-
Medical journals
- Career
Pachychoroid Disease of the Macula – Case Report
Authors: A. Stěpanov; J. Studnička; N. Jirásková
Authors‘ workplace: Fakultní nemocnice Hradec Králové, Oční klinika, přednostka prof. MUDr. Naďa Jirásková, Ph. D., FEBO ; Univerzita Karlova, Lékařská fakulta v Hradci Králové, katedra očního lékařství
Published in: Čes. a slov. Oftal., 74, 2018, No. 5, p. 208-212
Category: Case Report
doi: https://doi.org/10.31348/2018/5/6Overview
Pachychoroid disease of the macula includes four stages: pachychoroid pigment epitelopathy, central serous chorioretinopathy, pachychoroid neovasculopathy, and polypoid choroidal vasculopathy. Their basic common features are increase in choroidal thickness ≥ 300 μm, pathologically dilated choroidal vessels in the Haller’s layer with thinning of Sattle's layer and choriocapillaris layer. We report the case of a woman at the age of sixty-one, in which we have monitored the complete development of this disease within 8 years.
Key words:
pachychoroid disease of the macula, choroidal neovascular membrane, choroid, EDI-OCT
INTRODUCTION
Rapid progress in imaging methods of the posterior segment has brought a new insight into the etiology and pathogenesis of chorioretinal pathologies. A prime example of this trend is the development of optical coherence tomography (OCT). Following the first mention of examination of the retina using the OCT method in 1991 (10), further development led to a marked improvement in the resolution of images, and also to better quality display of the retinal structure (1, 5, 7).
In 2008, Spaide et al. introduced the EDI (Enhanced Depth Imaging) module, with improved display of the depth of the incision (fig. 1) (19), which enabled even more precise structural and functional analysis of the retina and choroid (2). Evaluation with the aid of EDI-OCT has brought a new insight into the pathogenesis of a range of pathologies.
1. Signs of central serous chorioretionpathy of the left eye 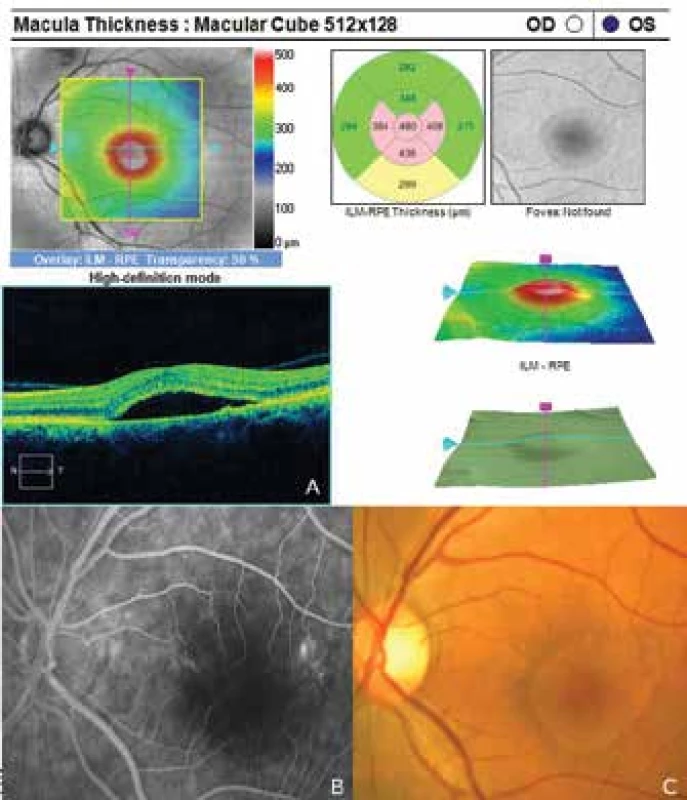
In 2013 Warrow et al. first used the term “pachychoroid”, and defined it as an abnormal and persistent increase of choroidal thickness > 300 µm (23). Further typical symptoms in connection with the presence of “pachychoroid” disease are dilation of the capillaries of the Haller's layer and thinning of the choriocapillaris and Sattler's layer (4, 13, 24). It is assumed that this condition has an autosomal dominant type of heredity, and that pachychoroid disease of the macula may occur in connection with the further effect of various exogenous and/or endogenous factors.
CASE REPORT
A 61-year-old woman was examined at the Department of Ophthalmology at the University Hospital in Hradec Králové in October 2009 due to deterioration of vision in the left eye. It ensues from her personal anamnesis that she had hitherto been receiving treatment for venous thrombosis of the right calf, and regularly used Clexane 0.4 ml/day. The previous ocular anamnesis was without any remarkable features.
The patient's ocular complaints had persisted for 2 months, in which she had noticed a progressively enlarging grey spot in the centre of the visual field of the left eye. Initial visual acuity in the left eye was 6/20, which was not improved by correction. Visual acuity in the right eye was 6/6 natural. The finding on the anterior segment of the eye was bilaterally commensurate to age. Fine pigment clusters were evident in the central landscape of the right eye, in the left eye a rounded transparent deposit was present in the macula, with discoid yellowish precipitates (fig. 1C). EDI-ICT (Zeiss, Cirrus) of the central landscape of the left eye showed serous ablation of the neuroretina (fig. 1A), thickening of the choroid (choroidal thickness was 345 µm – fig. 2). Central retinal thickness (CRT) was 480 µm. Fluorescence angiography (FAG) demonstrated mottled hypefluorescence in the temporal section of the macula and one point of grading fluorescence (fig. 1B). On the basis of the objective finding and the patient's anamnesis, a diagnosis of central serous chorioretinopathy (CSC) was determined in the left eye, and pigment epitheliopathy of the macula in the right eye. Direct photocoagulation of the saturation point was also performed in the left eye, with a subsequent improvement of the condition. Visual acuity in the left eye 2 months after laser therapy was 6/7.5. However, in June 2014 there followed recurrence of ablation of the neuroretina in the macula of the left eye (fig. 3), on FAG there was an evident enlargement of the surface of mottled hyperfluorescence in the temporal section of the macula. Visual acuity was 20/63. On the basis of the results of FAG, photodynamic therapy (PDT) was performed with verteporfin (Visudyne, Novartis), with half exposure time. The applied therapy was again followed by a subjective and objective improvement of the finding. Visual acuity in the left eye 5 months after PDT improved to 20/32. OCT of the central landscape of the left eye (fig. 4) demonstrated an improvement of the condition and reattachment of the neuroretina to the retinal pigment epithelium, CRT was 189 µm. In March 2016 the patient reported to the centre due to a further deterioration of visual acuity in the left eye (20/63). On the ocular fundus there was again evident edema of the central landscape, hard exudates and mottled intraretinal haemorrhages (fig. 5C). OCT-angiography demonstrated the presence of vascularisation of veins of the Haller's layer (tangled network) (fig. 5B) and ablation of the RPE (fig. 5A). The condition was concluded as pachychoroid neovasculopathy with complicated occult choroidal neovascular membrane (CNV). A decision was taken to perform a further session of PDT with half time exposure and subsequent treatment with micropulse laser. The procedure was performed without complications, but the condition continued to deteriorate. In March 2017 the finding was complicated by haemorrhage beneath the RPE (fig. 6A, C). Performed FAG demonstrated a finding of a polypoid lesion and through-growing choroidal capillaries in the temporal section of the macula (fig. 6B). The condition was concluded as polypoid choroidal vasculopathy, and commencement of treatment with Lucentis was recommended in a pro re nata regime (total 9 injections). During the course of treatment there was a pronounced improvement of the finding on the ocular fundus of the left eye – a reduction of RPE ablation was recorded, the haemorrhage was absorbed (fig. 7). Central visual acuity remained at the level of 20/100 during the course of treatment with Lucentis. At present we are continuing with the treatment.
2. Pachychoroid, thickness of choroidea 342 μm 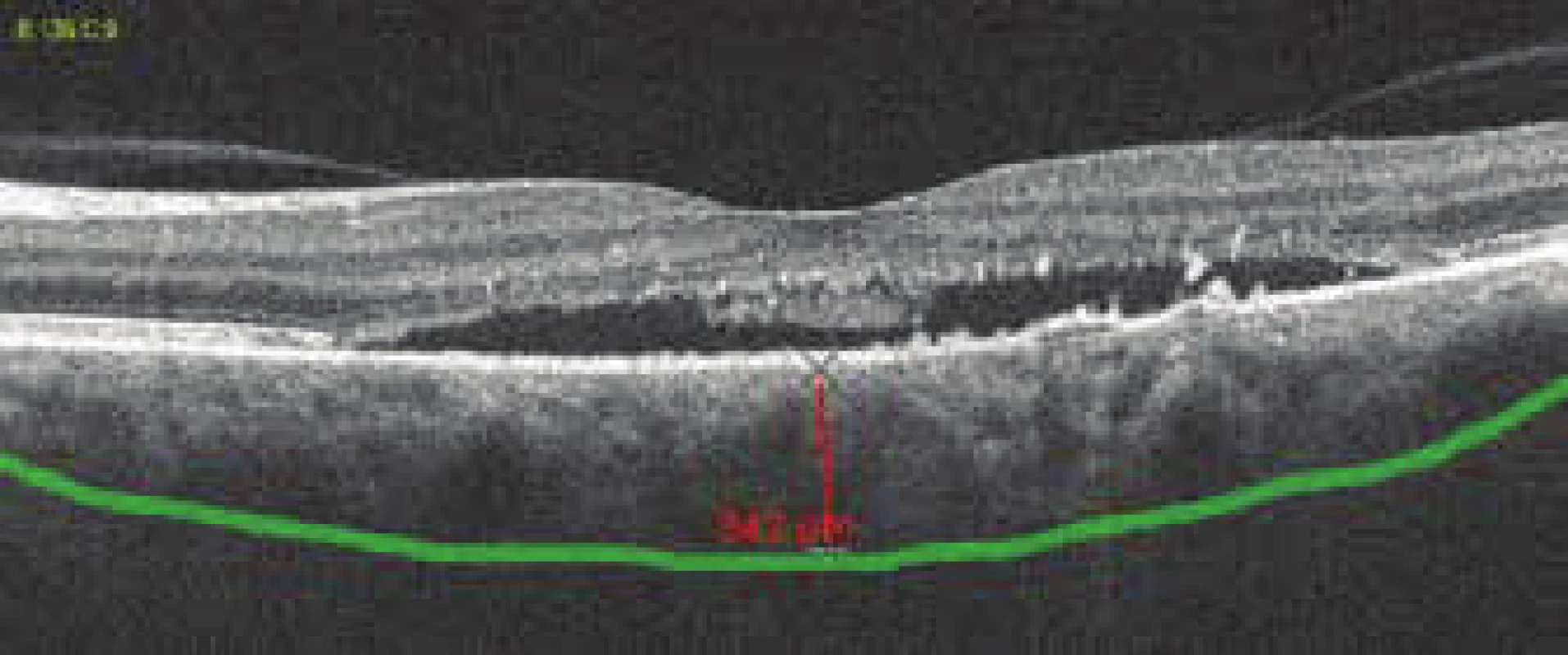
3. Recurrence of central serous chorioretinopathy after 5 years 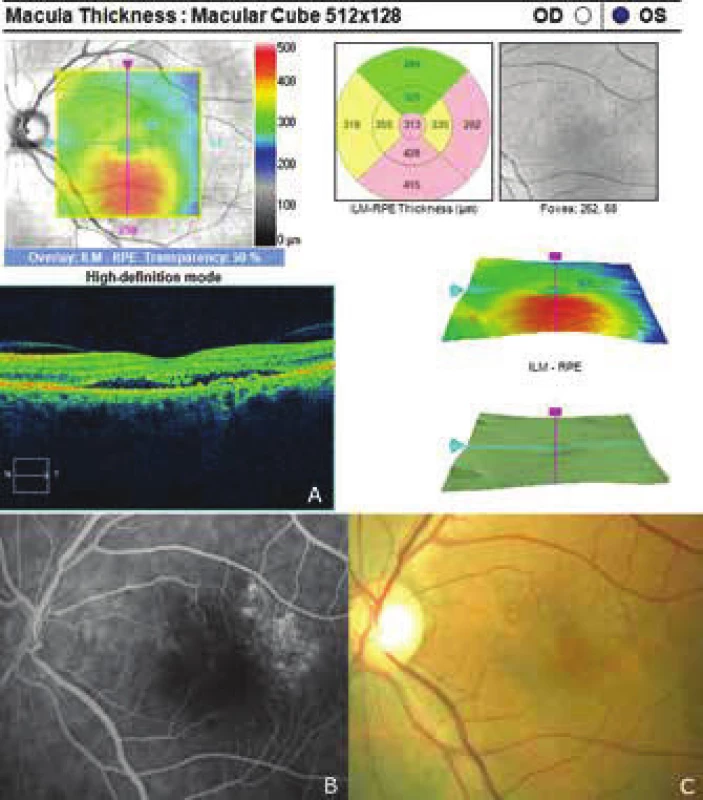
4. Improvement of condition after PDT, reduction of ablation of neuroretina 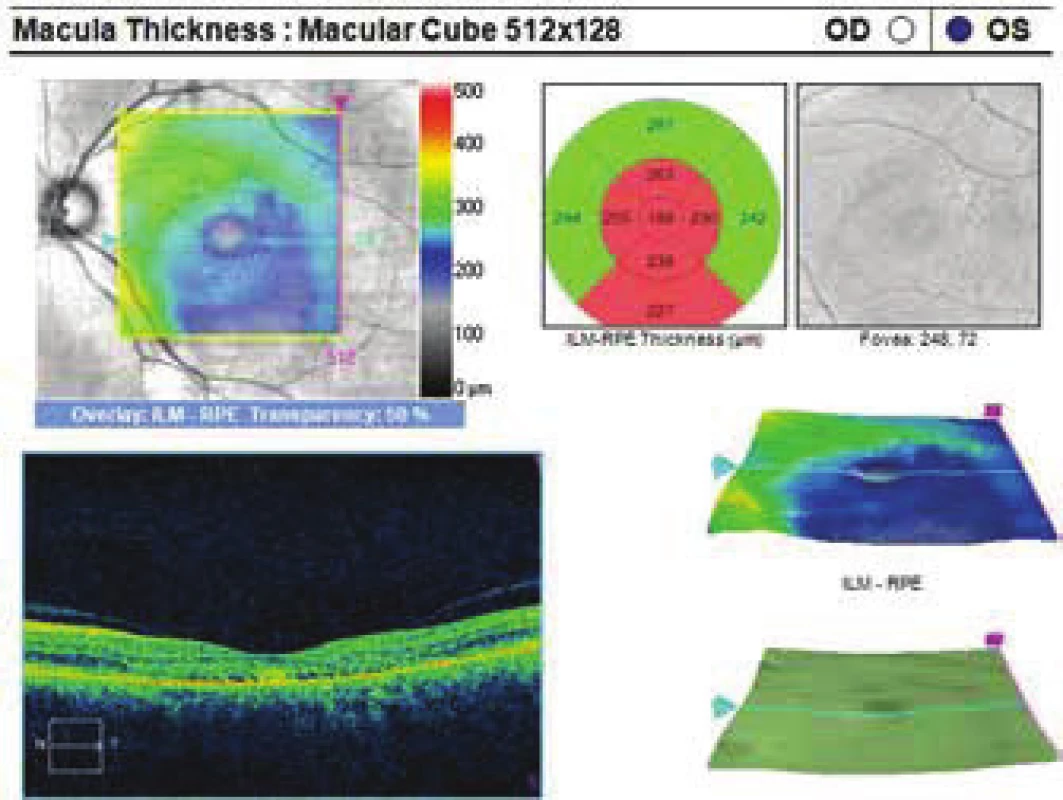
5. Finding of pachychoroid neovasculopathy. The red arrow indicates vascularisation of the veins in the Haller's layer on angio- -OCT (tangled network) 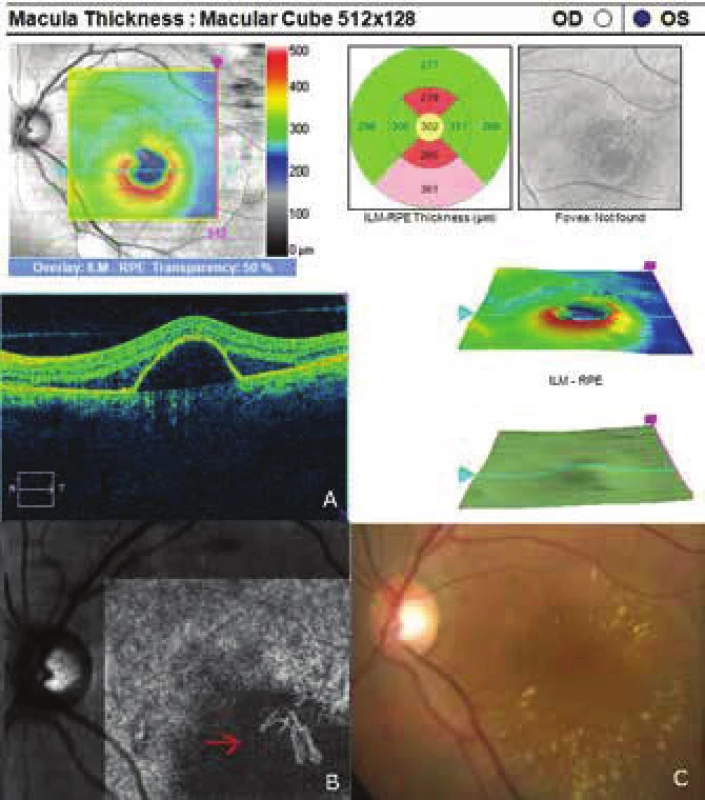
6. Finding of polypoid choroidal vasculopathy 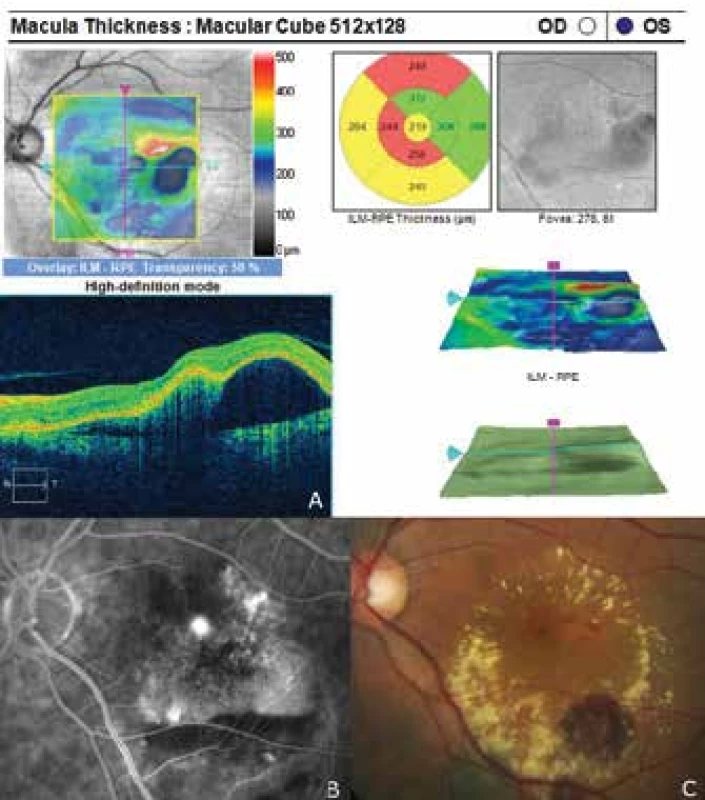
7. Improvement of condition of left eye following commencement of anti-VEGF treatment 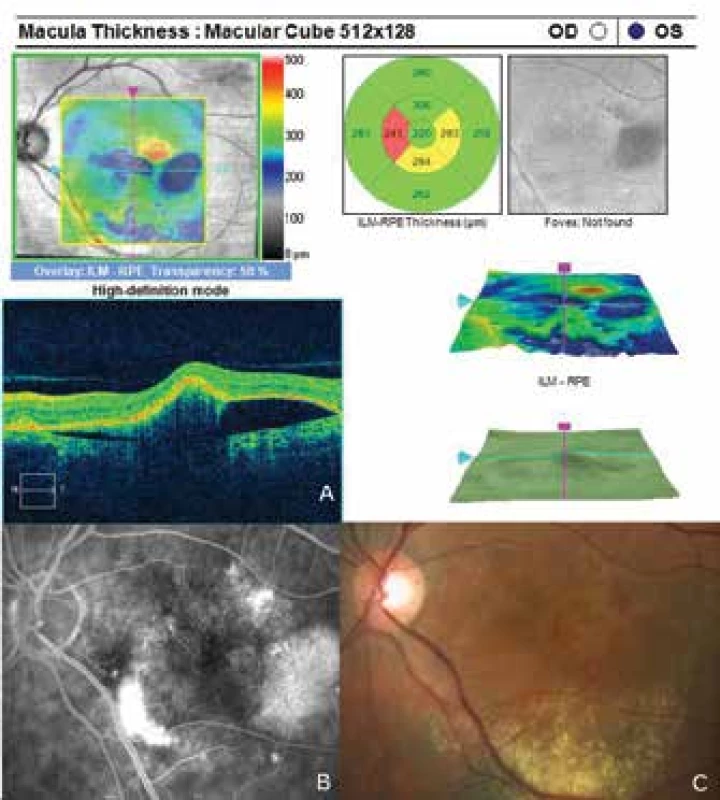
DISCUSSION
The group of pathologies included among pachychoroid diseases of the macula include central serous chorioretinopathy, in which a role is played by choroidal congestion and hyperpermeability and subsequent onset of serous detachment of the neuroretina and RPE, pachychoroid pigment epitheliopathy, which represents forme fruste CSC, pachychoroid neovasculopathy, which is characterised by the presence of ocult CNV, and finally polypoid choroidal vasculpathy, which is considered the most advanced form of pachychoroid disease of the macula (16, 20). With the progression of the pathology a progressive transition takes place to the next stage of the disease (3). We documented this also in our patient.
Photodynamic therapy with verteporfin is one of the treatment methods used for patients with central serous chorioretinopathy (21). It is known that the primary effect of PDT is the formation of singlet oxygen and free radicals with subsequent damage to the endothelial choriocapillaris (18).
This damage leads to an activation of throbocytes and the development of thrombosis in the choroidal channel, with an accompanying reduction of permeability of the veins in the choroid in the treated area. A further positive effect of this treatment in patients with CSC is the generation of inflammatory reaction. Verteporfin enters into the serous submacular fluid, and its activation releases pro-inflammatory factors which induce permanent adhesion between the neuroepithelium of the retina and the RPE (17). Vysloužilová et al. verified the effectiveness of PDT treatment with Visudyne in a reduced regime on 28 patients with CSC, with a favourable result (22). In our patient also, PDT treatment with reduced time had a positive effect. For further manifestations of the disease, repeated PDT treatment was no longer effective, and as a result intravitreal applications of Lucentis were commenced.
The development of occult CNV is a typical manifestation of pachychoroid neovasculopathy (15). Matsumoto et al. in their study described the benefit of intravitreal therapy with VEGF inhibitors in the case of this complication (14). In our patient we recorded the occurrence of occult CNV as a transition to pachychoroid neovasculopathy (PNV), and treatment with Lucentis was accompanied by an improvement of the anatomical finding and stabilisation of visual acuity. Despite this, however, we were unfortunately not able to prevent the progression of the condition and transition to polypoid choroidal vasculopathy (PCV).
A number of studies have evaluated the effect of anti-VEGF treatment on patients with PCV (8, 11, 12). Kokame et al. stated that monthly intravitreal injections of ranibizumab led to stabilisation of vision in all patients with PCV after 6 months of treatment, and the anatomical finding in the neuroretina also improved in 80% of eyes (11). Nevertheless, a reduction of polyps of the choroid was recorded in only 33% of patients. Lai et al. demonstrated that initial monthly treatment with bevacizumab in patients with PCV had a limited effect on the regression of polyps, since the polyps persisted in all patients after 3 months of treatment despite a functional improvement and reduction of central retinal thickness (12). Hikichi et al. presented their results of a two-year prospective study with the use of monotherapy with ranibizumab in a “pro re nata” regime on 75 patients with PCV (9). Despite the promising result of functional improvement, the finding of polyps persisted in all patients throughout the entire period of treatment. Treatment of PCV with VEGF blockers has a lesser anatomical effect than in the case of CNV types 1 and 2. Also in the case of our patient the polyp lesions of the choroid are still present despite the applied treatment.
The increased thickness of the choroid and the absence of drusens differentiate pachychoroid disease of the macula from ARMD and other degenerative diseases which predispose the patient to the onset of CNV (6). In our case the patient did not have retinal drusens accompanying a choroidal thickness of 345 µm, which enabled us to determine a diagnosis of pachychoroid disease of the macula and exclude ARMD.
CONCLUSION
Pachychoroid disease of the macula is an acquired pathology with a presumed genetic predisposition, a typical finding of abnormal and persistent increase in choroidal thickness > 300 µm. In our case we recorded the complete development of the clinical picture of this pathology over the course of eight-year observation. Upon the application of anti-VEGF treatment there was an improvement of the anatomical condition on the ocular fundus. The incomplete return of visual acuity in the affected eye is linked to advanced changes in the macula
Rychlý pokrok v zobrazovacích metodách zadního očního segmentu přinesl nový pohled na etiologii a patogenezi chorioretinálních onemocnění. Názorným příkladem tohoto trendu je vývoj optické koherenční tomografie (OCT). Po první zmínce o vyšetření sítnice metodou OCT v roce 1991 [10], vedl další rozvoj k výraznému zlepšení rozlišení snímků a také ke kvalitnějšímu zobrazení struktury sítnice [1,5,7].
Spaide a kol. uvedl v roce 2008 OCT modul EDI (Enhanced Depth Imaging) s vylepšeným zobrazením hloubky řezu (obrázek 1) [19], který umožnil ještě přesnější strukturální a funkční analýzu sítnice a cévnatky [2]. Hodnocení pomoci EDI-OCT přineslo nový pohled na patogenezi řady onemocnění.
V roce 2013 Warrow a kol. poprvé použil pojem „pachychoroid“ a definoval ho jako abnormální a trvalý nárůst tloušťky cévnatky ≥ 300 μm [23]. Další typické známky spojené s přítomností „pachychoroidu“ jsou dilatace cév Hallerovy vrstvy a ztenčování vrstvy choriokapilaris a Sattlerovy vrstvy [4,13,24]. Předpokládá se, že tento stav má autozomálně dominantní typ dědičnosti a v závislosti na dalším působení různých exogenních a/nebo endogenních faktorů může vzniknout pachychoroidní choroba makuly.
The authors of the study declare that no conflict of interest exists in the compilation, theme and subsequent publication of this professional communication, and that it is not supported by any pharmaceuticals company.
Received: 12. 7. 2018
Accepted: 3. 11. 2018
Available on-line: 27. 3. 2019
MUDr. Alexandr Stepanov, Ph.D., FEBO
Oční klinika FN Hradec Králové
Sokolská 581, 500 05 Hradec Králové
Sources
1. Alam, S., Zawadzki, RJ., Choi, S. et al.: Clinical application of rapid serial fourier-domain optical coherence tomography for macular imaging. Ophthalmology, 113(8); 2006 : 1425–31.
2. Azar, G., Wolff, B., Mauget-Faÿsse, M. et al.: Pachychoroid neovasculopathy: aspect on optical coherence tomography angiography. Acta Ophthalmol, 95(4); 2017 : 421-427.
3. Dansingani, KK., Balaratnasingam, C., Naysan, J. et al.: En face imaging of pachychoroid spectrum disorders with swept-source optical coherence tomography. Retina, 36(3); 2016 : 499–516.
4. de Jong, EK., Breukink, MB., Schellevis, RL. et al.: Chronic central serous chorioretinopathy is associated with genetic variants implicated in age-related macular degeneration. Ophthalmology, 122(3); 2015 : 562-70.
5. Drexler, W., Morgner, U., Ghanta, RK. et al.: Ultrahigh-resolution ophthalmic optical coherence tomography. Nat Med, 7(5); 2001 : 636.
6. Gemenetzi, M., De Salvo, G., Lotery, AJ.: Central serous chorioretinopathy: an update on pathogenesis and treatment. Eye, 24; 2010 : 1743–1756.
7. Gloesmann, M., Hermann, B., Schubert, C. et al.: Histologic correlation of pig retina radial stratification with ultrahigh-resolution optical coherence tomography. Invest Ophthalmol Vis Sci, 44(4); 2003 : 1696–703.
8. Gomi, F, Sawa, M, Sakaguchi, H. et al.: Efficacy of intravitreal bevacizumab for polypoidal choroidal vasculopathy. Br J Ophthalmol. 92; 2008 : 70–73.
9. Hikichi, T., Higuchi, M., Matsushita, T. et al.: Results of 2 years of treatment with as-needed ranibizumab reinjection for polypoidal choroidal vasculopathy. Br J Ophthalmol. 97; 2013 : 617–621.
10. Huang, D., Swanson, EA., Lin, CP. et al.: Optical coherence tomography. Science, 254(5035); 1991 : 1178–81.
11. Kokame, GT., Yeung, L., Lai, JC.: Continuous anti-VEGF treatment with ranibizumab for polypoidal choroidal vasculopathy: 6-month results. Br J Ophthalmol. 94; 2010 : 297–301.
12. Lai, TY., Chan, WM., Liu, DT. et al.: Intravitreal bevacizumab (Avastin) with or without photodynamic therapy for the treatment of polypoidal choroidal vasculopathy. Br J Ophthalmol. 92; 2008 : 661–666.
13. Lehmann, M., Bousquet, E., Beydoun, T.: Pachychoroid: an inherited condition? Retina, 35(1); 2015 : 10-6.
14. Matsumoto, H., Hiroe, T., Morimoto, M. et al.: Efficacy of treat-and-extend regimen with aflibercept for pachychoroid neovasculopathy and Type 1 neovascular age-related macular degeneration. Jpn J Ophthalmol, 62(2); 2018 : 144-150.
15. Pang, CE., Freund, KB.: Pachychoroid neovasculopathy. Retina, 35(1); 2015 : 1–9.
16. Rosa, DM., Kunal, K., Dansingani, K.: The pachychoroid clinical spectrum. [online] Retina Today. May/June 2016. Dostupné na www: <http://retinatoday.com/2016/06/the-pachychoroid-clinical-spectrum/> .
17. Ruiz-Moreno, JM., Lugo, FL., Armadá, F., et al.: Photodynamic therapy for chronic central serous chorioretinopathy. Acta Ophthalmol, 88(3) ; 2010 : 371-6.
18. Schmidt-Erfurth, U., Laqua, H., Schlötzer-Schrehard, U. et al.: Histopathological changes following photodynamic therapy in human eyes. Arch Ophthalmol, 120(6); 2002 : 835-44.
19. Spaide, RF., Koizumi, H., Pozzoni, MC.: Enhanced depth imaging spectral-domain optical coherence tomography. Am J Ophthalmol, 146(4); 2008 : 496–500.
20. Stepanov, A., Studnička, J., Středová, M. et al.: Pachychoroidní choroba makuly. Cesk Slov Oftalmol, 1; 2018 : 72–6.
21. van Dijk, EHC, Dijkman, G., Boon, CJF.: Photodynamic therapy in chronic central serous chorioretinopathy with subretinal fluid outside the fovea. Graefes Arch Clin Exp Ophthalmol, 255(10); 2017 : 2029-35.
22. Vysloužilová, D., Kolář, P., Matušková, V.: Náš přístup k léčbě centrální serózní chorioretinopatie. In: Sborník prací 13. Vejdovského olomouckého vědeckého dne. Olomouc. Olomouc: Univerzita Palackého Olomouc, 2012. s. 59-60.
23. Warrow, DJ., Hoang, QV., Freund, KB.: Pachychoroid pigment epitheliopathy. Retina, 33(8); 2013 : 1659–1672.
24. Yoneyama, S., Sakurada, Y., Kikushima, W. et al.: Genetic factors associated with choroidal vascular hyperpermeability and subfoveal choroidal thickness polypoidal choroidal vasculopathy. Retina, 36(8); 2016 : 1535-41.
Labels
Ophthalmology
Article was published inCzech and Slovak Ophthalmology

2018 Issue 5-
All articles in this issue
- Ocular Manifestations of Granulomatosis with Polyangiitis
- Yield of Display Modules of Corneal Tomography for Early Diagnosis of Corneal Ectasia
- Changes of Central Corneal Thickness in Normotensive and Hypertensive Glaucoma
- Pilot Results of Implantation of the New Hydrophobic Intraocular Lens Zeiss LUCIA 611P in the Czech Republic
- Merkel Cell Carcinoma of the Eyelids (Clinical-Histological Study)
- Pachychoroid Disease of the Macula – Case Report
- Czech and Slovak Ophthalmology
- Journal archive
- Current issue
- Online only
- About the journal
Most read in this issue- Ocular Manifestations of Granulomatosis with Polyangiitis
- Merkel Cell Carcinoma of the Eyelids (Clinical-Histological Study)
- Pachychoroid Disease of the Macula – Case Report
- Pilot Results of Implantation of the New Hydrophobic Intraocular Lens Zeiss LUCIA 611P in the Czech Republic
Login#ADS_BOTTOM_SCRIPTS#Forgotten passwordEnter the email address that you registered with. We will send you instructions on how to set a new password.
- Career

