-
Medical journals
- Career
INTRAOPERATIVE FAT GRAFTING INTO THE PECTORALIS AND LATISSIMUS DORSI MUSCLES-NOVEL MODIFICATION OF AUTOLOGOUS BREAST RECONSTRUCTION WITH EXTENDED LATISSIMUS DORSI FLAP
Authors: L. Streit 1; L. Dražan 1; M. Schneiderová 2; T. Kubek 1; P. Sin 1; K. Veselý 3; O. Coufal 4,5; J. Veselý 1
Authors‘ workplace: Department of Plastic and Aesthetic Surgery, St. Anne's University Hospital Brno and Faculty of Medicine, Masaryk University, Brno, Czech Republic 1; Department of Radiology, Masaryk Memorial Cancer Institute, Brno, Czech Republic 2; Department of Pathological Anatomy, St. Anne's University Hospital Brno and Faculty of Medicine, Brno, Czech Republic 3; Department of Surgical Oncology, Masaryk Memorial Cancer Institute, Brno, Czech Republic 4; Department of Comprehensive Cancer Care, Medical Faculty, Masaryk University, Brno, Czech Republic 5
Published in: ACTA CHIRURGIAE PLASTICAE, 59, 2, 2017, pp. 72-81
INTRODUCTION
The latissimus dorsi flap is a reliable and one of the most commonly used methods of both immediate and delayed breast reconstruction. Latissimus dorsi flap reconstruction was introduced first by Tansini in 1906 1 and popularized by Olivari and Schneider in the late 1970s 2,3. Its use is traditionally limited by the volume of transferred tissue and by the desired size of the breast. The volume of the breast is therefore routinely created by silicone prosthesis in one stage as described by Bostwick and colleagues 4,5. In this concept, the disadvantages associated with the use of the latissimus dorsi flap (donor site morbidity) and disadvantages of silicon implant (risks of infection, extrusion, rupture, capsular contracture and a recently suggested association with anaplastic large-cell lymphoma) occur both together.
The need for an implant may be avoided if the latissimus dorsi flap is harvested as extended when the whole muscle is taken. Hokin in 1983 and Marshall in 1984 increased the volume of the latissimus dorsi flap as they included lumbar fascia 6,7. Germann in 1996 8 and Delay in 1998 9 introduced the technique of latissimus dorsi flap harvesting when the supplementary volume of the flap was increased by the subfascial fat localized in scapular and parascapular region, in addition to the lumbar fat. Delay also defined individual fatty zones adjacent to the latissimus dorsi muscle anatomically and he popularized this reconstructive approach 10. In addition, Delay improved the options for autologous breast reconstruction by combining extended latissimus dorsi flap with abdominal advancement flap allowing breast reconstruction with one final horizontal scar 11.
Because of low morbidity, low complication rate, very good results and an excellent acceptance by the patients, autologous fat grafting has recently become recognized as an essential tool in breast reconstructive surgery. Sinna and colleagues demonstrated in consecutive sample of 200 patients that lipomodelling improves outcomes after extended latissimus dorsi flap reconstructions by augmenting breast volume and by enhancing its shape 12–14. Finally, Santanelli and colleagues have recently reported their technique of latissimus dorsi flap reconstruction associated with intraoperative fat grafting to the subcutaneous tissue of the latissimus dorsi flap as an alternative for entirely autologous breast reconstruction 15.
The aim of this study is to present a new concept of autologous breast reconstruction with the extended latissimus dorsi flap associated with simultaneous fat grafting into the pectoralis and latissimus dorsi muscles.
PATIENTS AND METHODS
Between April 2013 and February 2016, 14 patients underwent secondary unilateral total breast reconstruction with extended latissimus dorsi flap associated with primary fat grafting into the pectoralis major and latissimus dorsi muscles (Table 1). This surgical technique was indicated for patients after mastectomy requiring breast reconstruction desiring small or moderate breast volume, patients who refused scars on the abdomen or patients with the contraindication for abdominal free flap such as: 1) multiple abdominal scars, 2) previously harvested abdominal flap or abdominoplasty, 3) patients with a history of deep venous thrombosis or pulmonary embolism (Table 2).
1. Patients and operative data. RT: adjuvant radiotherapy after mastectomy, AAF: abdominal advancement flap, PM: pectoralis muscle, LTD: latissimus dorsi muscle. No adipose tissue was applied into the latissimus dorsi muscle in the cases when it was too thin (patients No. 2, 3, 5, 12). And also no fat graft was applied into the pectoralis muscle in one patient with the history of implant based breast reconstruction with atrophic pectoralis muscle (patients No. 12) 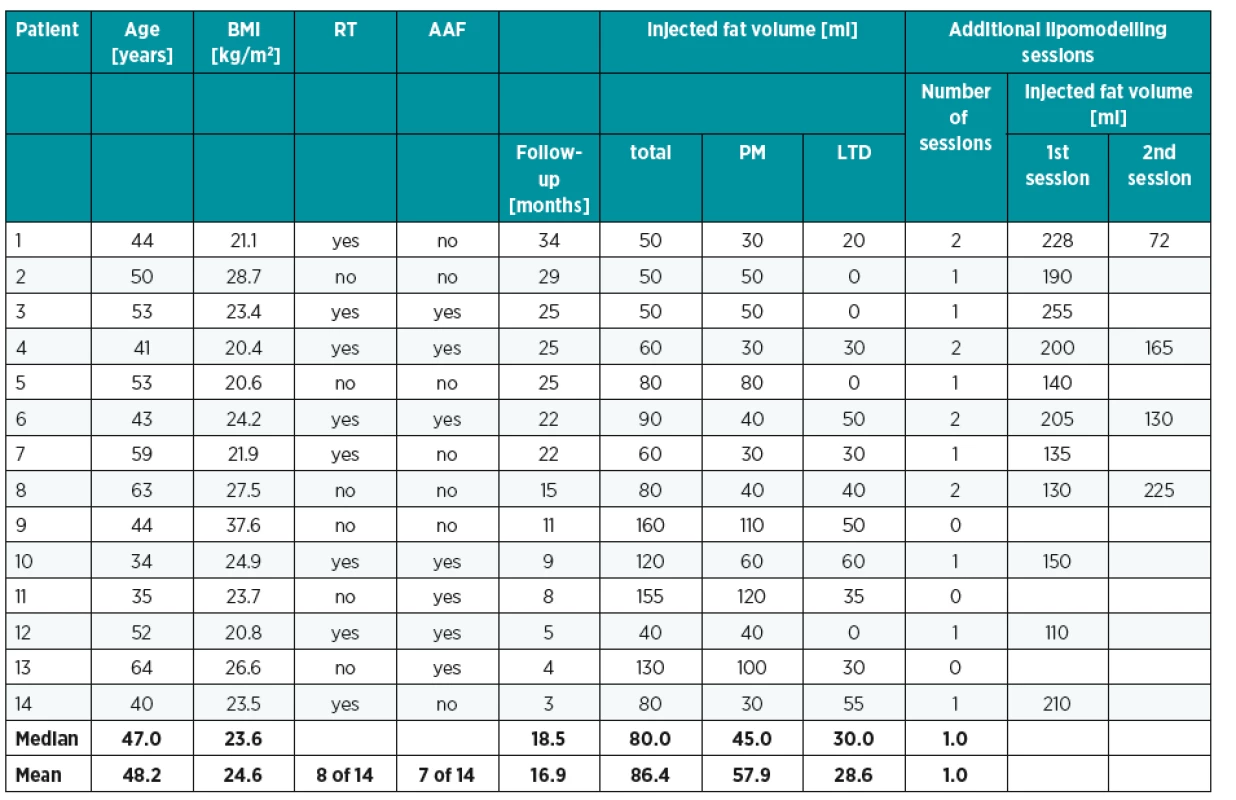
2. Choice of autologous latissimus reconstruction over abdominal flap 
Preoperative design
Planning begins in the chest in the front view. The median intermammary line, the inframammary crease and the foreseeable extent of the breast is designed in the post-mastectomy chest using opposite breast as a model. In the case of serious hypertrophy or ptosis of the preserved breast, patients are informed about the need of contralateral symmetrization in the second stage to obtain appropriate results. In cases with sufficient laxity of the skin below the submammary fold, the abdominal advancement flap may be conveniently used to reconstruct the skin of the lower part of the breast – this maneuver allows to reduce the minimum size of latissimus dorsi flap skin paddle and to reduce the length of the scar on the back respectively.
The bra strap area is then marked on the patient’s back in the upright position. The horizontal or slightly oblique skin paddle is drawn on the middle of the bra strap area (Fig. 1), the pinch technique is used to determinate its maximum width. In the case, when the abdominal advancement flap is planned simultaneously, we minimize the size of the skin paddle (6 x 14 cm approximately). In other cases, the size of the skin paddle is outlined with respect of the extent of the missing skin on the breast.
1. Latissimus dorsi flap planning. The bra strap area is marked first when the patient is wearing her bra. The horizontal or slightly transverse skin paddle is localized in the middle of the bra strap area 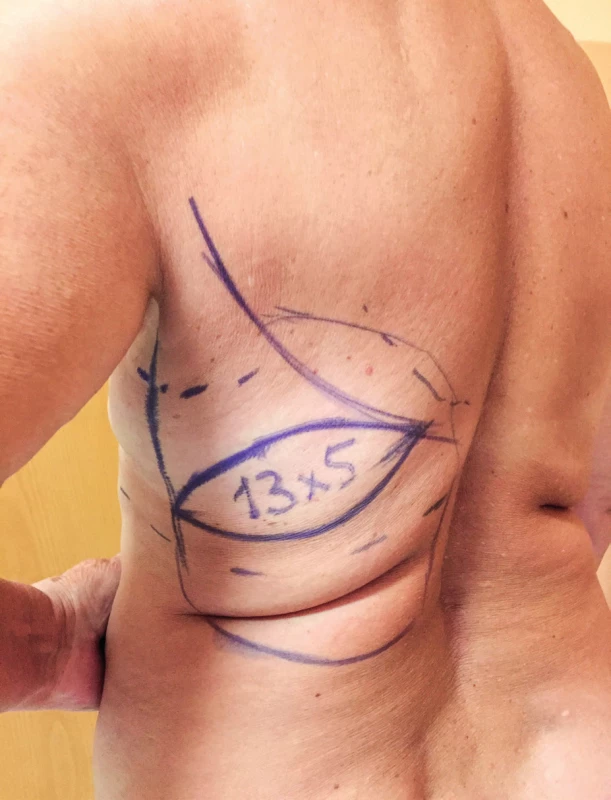
Donor site for fat harvesting is marked according to the patient’s wish and expected volume of fat graft (100–150 ml of processed adipose tissue should be prepared). Lower abdomen and inner thighs are preferred harvesting sites because of easy positioning of the patient in the supine position. We do not recommend fat harvesting from the flanks because of the risk of blood supply deterioration of the elevated skin after latissimus dorsi flap harvesting due to instillation of tumescent solution (with epinephrine) and liposuction in closely surrounding area.
Surgical technique
Latissimus dorsi flap harvesting was performed simultaneously with subcutaneous undermining of the anterior thorax in lateral decubis position. We used modification of harvesting technique described by Delay 10. The volume of the latissimus dorsi flap was extended by harvesting: 1) adipose tissue of the paddle below the superficial fascia retained on the entire surface of the muscle, 2) the scapular fat pad, 3) the fat adjacent to the anterior part of the muscle. On the contrary, we did not harvest supra-iliac fat pad.
Once the flap was raised and the pedicle clearly visible and free, a subtotal section of the muscle was performed distally and just a small muscular bridge was left to protect the vascular pedicle. The thoracodorsal nerve was preserved. The completely raised flap was then passed underneath the skin bridge anteriorly into the dissected subcutaneous pocket on the chest. The donor site was closed primarily.
Then, the patient was turned to the supine position and subcutaneous undermining of the anterior thorax was finalized. The abdominal advancement flap was dissected and a new inframammary fold was created in the patients who were scheduled for it.
Adipose tissue was harvested preferably from the lower abdomen or inner thighs region. Low-volume tumescent liposuction was used (500 ml of normal saline with 1 : 500 000 of epinephrine). Adipose tissue was aspirated manually into 30-ml Luer-lock syringes using 3.5 mm cannula (model PLA187, Pouret Medical, France) and it was centrifuged at 1200 g for 3 minutes directly in 30ml syringes.
Processed fat was injected first into the pectoralis muscle from several points (4–6 points) longitudinally and transversely relative to the orientation of muscle fibers with 10 ml syringes using 2 mm cannulas (model PLA189, Pouret Medical, France). In this way, the fat was applied first into the deep layer, then into the middle layer and finally into the superficial layer of the muscle. Because the cannula and the injected fat are visible between the muscle fibers (during application into the middle and superficial layers) the fat transfer can be done accurately under visual control till the formation of a small bulging – autoprosthesis. Fat was analogically injected between superficial muscle fibers and also into the adipose layer of the skin paddle of the latissimus dorsi flap (Fig. 2). After fat injection, the latissimus dorsi flap was secured to the breast pocket so that the originally distal part of the flap created volume of the décolleté area. The latissimus dorsi flap was then folded in the bottom part of newly reconstructed breast to form extra bulk. Breast skin was finally draped over the new mound and the latissimus dorsi skin paddle was partially or totally de-epithelialized. Two suction drains were positioned before skin closure.
2. Application of the processed fat (a) into the pectoralis muscle and (b, c) into the latissimus dorsi muscle 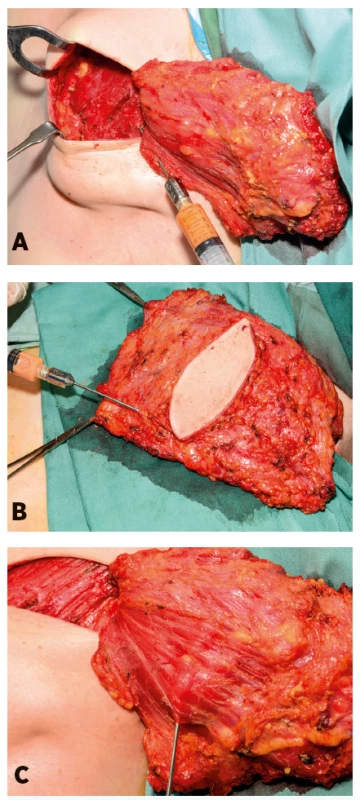
One or two sessions of lipomodelling were performed secondary at least 3 months after the previous surgery under general anesthesia. Contralateral symmetrizing mastopexy or reduction mammoplasty was performed 3-6 months after breast reconstruction, and finally in the following 3 months, nipple reconstruction using local flaps and areolar reconstruction using tattooing were performed under local anesthesia, if required by the patient.
Follow-up
All the patients were examined 1) clinically 2 weeks postoperatively, 2) clinically and by ultrasound shortly before each additional session of lipomodelling (when performed) and finally 3) clinically and by ultrasound at least 3 months postoperatively (after latissimus dorsi breast reconstruction or after final session of lipomodelling). The size and the number of fat necroses were evaluated by ultrasonography by a single experienced radiologist. Photographs were taken during each follow-up visit.
RESULTS
Breast reconstruction using the extended latissimus dorsi flap associated with fat grafting was performed in 14 patients. The mean patient age was 48.2 years (range, 34 to 64 years). The mean body mass index was 24.6 kg/m2 (range, 20.4 to 37.6 kg/m2) and 8 of these 14 patients had a history of adjuvant radiotherapy. In 7 patients, abdominal advancement flap was used to reconstruct the lower pole of the breast. Patient and operative data are shown in Table 1. The mean injected fat volume was 86.4 ml (range, 50 to 160 ml) when majority of volume was injected into the pectoralis major muscle. The mean injected fat volume to the pectoralis muscle was 57.9 ml.
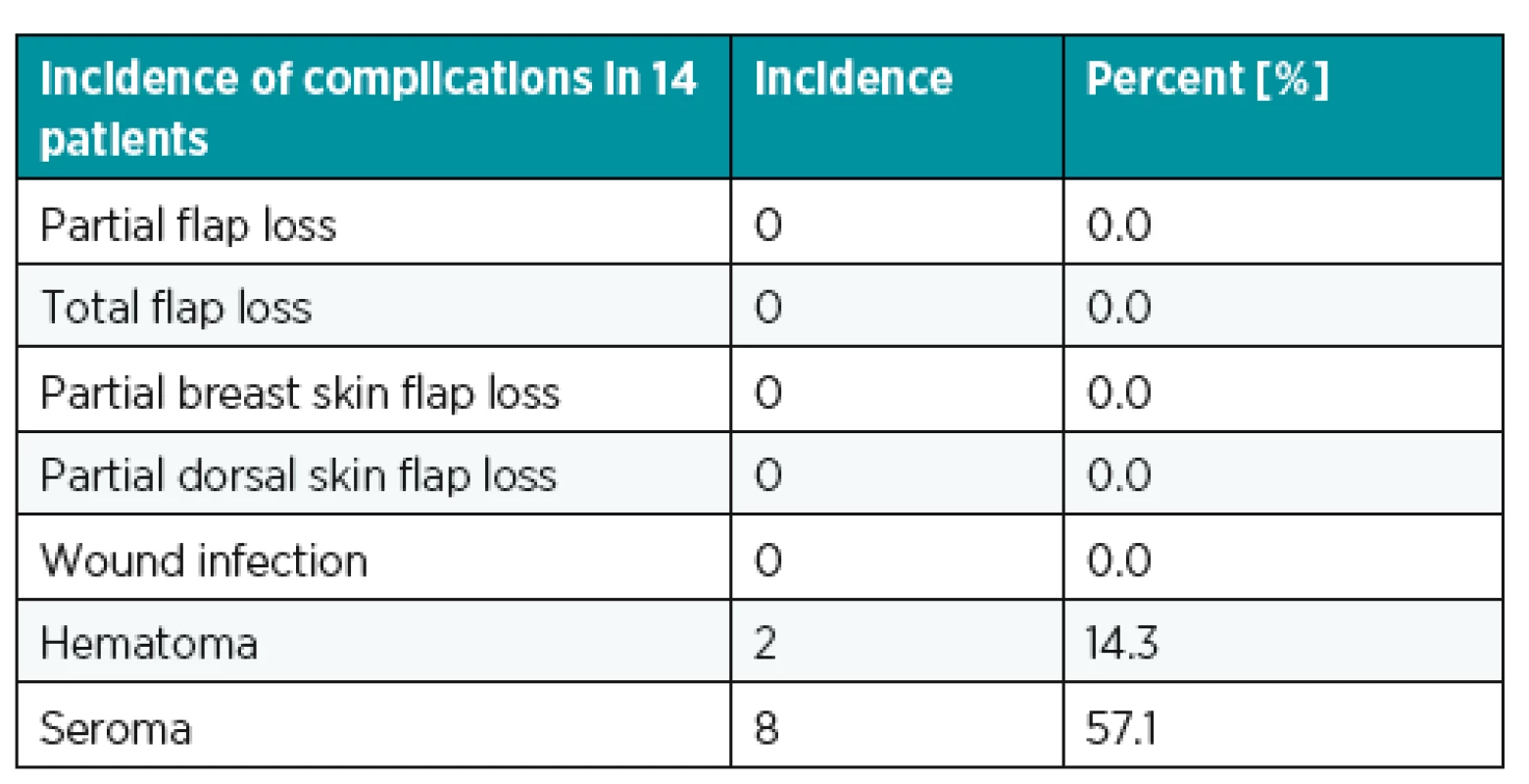
Suction drains were removed at around 6th postoperative day (range, 5th to 8th) when the fluid drainage from individual drain was less than 20 ml per 24 hours and when the patients were discharged from the hospital. The most common complication was seroma in the donor site (57.1%) and the most serious complication was dorsal hematoma with the need of early surgical revision. No fat grafting related complications were observed in either the recipient or the donor site (Table 3).
Eleven patients required an additional session of lipomodelling to enhance final breast volume and to improve breast shape while two sessions were need in 4 patients to obtain desired results. Then, 3 patients required symmetrizing surgery because of contralateral breast ptosis (2 patients) or relative hypertrophy (1 patient). Vertical scar mastopexy or wise pattern reduction mammoplasty was performed in these cases 3–6 months after breast reconstruction.
Postoperative ultrasound images were acquired for 10 patients. Specifically, number and size of oil cysts (oil liponecrotic pseudosysts) and fat necroses were evaluated separately for muscular layer (composed of pectoralis and latissimus dorsi muscles) and for subcutaneous layer of the reconstructed breast. We also evaluated maximal thickness of these layers and also overall maximal thickness of the reconstructed breast (Fig. 3, Table 4). Incidence and the size of oil cysts and fat necroses were significantly lower in muscular layer as compared with the subcutaneous layer. The muscular layer after fat grafting appeared as a homogeneous structure with significant lipomatous changes in comparison with normal skeletal muscle. The thickness of the muscular layer was expanded in additional stages of fat grafting. The resulting breast volume was made up mostly of the muscular layer augmented by fat grafting.
3. Typical postoperative ultrasound image. Muscular layer (ML) after fat grafting is shown as a homogeneous structure with significant lipomatous changes without oil cysts or fat necroses. The pectoralis (PM) and the latissimus dorsi (LTD) muscles can be distinguished according to the orientation of muscle fibers. There are single medium size oil cysts (OC) in the subcutaneous layer (SL) 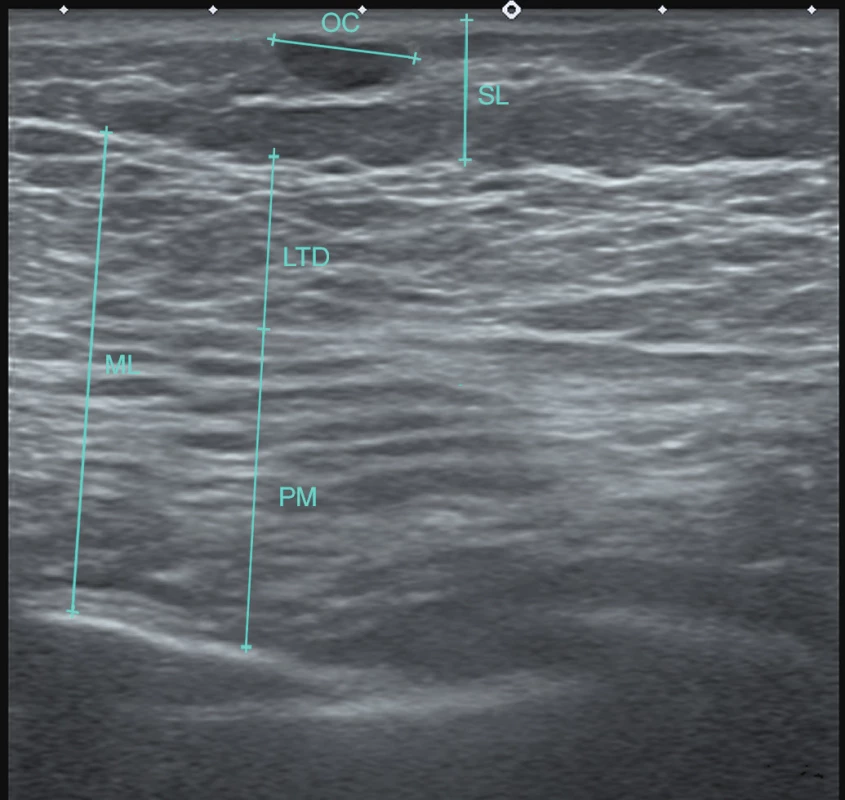
4. Evaluation of the postoperative breast ultrasound examination. The size of fat necrosis or oil liponecrotic pseudocysts is expressed by different letters: S – small (up to 10 mm in diameter), M – medium (10 to 20 mm in diameter) and L – large (more than 20 mm in diameter). The number of these fat necroses or oil liponecrotic pseudocysts is expressed by the number of these letters: one letter = single, two letters = sporadic (2–10), three letters = frequent (more than 10) and finally hyphen = none 
Preoperative and postoperative photographs are shown (Figs 4, 5). Satisfactory results were obtained and the scars in the donor area were well hidden under the bra strap and no contour defects were observed.
4. Patient 6: A 43-years-old women 48 months after mastectomy and adjuvant radiotherapy. She refused to have a scar on her abdomen. She underwent latissimus dorsi breast reconstruction associated with fat grafting into the pectoralis (40 ml) and latissimus dorsi (50ml) muscles, and with abdominal advancement flap. Two additional sessions of lipomodelling (205 and 130 ml) were performed to increase breast volume. One resulting horizontal scar on the breast and dorsal scar hidden in the bra. Preoperative and postoperative frontal, oblique and dorsal views, follow-up 22 months 
5. Patient 3: A 53-years-old women 18 mounts after mastectomy and adjuvant radiotherapy. She was not considered a good candidate for a DIEP flap breast reconstruction because she was very thin and a strong smoker. She underwent latissimus dorsi breast reconstruction associated with fat grafting into the pectoralis muscle (50 ml), and with abdominal advancement flap (allowing to reduce the size of the skin paddle and thus the length of the scars on her back). One additional session of lipomodelling (255 ml) was performed to increase breast volume. Preoperative and postoperative frontal and oblique views, follow-up 25 months 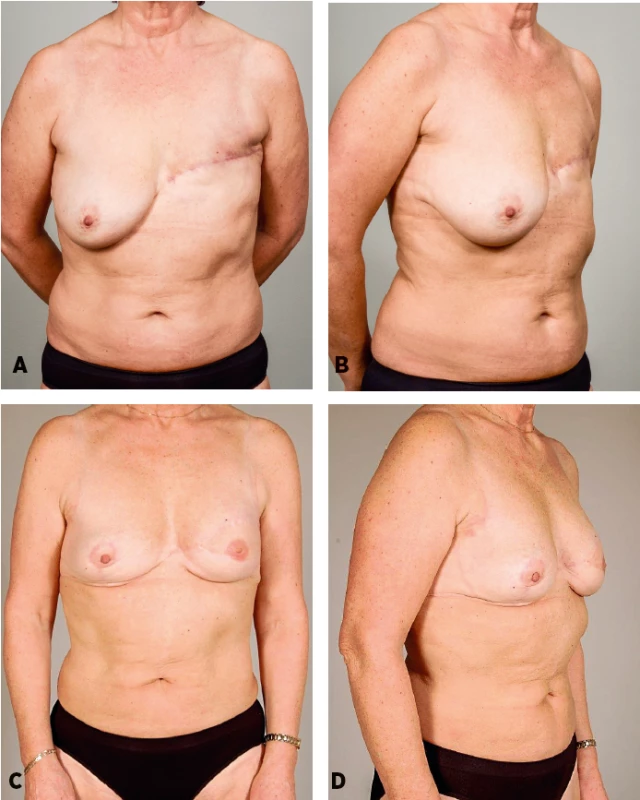
DISCUSSION
The breast reconstruction after mastectomy represents an integral component of the complex oncological care of breast cancer patients restoring physical and psychological integrity and improving women’s quality of life 16. Choice of reconstructive technique is a multifactorial decision based on several factors including patients’ age, body type, comorbidities, history of adjuvant radiotherapy and the size of contralateral breast. This decision is considerably influenced by the surgeons’ preference and experience. Implant based breast reconstruction is generally associated with lower invasiveness in the sense of donor site morbidity. In spite of that, autologous techniques are often preferred for the disadvantages of using silicone implants (risks of infection, extrusion, rupture, capsular contracture and a recently suggested association with anaplastic large-cell lymphoma 17). The abdominal free flaps and the latissimus dorsi flap are two most common autologous techniques today. The abdominal free flaps are often considered to be the gold standards for autologous breast reconstruction. In our department, we perform approximately 130 abdominal free flap breast reconstructions per year representing approximately 80% of autologous reconstructions 18–20.
We believe that current approaches to breast reconstruction based on the latissimus dorsi flap represent valuable alternatives for the patients who are not good candidates for abdominal based flaps, or even primary option for the patients who refuse to have a scar on the abdomen, or do not wish to undergo more demanding microsurgical procedures with the risk of flap loss. We also suggest the latissimus dorsi flap as a preferable alternative to the abdominal free flaps in patients with small skin excess in the lower abdomen when the resulting scar is usually pushed upwards above the edge of the underwear. In our department, latissimus dorsi breast flap represents approximately 10% of all performed autologous breast reconstructions.
Lindegren and the colleagues compared DIEP and latissimus dorsi flaps breast reconstruction with regards to the satisfaction with the aesthetic outcomes between the patients and plastic surgeons. The patients were more satisfied with latissimus dorsi flap reconstruction, which was related probably to the donor-site scar. Surgeons favored DIEP flap reconstruction with regards to the size and shape of the breast 21.
Santanelli and colleagues have recently reported their technique of latissimus dorsi flap volume expansion with intraoperative fat grafting to the subcutaneous tissue of the skin paddle of the latissimus dorsi flap as an alternative for entirely autologous breast reconstruction. In their study, the mean size of the harvested skin paddle was 19.7 x 11.0 cm and the mean injected fat volume was 101 ml 15.
We believe that too extensive harvest of skin paddle may result in more obvious disfigurement in the donor site area. Therefore, we have made an effort to minimize this disfigurement and the size of the scar on the back; the size of the skin paddle was up to 6 x 14 cm in the majority of the patients. Instead, we supplemented latissimus dorsi flap volume by adipose tissue from the areas where reduction of volume was beneficial. Furthermore, we injected fat into the pectoralis muscle. During our initial experiences, quantities of transferred adipose tissue were small, but we have gradually increased these quantities to 60 ml injected into the latissimus dorsi and to 120 ml injected into pectoralis muscles (see Table 1). We demonstrated that the capacity of pectoralis muscle to accept volume of the fat graft is about 2 times higher. We believe that this capacity is related to the initial thickness of the muscle layer while the pectoralis muscle is noticeably thicker than the latissimus muscle.
Ultrasound did not reveal higher incidence of fat necrosis in the muscular layer associated with the increasing volumes of the fat graft. Furthermore, number of fat necroses (cystoids) was significantly lower in muscular layer comparing with subcutaneous layer (Figure 3, see Table 4). These findings indicate that skeletal muscle is suitable and well-vascularized recipient tissue. Since the pectoralis muscle is exposed during breast reconstruction, fat graft can be applied under visual control, which enables application of fat more evenly in layers and thus to transfer higher volumes of adipose tissue. Analogically, fat graft is injected under visual control during immediate breast reconstruction by fat grafting 22–24, or without visual control during the secondary reconstructions 24,25.
Skeletal muscle seems to be eligible recipient tissue for adipose tissue grafting because it contains natural adipocytes 26.We had the opportunity to observe the pectoralis muscle in the 49-years-old patient 6 months after application of fat when we narrowed the breast base secondary after secondary breast reconstruction with lipomodelling (patient had skin excesses laterally because she had suffered from breast hypertrophy before mastectomy). After obtaining informed consents, we took small skeletal muscle samples (approximately 1 x 1 x 2 cm) from the pectoralis muscle after fat grafting and from the serratus anterior muscle (without fat grafting). Macroscopically, we observed lipomatous changes of the pectoralis muscle. Subsequently, the samples of the muscle were processed with the standard formalin – paraffin technique. 4 µm thick sections were cut with microtome, routinely stained with haematoxylin and eosin, and evaluated under the light microscope. In both samples, muscular component was well preserved. But we observed significantly higher number of adipocytes (respectively areas of adipose tissue) between muscle fibers in the sample taken form the pectoralis muscle after fat grafting. In the muscle sample of serratus anterior muscle from the same patient, we detected only a small number of adipocytes (Fig. 6).
6. Histological examination of the muscles: (a) the pectoralis muscle after fat grafting with lipomatous changes, (b) the serratus anterior muscle - the control sample from the same patient (without fat grafting). Haematoxylin eosin staining. Scale bar 100 mm. 
Adipocytes are found in the skeletal muscle naturally and fat content within muscles increases with age and atrophy and finally in paresis 27. Therefore, it is possible to understand the fat transfer into the muscle as an enlargement of its “adipose component” rather than as an application of tissue that in general does not belong to the skeletal muscle.
Capacity of the donor site to accept the fat graft is closely related to the initial volume of well-vascularized donor tissues. We believe that the degree of volume absorption is low when the transferred fat is covered by well-vascularized tissue like skeletal muscle. Based on our experiences we estimate the absorption about 30% in this case.
We can imagine a further increase in the quantity of the transferred fat up to the maximum of 140–160 ml into the pectoralis muscle and 60–70 ml into the latissimus dorsi muscle in normal-weight or slightly overweight patients. But we assume that the risk of complications associated with poor engraftment of the fat could increase beyond an acceptable level when transferred quantities were even higher. According to our experience, the best results are obtained in patients with BMI 20–26. Very thin patients usually have thinner recipient muscles. Furthermore, these very thin patients may not have enough adipose tissue in the donor sites.
The lack of a control group is the weakness of our study. Further prospective controlled clinical trials are needed to confirm our findings.
CONCLUSIONS
Our new modification of breast reconstruction with extended latissimus dorsi flap with immediate fat grafting into the pectoralis and latissimus dorsi muscles is a safe and reliable method for entirely autologous secondary breast reconstruction. Pectoralis and latissimus dorsi muscles were shown to be reliable and safe recipients for grafted fat. Fat grafting into the pectoralis and latissimus dorsi muscles during extended latissimus dorsi flap reconstruction allows reconstructing additional breast volume. We believe that our new concept may potentially reduce the number of required additional lipomodelling sessions, which often follow latissimus dorsi breast reconstruction.
Funding: By 1) Ministry of Health, Czech Republic - Conceptual Development of Research Organization (Masaryk Memorial Cancer Institute – IN: 00209805) and by 2) the Ministry of Education, Youth and Sports, Czech Republic – National Program of Sustainability I – LO1413.
Conflicts of interest: None declared.
Ethical approval: Not required.
Corresponding author:
Libor Streit, M.D., Ph.D.
Department of Plastic and Aesthetic Surgery, St. Anne’s University Hospital Brno and Faculty of Medicine, Masaryk University
Berkova 34, 612 00 Brno
Czech Republic
E-mail: liborstreit@gmail.com
Sources
1. Tansini I. Sopra il mio nuovo processo di amputazione della mammella. Gazz Med Ital. 1906,57 : 141.
2. Olivari N. The latissimus flap. Br J Plast Surg. 1976 Apr;29(2):126-8.
3. Schneider WJ, Hill HL Jr, Brown RG. Latissimus dorsi myocutaneous flap for breast reconstruction. Br J Plast Surg. 1977 Oct;30(4):277-81.
4. Bostwick J, Vasconez LO, Jurkiewicz MJ. Breast reconstruction after a radical mastectomy. Plast Reconstr Surg. 1978 May;61(5):682-93.
5. Bostwick J, Nahai F, Wallace JG, Vasconez LO. Sixty latissimus dorsi flaps. Plast Reconstr Surg. 1979 Jan;63(1):31-41.
6. Hokin JA. Mastectomy reconstruction without a prosthetic implant. Plast Reconstr Surg. 1983 Dec;72(6):810-18.
7. Marshall DR, Anstee EJ, Stapleton MJ. Soft tissue reconstruction of the breast using an extended composite latissimus dorsi myocutaneous flap. Br J Plast Surg. Jul 1984;37(3):361-368.
8. Germann G, Steinau HU. Breast reconstruction with the extended latissimus dorsi flap. Plast Reconstr Surg. 1996 Mar;97(3):519-26.
9. Delay E, Gounot N, Bouillot A, Zlatoff P, Comparin JP. Breast reconstruction with the autologous latissimus dorsi flap. Preliminary report of 60 consecutive reconstructions. Ann Chir Plast Esthet. 1997 Apr;42(2):118-30.
10. Delay E, Gounot N, Bouillot A, Zlatoff P, Rivoire M. Autologous latissimus breast reconstruction: a 3-year clinical experience with 100 patients. Plast Reconstr Surg. 1998 Oct;102(5):1461-78.
11. Delay E, Jorquera F, Pasi P, Gratadour AC. Autologous latissimus breast reconstruction in association with the abdominal advancement flap: a new refinement in breast reconstruction. Ann Plast Surg. 1999 Jan;42(1):67-75.
12. Sinna R, Delay E, Garson S, Delaporte T, Toussoun G. Breast fat grafting (lipomodelling) after extended latissimus dorsi flap breast reconstruction: A preliminary report of 200 consecutive cases. J. Plast. Reconstr. Aesthetic Surg. 2010;63(11):1769-1777.
13. Delay E, Garson S, Tousson G, Sinna R. Fat injection to the breast: technique, results, and indications based on 880 procedures over 10 years. Aesthet Surg J. 2009 Sep-Oct;29(5):360-76.
14. Delay E, Streit L, Toussoun G, La Marca S, Ho Quoc C. Lipomodelling: an important advance in breast surgery. Acta Chir Plast. 2013;55(2):34-43.
15. Santanelli di Pompeo F, Laporta R, Sorotos M, Pagnoni M, Falesiedi F, Longo B. Latissimus dorsi flap for total autologous immediate breast reconstruction without implants. Plast Reconstr Surg. 2014 Dec;134(6):871e-9e.
16. Dražan L, Veselý J, Hýža P, Kubek T, Foretová L, Coufal O. Surgical prevention of breast carcinoma in patients with hereditary risk. Klin Onkol. 2012;25 Suppl:S78-83.
17. Kim B, Predmore ZS, Mattke S, van Busum K, Gidengil CA. Breast Implant-associated Anaplastic Large Cell Lymphoma: Updated Results from a Structured Expert Consultation Process. Plast Reconstr Surg Glob Open. 2015 Feb 6;3(1):e296.
18. Veselý J, Stupka I, Drazan L, Holusa P, Licata P, Corradini B. DIEP flap breast reconstruction-new experience. Acta Chir Plast. 2001;43(1):3-6.
19. Drazan L, Vesely J, Hyza P, Castagnetti F, Stupka I, Justan I, Novak P, Monni N. Bilateral breast reconstruction with DIEP flaps: 4 years‘ experience. J Plast Reconstr Aesthet Surg. 2008 Nov;61(11):1309-15.
20. Hyza P, Streit L, Vesely J, Stafova D, Sin P. New technique of immediate nipple reconstruction during immediate autologous DIEP or MS-TRAM breast reconstruction. Ann Plast Surg. 2015 Jun;74(6):645-51.
21. Lindegren A, Halle M, Docherty Skogh AC, Edsander-Nord A. Postmastectomy breast reconstruction in the irradiated breast: a comparative study of DIEP and latissimus dorsi flap outcome. Plast Reconstr Surg. 2012 Jul;130(1):10-8.
22. Al-Kalla T, Komorowska-Timek E. Breast total male breast reconstruction with fat grafting. Plast Reconstr Surg Glob Open. 2014 Dec 5;2(11):e257.
23. Khouri RK, Rigotti G, Khouri RK Jr, Cardoso E, Marchi A, Rotemberg SC, Baker TJ, Biggs TM. Tissue-engineered breast reconstruction with Brava-assisted fat grafting: a 7-year, 488-patient, multicenter experience. Plast Reconstr Surg. 2015 Mar;135(3):643-58.
24. Ho Quoc C, Piat JM, Carrabin N, Meruta A, Faure C, Delay E. Breast reconstruction with fat grafting and BRAVA(®) pre-expansion: Efficacy evaluation in 45 cases. Ann Chir Plast Esthet. 2016 Jun;61(3):183-9.
25. Mestak O, Mestak J, Bohac M, Edriss A, Sukop A. Breast Reconstruction after a Bilateral Mastectomy Using the BRAVA Expansion System and Fat Grafting. Plast Reconstr Surg Glob Open. 2013 Dec 6;1(8):e71.
26. Guo Z, Mishra P, Macura S. Sampling the intramyocellular triglycerides from skeletal muscle. J Lipid Res. 2001 Jul;42(7):1041-8.
27. English C, Thoirs K, Coates A, Ryan A, Bernhardt J. Changes in fat mass in stroke survivors: a systematic review. Int J Stroke. 2012 Aug;7(6):491-8.
Labels
Plastic surgery Orthopaedics Burns medicine Traumatology
Article was published inActa chirurgiae plasticae

2017 Issue 2-
All articles in this issue
- Editorial
- COMPLICATIONS OF LOWER EXTREMITY HEMATOMAS IN PATIENTS WITH PRE-INJURY WARFARINE USE
- SURGICAL CORRECTION OF LABIA MINORA HYPERTROPHY, A PERSONAL TECHNIQUE
- SUTURE VERSUS FIBRIN GLUE MICRONEURAL ANASTOMOSIS OF THE FEMORAL NERVE IN SPRAGUE DEWLY RAT MODEL. A COMPARATIVE EXPERIMENTAL ASSESSMENT OF THE CLINICAL, HISTOLOGICAL AND STATISTICAL FEATURES
- INTRAOPERATIVE FAT GRAFTING INTO THE PECTORALIS AND LATISSIMUS DORSI MUSCLES-NOVEL MODIFICATION OF AUTOLOGOUS BREAST RECONSTRUCTION WITH EXTENDED LATISSIMUS DORSI FLAP
- HAS A GLOMUS TUMOR ALWAYS A QUICK DIAGNOSIS?
- CURRENT CONCEPTS IN PERIPHERAL NERVE INJURY REPAIR
- SUBACUTE ARTERIAL BLEEDING AFTER SIMULTANEOUS MASTOPEXY AND BREAST AUGMENTATION WITH IMPLANTS: CASE REPORT
- AUTOLOGOUS FAT TRANSFER, BREAST LIPOMODELLING AND FAT TRANSFER TO THE FACE: CURRENT GOLD STANDARDS AND EMERGING NEW DATA
- Acta chirurgiae plasticae
- Journal archive
- Current issue
- Online only
- About the journal
Most read in this issue- SURGICAL CORRECTION OF LABIA MINORA HYPERTROPHY, A PERSONAL TECHNIQUE
- INTRAOPERATIVE FAT GRAFTING INTO THE PECTORALIS AND LATISSIMUS DORSI MUSCLES-NOVEL MODIFICATION OF AUTOLOGOUS BREAST RECONSTRUCTION WITH EXTENDED LATISSIMUS DORSI FLAP
- COMPLICATIONS OF LOWER EXTREMITY HEMATOMAS IN PATIENTS WITH PRE-INJURY WARFARINE USE
- HAS A GLOMUS TUMOR ALWAYS A QUICK DIAGNOSIS?
Login#ADS_BOTTOM_SCRIPTS#Forgotten passwordEnter the email address that you registered with. We will send you instructions on how to set a new password.
- Career