-
Medical journals
- Career
Pre-exposure prophylaxis, a new approach for HIV prevention: experience from the HIV Center of the Military University Hospital Prague
Authors: Z. Bartovská; M. Zlámal; P. Janská; M. Holub
Authors‘ workplace: Ústřední vojenské nemocnice Praha
Published in: Epidemiol. Mikrobiol. Imunol. 69, 2020, č. 1, s. 38-41
Category: Short Communication
Overview
Aims: To present our experience with pre-exposure prophylaxis (PrEP) at the HIV Center of the Military University Hospital Prague, to characterize a set of PrEP candidates, to assess the efficiency and safety of PrEP and to measure the incidence of sexually transmitted diseases (STDs) in the studied cohort.
Material and methods: In total, 99 PrEP candidates (98 men who have sex with men and one woman) were monitored during an 18-month period. An initial examination included acquiring a history, performing a physical examination, screening for HIV (serology) and other STDs and performing renal function tests.
Results: In total, 81 candidates (in the cohort of 99 candidates) received PrEP. During the course of the 18-month period, none of the clients were newly infected with HIV, and no side effects of the provided medication were recorded. A total of 21% of the 99 PrEP candidates were initially tested positive for STDs, including two clients with HIV. A total of 14% of the 81 PrEP clients contracted an STD during the monitoring period.
Conclusions: Experience amassed at the HIV Center of the Military University Hospital Prague over an 18-month period documented the demand for PrEP, assessed PrEP efficacy, indicated good tolerance and suggested that PrEP played a role in the decline in the HIV incidence in the Czech Republic. In addition, several new HIV cases and cases of other asymptomatic STDs were also detected.
Keywords:
HIV – Pre-exposure prophylaxis – Sexually transmitted diseases
INTRODUCTION
An estimated worldwide total of 36.9 million people were living with human immunodeficiency virus (HIV) at the end of 2018. That same year, 1.8 million people became newly infected [1]. These data confirm a slightly positive trend in the decline of HIV prevalence, which has been globally apparent from the second half of the 1990s. A cumulative total of 3,446 HIV cases have been reported in the Czech Republic as of the end of April 2019. In the Czech Republic, the HIV incidence has significantly increased over the past several years, with a maximum of 286 new cases reported in 2016. Since that time, there has been a slow decline over the past two years (Figure 1) [2]. High-risk subpopulations in the Czech Republic include men who have sex with men (MSM), sexual partners of persons with HIV with a detectable viral load, sex workers and intravenous drug users (IVDUs). Prevention measures are thus focused on these four groups.
1. New cases of HIV infection in the Czech Republic over the last 34 years [2] ![New cases of HIV infection in the Czech Republic over the last 34 years [2]](https://pl-master.mdcdn.cz/media/cache/media_object_image_small/media/image_pdf/6e1995b09210459aad590b2c451f3f06.png)
Key preventive measures include counseling on safer sex and, for IVDUs, harm reduction programs. A new prevention method for people who are HIV-negative and at high risk for HIV exposure is pre-exposure prophylaxis (PrEP). Pre-exposure prophylaxis is the administration of a combination of antiretroviral medications, specifically emtricitabine/tenofovir disoproxil 200/245 mg. PrEP is derived from experience with post-exposure prophylaxis (PEP) in health care professionals following needle-stick injuries and in individuals after possible sexual exposure and from prevention of HIV vertical transmission. The efficacy of PrEP for HIV transmission prevention has been shown by studies on different high-risk subpopulations, especially MSM [3]. Some studies were discontinued early as the HIV transmission rate was proven to be reduced by 86%, and all study participants were subsequently offered PrEP [4, 5].
Based on clinical study results, PrEP has been recommended by the World Health Organization (WHO) as an additional prevention choice, as part of a combination HIV prevention approach, since 2015 [6]. As a result, PrEP is currently used around the world, with government funding available in selected countries. In the Czech Republic, the authorized medicinal product emtricitabine/tenofovir disoproxil 200/245 mg was approved for PrEP use in August 2016. Subsequently, a guideline published by HIV specialists in Pilsen in November 2017 was acknowledged by the Society of Infectious Diseases of the Czech Medical Association of J. E. Purkyně in Prague in May 2018 [7]. Infectious diseases specialists with experience in the diagnosis and therapy of HIV and sexually transmitted diseases (STDs) are currently authorized to provide PrEP in the Czech Republic. The drugs must be paid for by each patient. Requirements for PrEP include examinations prior to the administration of PrEP and during treatment, including a physical examination, STD screening (including serology tests for HIV and hepatitis B and C (HB, HC) and renal function tests.
In accordance with internationally recommended guidelines and the abovementioned Czech guidelines, we prescribed emtricitabine/tenofovir disoproxil 200/245 mg for daily or on-demand dosing (two tablets 2 to 24 hours before a possible sexual exposure and then one tablet per day for two consecutive days). This drug combination meets key PrEP requirements: it is highly effective, even in the event of a missed dose, while exhibiting low toxicity and a minimal risk of side effects (e.g., reversible renal insufficiency and bone metabolism changes). Furthermore, dosing is simple, and interactions with other drugs, food or alcohol are uncommon.
MATERIAL AND METHODS
A total of 99 PrEP candidates were examined between 1st of October 2017 and 31st of March 2018 at the Department of Infections Diseases, First Faculty of Medicine, Charles University and Military University Hospital Prague. In total, 57 of the candidates were sent by Dům světla (House of Light; a social center of a non-govermental organization providing anonymous free STD testing and counseling) following anonymous STD screening, 37 candidates requested PrEP themselves, and five candidates were recommended to take PrEP because they had been given PEP previously. All candidates were aged 20–61 years (average 34 years) and included 98 MSM and one woman. Sixty-eight candidates were Czech nationals, while the remaining 31 were Slovak (14), Russian (5), French (2), Polish (2), Australian (1), Azerbaijani (1), Moroccan (1), Nigerian (1), Portuguese (1), Taiwanese (1), Ukrainian (1) and US (1) citizens. Twelve PrEP candidates reported HIV--positive partners, and five candidates had already been taking PrEP prescribed abroad (France, USA or Germany).
The initial examination of all candidates included acquiring a detailed history and performing a clinical examination focusing on the symptoms of HIV and other STDs. Baseline testing included serology testing for HIV, syphilis, and hepatitis B and C; detection of Chlamydia trachomatis and Neisseria gonorrhoeae by polymerase chain reaction (PCR) from urethral, pharyngeal and rectal swabs; urinalysis; and renal function tests. PrEP was offered to all candidates without medical contraindications (HIV positivity, renal failure, drug allergy and active HB infection). All PrEP clients received counseling on PrEP as an additional HIV prevention choice and safer sex counseling, including condom use. Candidates who tested positive for STDs were treated in cooperation with dermatovenerologists. Vaccination was provided to candidates according to their HB status. The clinical examination and laboratory tests were repeated in regular intervals during the monitoring period in agreement with the abovementioned Czech guidelines (serology testing for HIV every three months; renal function tests after the first three months and every six months thereafter; syphilis serology and PCR detection of Chlamydia trachomatis and Neisseria gonorrhoeae at least every six months and hepatitis B and C serology in seronegative clients once a year)[7].
RESULTS
Two candidates (in the cohort of 99 PrEP candidates) were initially tested positive for HIV. Of the remaining 97 candidates, 81 were administered PrEP until March 2019. Seventy-one clients used PrEP on a daily basis, and 10 clients used PrEP on demand. During the monitoring period, none of the clients were found newly infected with HIV, and no side effects of the provided medication were observed. The remaining 16 PrEP candidates postponed PrEP use due to STD treatment, in view of medication risks or the virological status of their HIV-positive partners.
Of the entire cohort, 47% of the PrEP candidates had a past history of STDs, and 12% had two STDs. A total of 21% of the PrEP candidates were initially tested positive for STDs (including HIV), and all had asymptomatic active infections. The prevailing STD in both history and initial testing was gonorrhea. A total of 14% of the 81 PrEP clients contracted an STD during the monitoring period, most frequently chlamydia or gonorrhea (Table 1 and 2).
1. Sexually transmitted diseases (STDs) in PrEP candidates 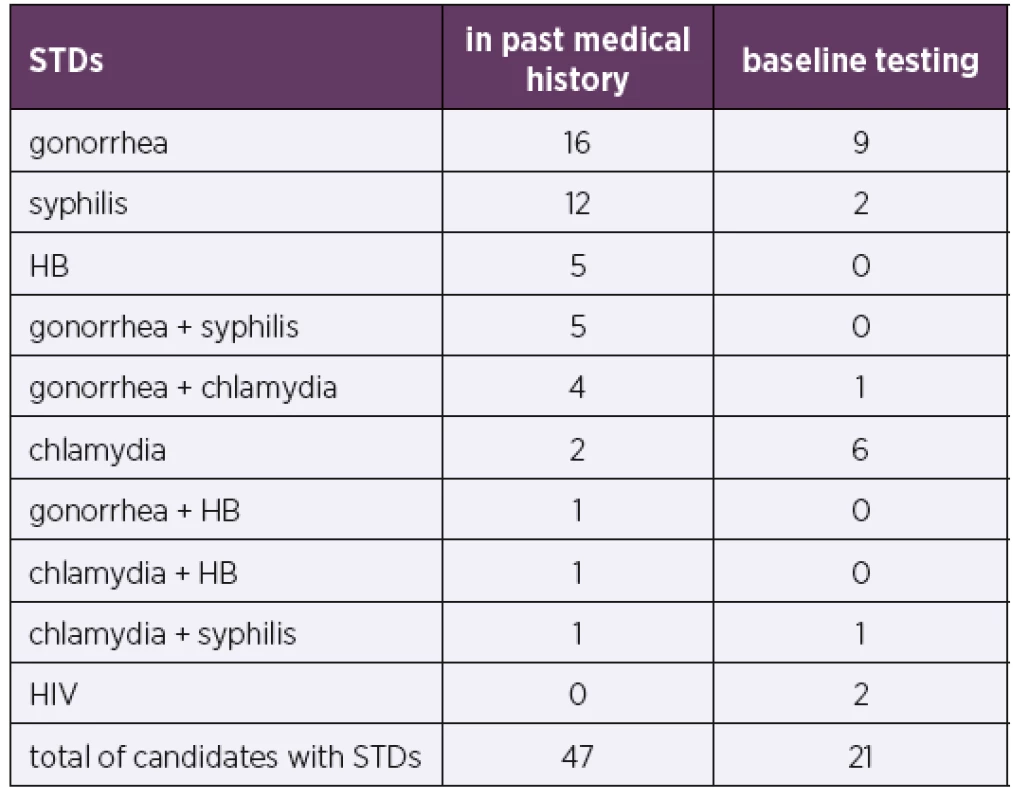
STDs – sexually transmitted diseases; HB – hepatitis B; HIV – human immunodeficiency virus
PrEP candidates (99)2. Sexually transmitted diseases (STDs) in PrEP clients 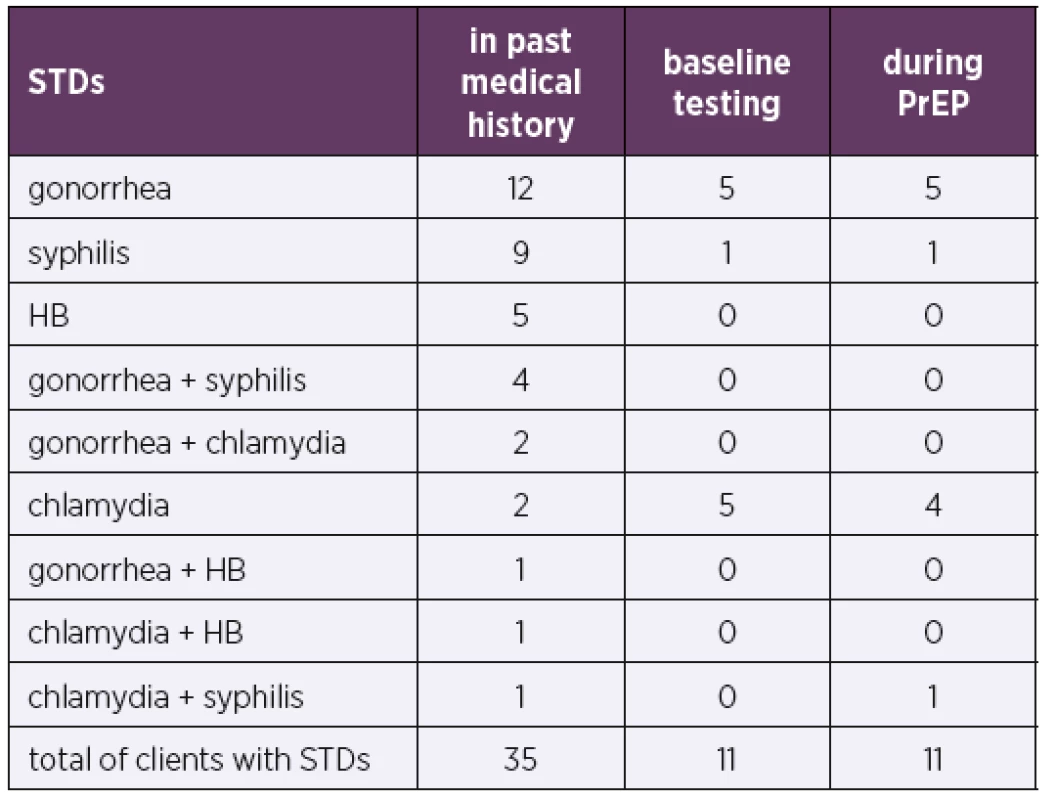
STDs – sexually transmitted diseases; HB – hepatitis B
PrEP clients (81)Baseline testing included tests for viral hepatitis. In total, 5% of the cohort of 99 PrEP candidates had a history of viral HB, 67% had been vaccinated against HB, and vaccination was recommended, and in some cases initiated, in the remaining candidates with negative HB serology.
DISCUSSION
Initial results from our HIV center indicate the existence of a high demand for PrEP, with PrEP prescribed to nearly 100 candidates over the course of the first 18 months since the launch of PrEP in the Czech Republic. Excluding one patient, all of our candidates were MSM, which is in agreement with epidemiological data [2]. Most candidates (68%) were Czech citizens, while the majority of foreigners (14%) were Slovak. The proportion of Slovaks, though mostly living and working in the Czech Republic, may be an indication of the unsatisfactory situation of preventive programs in Slovakia. A substantial number of candidates were sent by a social center following STD screening, which indicates that such organizations play an important role in HIV prevention.
In total, 82% of all candidates had initiated PrEP by the end of March 2019, with a majority preferring daily dosing. During the monitoring period, none of the clients were found newly infected with HIV, and no side effects of the provided medication were recorded, which highlights the efficiency and safety of the procedure. Our present experience confirms the importance of PrEP as an additional prevention choice and is in agreement with clinical studies [3, 4, 5].
Our data suggest that the introduction of PrEP in the Czech Republic, along with the newly introduced international recommendation calling for the start of antiretroviral therapy immediately after HIV diagnosis, may play a part in the recently observed decline in the incidence of new HIV cases (see Figure 1). The preceding increase in the HIV incidence in the Czech Republic reflected an epidemic in the MSM population (see Figure 1) [2].
Importantly, many new (mostly asymptomatic) STDs were detected in our cohort, both during initial exa-minations and during the course of further monitoring linked to the administration of PrEP. Studies show a mild increase in STD incidence in PrEP clients [8]. Possible interpretations of this trend include PrEP clients engaging in relatively risky behavior (including sex without condoms), increased STD screening during PrEP use and an overall increase in the incidence of STDs [9]. Nevertheless, a higher STD rate does not imply questioning the effectiveness of PrEP for HIV prevention. Recent studies also highlight the importance of frequent STD testing and early treatment of detected STDs [8]. The prevailing STD detected in our cohort was gonorrhea, followed by chlamydia, which is in agreement with STD incidence data in the Czech Republic [10]. An important outcome of baseline testing before PrEP administration was the detection of two new HIV cases in our cohort.
CONCLUSION
Our experience with PrEP documents a demand for this additional prevention choice, demonstrate its efficiency, indicates good tolerance and suggests that PrEP is playing a role in the decline in the HIV incidence in the Czech Republic over the past two years. The results also indicate that it is desirable to increase PrEP availability to people who are at an increased risk of HIV infection. To do so, greater awareness is required among both PrEP candidates and healthcare professionals. In addition, PrEP availability may also be increased by means of better financial affordability and by extending the authority to prescribe PrEP to other specializations and ambulatory care settings.
Do redakce došlo dne 7. 7. 2019.
MUDr. Zofia Bartovská, Ph.D.
Klinika infekčních nemocí 1. LF UK a ÚVN
U Vojenské nemocnice 1200
169 02 Praha 6
e-mail: zofia.bartovska@uvn.cz
Sources
1. World Health Organization. Data and statistics [Internet].2019 [cited 17 December 2019]. Dostupné na www: https://www.who.int/hiv/data/en/.
2. SZU-NRL pro HIV/AIDS. Nové případy infekce HIV v České republice v jednotlivých letech [Internet]. 2019 [cited 16 December 2019]. Dostupné na www: http://www.szu.cz/uploads/documents/CeM/HIV_AIDS/rocni_zpravy/2018/Grafy_k_tiskove_zprave_NRL_pro_HIV_AIDS_Trendy_vyvoje_a_vyskyt_HIV_AIDS_v_CR_v_roce_2018.pdf
3. Grant RM, Lama JR, Anderson PL, et al. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N Engl J Med, 2010;363(27):2587–2599.
4. Dolling DI, Desai M, Mc Owan A, et al. An analysis of baseline data from the PROUD study: an open-label randomised trial of pre-exposure prophylaxis. Trials, 2016;17 : 163.
5. Molina JM, Capitant C, Spire B, et al. On-Demand Preexposure Prophylaxis in Men at High Risk for HIV-1 Infection. N Engl J Med, 2015;373(23):2237–2246.
6. World Health Organization. Guideline on when to start antiretroviral therapy and on pre-exposure prophylaxis for HIV [Internet]. 2015 [cited 16 December 2019]. Dostupné na www: https://www.who.int/hiv/pub/guidelines/earlyrelease-arv/en/.
7. Dlouhý P. Pracovní postup pro poskytování preexpoziční profylaxe infekce HIV [Internet]. 2018 [cited 16 December 2019]. Dostupné na www: https://www.infekce.cz/PPPrEP18.htm.
8. Traeger MW, Cornelisse VJ, Asselin J, et al. Association of HIV Preexposure Prophylaxis With Incidence of Sexually Transmitted Infections Among Individuals at High Risk of HIV Infection. JAMA, 2019;321(14):1380–1390.
9. Montaño MA, Dombrowski JC, Dasgupta S, et al. Changes in Sexual Behavior and STI Diagnoses Among MSM Initiating PrEP in a Clinic Setting. AIDS Behav, 2019;23(2):548–555.
10. Regionální zpravodajství NZIS - Česká republika. Incidence – hlášené případy pohlavních nemocí podle věku a pohlaví [Internet]. 2019 [cited 16 December 2019]. Dostupné na www: https://reporting.uzis.cz/cr/index.php?pg=statisticke-vystupy--morbidita--incidence-dle-diagnoz--incidence-hlasene-pripady-pohlavnich-nemoci-podle-veku-a-pohlavi®ion=olk&year=2016.
Labels
Hygiene and epidemiology Medical virology Clinical microbiology
Article was published inEpidemiology, Microbiology, Immunology

2020 Issue 1-
All articles in this issue
- Biofilm-producing potential of urinary pathogens isolated from chronic and recurrent urinary tract infections and impact of biofilm on gentamicin and colistin in vitro efficacy
- Therapeutic potential of bacteriophages for staphylococcal infections and selected methods for in vitro susceptibility testing of staphylococci
- Clonal characterisation of Streptococcus pneumoniae strains using MLST and MLVA – Can MLVA improve the characterisation?
- The potential for use of non-thermal plasma in microbiology and medicine
- Biological agents of bioterrorism – preparedness is vital
- The first confirmed detection of Staphylococcus argenteus in the Czech Republic
- Vzpomínka na MUDr. Martinu Havlíčkovou, CSc.
- Zemřel doc. MUDr. Zdeněk Ježek, DrSc., Rytíř českého lékařského stavu
- Pre-exposure prophylaxis, a new approach for HIV prevention: experience from the HIV Center of the Military University Hospital Prague
- Epidemiology, Microbiology, Immunology
- Journal archive
- Current issue
- Online only
- About the journal
Most read in this issue- Therapeutic potential of bacteriophages for staphylococcal infections and selected methods for in vitro susceptibility testing of staphylococci
- Biological agents of bioterrorism – preparedness is vital
- The first confirmed detection of Staphylococcus argenteus in the Czech Republic
- Pre-exposure prophylaxis, a new approach for HIV prevention: experience from the HIV Center of the Military University Hospital Prague
Login#ADS_BOTTOM_SCRIPTS#Forgotten passwordEnter the email address that you registered with. We will send you instructions on how to set a new password.
- Career

