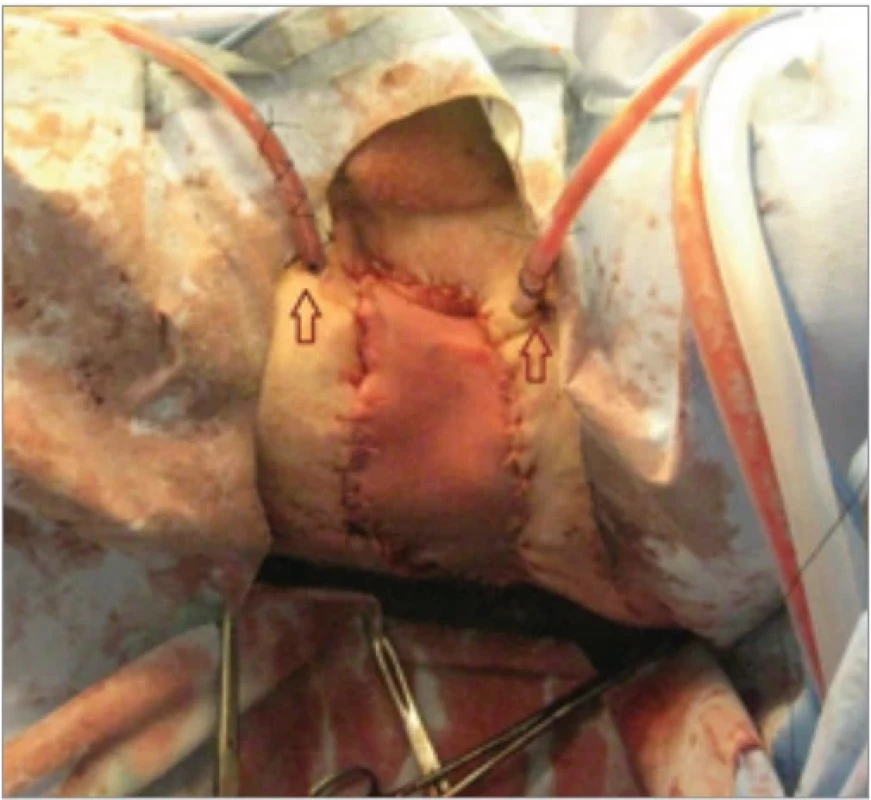
5. Patient 1 – 3.5 years post-operative. 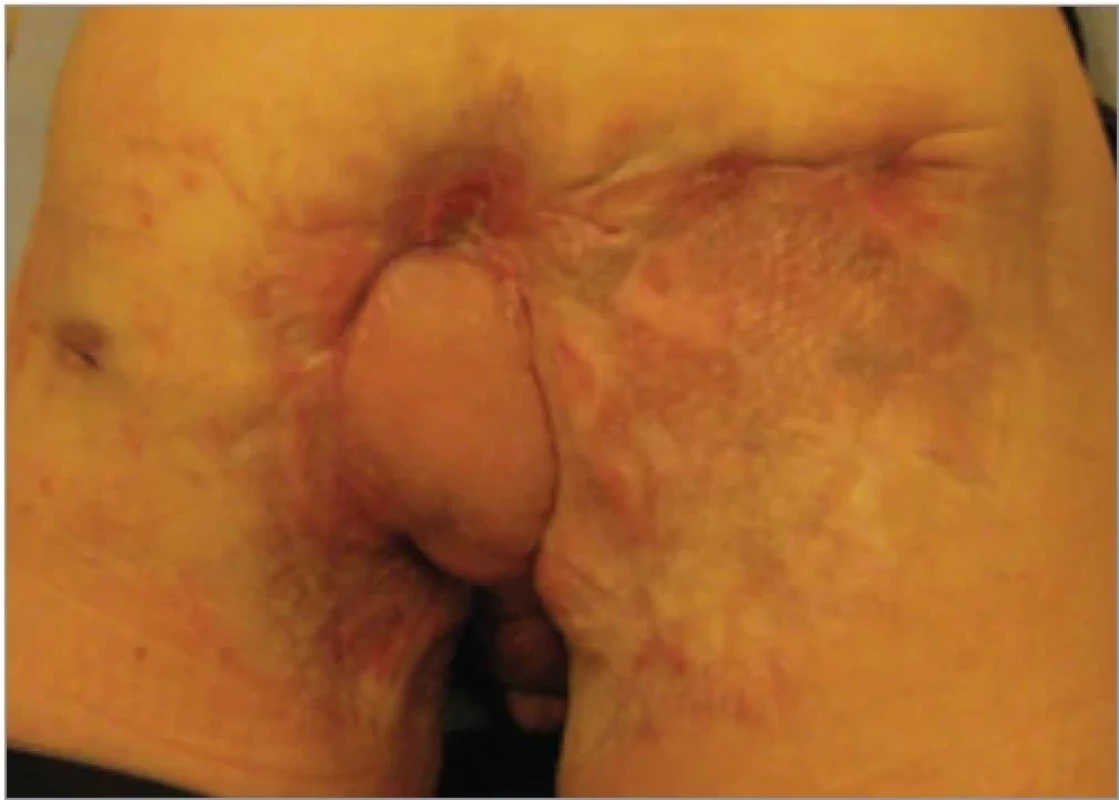
6. Patient 2 – preoperative skin markings, donor-site. 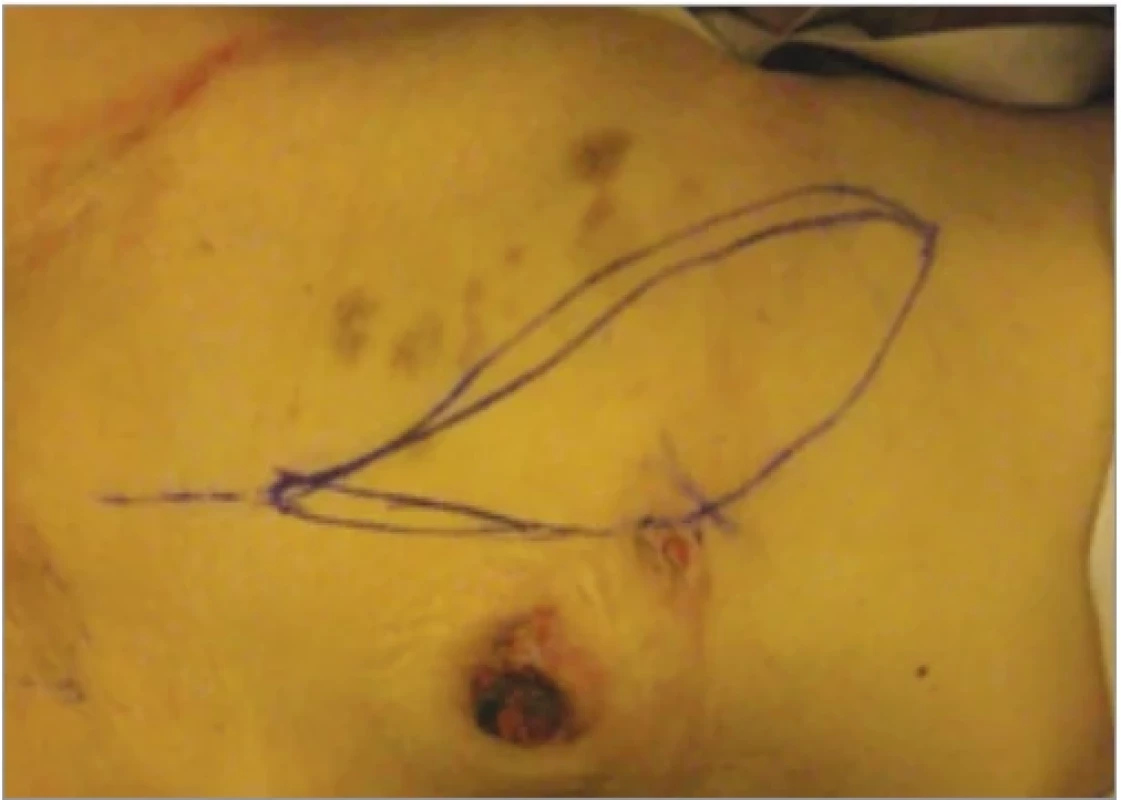
While hospitalized, the patient developed a superficial parietal abscess at the donor site. This was managed with surgical drainage, bacteriological sampling, and targeted antibiotic therapy, leading to both clinical and biological improvement.
Additionally, despite abdominal wall reinforcement with a polyglactin mesh, the patient developed an uncomplicated incisional hernia which was managed conservatively.
Two areas of minimal dehiscence were noted along the flap’s circumference which after bi-daily local wound care, improved considerably. The patient was discharged from hospital on day 22. He was initially kept under close observation with frequent consultations, which were gradually spaced out. The patient is now seen annually for routine follow-up.
At the time of writing, the patient has been followed for three and a half years without any recurrence of hidradenitis suppurativa or flap-related complications (Fig. 5).
Patient 2
A 67-year-old woman, diagnosed with hidradenitis suppurativa at the age of 47, presented with focalized lesions affecting the intergluteal cleft and bilateral inguinal regions. Her medical history was significant for active smoking, obesity, unilateral carotid stenosis, high blood pressure, and severe lower extremity arterial disease. Her disease course was characterized by multiple exacerbations requiring drainage procedures and antibiotic treatment (long - -term doxycycline treatment), along with extensive local wound care.
She was treated with the same biologic protocol (adalimumab) as Patient 1.
Surgical management was performed under general anesthesia. Incisions were made according to preoperative markings, isolating a skin paddle overlying the rectus abdominis muscle (Fig. 6). The fascial sheath of the muscle was incised, and a caudal dissection was carried out to reach the flap’s pivot point at the deep inferior epigastric pedicle, while preserving the pubic insertion of the muscle. The flap was then tunneled through a preformed window to reach the perineal defect and positioned within the intergluteal cleft. Before the fixation of the flap, the inguinal lesions were addressed. Following their excision, dissection was extended to the adductor muscles to ensure a viable recipient bed for bilateral skin graft placement (Fig. 7). In the final stage of the operation, the patient was repositioned in the prone position to facilitate posterior fixation of the flap (Fig. 8, 9).
7. Inguinal lesions are excised; the dissection is extended to the adductor muscles to prepare a recipient bed for bilateral skin graft placement. 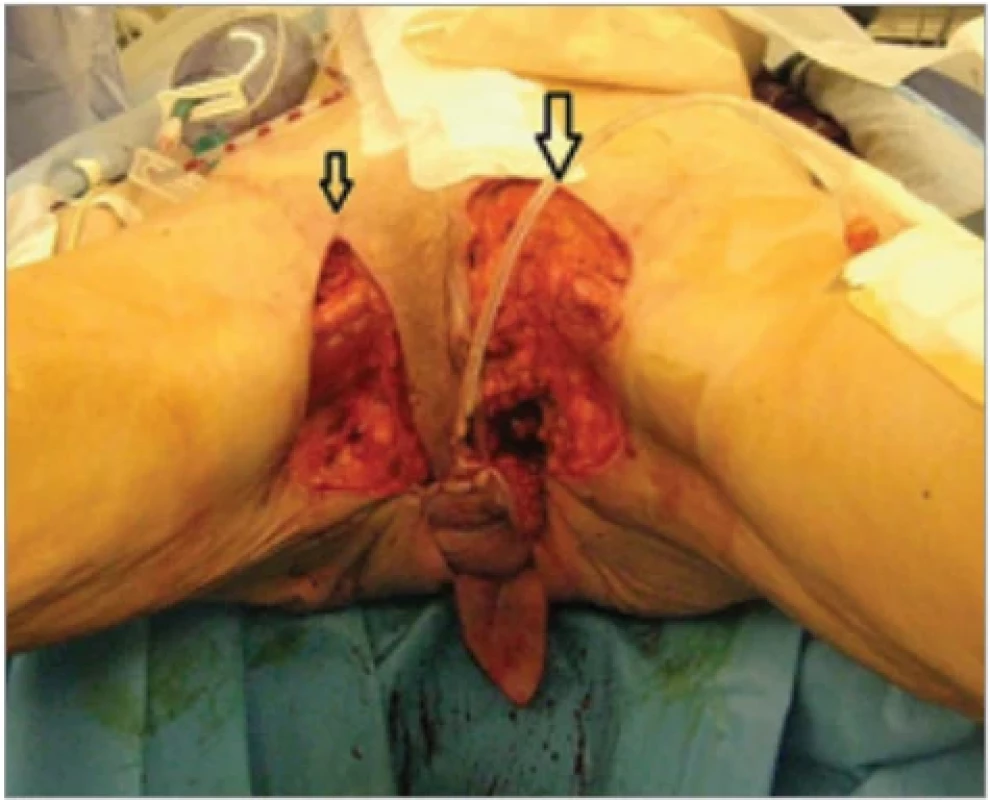
8. Flap’s position after tunneling to the perineal region; note that the patient was positioned in a prone position. 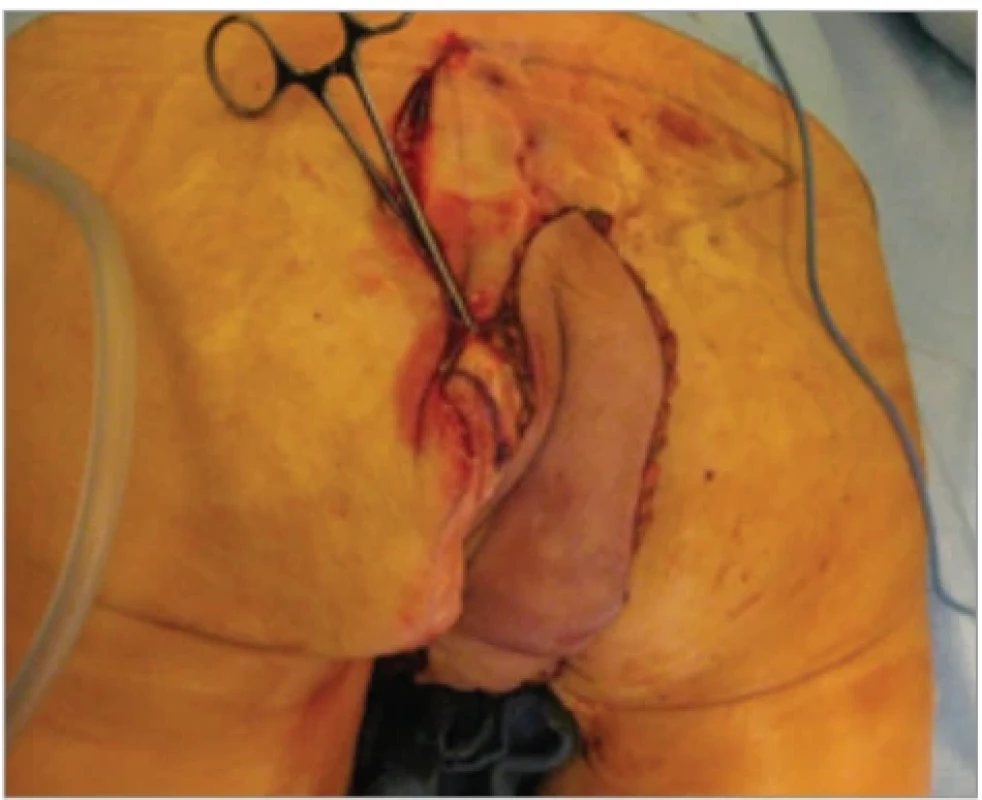
9. Flap’s position after its defi nitive fi xation. 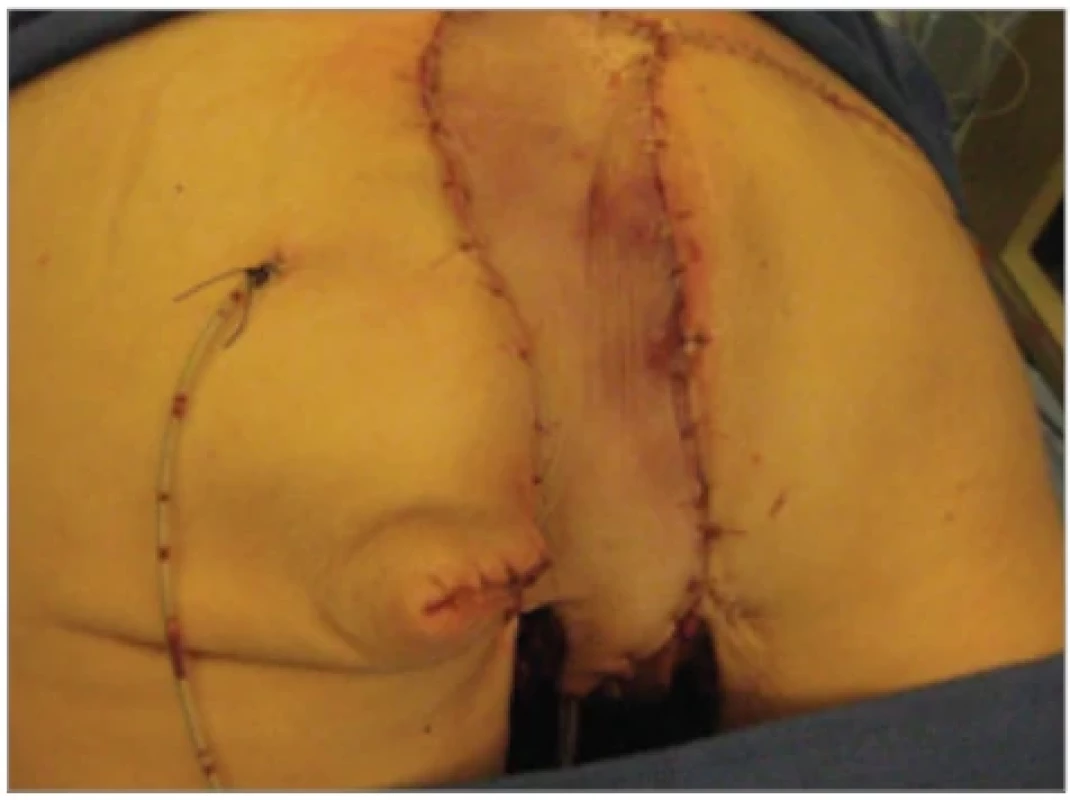
10. Patient 2 – 2.5 years post-operative. 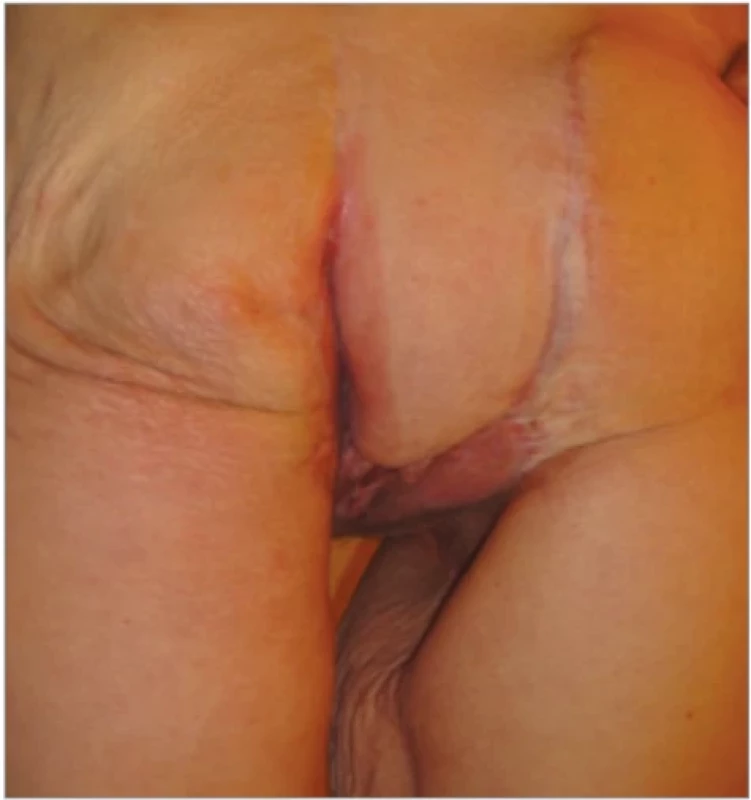
To reinforce the abdominal wall and reduce the risk of an incisional hernia, an absorbable polyglactin mesh was placed also placed in this patient.
No complications were noted at the donor site, and the bilaterally grafted inguinal regions showed uneventful healing. In the immediate postoperative period, the flap exhibited signs of venous congestion, necessitating heparin therapy and local wound management. A vacuum-assisted closure device was initially placed; however, its use was discontinued due to flap maceration and poor patient tolerance. Local wound care with alginate dressings was introduced. The patient was discharged from the hospital after 2 weeks of satisfactory wound progression. However, after discharge, presumed inadequate access to proper local wound care and a period of relative malnutrition negatively impacted the flap’s evolution, leading to deterioration and necessitating readmission. During this second hospitalization, which lasted 2 weeks, nutritional support was optimized, and appropriate wound management was re-established. The patient was followed closely with frequent consultations, which were gradually spread out over time. She is now seen annually for follow-up. The patient has now been followed up for two and a half years without any recurrence of hidradenitis suppurativa or flap-related complications (Fig. 10).
Discussion
Hidradenitis suppurativa (HS) is a chronic, recurrent inflammatory skin disorder that primarily affects intertriginous areas rich in apocrine glands and terminal hair follicles [1].
Prevalence estimates range from one to four percent, though HS is likely underdiagnosed due to its clinical resemblance to other conditions, particularly during early stages. Also, misdiagnosis as recurrent furunculosis or simple “boils” is common [2]. Although the exact pathogenesis remains incompletely understood, recent evidence supports the prevailing theory that HS is a chronic follicular occlusive disease involving the follicular portion of the folliculopilosebaceous unit [3]. The disease affects more frequently women, with a reported female-to-male ratio of approximately 3.5 : 1, and the median age at diagnosis is 31 years [7]. The axillae represent the most frequently affected area, followed by the inguinal folds, inner thighs, perineal and perianal regions, inframammary and mammary areas, buttocks, pubic region, scrotum, vulva, and, less frequently, the trunk, scalp, and retroauricular regions.
The clinical manifestations of HS are variable and progressive, ranging from recurrent inflamed nodules and abscesses to the development of draining sinus tracts (also referred to as skin tunnels) and dense bands of scar tissue. The associated symptoms are pain, malodor, purulent drainage, and disfigurement, and can significantly impair quality of life and exert a profound psychosocial burden on affected individuals [4].
The diagnosis of HS is clinical and is based on patient history and characteristic skin findings, with chronicity and recurrence as key hallmarks [8].
Onset during adolescence or early adulthood, recurrent or persistent lesions, and a positive family history support the diagnosis. HS has also been reported at a higher prevalence in patients with seronegative arthritis and Crohn’s disease with both conditions sharing clinical and histopathological features such as sinus tracts, granulomatous inflammation, and scarring [9].
Skin biopsy is generally not required for the diagnosis of HS but may be helpful in uncertain cases to exclude other conditions. Radiologic imaging is also not routinely indicated; however, magnetic resonance imaging can be valuable in assessing the extent of severe anogenital involvement especially in the preoperative setting [10].
Guidelines for the diagnosis, evaluation, and management of HS were published by the United States and Canadian Hidradenitis Suppurativa Foundations in 2019 [5], and more recently by the European S2k guidelines in 2024 [6]. These guidelines aim to standardize care, synthesize available evidence, and support clinicians in selecting and implementing evidence-based therapies. Treatment strategies for HS are broadly categorized into medical and surgical approaches. Management is guided by disease severity, often assessed using the Hurley staging system (Tab. 1) which, despite its simplicity and widespread clinical use, lacks sensitivity to change and is therefore unsuitable for clinical trial endpoints [5,6]. To address this, more responsive tools such as the Hidradenitis Suppurativa Clinical Response (HiSCR) [11] and the International Hidradenitis Suppurativa Severity Score System (IHS4) [12] have been developed and validated, with their use endorsed by international guidelines.
1. Hurley staging system for hidradenitis suppurativa [6,8]. ![Hurley staging system for hidradenitis suppurativa [6,8].](https://pl-master.mdcdn.cz/media/cache/media_object_image_small/media/image/76309c3feb146c1c62631fad316862fb.png)
Regardless of disease severity, patient education, smoking cessation, weight loss, psychosocial support, and appropriate management of lesions, pain, and comorbidities are essential components of care [5,6].
Medical management aims to reduce lesion development, control disease progression, and treat acute symptomatic lesions. This involves a range of therapies (e. g. topical and systemic antibiotics, biologic agents) which are extensively detailed in current guidelines and fall beyond the scope of this case report.
Surgical management targets the removal of active disease foci and chronic scarred tissue and is typically reserved for advanced or refractory HS [13]. Procedures range from localized excision of lesions to wide excision of extensively affected areas. The surgical approach must be individualized and tends to become more aggressive in higher-stage disease, particularly when exclusive medical therapy fails. Importantly, surgery should not be used in isolation; evidence showed that optimal outcomes are achieved through a multidisciplinary approach combining surgery with medical treatment and lifestyle modifications [14].
A variety of surgical techniques have been reported in the literature [13–20]. Punch debridement is typically used for small, active lesions to evacuate newly inflamed nodules, while surgical unroofing (also known as deroofing) offers a more conservative option for persistent nodules and sinus tracts in Hurley stage I or II [13]. For patients with Hurley stage II or III disease, the STEEP procedure (skin-tissue-sparing excision with electrosurgical peeling) has been introduced as an alternative to wide excision, allowing for the removal of affected tissue while preserving surrounding healthy skin [15].
Wide excision with reconstruction is considered a last-resort treatment for extensive and severe hidradenitis suppurativa (Hurley stage III), particularly when exclusive medical therapy and conservative surgical options have failed. In a cohort study of 253 patients, Deckers et al. reported that wide excision followed by secondary healing achieved remission in up to 50% of cases at a median follow-up of 36 months [16]. Favorable outcomes have also been observed with wide excision and reconstruction using pedicled thoracodorsal artery perforator flaps in high-stage axillary HS lesions [17]. A recent meta-analysis further supports this approach, demonstrating that wide excision combined with flap-based reconstruction is associated with a lower rate of postoperative recurrence compared to conservative surgical techniques, although the increased morbidity of more extensive procedures must be carefully considered [18].
In this report, we present two patients with severe and recurrent hidradenitis suppurativa lesions affecting the perineal, inguinal regions, and intergluteal cleft. Given the aggressive course of their disease, the exhaustion of medical and conservative options, and the frequency of relapses, an extensive surgical intervention was deemed necessary following multidisciplinary consultation and thorough discussion with both patients. Reconstruction following abdominoperineal resection was performed using a pedicled Taylor’s flap, chosen for its ability to effectively fill the resultant dead space with the rectus abdominis muscle. This approach is supported in the literature as a reliable option in similar reconstructive contexts, with favorable outcomes [19]. However, a recognized limitation of the Taylor’s flap is the iatrogenic weakening of the abdominal wall due to the removal of the rectus abdominis muscle and its sheath, as illustrated in the case of the first patient. An alternative technique, such as the deep inferior epigastric perforator (DIEP) flap, may help mitigate this complication. Unlike the transverse rectus abdominis muscle (TRAM) flap, the DIEP flap has been associated with reduced donor-site morbidity [20].
Conclusion
Hidradenitis suppurativa in its advanced, refractory form remains a therapeutic challenge, especially when extensive perineal involvement and multiple comorbidities are present. In the two cases presented, exclusive medical and conservative surgical therapies were ineffective in achieving long-term control. Abdominoperineal resection, albeit radical, offered a definitive solution in both patients. Reconstruction using a pedicled oblique rectus abdominis myocutaneous flap (Taylor’s flap) proved to be a reliable option for covering the extensive perineal defect, even in patients with significant comorbidities. These cases highlight the value of a multidisciplinary approach and demonstrate that, in highly selected patients, radical resection followed by tailored reconstructive strategies can be considered a salvage option when all other therapies have been exhausted.
Roles of the authors
Ismaïl Ben Ayad: conceptualization, methodology, writing – original draft and review, editing; François Chateau: investigation, validation. Both authors have reviewed and approved the final manuscript, and each author agrees to be held accountable for all aspects of the research.
Disclosure
The authors have no conflicts of interest to disclose. The authors declare that this study has received no financial support. All procedures performed in this study involving human participants were in accordance with ethical standards of the institutional and/or national research committee and with the Helsinki declaration and its later amendments or comparable ethical standards.
Sources
1. Revuz J. Hidradenitis suppurativa. J Eur Acad Dermatol Venereol. 2009, 23 (9): 985–998.
2. Prens LM., Bouwman K., Troelstra LD., et al. New insights in hidradenitis suppurativa from a population-based Dutch cohort: prevalence, smoking behaviour, socioeconomic status and comorbidities. Br J Dermatol. 2022, 186 (5): 814–822.
3. Zouboulis CC., Benhadou F., Byrd AS., et al. What causes hidradenitis suppurativa ?–15 years after. Exp Dermatol. 2020, 29 (12): 1154–1170.
4. Kouris A., Platsidaki E., Christodoulou C., et al. Quality of life and psychosocial implications in patients with hidradenitis suppurativa. Dermatology. 2016, 232 (6): 687–691.
5. Alikhan A., Sayed C., Alavi A., et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part II: topical, intralesional, and systemic medical management. J Am Acad Dermatol. 2019, 81 (1): 91–101.
6. Zouboulis CC., Bechara FG., Fritz K., et al. S2k guideline for the treatment of hidradenitis suppurativa / acne inversa – short version. J Dtsch Dermatol Ges. 2024, 22 (6): 868–889.
7. Canoui-Poitrine F., Le Thuaut A., Revuz JE., et al. Identification of three hidradenitis suppurativa phenotypes: latent class analysis of a cross-sectional study. J Invest Dermatol. 2013, 133 (6): 1506–1511.
8. Jemec GB. Clinical practice. Hidradenitis suppurativa. N Engl J Med. 2012, 366 (2): 158–164.
9. Van der Zee HH., van der Woude CJ., Florencia EF., et al. Hidradenitis suppurativa and inflammatory bowel disease: are they associated? Results of a pilot study. Br J Dermatol. 2010, 162 (1): 195–197.
10. Griffin N., Williams AB., Anderson S., et al. Hidradenitis suppurativa: MRI features in anogenital disease. Dis Colon Rectum. 2014, 57 (6): 762–771.
11. Kimball AB., Jemec GB., Yang M., et al. Assessing the validity, responsiveness and meaningfulness of the Hidradenitis Suppurativa Clinical Response (HiSCR) as the clinical endpoint for hidradenitis suppurativa treatment. Br J Dermatol. 2014, 171 (6): 1434–1442.
12. Zouboulis CC., Tzellos T., Kyrgidis A., et al. Development and validation of the International Hidradenitis Suppurativa Severity Score System (IHS4), a novel dynamic scoring system to assess HS severity. Br J Dermatol. 2017, 177 (5): 1401–1409.
13. Kohorst JJ., Baum CL., Otley CC., et al. Surgical management of hidradenitis suppurativa: outcomes of 590 consecutive patients. Dermatol Surg. 2016, 42 (9): 1030–1040.
14. Saunte DML., Jemec GBE. Hidradenitis suppurativa: advances in diagnosis and treatment. JAMA. 2017, 318 (20): 2019–2032.
15. Blok JL., Spoo JR., Leeman FWJ., et al. Skin-Tissue-sparing Excision with Electrosurgical Peeling (STEEP): a surgical treatment option for severe hidradenitis suppurativa Hurley stage II/III. J Eur Acad Dermatol Venereol. 2015, 29 (2): 379–382.
16. Deckers IE., Dahi Y., van der Zee HH., et al. Hidradenitis suppurativa treated with wide excision and second intention healing: a meaningful local cure rate after 253 procedures. J Eur Acad Dermatol Venereol. 2018, 32 (3): 459–462.
17. Elgohary H., Nawar AM., Zidan A., et al. Outcome of pedicled thoracodorsal artery perforator flap in the surgical treatment of stage II and III hidradenitis suppurativa of axilla. Ann Plast Surg. 2018, 81 (6): 688–693.
18. Riddle A., Westerkam L., Feltner C., et al. Current surgical management of hidradenitis suppurativa: a systematic review and meta-analysis. Dermatol Surg. 2021, 47 (3): 349–354.
19. Boccara D., Lefevre JH., Serror K., et al. Short - and long-term morbidity after Taylor flap (oblique rectus abdominis flap) for perineal reconstruction after abdominoperineal resection: a single-center series of 140 patients. J Plast Reconstr Aesthet Surg. 2024, 93 : 163–169.
20. Qiu SS., Jurado M., Hontanilla B. Comparison of TRAM versus DIEP flap in total vaginal reconstruction after pelvic exenteration. Plast Reconstr Surg. 2013, 132 (6): 1020e–1027e.
Ismaïl Ben Ayad, MD, MSurg
Avenue Hippocrate 10
1200 Brussels
Belgium
chirurgie.iba@gmail.com, ismail.benayad@uclouvain.be
Submitted: 11. 4. 2025
Accepted: 22. 11. 2025
Labels
Plastic surgery Orthopaedics Burns medicine Traumatology
Article was published inActa chirurgiae plasticae

2025 Issue 4-
All articles in this issue
- Will robots replace us?
- Association between the maxillofacial injury severity score to helmet types, neutrophil lymphocyte ratio and length of stay
- Anterior approach to posterior auricular artery in microvascular anastomosis – a new concept in replanting amputated ears – experience from four successful cases and literature review
- Treatment and outcomes of rectus diastasis in post-bariatric patients and postpartum women – a systematic review
- Taylor’s flap reconstruction following abdominoperineal resection for advanced hidradenitis suppurativa – a case series and literature review
- Intraoperative cone beam computed tomography in craniomaxillofacial traumatology – a case report
- Digital retractile keloid in a context of spontaneous keloids – a case report
- Extensive keloid scar of the left auricle – combined treatment of excision followed by brachytherapy: a case report
- Post-otoplasty keloid scars – treatment strategies and case reports
- Large urethral false passage during a routine catheterization – low threshold for better outcome
- Acta chirurgiae plasticae
- Journal archive
- Current issue
- Online only
- About the journal
Most read in this issue- Treatment and outcomes of rectus diastasis in post-bariatric patients and postpartum women – a systematic review
- Anterior approach to posterior auricular artery in microvascular anastomosis – a new concept in replanting amputated ears – experience from four successful cases and literature review
- Association between the maxillofacial injury severity score to helmet types, neutrophil lymphocyte ratio and length of stay
- Taylor’s flap reconstruction following abdominoperineal resection for advanced hidradenitis suppurativa – a case series and literature review
Login#ADS_BOTTOM_SCRIPTS#Forgotten passwordEnter the email address that you registered with. We will send you instructions on how to set a new password.
- Career

