-
Medical journals
- Career
Assessment of colorectal anastomosis perfusion with confocal laser endomicroscopy − an experimental study
Authors: J. Ptáčník 1,2; J. Kalvach 1,3; M. Kollár 4; J. Martínek 5; Š. Juhás 1; J. Juhásová 1; H. Kristianova 1,2; O. Ryska 1,6
Authors‘ workplace: Ústav živočišné fyziologie a genetiky Akademie věd České republiky, v. v. i., Liběchov 1; I. chirurgická klinika – hrudní, břišní a úrazové chirurgie, Všeobecná fakultní nemocnice, Praha 2; Chirurgická klinika 2. lékařské fakulty Univerzity Karlovy a Ústřední vojenské nemocnice, Praha 3; Pracoviště klinické a transplantační patologie, Institut klinické a experimentální medicíny, Praha 4; Klinika hepatogastroenterologie, Praha 5; Royal Lancaster Infirmary, UHMB NHS Foundation Trust, Lancaster 6
Published in: Rozhl. Chir., 2022, roč. 101, č. 12, s. 593-598.
Category: Original articles
doi: https://doi.org/10.33699/PIS.2022.101.12.593–598Overview
Introduction: Early diagnosis of complicated healing of colorectal anastomosis can increase the chance for salvage surgery and thus reduce overall morbidity. Confocal laser endomicroscopy (CLE) enables in vivo assessment of tissue perfusion without disturbing its integrity. This experimental study evaluates the potential of CLE for postoperative monitoring of colorectal anastomosis.
Methods: A hand-sewn colorectal anastomosis was performed in 9 pigs. The animals were subsequently divided into groups with normal (N=3) and ischemic anastomosis (N=6). Microscopic signs of hypoperfusion were evaluated postoperatively at regular intervals using CLE.
Results: Uneven saturation of the images was evident in the group with ischemic anastomosis. The epithelium had inhomogeneous edges and more numerous crypt branching was visible. Tissue oedema quantified as the number of crypts per visual field was already more extensive at the first measurement after induction of ischemia. There was also a significant difference between the values measured before and 10 minutes after ischemia – 8.7±1.9 vs. 6.0±1.1 (p=0.013).
Conclusion: Postoperative monitoring of the colorectal anastomosis using CLE enables prompt detection of perfusion disorders.
Keywords:
anastomotic leak – Perfusion – confocal laser endomicroscopy – colorectal anastomosis
INTRODUCTION
Following rectal resection with low anastomosis, anastomotic leak (AL) may occur in up to 20% of patients [1]. This complication is associated not only with a risk of reoperation, longer hospitalization, longer recovery and increased total treatment cost, but also a higher mortality and worse oncological outcomes. Various protective measures (protective stoma, assessment of perfusion of the resection margins) which would potentially decrease AL rate were evaluated in the past. However, the outcomes remain controversial [2,3].
Conventional surgical treatment of clinically significant AL with diffuse peritonitis most often requires laparotomy, disconnecting the anastomosis and creating a terminal colostomy. In clinical practice, restoration of bowel continuity in such patients is only achieved in 30% of cases [4]. An alternative is to attempt to salvage the anastomosis [5]. Early diagnosis of AL prior to sepsis development is essential.
In the past, several predictors of complicated anastomosis healing were assessed, such as the levels of acute phase proteins and proinflammatory cytokines, evaluation of metabolites and bacterial colonization in the peritoneal exudate, and evaluation of oxygenation around the anastomosis. However, none of these methods enables reliable identification of AL before development of systemic inflammatory response, thus the window suitable for attempting to salvage the anastomosis [6–11].
Confocal laser endomicroscopy (CLE) enables in vivo monitoring of the microcirculation at the anastomotic site. CLE utilises the physical principle of a confocal microscope, the light source is a laser, which illuminates the specimen through a confocal screen. Not only light, but also fluorescent radiation emitted from the specimen passes through this system. In the case of CLE, radiation refracted from the tissue is subsequently processed by software, which creates a complete histological image seen on the monitor. After application of the contrast agent (fluorescein), the bowel mucosa is scanned with high resolution at 1000× magnification. The endomicroscopic probe may be inserted directly or through endoscope and may be left in situ for continuous monitoring [12]. The benefit of this technique of microcirculation monitoring was documented on a septic experimental model [13]. CLE has not yet been used for postoperative anastomosis monitoring.
The aim of this study was to evaluate postoperative monitoring of colorectal anastomosis using CLE on an experimental model. The secondary aim was to establish objective criteria to evaluate ischemic changes at the anastomotic site.
METHODS
A total of 9 laboratory pigs with an average weight of 43 (35–55) kg were included in the study. All animals were transported to the experiment location (Institute of Animal Physiology and Genetics, Czech Academy of Sciences, Libechov) at least five days prior to surgery in order to ensure necessary acclimation. The project protocol (no. 18-2017, 42-2018) was approved by the expert ethical committee of Czech Academy of Science in accordance with valid legal regulations (no. 246/1992, 207/2004).
Anesthesia
The animals were fed standard feed until the night before the planned surgery. The pigs were premedicated by intramuscular injection containing a mixture of Tiletamine-Zolazepam 4 mg/kg (Zoletil 100, Virbac, Carros, France), Ketamine 10 mg/kg (Narketan 10, Ventoquinol, Paris, France) and Xylazine 2 mg / kg (Rometar 2%, SpofaDental Inc, Jicin, Czech Republic). The animals were then transported to the operating room and intubated. General anaesthesia was induced and maintained by Isoflurane (Isofluran, Piramal Critical Care, PA, USA) and Fentanyl 150–200 mg/hour (Fentanyl - Janssen, Piramal Critical Care, PA, USA). Dosage was adjusted based on reflex evocation.
Surgery
A low midline laparotomy was performed, and the distal sigmoid and rectum were mobilised. The colon was transected at the level of the proximal rectum leaving the mesocolon intact. Hand-sewn end-to-end colorectal anastomosis was then constructed in one layer using Vicryl 3-0 (Ethicon US LLC, OH, USA). The animal was then placed in either the control group with a normal anastomosis (N=3) or in the ischemic group (N=6). Ischemia was induced by ligation of the inferior mesenteric artery. The bowel was subsequently skeletonized 4 cm orally and aborally from the anastomosis.
Anastomosis Monitoring
The Gastro-Flex UHD probe (Cellvizio, Mauna Kea Technologies, Paris, France) was used to evaluate perfusion, it was introduced into the anastomosis using a two-channel endoscope (GIF 2T160; Olympus Medical, Tokyo, Japan). The areas 0.5–1cm above and under the anastomosis were always scanned. The image was analysed by the original processor Cellvizio with monitor (Mauna Kea Technologies, Paris, France). Two minutes prior to the CLE examination, fluorescein was administered intravenously (1.0–5.0 ml of 10% solution). The scanning was done immediately after surgery and then in 30-minute intervals during following three hours.
In six animals, ischemia was induced to the anastomosis 20 minutes after the operation using the above-mentioned technique. Subsequent assessment was done ten minutes after ischemia induction (meaning 30 minutes after the operation). Monitoring in the control group was performed similarly, except there was no further intervention.
Scans obtained from the acquired short video sequences were then evaluated for the presence of ischemic changes – image intensity, character of the interstitium, cell appearance, crypt branching and epithelial borders. The extent of oedema was objectively evaluated by calculating the number of vertically captured crypts in one visual field. At least 15 visual fields were counted from each scan. The values obtained from normal and ischemic anastomoses were then compared.
Perioperative care
During the experiment, all animals received an infusion of balanced crystalloids or glucose (55 mg/l) to maintain homeostasis. Systolic, diastolic and mean arterial pressures were continuously monitored to rule out causes of possible splanchnic hypoperfusion. Animals were actively warmed during the experiment. After the final measurements were performed, the experiment was terminated by deepening the sedation and intravenous administration of KCl.
Statistics
Based on the type of data distribution, they are presented as an means with standard deviation. Student ´s T-test was used to compare control and ischaemic groups and to compare data obtained before and after ischemia induction. A value of p<0,05 was considered statistically significant. Data was processed using software MedCalc 19.2 (MedCalc Software Ltd, Belgium).
RESULTS
The surgery and subsequent monitoring were carried out without adverse events or serious technical problems in all nine pigs. All animals were hemodynamically stable throughout the entire experiment.
The histological image obtained using CLE in animals with ischemic anastomosis showed characteristic signs. Non-homogenous image saturation with hypodensity in the crypts was typical. The epithelium had uneven edges. There was an apparent halo around the cells, which was not present in animals with a normal anastomosis. During the monitoring course after ischemia induction, increasingly more frequent crypt branching was observed signifying a gradual breakdown of epithelial structure (Fig. 1, 2).
1. Confocal laser endomicroscopy imaging of normal colorectal anastomosis 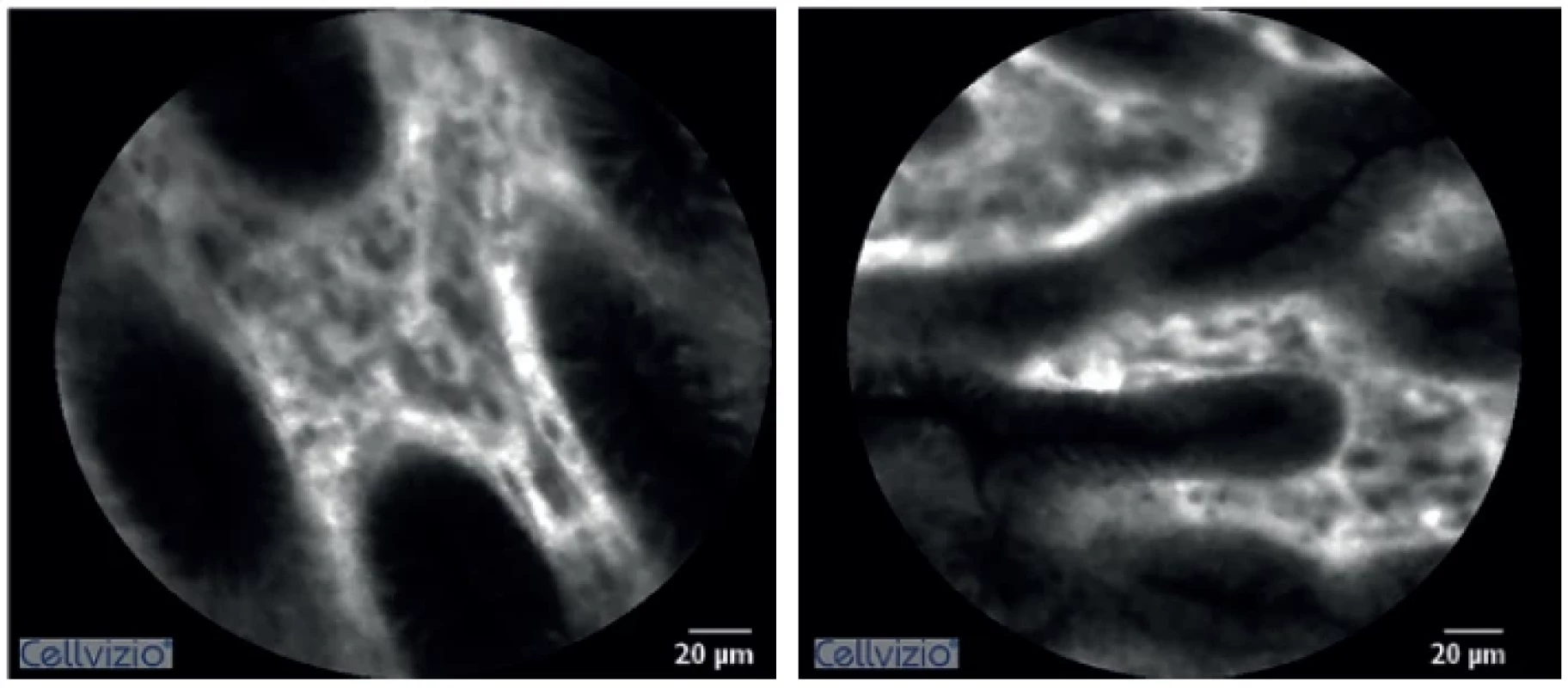
2. Confocal laser endomicroscopy imaging of ischemic colorectal anastomosis 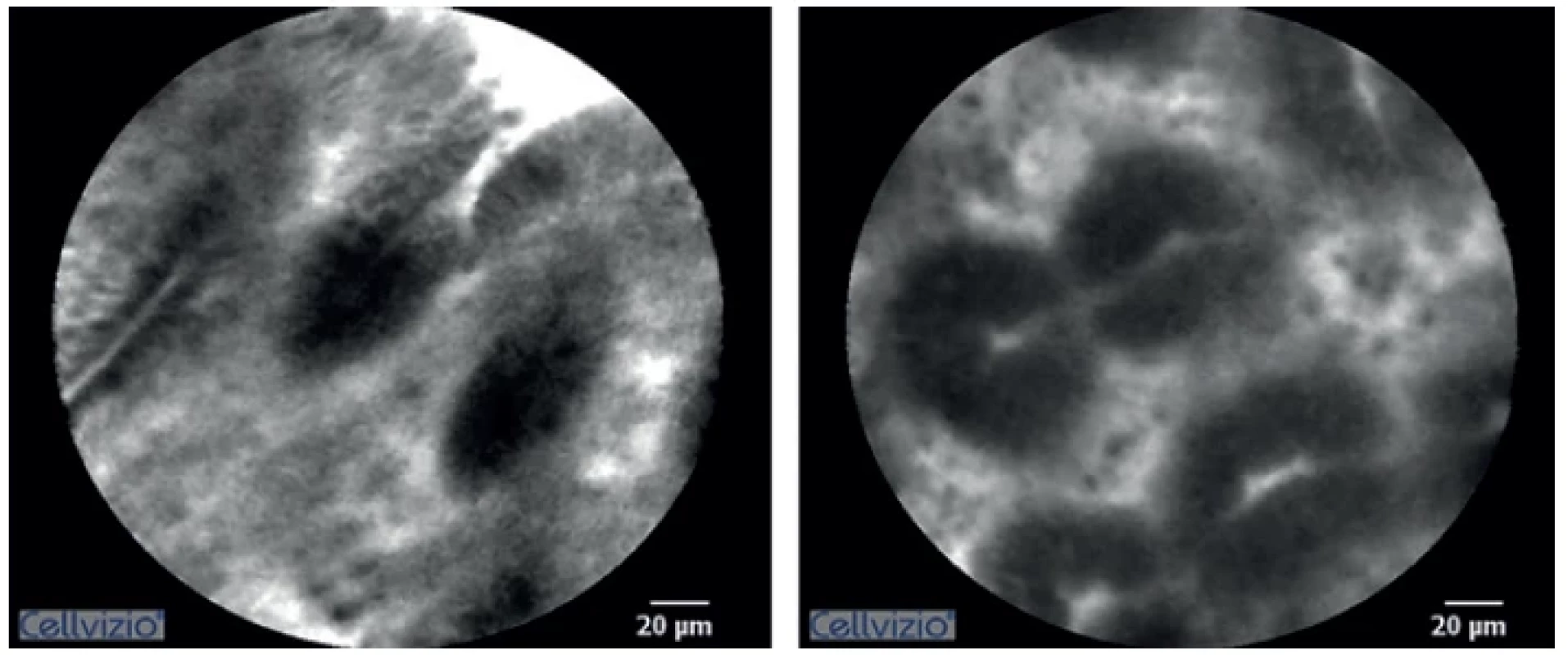
(Uneven saturation of the image is noticeable – in the lower part of the epithelium. Image hyperintensity can be seen in the crypt. The epithelium does not have homogeneous borders. Increased crypt branching is visible) When evaluating oedema, a statistically significant difference was seen between both groups, already at the first measuring after inducing ischemia (Graph 1). Similarly, a statistically significant difference was demonstrated between the baseline and values measured 10 minutes after ischemia induction – 8.7±1.9 vs. 6.0±1.1 (p=0.013).
1. Mean number of crypts (incl. standard deviation) per field assessed by confocal laser endomicroscopy in groups with normal (N=3) and ischemic anastomosis (N=6). 
DISCUSSION
Proactive postoperative monitoring and early diagnosis of dehiscence may contribute to ideal timing of reintervention and thus improve the outcomes. Treatment of AL prior to the development of sepsis or septic shock enables a less invasive approach. In such situations, transanal surgical or endoscopic treatment of the dehiscence, or possibly construction of a new anastomosis, may be considered [4–11].
In the past, a number of AL predictors were assessed, such as serum or peritoneal levels of acute phase proteins or proinflammatory cytokines. Intraperitoneal levels of various metabolites were also evaluated using microdialysis [6]. Changes in above-mentioned markers observed early in the postoperative period (1.–3. postoperative day) may, however, be affected by reaction to perioperative stress or normal healing processes. Indication for early surgical intervention based on these variables remains problematic. Additionally, significant differences between patients with and without AL are usually observed after the 6. postoperative day, when the clinical signs of dehiscence are usually manifested [14].
Adequate bloods supply or more precisely adequate oxygenation of anastomosis can be also assessed with the aim to predict impaired healing. The viability of resection margins may currently be evaluated using fluorescence angiography or hyperspectral imaging. The latter also enables quantification of tissue oxygenation with no need for contrast agent administration [10,15,16]. The viability of resection margins may also be evaluated using classical endoscopy with a significantly lower specificity and sensitivity [9]. These techniques are currently used mainly for intraoperative assessment.
Nevertheless, hypotension with splanchnic hypoperfusion or oedema which worsens tissue oxygenation may occur in later stages. Taking this into account, postoperative, preferably continual, monitoring seems to be more beneficial, albeit technically more demanding. Microdialysis can be used for continuous monitoring of metabolism at the anastomosis. This technique has already been verified in experimental study on intact bowel, as well as on a model of ischemic anastomosis, where the studied metabolites reflected the actual ratio between aerobic and anaerobic metabolism [14,17].
Confocal laser endomicroscopy (CLE) is a new, rapidly developing technique enabling in vivo tissue visualisation in real time with approximately 1000x magnification and 1 micrometre resolution. The visual field size ranges between 240 and 600 μm. CLE is based on irradiation of the surface (mucosa) by laser with low output and subsequent scanning of the refracted fluorescent light (12 scans with resolution 30 000 pixels per second) after intravenous application of contrast agent (Fluorescein). The scans or video sequences obtained may be assessed directly during the examination. Currently, CLE is most often used in combination with endoscopy, the probe is introduced directly to the examination site via the working channel of the endoscope. In digestive surgery and gastroenterology, CLE may be used in patients with esophageal, stomach or duodenal lesions, or to evaluate disease activity in patients with inflammatory bowel disorders (IBDs) [18,19]. The probe may also be placed directly on the tissue to verify, for example, perioperative negativity of resection margins. Unlike biopsy, CLE does not damage the examined tissue and does not require further processing. Compared to perioperative histology, CLE assessment is easier and enables evaluation of several areas, which decreases the risk of false negative findings [20].
Potential of CLE to be used for postoperative monitoring of bowel anastomosis has not been evaluate to date. Our study demonstrated that this technique is able to detect changes caused by impaired perfusion of anastomosis promptly. Characteristic signs were visible within 20 minutes after ischemia induction. Liu et al assessed blood supply of the intestinal anastomosis during surgery in their experimental study. The authors measured red blood cell velocity in capillaries to compare two groups of rabbits with normal and decreased anastomotic perfusion. They came to the conclusion that CLE enables intraoperative assessment of perfusion not only qualitatively, which is also possible with other techniques, but also quantitatively [21].
During our study, a new technique of assessing tissue oedema, which is closely associated with perfusion disorders and degree of oxygenation, was developed. Differences in overall number of crypts in the visual field were statistically significant 10 minutes after anastomotic ischemia. There is no risk of injury to the anastomosis during endomicroscopy and it is possible to repeat it in relatively short intervals. In the postoperative course, in the case of dehiscence, this method could assess the extent of ischemia or tissue damage and help to estimate chances for anastomotic salvage.
Correlation between the animal model of ischemic anastomosis and clinical practice can be considered as limitation of this study. This is the first evaluation of postoperative monitoring with CLE, therefore uniform and reliable model was required. Partial ischemia would surely be closer to conditions appearing in human medicine, however, wouldn’t suit the needs of this experiment due to high variability. This project also aimed to define measurable criteria of ischemia and therefore model with evident and prompt tissue changes was needed. Relatively small cohort can be also seen as study limitation. However, changes detected by CLE were so significant, that it was possible to demonstrate its efficacy even on a smaller group of subjects. Under these circumstances, enrolling more animals would be unethical.
Prior to clinical evaluation, it is necessary to solve several technical problems, including positioning and immobilization of the CLE probe in the area of anastomosis. Another problem may be to ensure a stable position of the probe once bowel function restores after surgery. On the other hand, has been more indications for CLE nowadays, which obviously increased its availability. This type of monitoring also fits into the concept of smart drains [22].
CONCLUSION
This experimental study confirmed CLE as a novel technique suitable for postoperative monitoring of colorectal anastomosis. New technique allowing quantification of microscopic signs of hypoperfusion was introduced.
Supported by grant NV19-08-00449 (Czech Health Research Council)
Conflict of interests
The authors declare that they have not conflict of interest in connection with this paper and that the article has not been published in any other journal, except congress abstracts and clinical guidelines.
MUDr. Ondřej Ryska, PhD, FEBS
Biskupcova 89
130 00 Praha 3
e-mail: ondrejryska@centrum.czRozhl Chir. 2022;101 : 593–598
Sources
1. Sciuto A, Merola G, De Palma G, et al. Predictive factors for anastomotic leakage after laparoscopic colorectal surgery. World J Gastroenterol. 2018 Jun 7;24(21):2247 – 2260. doi:10.3748/wjg.v24.i21.2247.
2. Frye J, Bokey EL, Chapuis PH, et al. Anastomotic leakage after resection of colorectal cancer generates prodigious use of hospital resources. Colorectal Dis. 2009;11 : 917–920. doi:10.1111/j.1463 - 1318.2008.01728.x.
3. Fraccalvieri D, Biondo S, Saez J, et al. Management of colorectal anastomotic leakage: differences between salvage and anastomotic takedown. Am J Surg. 2012 Nov;204(5):671–676. doi: 10.1016/j. amjsurg.2010.04.022.
4. Khan AA, Wheeler JM, Cunningham C, et al. The management and outcome of anastomotic leaks in colorectal surgery. Colorectal Dis. 2008 Jul;10(6):587–592. doi:10.1111/j.1463-1318.2007.01417.x.
5. Borstlap WAA, Musters GD, Stassen LPS, et al. Vacuum-assisted early transanal closure of leaking low colorectal anastomoses: the CLEAN study. Surg Endosc. 2018 Jan;32(1):315–327. doi:10.1007/s00464 - 017-5679-6.
6. Matthiessen P, Strand I, Jansson K, et al. Is early detection of anastomotic leakage possible by intraperitoneal microdialysis and intraperitoneal cytokines after anterior resection of the rectum for cancer? Dis Colon Rectum 2007 Nov;50(11):1918 – 1927. doi: 10.1007/s10350-007-9023-4.
7. Millan M, García-Granero E, Flor B, et al. Early prediction of anastomotic leak in colorectal cancer surgery by intramucosal pH. Dis Colon Rectum 2006 May;49(5):595–601. doi:10.1007/s10350 - 006-0504-7
8. Fouda E, El Nakeeb A, Magdy A, et al. Early detection of anastomotic leakage after elective low anterior resection. J Gastrointest Surg. 2011 Jan;15(1):137–144. doi:10.1007/s11605-010-1364-y.
9. Daams F, Wu Z, Lahaye MJ. Prediction and diagnosis of colorectal anastomotic leakage: A systematic review of literature. World J Gastrointest Surg. 2014 Feb 27;6(2):14–26. doi:10.4240/wjgs.v6.i2.14.
10. Sherwinter DA, Gallagher J, Donkar T, et al. Intra-operative transanal near infrared imaging of colorectal anastomotic perfusion: a feasibility study. Colorectal Dis. 2013 Jan;15(1):91–96. doi:10.1111/ j.1463-1318.2012.03101.x.
11. Birke-Sorensen H, Andersen NT. Metabolic markers obtained by microdialysis can detect secondary intestinal ischemia: an experimental study of ischemia in porcine intestinal segments. World J Surg. 2010 May;34(5):923–932. doi:10.1007/ s00268-010-0502-8.
12. Martínek J, Kollár M, Krajčíová J, et al. Confocal laser endomicroscopy in diagnosing indeterminate biliary strictures and pancreatic lesions − a prospective pilot study. Rozhl Chir. 2020;99 : 258–265 doi:10.33699/PIS.2020.99.6.258–265.
13. Schmidt C, Lautenschläger C, Petzold B et al. Confocal laser endomicroscopy reliably detects sepsis-related and treatment - associated changes in intestinal mucosal microcirculation. Br J Anaesth. 2013 Dec;111(6):996–1003. doi:10.1093/ bja/aet219.
14. Ellebaek Pedersen M, Qvist N, Bisgaard C, et al. Peritoneal microdialysis. Early diagnosis of anastomotic leakage after low anterior resection for rectosigmoid cancer. Scand J Surg. 2009;98(3):148–154. doi:10.1177/145749690909800304.
15. Benčurik V, Škrovina M, Martínek L, et al. Intraoperative fluorescence angiography and risk factors of anastomotic leakage in mini-invasive low rectal resections. Surgical Endosc. 2021;35 : 5015–5023. doi:10.1007/s00464-020-07982-x.
16. Jansen-Winkeln B, Dvorak M, Köhler H, et al. Border line definition using hyperspectral imaging in colorectal resections. Cancers (Basel) 2022 Mar;14(5):1188. doi:10.3390/cancers14051188.
17. Ryska O, Kalvach J, Pazin J, et al. Surface microdialysis for detection of colorectal anastomosis ischemia—An experimental study. J Surg Res. 2021 May;261 : 293–300. doi:10.1016/j.jss.2020.12.021.
18. Li WB, Zuo XL, Li CQ, et al. Diagnostic value of confocal laser endomicroscopy for gastric superficial cancerous lesions. Gut 2011 Mar;60(3):299–306. doi:10.1136/ gut.2010.223586.
19. Karstensen JG, Klausen PH, Saftoiu A, et al. Molecular confocal laser endomicroscopy: a novel technique for in vivo cellular characterization of gastrointestinal lesions. World J Gastroenterol. 2014 Jun 28;20(24):7794–800. doi:10.3748/wjg. v20.i24.7794.
20. De Palma GD, Luglio G, Staibano S, et al. Perioperative characterization of anastomotic doughnuts with high-resolution probe-based confocal laser endomicroscopy in colorectal cancer surgery: a feasibility study. Surg Endosc. 2014 Jul;28(7):2072–2077. doi:10.1007/ s00464-014-3429-6.
21. Liu X, Tang Y, Li Z, et al. In vivo real-time assessment of the anastomotic blood supply in colorectal surgery using confocal laser endomicroscopy in an anastomotic model. Surg Endosc. 2022 Jun;36(6):4136 – 4144. doi:10.1007/s00464-021-08738-x.
22. Roser MV, Frank AHR, Henrichs L, et al. Evaluation of an integrated smart sensor system for real-time characterization and digitalization of postoperative abdominal drain output: A pilot study. Surg Innov. 2022 Jun;29(3):438–445. doi: 10.1177/15533506211031459.
Labels
Surgery Orthopaedics Trauma surgery
Article was published inPerspectives in Surgery

2022 Issue 12-
All articles in this issue
- Experimentální chirurgie
- Artificial neural networks and computer vision in medicine and surgery
- Domestic pig head and neck arteries from the viewpoint of imaging methods and experimental surgery
- Permanent intravenous access in experimental surgery – our experience
- Chirurgie v době koronavirové
- Introducing in vivo pancreatic cancer models for the study of new therapeutic regimens
- Assessment of colorectal anastomosis perfusion with confocal laser endomicroscopy − an experimental study
- Experimental surgery as part of the development of degradable biomaterials for cardiovascular surgery
- Chylothorax treatment with thoracic duct embolization
- Perspectives in Surgery
- Journal archive
- Current issue
- Online only
- About the journal
Most read in this issue- Domestic pig head and neck arteries from the viewpoint of imaging methods and experimental surgery
- Chylothorax treatment with thoracic duct embolization
- Introducing in vivo pancreatic cancer models for the study of new therapeutic regimens
- Permanent intravenous access in experimental surgery – our experience
Login#ADS_BOTTOM_SCRIPTS#Forgotten passwordEnter the email address that you registered with. We will send you instructions on how to set a new password.
- Career

