-
Medical journals
- Career
Mucormycosis occurring in an immunocompetent patient: a case report and review of literature
Authors: Aparna K Sanath 1; Meghanand T Nayak 1; Sunitha Jd 1; Shilpa Dutta Malik 1; Sanath Aithal 2
Authors‘ workplace: Department of Oral & Maxillofacial Pathology and Oral Microbiology, Teerthanker Mahaveer Dental College & Research Centre, TMU campus, Delhi road, Bagadpur, Moradabad, Uttar Pradesh. India 1; Department of Dermatology, Venereology & Leprology, Teerthanker Mahaveer Medical College & Research Centre, TMU campus, Delhi road, Bagadpur, Moradabad, Uttar Pradesh. India 2
Published in: Čes.-slov. Patol., 56, 2020, No. 4, p. 223-226
Category:
Overview
Mucormycosis is an opportunistic fungal infection with a high fatality rate and is the third most common fungal infection that is invasive in nature, next to candidiasis and aspergillosis. The condition is generally vasotropic and angio-invasive in nature. It gets disseminated to a wider area locally and also exhibits a distant spread. It is usually associated with medically compromised patients. However, mucormycosis in immunocompetent individuals is gaining attention as several cases have been reported throughout the world with a high incidence of such cases being reported from the Indian subcontinent. It is attributed to the poor socio-economic status and triggered by the local trauma due to unhygienic setup or poor health care. The pathway of pathogenesis is not clearly understood in immunocompetent patients and therefore becomes a matter of great concern. Here, we report one such case of mucormycosis affecting the maxillary region following tooth extraction in a 42-year-old male.
Keywords:
mucormycosis – Immunocompetent – Tooth extraction – Fungal hyphae
Mucormycosis (Zygomycosis) is an opportunistic fungal infection involving several areas of the body, caused by saprophytic fungus. Three variants of mucormycosis are identified, namely: cutaneous, rhino-cerebral and pulmonary variants; but the rhinocerebral form is more relevant to dental healthcare providers as it represents almost one-third to half of all the cases. The rhino-cerebral form is further subdivided into two subtypes: a highly fatal rhino-orbito-cerebral form involving the ophthalmic and internal carotid arteries, which is invasive; and a less fatal rhino-maxillary form, which involves the sphenopalatine and greater palatine arteries that result in thrombosis of the nasal turbinate and palatal necrosis. It is generally vasotropic, hence becoming angio-invasive and disseminated. Mucormycosis is usually associated with medically compromised patients. In an immunocompetent host, these infections generally progress locally, directly to the adjacent tissue and rarely disseminate. This fungus is ubiquitous in nature and inhalation of the spores is the common route of infection. Oral mucosal epithelium and endothelium act as a barrier against tissue - and angio-invasion (1). In such cases, the palate may be infected with Zygomycetes after minor oral trauma, like tooth extraction leading to primary palatal mucormycosis which tends to spread superiorly. Once, the spores of the fungus enter through a minor trauma, it is believed to have the capacity to secrete toxins and proteases that damage the endothelial cells in the oral mucous membrane (1). Mignogna et al. hypothesized that a chronic local insult may act as a predisposing factor for mucormycosis to develop in apparently healthy or immunocompetent individuals (2). This hypothesis was further supported by other authors by the fact that the chronic local insult causes an alteration in the first line barrier defense due to an impairment in the mucous membrane which makes an individual vulnerable to fungal infection (3). Furthermore, it was substantiated and hypothesized by Schleimer RP et al that in patients with chronic sinusitis, molecules like S100, SPINK5 and members of the epidermal differential complex, necessary in barrier maintenance of upper airways and sinuses are reduced (4). These factors hence give an explanation for possible mucormycosis in immunocompetent individuals. The confirmatory diagnosis is usually possible with the biopsy specimen. The hyphae of the fungus can be detected by using hematoxylin and eosin (H&E), Gomori’s methenamine silver (GMS), periodic acid-Schiff (PAS) or calcofluor white stains. Sabouraud’s dextrose agar can be used to detect the fungal growth in a culture medium. However, negative culture results may often appear despite positive histologic findings. Here, we present a case of mucormycosis in an apparently immunocompetent patient, following dental extraction.
CASE REPORT
A 42-year-old male patient presented to the outpatient department with pain and swelling in the right cheek region for 4 months. The patient gave a history of the extraction of a maxillary right posterior tooth in a rural health center about a year back. He was a known smoker for the past 20 years but had no history of any systemic illnesses. On examination, the patient’s oral hygiene was poor with halitosis. He presented with ulceration and erythema in the labial and palatal mucosa in relation to the extracted site in the maxillary right first premolar region. Posteriorly, the lesion extended to the maxillary right first molar region. An area of exposed maxillary bone was also evident (Fig. 1). On palpation, the lesion was tender and the swabs made from the lesional area were sent for a culture test. The culture was negative for any fungal growth. On routine blood investigation, his blood sugar was found to be within normal range. A biopsy of the affected area was performed and sent for histopathologic evaluation. Histopathological examination revealed numerous PAS-positive, large, non-septate, branching hyphae, with blood vessels, areas of hemorrhage and necrosis (Fig. 2). Based on the histopathology, a diagnosis of mucormycosis was made. The patient was administered Amphotericin-B anti-fungal therapy for 6 weeks and the necrotic fragments along with the dead, sequestered bone was surgically debrided. The patient was followed up for 6 months and no signs and symptoms of recurrence were noted.
1. Ulceration in the alveolar mucosa with unhealed socket and denuded bone in relation to tooth #14. 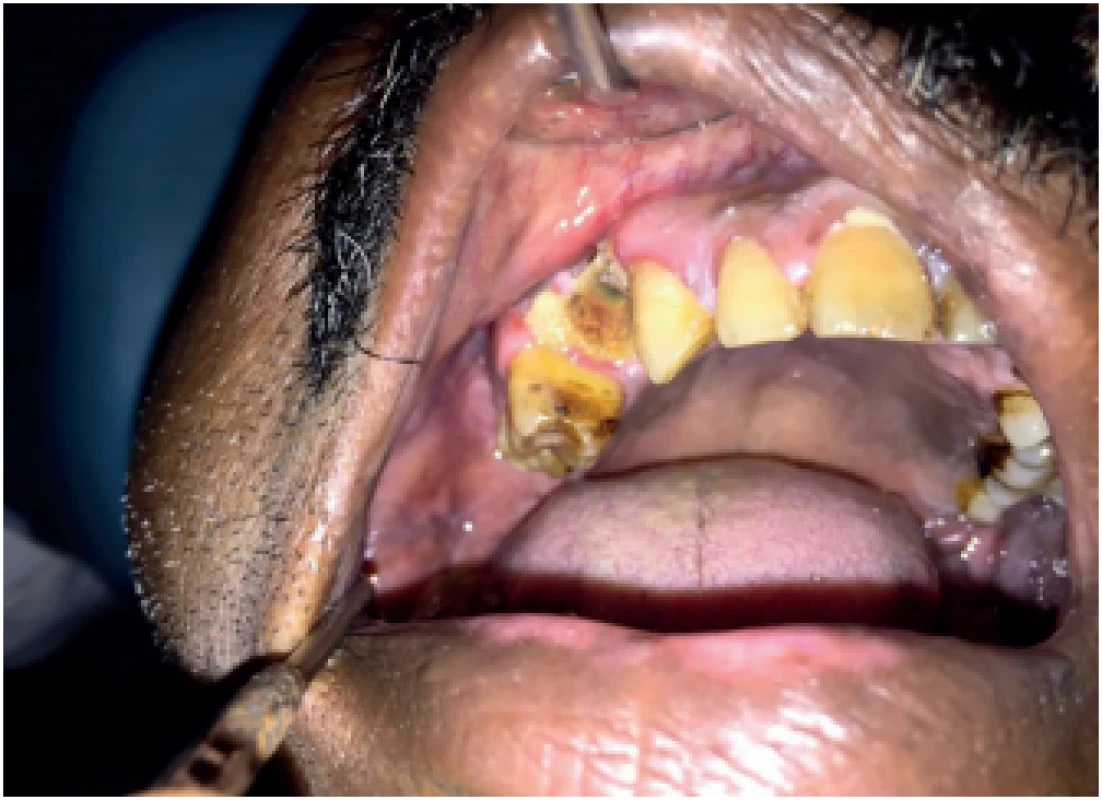
2. Coenocytic (aseptate), branching, fungal hyphae within necrotic granulation tissue, with small vessels angioinvasion (A - PAS, 400x, B - H&E, 100x, C - H&E, 400x). 
DISCUSSION
Mucormycosis is a fatal and rapidly progressive opportunistic infection caused by fungi belonging to the class Zygomycetes/Phycomycetes, order Mucorales. This condition was first described in human beings by Paltauf in the year 1885 (5), but it is being reported with increasing frequency only in the past two decades. According to the revised taxonomy of the fungi, the phylum Zygomycota, subphylum Zygomycotina, and class Zygomycetes have been replaced by the phylum Glomeromycota, with subphylum Mucormycotina which includes the order Mucorales (6). Mucorales are considered as the etiologic factor of the mucormycosis infection. Hence the term mucormycosis is preferred over zygomycosis. In dentistry, this condition gains increasing interest because of its manifestation in the facial and oral tissue before affecting other areas. Mucormycosis is the third most common invasive fungal infection, following aspergillosis and candidiasis, and accounts for 8.3–13.0% of all fungal infections (7). These fungi are ubiquitous, usually harmless and become pathogenic in humans under certain conditions like immunosuppression; steroids, antibiotic, cytotoxic therapy; and diabetic acidosis. The other predisposing factors include malignancy, renal failure, liver transplant, blood dyscrasias, burns, malnutrition and repeated hospitalization (8). However, in the past few years, mucormycosis in immunocompetent or in apparently healthy individuals showing no identifiable risk factors have been reported (9) (Tab. 1).
1. Reported cases in immunocompetent patients# 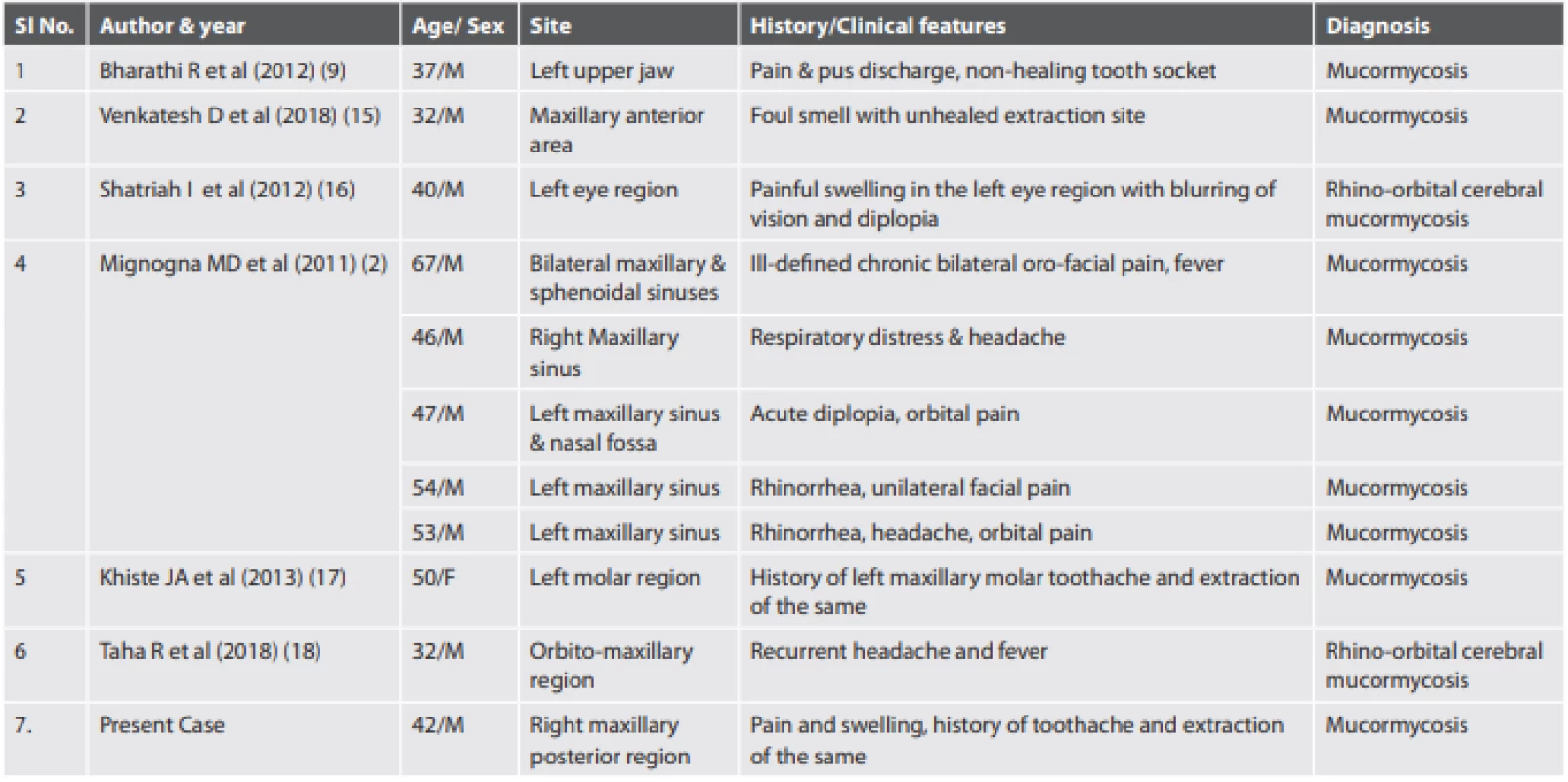
#Note: Immunocompetent patients – not having any known systemic illness. The disease may present with various manifestations with a predilection for the paranasal sinuses. Mucorales are aerobic in nature, but they can survive in-vitro for 2–5 days. They are usually found in soil, bread molds, and decayed matter. These organisms can usually be cultured from the oral cavity, throat, nasal cavity and stools of any healthy person. Hence every individual is potentilally exposed to it and may inhale the spores. The spores are then transported to the pharynx by the nasal ciliary system and is cleared by the gastrointestinal tract. Similarly, the phagocytes clear the spores inhaled into the lungs. In vulnerable individuals, the infection begins in the middle and inferior nasal turbinates. From the nasal cavity, the infection spreads to the paranasal sinus region and then to the retro-orbital region, either via direct extension or through ethmoidal, lacrymal, or angular vessels. Infection can also spread to the brain through the cribriform plate and the ophthalmic vessels. Following spread, the function of the cranial nerves II, III, IV, and VI may be impaired and can lead to ptosis, proptosis, pupillary dilatation, visual loss, periorbital cellulitis, and numbness. Intraorally, ulcer with a black and necrotic surface (eschar formation) having raised erythematous borders with areas of denudation can be seen. Other associated signs and symptoms can be a nasal obstruction with blood discharge, facial pain, headache, visual disturbances, and facial paralysis. If there is cranial vault involvement, it can lead to blindness, lethargy, seizures or can even lead to death. According to Moye and Cardill, black eschar formation is due to the thrombosis of the blood vessels (10). Fungal hyphae show an affinity for blood vessels and thus invade the tissues (10). Direct penetration into blood vessels and intravascular growth of the fungus leads to thrombosis and necrosis of the adjacent tissues.
Oral manifestations are usually seen in the palate, however other areas like tooth extraction site and tongue have also been reported. Clinically areas of ischemic necrosis with eschar (11) formation in the mucoperiosteum and areas of bony denudation are seen. According to Adam et al, the mortality rate of mucormycosis affecting various regions of the body is as follows: cutaneous mucormycosis – 16%, rhinocerebral – 67%, pulmonary – 83%, gastrointestinal and disseminated – 100% (12). Clinically, oral mucormycosis may be difficult to be differentiated from oral aspergillosis and other etiologies of palatal ulcers such as squamous cell carcinoma, gummatous necrosis of tertiary syphilis, or tuberculosis (Tab. 2) (13-18). Tissue cultures are positive in only 23% of cases and histopathologic findings aid in diagnosis (13,19). Swabs of discharge or abnormal tissue are not helpful as Mucorales can be colonizers. Therefore, a histopathologic diagnosis is of great importance. H&E or PAS stained histopathological examination reveals typical non-septate hyphae with variably thin, often collapsed or twisted walls showing branching at irregular angles and intervals, invading the blood vessel walls. However, in recent times, molecular techniques like PCR-based procedures aid in detecting Mucorales DNA, not only in culture and tissue samples but also in plasma or serum or even urine samples (20). As the oral cavity houses a complex flora, a mixed infection may be commonly present in the affected patients which indicates a high level of immunosuppression. These recent PCR techniques, therefore help in detecting Mucormycosis in a very early stage and also differentiate them from other infections, for example aspergillosis (20). In the case discussed here, a negative culture result was obtained and the diagnosis was based on histopathologic findings. The patient being immunocompetent, the disease outcome had a favorable prognosis.
2. Characteristics of Mucormycosis versus Aspergillosis. 
In conclusion, the diagnosis of mucormycosis is challenging to date. The mortality rate of mucormycosis is quite high due to delayed diagnosis and lack of timely treatment. Therefore, a non-specific palatal ulcer could be considered as the presenting sign of mucormycosis and it is necessary for a dental practitioner to be alert to early signs and symptoms of this disease, especially when evaluating the patients within the high-risk group. The histopathological demonstration of Mucorales-like hyphae is essential for the diagnosis. The most effective treatment and management often require a combination of antifungal agents, along with the elimination of predisposing factors, followed by surgery.
ACKNOWLEDGMENTS
We would like to thank Prof. Dr. Pankaj Kukreja, Head of the Department, Department of Oral & Maxillofacial Surgery, TMDCRC, Teerthanker Mahaveer University, Moradabad for performing the surgical procedures.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest regarding the publication of this paper.
∗ Correspondence address:
Dr. Aparna K Sanath,
Associate Professor, Department of Oral & Maxillofacial Pathology and Oral Microbiology, Teerthanker Mahaveer Dental College & Research Centre,
TMU campus, Delhi road, Bagadpur,
Moradabad, Uttar Pradesh, INDIA 244001
tel.: +91 8762828167
e-mail: draparnasanath@gmail.com
Sources
- Kontoyiannis DP, Lewis RE. Invasive zygomycosis: update on pathogenesis, clinical manifestations, and management. Infect Dis Clin North Am 2006; 20(3): 581-607.
- Mignogna MD, Fortuna G, Leuci S, Adamo D et al. Mucormycosis in immunocompetent patients: a case-series of patients with maxillary sinus involvement and a critical review of the literature. Int J Infectious Diseases 2011; 15(8): e533-e540.
- Lane AP. The role of innate immunity in the pathogenesis of chronic rhinosinusitis. Curr Allergy Asthma Rep 2009; 9(3): 205-212.
- Schleimer RP, Kato A, Peters A, et al. Epithelium inflammation and immunity in the upper airways of humans: studies in chronic rhinosinusitis. Proc Am Thoracic Soc 2009; 6(3): 288-294.
- Kalaskar RR, Kalaskar AR, Ganvir S. Oral mucormycosis in an 18-month old child: a rare case report with a literature review. J Korean Assoc Oral Maxillofac Surg 2016; 42(2): 105-110.
- Hibbett DS, Binder M, Bischoff JF, et al. A higher-level phylogenetic classification of the Fungi. Mycol Res 2007; 111(5): 509-547.
- Oliveira FR, Couto NG, Bastos JO, Colleti J Junior, Carvalho WB. Abdominal mucormycosis in a child: a case report. Rev Soc Bras Med Trop 2016; 49(6): 796-798.
- Vahabzadeh-Hagh AM, Chao KY, Blackwell KE. Invasive oral tongue mucormycosis rapidly presenting after orthotopic liver transparent. Ear Nose Throat J 2019; 98(5): 268-270.
- Bharathi R, Arya AN. Mucormycosis in an immunocompetent patient. J Oral Maxillofac Pathol 2012; 16(2): 308-309.
- Garg R, Gupta VV, Ashok L. Rhinomaxillary mucormycosis: A palatal ulcer. Contemp Clin Dent 2011; 2(2): 119-123.
- DR Telles, N Karki, MW Marshall. Oral fungal infections - diagnosis and management. Dent Clin N Am 2017; 61(2): 319-349.
- Adam RD, Hunter G, DiTomosso J, Commerci G Jr. Mucormycosis: emerging prominence of cutaneous infections. Clinical Infectious Diseases 1994; 19(1): 67-76.
- Al Akhrass F, Debiane L, Abdallah L et al. Palatal mucormycosis in patients with hematologic malignancy and stem cell transplantation. Medical Mycology 2011; 49(4): 400–405.
- Doni BR, Peerapur BV, Thotappa LH, Hippargi SB. Sequence of oral manifestations in rhino-maxillary Mucormycosis. Ind J Dent Res 2011; 22(2): 331-335.
- Venkatesh D, Dandagi S, Chandrappa PR, Hema KN. Mucormycosis in immunocompetent patient resulting in extensive maxillary sequestration. J Oral Maxillofac Pathol 2018; 22(Suppl 1): S112-116.
- Shatriah I, Mohd-Amin N, Tuan-Jaafar TN, Khanna RK, Yunus R, Madhavan M. Rhino-orbito-cerebral Mucormycosis in an immunocompetent patient: Case report and review of literature. Middle East Afr J Ophthalmol 2012; 19(2): 258-261.
- Khiste JA, Bolde SA, Pandit GA, Tamboli NAS. Mucormycosis of maxillary sinus in immunocompetent patient masquerading as neoplasm: A case report. Int J Oral Maxillofac Pathol 2013; 4(4): 50-53.
- Taha R, Almteri T, Bahabri N, Almoallim H. Rhino-orbito-cerebral mucormycosis in immunocompetent young patient: case Report. Clin Med Rev Case Rep 2018; 5 : 207.
- Ferry AP, Abedi S. Diagnosis and management of rhino-orbitocerebral mucormycosis (phycomycosis). A report of 16 personally observed cases. Ophthalmology 1983; 90(9): 1096 – 1104.
- Millon L, Scherer E, Rocchi S, Bellanger A-P. Molecular strategies to diagnose mucormycosis. J Fungi 2019; 5(1): 24.
Labels
Anatomical pathology Forensic medical examiner Toxicology
Article was published inCzecho-Slovak Pathology

2020 Issue 4-
All articles in this issue
- Gastrointestinální trakt – WHO klasifikace, imunohistochemie a molekulární genetika
- Chtěl jsem být bankovní lupič, ale nevěděl jsem, kde se na to studuje
- ′ UROPATOLOGIE
- ′ PATOLOGIE SERÓZNÍCH POVRCHŮ
- ′ KARDIOPATOLOGIE
- ′ PATOLOGIE ORL OBLASTI
- ′ PULMOPATOLOGIE
- ′ PATOLOGIE GIT
- ′ HEPATOPATOLOGIE
- ′ PATOLOGIE MĚKKÝCH TKÁNÍ
- ′ PULMOPATOLOGIE
- ′ NEFROPATOLOGIE
- ′ GYNEKOPATOLOGIE
- ′ KARDIOPATOLOGIE
- ′ CYTODIAGNOSTIKA
- ′ NEUROPATOLOGIE
- Prof. MUDr. Ľudovít Danihel, PhD. jubiluje.
- Comments on the 5th edition of WHO classification of digestive system tumors – Part 1. Gastrointestinal tract
- Changes in histopathological classification of neuroendocrine tumors in 5th edition of WHO classification of gastrointestinal tract tumors (2019)
- Immunohistochemistry and molecular genetics in the differential diagnostics of mesenchymal lesions of gastrointestinal tract
- Right heart ventricle myocarditis induced by pulmonary thrombembolism
- ′ GYNEKOPATOLOGIE
- Mucormycosis occurring in an immunocompetent patient: a case report and review of literature
- Czecho-Slovak Pathology
- Journal archive
- Current issue
- Online only
- About the journal
Most read in this issue- Chtěl jsem být bankovní lupič, ale nevěděl jsem, kde se na to studuje
- Comments on the 5th edition of WHO classification of digestive system tumors – Part 1. Gastrointestinal tract
- Prof. MUDr. Ľudovít Danihel, PhD. jubiluje.
- Changes in histopathological classification of neuroendocrine tumors in 5th edition of WHO classification of gastrointestinal tract tumors (2019)
Login#ADS_BOTTOM_SCRIPTS#Forgotten passwordEnter the email address that you registered with. We will send you instructions on how to set a new password.
- Career

