-
Medical journals
- Career
Determination of the coating thickness of HPMC hard capsules by near-infrared reflectance spectroscopy
Authors: J. Muselík 1; K. Dvořáčková 1; M. Rabišková 1; A. Bartošíková 1; M. Rosendorfová 1; M. Dračková 2; L. Vorlová 2
Authors‘ workplace: University of Veterinary and Pharmaceutical Sciences Brno, Faculty of Pharmacy, Department of Pharmaceutics, Czech Republic 1; University of Veterinary and Pharmaceutical Sciences Brno, Faculty of Veterinary Hygiene and Ecology, Institute of Milk Hygiene and Technology, Czech Republic 2
Published in: Čes. slov. Farm., 2009; 58, 155-159
Category: Original Articles
Overview
The aim of this study was to develop the method for off-line determination of hard capsules coating thickness. The polymer films studied were Eudragit® L 12.5 and Eudragit® S 12.5 applied on hypromellose capsules surface in a bottom-spray fluid-bed coater equipped with a Wurster column. The coating thickness reference values for calibration were obtained from microscopic analysis. Calibration model was created using partial least squares regression with six factors for Eudragit® L film and five factors for Eudragit® S film. The calibration and validation results led to following parameters: determination coefficients R2 were more than 0.98 and standard error of cross validation was 6.6 μm (coat thickness range was 15–235 μm) for Eudragit® L film and 8.3 μm (coat thickness range was 30–230 μm) for Eudragit® S film, respectively. Obtained results confirmed suitability of near infrared spectroscopy for coating process quality control in pharmaceutical industry.
Key words:
film-coating – Eudragit® L and S – hard capsules – coating thickness – near-infrared spectroscopyIntroduction
Currently, coating of hard capsules plays an important role in drug administration due to a number of reasons such as the protection of capsule filling (against moisture, light, oxygen), easier identification, and especially the possibility of controlled drug release. A clear advantage is the independence of the capsule coating process on its contents 1). The pH-dependent coating is efficient in drug site-specific delivery to different parts of the gastrointestinal (GI) tract such as duodenum, ileum or colon. Some coating polymers exhibiting pH-dependent solubility belong to the anionic copolymers of methacrylic acid and methylmethacrylate or ethyl acrylate well known under the brand name Eudragit®. Eudragit® L 12.5 and Eudragit® S 12.5 are 12.5% (w/w) aqueous isopropyl alcohol solutions containing Eudragit® L 100 or S 100. Eudragit® L 12.5 is soluble in a dissolution medium above pH 6.0, thus enabling an enteric coating to protect drugs against degradation in the stomach or to prevent irritation of the gastric mucosa. Eudragit® S 12.5 is soluble in a medium of a higher pH value, i. e. above 7.0 2), thusappropriatefor ileac or colonic drug delivery that can be used for local treatment of a variety of intestinal diseases or oral bioavailability improvement of some drugs such as peptides and proteins 3). Capsules coated with Eudragit® S are also prospective for oral vaccines delivery near to the Peyer’s patches 4).
Traditionally, the film coating thickness on the surface of dosage forms (tablets, pellets and capsules) has been determined either by applying a specified amount of the coating material, that is by measuring the weight gain, or by microscopic analysis of the dosage form cross-section until the desired end point is achieved. These off-line methods allow the coating evaluation at the end of the process or if the coating operation is interrupted. The Food Drug Administration (FDA) initiated the control of manufacturing processes by real-time monitoring of critical manufacturing points. Real-time monitoring has been used in pharmaceutical unit operations such as drying, blending, content-uniformity testing, coating etc. 5). Near-infrared spectroscopy (NIRS) has gained wide acceptance within the pharmaceutical industry for raw material testing, product quality control and process monitoring 6). When compared to traditional methods, NIRS offers many advantages. It is an instantaneous, non-destructive method, requiring minimal or no sample preparation 6–8). Once calibrated, NIRS is simple to operate and is well suited for the determination of the major components in many types of samples. NIRS has a possibility of working in the reflectance mode and the use of fibre probe modules coupled to the spectrophotometer makes it useful for in-process or at-line monitoring 9).
It has been shown that NIRS can be successfully used to predict the required coating amount sprayed on tablets 7, 10) and pellets 11) surfaces or to evaluate the plastic primary packing thickness 12). For quantitative NIR spectroscopy, it is crucial to propose a calibration model. The objective of this study was to develop new methods based on near-infrared spectroscopy as an alternative for rapid, non-destructive and reproducible determination of coating thickness of two different polymer films Eudragit® L (EL) and Eudragit® S (ES) applied as isopropyl alcohol solutions on hard hypromellose capsules.
EXPERIMENTAL PART
Materials
Caffeine as the model drug (Jilin Province Shulan Synthetic Pharmaceutical Co., Ltd., Shulan City, China) in a dose of 100 mg together with the soluble filler α lactose monohydrate q. s. (Cerapharm, Vienna, Austria) were filled into hard HPMC capsules of size 4 – Vcaps® (Capsugel, Colmar, France). Eudragit® L 12.5 or Eudragit® S 12.5 (Evonik Degussa, Darmstadt, Germany) were used as the coating materials. Polyethylene glycol (PEG) 6000 (Sigma-Aldrich, Prague, Czech Republic) was added into the Eudragit® coating solution as the plasticizer. All materials were of Ph. Eur. quality.
Hard capsules filling process and evaluation of prepared capsules
Hypromellose hard capsules were filled in a manual filling machine with a caffeine and lactose monohydrate mixture which was prepared by homogenization of 10.0 g of the model substance and 2.3 g of lactose monohydrate to fill 100 pieces of hard capsules. Weight uniformity of capsules and caffeine content were evaluated according to Ph. Eur. 5.
Coating preparation
Two hundred filled capsules were subsequently coated in a Wurster-M 100 coater (Medipo ZT, s. r. o., Czech Republic) using either a 66.67 g Eudragit® 12.5 isopropyl alcohol solution (S or L) with a mixture of 5.05 g of a 33% water solution of PEG 6000 and 28.28 g of isopropyl alcohol to reach the solid content of polymer 8.3%, according to the producer’s recommendation. Subsequently, within the coating process, the samples of 20 hard capsules with an increasing coating amount were withdrawn for the subsequent evaluation. Overall, seven batches for EL and eight batches for ES were prepared, and the experiment was carried out twice.
Reference determination of the layer thickness
The coating thickness was determined by optical analysis using an optical microscope (DN 45, Lambda, Prague, Czech Republic) connected to a CCD camera (Alphaphot, Nikon, Tokyo, Japan) and operated by an Ia32 software. Twenty different positions of 3 randomly selected coated hard capsules from each prepared batch were measured. The results were expressed as the mean thickness and standard deviation for each tested capsule separately.
NIRS measurements
Near infrared diffusion reflectance spectra were acquired with a FT-NIR Nicolet Antaris (Thermo Electron Corporation, USA) spectrophotometer equipped with an integrating sphere. Solid samples were placed into the sampling cup and the spectra were recorded in the 10000–4000 cm-1 wavenumber range. A total of 60 scans were collected from both sides of each of the capsules and the average spectrum was stored. A total time measurement of one capsule was about 2 minutes. The NIR-data were analysed using a TQ Analyst software version 6.2.1.509 (Thermo Nicolet Corporation, Inc., USA).
RESULTS AND DISCUSSION
Calibration model development
Prior to any quantitative analysis, the NIR spectrometer requires calibration based on primary measurements of the characteristic being measured, since NIRS does not give an absolute value of this characteristic 13). The calibration process basically involves selection of a representative calibration sample set, spectra acquisition and determination of reference values, multivariate modelling and validation of the model by cross validation, set validation or external validation 6).
During the first stage, a calibration experiment was performed. This involved collecting a set of reference values of calibration samples. The calibration set included 21 coated capsules for an EL film (3 capsules from each batch of 7 produced) with a polymer film thickness in the range of approximately 15–235 μm and 24 coated capsules for ES film (3 capsules from each batch of 8 produced) with the polymer film thickness in the range of approximately 30–230 μm. The reference values of the polymer film thickness of coated capsules were determined using microscopic analysis and are listed in Tables 1 and 2.
1. Eudragit<sup>®</sup> L 12.5 thickness of hard capsule coating measured by microscopic analysis 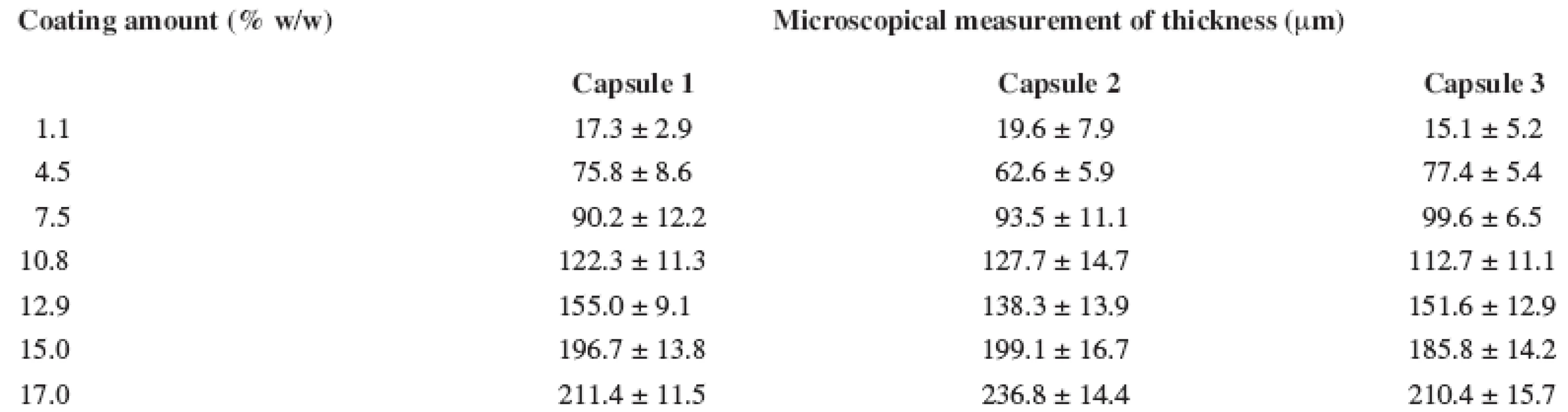
2. Eudragit<sup>®</sup> S 12.5 thickness of the hard capsule coating measured by microscopic analysis 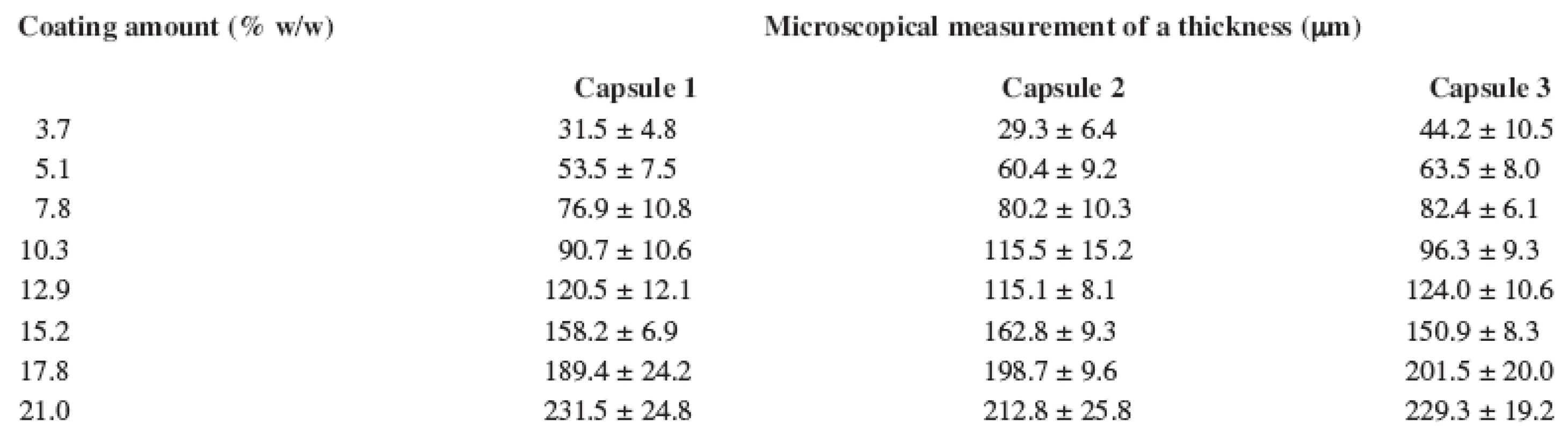
Since the NIR spectra consist of uncharacteristic, highly overlapping, broad, low absorption bands containing chemical and physical information of all sample components 6), their widespread use was somewhat hindered until multivariate calibration methods for withdrawing their analytically relevant information were developed. To perform quantitative NIR analysis, mathematical and statistical methods (i.e. chemometrics) are required to extract “relevant” information and reduce “irrelevant” information, i.e. interfering parameters. Principal component regression (PCR) and partial least-squares regression (PLS) are the two multivariate calibration methods most frequently used in this context 14).
In this study, the calibration model was constructed using the PLS algorithm, assigning the reference value of the coating thickness to each spectrum and entering these data into the TQ Analyst software. The range of wavenumbers suitable for the calculation of the calibration model was evaluated on the basis of the observation of intensity changes in the absorption bands in the spectrum of capsules with different amounts of the applied polymer material (Fig. 1).
Fig. 1. NIR spectra of separate EL film (–), separate ES film (– – –), hypromellose hard capsule (...) and capsules coated by EL film (-..-) and ES film (-.-). 
Calibration process using the PLS algorithm allows to find some extent of correlation for each wavenumber between the changes in the coating thickness and the changes in the intensity of absorption. The selected wavenumber regions were 9388–7475 cm-1 and 6194–5400 cm-1 for the EL film, and 8894–7185 cm-1 and 6133–5700 cm-1 region for the ES film, respectively.
PLS calibration models characteristic
The qualities of the developed models were checked by comparison of the reference values to the predicted values, and calculating, among others, the standard error of calibration (SE), the number of outliers and the number of PLS factors. The correlation equations in the form were found to be
Y = 0.458 + 0.9962 ×X (r = 0.9981, SE = 4.0) for EL film, [1]
Y = 1.327 + 0.9891 ×X (r = 0.9945, SE = 6.7) for ES film, [2]
where Y means the NIRS predicted thickness (μm) and X denotes the reference coating thickness values (μm). Chauvenet test was applied to determine the statistical significance of the outliers. No outliers were found in our calibration sets. PLS found the directions of greatest variability comparing both spectral and target property information with the new axes, called PLS components or PLS factors 6). The optimal number of factors used to build the calibration model was found to be six for the EL film and five for the ES film, indicating a low potential risk of overfitting the model, e.g. modelling the system noise.
Validation model
Validation was performed by calculating the standard error of prediction (SEP) of five validation capsules over the thickness range of 60–200 μm for the EL film and seven validation capsules over the thickness range of 60–200 μm for the ES film. The standard error of prediction was 7.5 μm for the EL film and 5.3 μm for the ES film.
The same samples from the calibration set were used to construct the validation model using cross validation 15). Cross validation (leave-one-out) was performed excluding one of the standards and calibration of the remaining data was constructed as a new model that was used for the quantification of the excluded standard. A simple regression analysis was performed to assess the linearity of the developed models (Fig. 2) and the standard error of cross validation (SECV) was calculated. The observed correlation equation describing the relationship between the PLS algorithm predicted coating thickness and a reference thickness
Fig. 2. The correlation plot of the reference film coating thickness (determined by microscopic analysis) and the PLS algorithm predicted film coating thickness by cross validation ( ■ EL, □ – ES) 
Y = 0.913 + 0.9921 ×X (r = 0.9949, SECV = 6.6) for EL film, [3]
Y = 2.723 + 0.9796 ×X (r = 0.9916, SECV = 8.3) for ES film, [4)
where Y is the NIRS predicted thickness (μm) and X is the reference coating thickness (μm).
CONCLUSION
A reliable and fast NIRS method was developed for off-line determination of the thickness of the polymer film applied to the hard capsules surface. The obtained results clearly demonstrated the feasibility of the NIRS methods for the determination of coating thickness of hard capsules. The connection of a NIR spectrophotometer and a coating equipment makes it possible to use this method as a part of the at-line quality control procedure in pharmaceutical industry.
This work was supported by IGA VFU 136/2008/ FaF and IGA Ministry of Healthcare of Czech Republic NS 10222-2/2009.
Address for correspondence:
Mgr. Jan Muselík, Ph.D.
Department of Pharmaceutics, University of Veterinary and Pharmaceutical Sciences Brno
Palackého 1–3, 612 42 Brno, Czech Republic
e-mail: muselikj@vfu.cz
Received 22 Juny 2009
Accepted 14 July 2009
Sources
1. Cole, E. T. et al.: Int. J. Pharm., 2002; 231, 83–95.
2. Schellekens, R. C. A. et al.: Pharm. Res., 2008; 25, 1828–1835.
3. Ishibashi, T. et al.: Int. J. Pharm., 1998; 168, 31–40.
4. Borges, O. et al.: J. Controll. Rel., 2006; 114, 348–358.
5. Pérez-Ramos, J. D. et al.: AAPS PharmSciTech, 2005; 6(1), www.aapspharmscitech.org.
6. Reich G.: Advan. Drug Delivery Rev., 2005; 57, 1109.
7. Andersson, M. et al.: J. Pharm. Biomed. Anal., 1999; 20, 27.
8. Luypaert, J., Massart, D. L., Heyden, Y, W.: Talanta, 2007, 72, 865.
9. Azzouz T., Tauler R.: Talanta, 2008; 74, 1201.
10. Kirsch, J. D., Drennen, J. K.: Pharm. Res., 1996; 13, 234.
11. Andersson, J. et al.: Anal. Chem., 2000; 72, 2099.
12. Laasonen, M. et al.: Eur. J. Pharm. Sci., 2004; 21, 493.
13. M. Blanco, J. et al.: Anal. Chim. Acta, 1999; 384, 207.
14. Pérez-Ramos, J. el al.: AAPS PharmSciTech, 2005; 6, E:20.D, www.aapspharmscitech.org.
15. Massart, D. L. et al: Handbook of Chemometrics and Qualimetrics: Part A. Amsterdam, Lausanne, New York, Oxford, Shannon, Singapore, Tokyo, Elsevier, 1997.
Labels
Pharmacy Clinical pharmacology
Article was published inCzech and Slovak Pharmacy

2009 Issue 4-
All articles in this issue
- Standard prescriptions for the formulation of medicinal preparations in pharmacies III Some possibilities of using isopropyl alcohol
- Determination of the constituents of propolis of different geographical origin
- Cardioprotective effect of 2’,3,4’--trihydroxychalcone in preclinical experiment
- Determination of the coating thickness of HPMC hard capsules by near-infrared reflectance spectroscopy
- The role of flavonoid osajin in renal ischemia-reperfusion model
- Effects of combined hormonal deprivation and fungal elicitation on the production of coumarins in cell suspension cultures of Angelica archangelica L.
- Studies of the properties of tablets from directly compressible isomalt
- Czech and Slovak Pharmacy
- Journal archive
- Current issue
- Online only
- About the journal
Most read in this issue- Standard prescriptions for the formulation of medicinal preparations in pharmacies III Some possibilities of using isopropyl alcohol
- Determination of the constituents of propolis of different geographical origin
- Studies of the properties of tablets from directly compressible isomalt
- Determination of the coating thickness of HPMC hard capsules by near-infrared reflectance spectroscopy
Login#ADS_BOTTOM_SCRIPTS#Forgotten passwordEnter the email address that you registered with. We will send you instructions on how to set a new password.
- Career

