-
Medical journals
- Career
Seroprevalence study of hepatitis E virus infection in two districts of the Czech Republic
Authors: P. Straková 1,2; B. Kříž 3; I. Rudolf 1,2; Z. Hubálek 1,2
Authors‘ workplace: Institute of Vertebrate Biology, v. v. i., Academy of Sciences of the Czech Republic, Brno 1; Department of Experimental Biology, Faculty of Science, Masaryk University, Brno 2; Centre for Epidemiology and Microbiology, National Institute of Public Health, Prague 3
Published in: Epidemiol. Mikrobiol. Imunol. 63, 2014, č. 2, s. 92-94
Category: Review articles, original papers, case report
Overview
The aim of this work was to retrospectively examine serum samples from 230 people living in two districts in the Czech Republic in year 1989 (Znojmo and Jihlava) for the IgG antibodies against hepatitis E virus (HEV). For examination, commercial ELISA kit DIA.PRO was used and 13 seropositive persons were found, i.e. 5.7% of those examined. The seropositivity rate was higher in women (6.8%) than in men (3.7%), but the difference was not statistically significant. The age of examined persons did not reveal a significant effect on the seropositivity rate: mean age of seropositive subjects was 38.0 years while that of seronegative persons was 39.4 years.
Keywords:
hepatitis E virus – ELISA – IgG antibodies – zoonotic potentialINTRODUCTION
The hepatitis E virus (HEV), causing viral hepatitis E (VHE), belongs to the genus Hepevirus in the family Hepeviridae. HEV consists of the positive single-stranded RNA (+ss RNA) with three open-reading frames (ORF 1, ORF 2, ORF 3); it is a non-enveloped virus and the length of its genome is 7.2 kb [7].
According to sequence analysis, there are four known genotypes: the genotype 1 is present in Asia and North Africa, the genotype 2 in Mexico and South America, the genotype 3 in North and South America and in several European countries and the genotype 4 in Asia [7, 15]. In addition, it is possible that there is a new genotype 5 for the avian HEV [12].
The genotypes 1 and 2 cause water-borne infections and are transmitted by faecal-oral route. They only infect humans and may cause large outbreaks unlike genotypes 3 and 4 that have zoonotic potential. In the latter case, the source of infection is raw or undercooked meat.
This virus infects many animal species both wild and domestic, e.g. domestic pig, poultry, dog, cat, sheep, goat, rodents, red deer, wild boar, mongoose, horse, primates or shellfish [15, 23]. For some of them, namely domestic pigs and wild boars, cases of zoonotic transmission have been described [14, 20].
Clinical features of VHE are very similar to those of hepatitis A. As the hepatitis A virus, it is transmitted by the faecal-oral route, but the incubation period is longer, the age of patients is higher, and it is more likely to cause fulminant disease. The serious aspect of VHE is its severity for pregnant women − when they become infected in the later stages of the pregnancy, the mortality rate might be very high [7]. In Europe, this infection can be serious for older men with cirrhosis, in patients with immunodeficiency and for potential development of chronic VHE after transplantations [13]. Person to person transmission is rare but HEV may cause epidemics. There is also a possible blood-borne transmission in drug users [19].
It was previously considered that VHE affects especially travellers to Asia or Africa because of HEV endemicity but recent records indicate also relatively high prevalence of VHE in humans in Europe or in North America although many cases are subclinical. HEV infections in humans in Europe have been caused by strains which are 99–100% identical to European swine HEV [15]. The transmission can be either by direct contact with pigs or other animals, or food-borne. The food-borne transmission was confirmed and described in Japan by consumption of undercooked pig liver or meat from wild boar or deer [25]. This finding can be also valid for Europe [5]. The laboratory diagnosis is aimed at detection of specific IgM or IgG antibodies against HEV in serum or at its RNA by nested reverse transcription PCR or real-time PCR [23]. At present, excepting China, there is no vaccine that can protect against infection caused by HEV.
In this study, we decided to carry out retrospective serosurvey of HEV antibodies in two districts of the Czech Republic.
MATERIALS AND METHODS
Blood samples
The serological survey was organized by the National Institute of Public Health (NIPH), Prague, and Regional/District Hygienic Services. Blood samples were collected by physicians (general practitioners) and were taken randomly from 230 healthy individuals of age 19–65 years, not suffering from any fever and showing no signs of immunodeficiency, in the districts Znojmo (108 persons) and Jihlava (122 persons) during 1989. The blood samples were centrifuged in the laboratory 3 to 6 hours after collection, and the separated sera were stored at -20 oC in the Serum Bank of Centre for Epidemiology and Microbiology, NIPH until use. Every blood sample taken was coded according to region, district, physician and person tested.
ELISA assay
In this study, we used a commercially available ELISA kit (DIA.PRO, Italy) for determination of IgG antibodies to HEV. The test was performed according to the manual provided by the producer. We had to calculate a cut-off value by using formula provided by producer: Cut-off value = Negative control OD + 0.350. These results were interpreted according to the table found in brochure as a ratio of the sample OD and the cut-off value found out from the formula mentioned before.
RESULTS
Overall, out of 230 examined persons, 13 (5.7%) were seropositive for HEV. The proportion of positive sera from the two districts was almost identical (Table 1). The seropositivity rate was higher in women (6.8%) than in men (3.7%), but the difference is not statistically significant (χ2 = 0.95; P = 0.33). There was no difference between the two districts: Jihlava district had 5.7% seropositive persons, Znojmo 5.6% (χ2 = 0.004; P = 0.95). The age of examined persons did not reveal a significant effect on the seropositivity rate: mean age of seropositive subjects was 38.0 years while that of seronegative persons was 39.4 years (t-value = 0.343; P = 0.731), and the medians were 33 and 40, respectively (Mann-Whitney nonparametric test: P = 0.775).
1. The number of positive and negative sera according to sex and district Tabulka 1. Počet pozitivních a negativních sér podle pohlaví a okresu 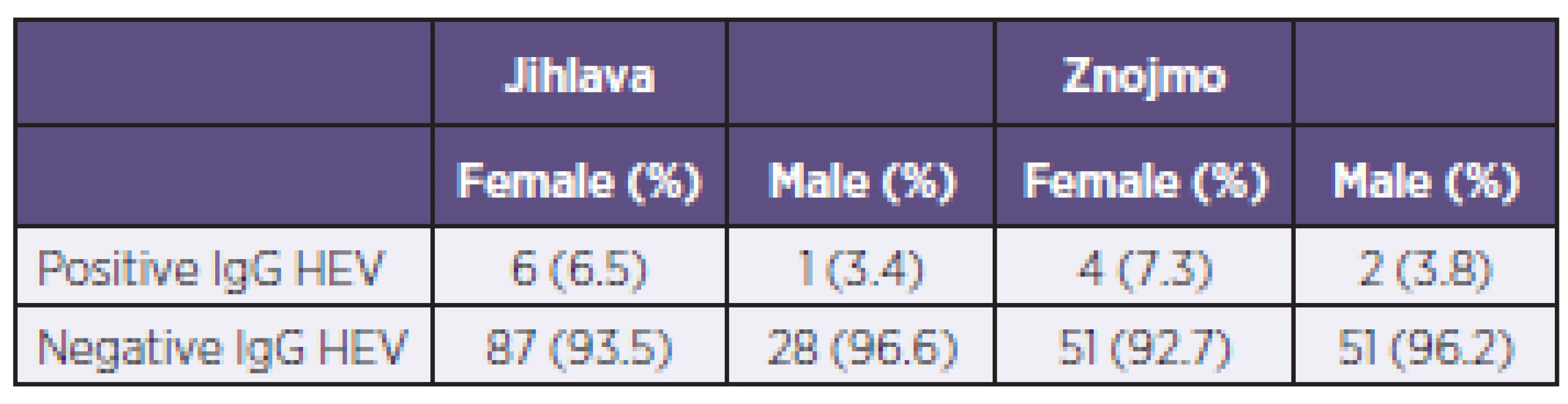
DISCUSSION
According to the WHO report [30], Albania and Kosovo reported the seroprevalence of HEV to be 9.7% and 2.5%, respectively. Moreover, HEV was responsible for acute hepatitis in 2.4% of patients in Albania and in 1.1% of patients in Hungary [30]. In Eastern Europe, most reports were published from Russia [17; 11; 1]. In Belarus, no HEV seropositivity was found in HIV infected persons and intravenous drug users [2]. In Moldova, 24.3% of swine farmers had antibodies to HEV [10]. In Western Europe, there are many reports from different countries (Germany, Spain, Italy, France, UK etc.). The antibodies against HEV have been found in a small proportion (0% to 10%) of healthy persons in most studies [4; 18]; in a few studies, higher rates of up to 20% were found, e.g. in Italy [6].
The number of reported VHE cases in the Czech Republic increases every year – from the 21 patients in 2002 to the 298 patients in 2012 [26]. From January to November 2013, there were 190 confirmed cases reported [27]. The situation in other European countries is similar: for example in Sweden, 84 confirmed cases of VHE during 2003–2012 and 13 cases in 2013 so far [28], in Finland 53 confirmed cases of VHE during 2003–2012 and six cases in 2013 so far [29], in France 280 confirmed cases in 2011. The reported morbidity rate might be biased by the growing interests of public health workers and increased surveillance for this disease.
The first serological survey for HEV in the Czech Republic was done by Pazdiora et al. [16]. They found antibodies against HEV in the sera of 17 subjects (from 269 studied), 10 positive people (from 36 studied) suffering from acute non-A, non-B, non-C hepatitis in the town of Plzen, 3 positive people (from 62 studied) in the group of gypsies without showing any symptoms of hepatitis and 4 positive people (from 171 studied) in the group of patients of venereological unit. Trmal et al. [21] described two epidemics caused by HEV in Ústí n. L. district in the Czech Republic. In the first epidemic, they found 13 infected people (from 80 studied) while in the second one, they found 8 infected people (from 16 studied). They all ate porcine products. The first group bought this food on the market and in the second group they got infected after pig-slaughter. The serological survey showed that most of these cases were inapparent and only 2 patients from the first group and 3 patients from the second group suffered from VHE. The porcine products were negative by PCR for HEV but the virus was isolated from the stool of pigs from the farms. Such local epidemics caused by porcine sausages are not rare in the world, they were described for example in France [3; 8].
According to Vasickova et al. [24], the most frequent genotype of HEV in the Czech Republic is genotype 3, being detected in both pigs and humans. Swine HEV strains are usually most homologous with human HEV from the same geographic areas, supporting the possible zoonotic transmission. Berto et al. [3] investigated HEV transmission dynamics in commercial pig farms in six different European countries (including Czech Republic – 10 pig farms of a total of 200 pigs of different age groups). They found out that in none of the weaners and fatters was detected HEV RNA. Only one grower out of 32, five sows out of 103 and one boar out of 28 were tested positive for HEV by real time PCR.
The seasonality of HEV has not been described but due to many cases caused by eating infected pork meat or pig offal after pig slaughter, the higher number of infected people occurs during cold winter months [22]. HEV is circulating in many pig farms in Europe and presents epidemiological risk for public health especially in those countries where pork products are eaten undercooked or raw [3].
Taking into account the age of patients in the Czech Republic, the increasing number of patients can be seen above from the age of 35 and the significant increase was observed in the people over 50 [22]. Norder et al. [15] studied Swedish patients infected with genotype 3 strains and these individuals were mainly males and 20–25 years older than patients infected with genotype 1. The similar results were obtained in the studies done by Sylvan et al. [19] and Dalton et al. [9]. We highlight the importance of HEV sequencing in diagnostics because the knowledge of genotypes and their geographical distribution would be helpful in molecular epidemiology and risk assessment of this neglected disease as well [30].
CONCLUSIONS
The hepatitis E virus occurs in the population of the Czech Republic. Its transmission by faecal-oral route seems less likely in Czechland. Some of the HEV infections could be imported from endemic countries, but nowadays we also have to consider autochthonous infections because of the known zoonotic potential of the virus in our country.
Most of our seropositive subjects were people of middle age or elderly. There was a higher proportion of seropositive women than men, although the result was statistically insignificant.
General practitioners and clinicians should consider the diagnosis VHE when other viral hepatitides (A, B and C) have been excluded. In additien, it is very important to improve and validate the diagnostic methods for HEV infection to guarantee the sensitivity and specificity of the results.
Do redakce došlo dne 13. 1. 2014.
Adresa pro korespondenci:
Prof. RNDr. Zdenek Hubálek, DrSc.
Ústav biologie obratlovců AVČR, v.v.i.
Klášterní 2
691 42 Valtice
e-mail: zhubalek@brno.cas.cz
Sources
1. Abe K, Hayakawa E, Sminov AV, Rossina AL et al. Molecular epidemiology of hepatitis B, C, D and E viruses among children in Moscow, Russia. J Clin Virol, 2004;30 : 57–61.
2. Balayan MS, Fedorova OE, Mikhailov MI, Rytick PG et al. Antibody to hepatitis E virus in HIV-infected individuals and AIDS patients. J Viral Hepat, 1997;4 : 279–283.
3. Berto A, Backer JA, Mesquita JR, Nascimento MS et al. Prevalence and transmission of hepatitis E virus in domestic swine populations in different European countries. BMC Research Notes, 2012;5 : 190.
4. Boutrouille A, Bakkali-Kassimi L, Crucière C, Pavio N. Prevalence of anti-hepatitis E virus antibodies in French blood donors. J Clin Microbiol, 2007;45 : 2009–2010.
5. Bouwknegt M, Lodder-Verschoor F, van der Poel WH, Rutjes SA, de Roda Husman AM. Hepatitis E virus RNA in commercial porcine livers in The Netherlands. J Food Prot, 2007;70 : 2889–2895.
6. Cacopardo B, Russo R, Preiser W, Benanti F et al. Acute hepatitis E in Catania (eastern Sicily) 1980-1994. The role of hepatitis E virus. Infection, 1997;25 : 313–316.
7. Collier L. Human Virology, 4th ed., Oxford Univ. Press Inc., New York, 2011, 400 pp.
8. Colso P, Borentain P, Queyriaux B, Kaba M et al. Pig liver sausage as a source of hepatitis E transmission to humans. J Infect Dis, 2010;202 : 825–834.
9. Dalton HR, Stableforth W, Thurairajah P, Hazeldine S et al. Autochthonous hepatitis E in Southwest England: natural history, complications and seasonal variation, and hepatitis E virus IgG seroprevalence in blood donors, the elderly and patients with chronic liver disease. Eur J Gastroenterol Hepatol, 2008;20 : 784–790.
10. Drobeniuc J, Favorov MO, Shapiro CN, Bell BP et al. Hepatitis E virus antibody prevalence among persons who work with swine. J Infect Dis, 2001;184 : 1594–1597.
11. Fedorova OE, Balaian MS, Mikhaĭlov MI, Pavlova IP et al. Hepatitis E in a nonendemic region: antibodies to hepatitis E virus in various groups of residents. Vopr Virusol, 1996;41 : 104–107.
12. Haqshenas G, Huang FF, Fenaux M, Guenette DK et al. The putative capsid protein of the newly identified avian hepatitis E virus shares antigenic epitopes with that of swine and human hepatitis E viruses and chicken big liver and spleen disease virus. J Gen Virol, 2007;83 : 2201–2209.
13. Kamar N, Bendall R, Legrand-Abravanel F, Xia NS, Ijaz S, Izopet J et al. Hepatitis E. Lancet, 2012;379(9835):2477–2488.
14. Meng XJ. Novel strains of hepatitis E virus identified from humans and other animal species: is hepatitis E a zoonosis? J Hepatol, 2000;33 : 842–845.
15. Norder H, Sundqvist L, Magnusson L, Østergaard Breumm S et al. Endemic hepatitis E in two Nordic countries. Euro-Surveill, 2009;14 : 1–9.
16. Pazdiora P, Nĕmeček V, Topolcan O. První výsledky sledování protilátek proti viru hepatitidy E u vybraných skupin populace v Západočeském kraji. Epidemiol Mikrobiol Imunol, 1996;45 : 117–118.
17. Shakhgildian IV, Onishchenko GG, Schastnyĭ EI, Khukhlovich PA. et al. The current problems in the epidemiology and prevention of enteric viral hepatitis in Russia. Zh Mikrobiol Epidemiol Immunobiol, 1994;5 : 20–25.
18. Stroffolini T, Menchinelli M, Dambruoso V, Menniti Ippolito F et al. Prevalence of hepatitis E in a central Italian town at high endemicity for hepatitis C virus. Ital J Gastroenterol, 1996;28 : 523–525.
19. Sylvan SP. The high rate of antibodies to hepatitis E virus in young, intravenous drug-abusers with acute hepatitis B-virus infection in a Swedish community: a study of hepatitis markers in individuals with intravenously or sexually acquired hepatitis B-virus infection. Scand J Infect Dis, 1998;30 : 429–430.
20. Tei S, Kitajima N, Takahashi K, Mishiro S. Zoonotic transmission of hepatitis E virus from deer to human beings. Lancet, 2003;362 : 371–373.
21. Trmal J, Pavlík I, Vašíčková P, Matějíčková L et al. Epidemie virové hepatitidy E v České republice? Epidemiol Mikrobiol Imunol, 2012;61 : 15–20.
22. Trmal J, Beneš Č, Trnková M. Odlišnosti výskytu virových hepatitid A a E v České republice. Epidemiol Mikrobiol Imunol, 2013;62 : 19–25.
23. Vasickova P, Psikal I, Kralik P, Widen F et al. Hepatitis E virus: a review. Veterinarni Medicina, 2007;52 : 365–384.
24. Vasickova P, Slany M, Chalupa P, Holub M et al. Detection and phylogenetic characterization of human hepatitis E virus strains, Czech Republic. Emerg Infect Dis, 2011;17 : 917–919.
25. Yazaki Y, Mizuo H, Takahashi M, Nishizawa T et al. Sporadic acute or fulminant hepatitis E in Hokkaido, Japan, may be food-borne, as suggested by the presence of hepatitis E virus in pig liver as food. J Gen Virol, 2003;84 : 2351–2357.
26. EPIDAT (2012): National Institute of Public Health: http://www.szu.cz/publikace/data/vybrane-infekcni-nemoci-v-cr-v-letech-2003-2012-absolutne, on 7 Oct. 2013.
27. EPIDAT (2013): National Institute of Public Health: http://www.szu.cz/publikace/data/kumulativni-nemocnost-vybranych-hlasenych-infekci-v-ceske, on 12 Dec. 2013.
28. Smittskyddsinstitutet: Hepatitis E data/statistics. http://www.smittskyddsinstitutet.se/in-english/statistics/hepatitis-e/, on 12. Dec. 2013.
29. Terveyden ja hyvinvoinnin laitos: Hepatitis E virus data: http://www3.thl.fi/stat/, on 12. Dec. 2013.
30. WHO (2010): The global prevalence of Hepatitis E virus infection and susceptibility: A systematic review: http://whqlibdoc.who.int/hq/2010/WHO_IVB_10.14_eng.pdf
Labels
Hygiene and epidemiology Medical virology Clinical microbiology
Article was published inEpidemiology, Microbiology, Immunology

2014 Issue 2-
All articles in this issue
- Phylogenetic and molecular analysis of A/H1N1pdm influenza viruses isolated in the epidemic season 2012/2013 from hospitalised patients with symptoms of influenza-like illness
- An increase in the prevalence of syphilis in women in Eastern Bohemia – 30 years of surveillance
- Diagnosis of Clostridium difficile infections: Comparative study of two immuno enzyme assays with confirmation by PCR and culture followed by PCR ribotyping
- A point prevalence survey of healthcare-associated infections in the Slovak Republic – a part of the EU project
- Nosocomial transmission of listeriosis
- Diversity of human Salmonella isolates in the South Moravian Region in 2009–2012
- Candida dubliniensis in clinical specimens and possibilities for identification
- Natural antibodies against α(1,3) galactosyl epitope in the serum of cancer patients
- Cytolethal distending toxins
- Evaluation of the importance of a ready-made, gentamicin-impregnated spacer in relation to bacteriological findings in patients with periprosthetic joint infections
- Q fever – an occupational disease leading to disability – case report
- Measles re-emerging in the Ústí Region
- Predicted strain coverage of a new protein-based meningococcal vaccine in the Czech Republic
- Post-mortem analysis of Candida albicans breakthrough infection during echinocandin treatment in haematopoietic stem cell transplant recipient
- Viral gastroenteritis in Eastern Bohemia Region of the Czech Republic
- Seroprevalence study of hepatitis E virus infection in two districts of the Czech Republic
- Epidemiology, Microbiology, Immunology
- Journal archive
- Current issue
- Online only
- About the journal
Most read in this issue- Candida dubliniensis in clinical specimens and possibilities for identification
- A point prevalence survey of healthcare-associated infections in the Slovak Republic – a part of the EU project
- Q fever – an occupational disease leading to disability – case report
- Diagnosis of Clostridium difficile infections: Comparative study of two immuno enzyme assays with confirmation by PCR and culture followed by PCR ribotyping
Login#ADS_BOTTOM_SCRIPTS#Forgotten passwordEnter the email address that you registered with. We will send you instructions on how to set a new password.
- Career

