-
Medical journals
- Career
Synthesis and antimicrobial properties of 2-[(7-ethyl-3-methylxanthin-8-yl)sulfanyl]acetohydrazide derivatives
Authors: Dmytro Ivanchenko 1; Mykola Romanenko 2; Olha Pakhomova 1; Oleksandra Cherchesova 1; Natalia Rudko 2; Natalia Voronova 3; Horban Valerii 3
Authors‘ workplace: Department of Psychology, Ukraine Sigmund Freud University, Ukraine. 1; Department of Clinical Laboratory Diagnostics and Biological Chemistry, Zaporizhzhia State Medical and Pharmaceutical University, Ukraine 2; Department of General and Applied Ecology and Zoology, Zaporizhzhia National University, Ukraine 3
Published in: Čes. slov. Farm., 2026; 75, 14
Category: Original Articles
doi: https://doi.org/10.36290/csf.2026.011Overview
Antimicrobial resistance (AMR) represents a major global health threat, driving the urgent search for new therapeutic agents. Hybrid molecules based on xanthine and 1,2,4-triazole scaffolds present a promising direction due to their potential multi-target antimicrobial activities. This study aimed at synthesizing new 2-[(7-ethyl-3-methylxanthin-8-yl)sulfanyl]acetohydrazide derivatives containing 1,2,4-triazole moieties, characterizing their structures, and evaluating their antimicrobial and antifungal properties.
A series of previously undescribed compounds was synthesized using N-substituted isothiocyanates and further S-alkylation/cyclization strategies. The structures were confirmed via 1H NMR spectroscopy, elemental analysis; mass spectrometry was additionally used for representative derivative 14. Antimicrobial activity was assessed against E. coli, S. aureus, P. aeruginosa and C. albicans by broth dilution methods to determine minimum inhibitory and bactericidal/fungicidal concentrations.
All synthesized derivatives were structurally characterized with high confidence. Most compounds exhibited weak to moderate antibacterial and antifungal activity. Notably, the amide and nitrile derivatives displayed moderate antifungal activity against C. albicans (MIC 25 μg/mL), comparable to that of ketoconazole (MIC 25 μg/mL). Structure–activity analysis indicated that S-substitution and side-chain modifications improve antimicrobial potency.
New xanthine-triazole hybrid molecules were synthesized and characterized, showing that selected derivatives possess promising antimicrobial and antifungal properties. These findings support further optimization and biological exploration of such hybrids as candidates to combat antimicrobial resistance.
Keywords:
antimicrobial activity – synthesis – triazole – xanthine derivatives – hybrid molecules
Introduction
Antimicrobial resistance (AMR) has emerged as one of the most critical global public health challenges of the 21st century, surpassing many traditional health concerns in its impact on mortality and healthcare systems. The World Health Organization classifies AMR among the top public health threats, with bacterial AMR directly responsible for 1.27 million global deaths in 2019 and associated with an additional 4.95 million deaths (1, 2).
Projections indicate that, without intervention, AMR could lead to 1.91 million attributable deaths and over 8.22 million associated deaths annually by 2050 (2). The economic burden is staggering, with the World Bank estimating US$1 trillion in additional healthcare costs by 2050 and annual GDP losses of US$1-5.2 trillion by 2050 (3). This crisis stems from antimicrobial misuse in humans, animals, and agriculture, compounded by poor infection control, inadequate sanitation, and global mobility facilitating pathogen spread.
Bacterial resistance mechanisms include genetic alterations (e. g., mobile genetic elements and mutations) and phenotypic adaptations (e. g., tolerance, persistence, and biofilms) (4). Biofilms, protected by extracellular polymeric substances (EPS), pose particular challenges by limiting antibiotic diffusion and promoting resistance gene transfer, making them key reservoirs for multidrug-resistant infections. The EPS matrices act as diffusion-limiting barriers to conventional antibiotics, while facilitating horizontal gene transfer through close cell-to-cell contact within biofilm communities (5–8).
To counter AMR limitations in conventional antibiotics, hybrid molecules combining multiple pharmacophores for synergistic, multitarget effects are promising. Purine and xanthine derivatives, structurally akin to alkaloids like caffeine and theophylline, exhibit diverse activities, including interference with bacterial DNA synthesis and repair (9). Synthetic xanthines (e. g., aciclovir, 6-mercaptopurine) show broad-spectrum antimicrobial potential with low toxicity (10). Recent studies have demonstrated that xanthine-based compounds can exhibit strong broad-spectrum antibacterial activity, weak hemolytic activity, and low cytotoxicity against mammalian cell lines, while maintaining potent in vivo efficacy against drug-resistant bacterial strains through mechanisms involving DNA synthesis inhibition and cell wall disruption (11–13).
Similarly, 1,2,4-triazole derivatives display antibacterial and antifungal properties, with S-substituted variants targeting enzymes, generating reactive oxygen species, and disrupting membranes (14, 15). The 1,2,4-triazole nucleus is well-recognized for its diverse biological activities, and structure-activity relationship studies have shown that modifications to triazole scaffolds can significantly influence antimicrobial potency and selectivity (16).
Hybridizing xanthine and triazole moieties offers synergistic potential against resistant strains through multiple mechanisms of action. This molecular hybridization strategy leverages the individual biological activities of each pharmacophore while potentially achieving enhanced antimicrobial effects. Structure-activity relationship studies indicate that substituents on these scaffolds (e. g., electron-donating groups on triazoles, alkyl modifications on xanthines) enhance potency and selectivity (14). However, comprehensive evaluations of such hybrids remain limited, highlighting a research gap in this promising area.
This study aimed at developing synthetic approaches for previously undescribed 7-ethyl-3-methylxanthine derivatives containing 1,2,4-triazole moieties at position 8, characterizing the obtained compounds, and assessing their antimicrobial potential as novel therapeutic agents against multidrug-resistant pathogens.
Materials and methods
Chemistry
All investigated compounds were synthesized at the Biochemistry Department at Zaporizhzhia State Medical and Pharmaceutical University. The structures of the synthesized compounds were confirmed by physicochemical methods. Melting points were set in open capillaries using a „Stanford Research Systems Melting point Apparatus 100“ (SRS, USA). Elemental analysis (C, H, N) was performed using an „Elementary vario EL cube“ analyzer (Elementary Analysensysteme, Germany). 1H NMR spectra were recorded on a „Bruker SF-400“ spectrometer (operating frequency 400 MHz, solvent DMSO, internal standard – TMS; Bruker Corporation, USA). Mass spectra were recorded on a Varian 1 200L mass spectrometer (Varian, Inc., USA) using electron impact ionization (70 eV) with direct sample insertion.
Antibacterial and antifungal activity
The antimicrobial and antifungal assays were performed using standard strains of Escherichia coli (ATCC 25922), Staphylococcus aureus (ATCC 25923), Pseudomonas aeruginosa (ATCC 27853) and Candida albicans (ATCC 885-653), which were obtained at the bacteriological laboratory of Zaporizhia Regional Laboratory Center of the State Epidemiological Service of Ukraine.
The stock solutions were prepared by dissolving 1.0 mg of each investigated substance in 1.0 mL of DMSO. Mueller–Hinton broth (4.0 mL) (for S. aureus ATCC 25923, E. coli ATCC 25922, P. aeruginosa ATCC 27853) or Sabouraud broth (for C. albicans ATCC 885-653) were added to the stock solution to obtain the necessary dilution containing the respective substance at 200 µg/mL concentration. A series of twofold dilutions was used to obtain three additional solutions with the concentrations of 100, 50 and 25 µg/mL respectively, each in a volume of 1 mL. An aliquot of 0.1 mL of the microbial suspension was added to each tube. The test tubes after inoculation with S. aureus, E. coli and P. aeruginosa were incubated at 37 ± 1°C for 16–24 h, after inoculation with C. albicans – at 28 ± 1 °C for 44-48 h. MIC (minimum inhibitory concentration) was defined as the lowest concentration showing no visible microbial growth. Although EUCAST/CLSI guidelines recommend testing six or more concentrations for precise MIC determination, the present study employed a four-point screening to assess relative antimicrobial potency.
For the determination of the minimum bactericidal concentration (MBC) and minimum fungicidal concentration (MFC), 0.1 mL from each tube showing no visible growth at or above the MIC was streaked onto Mueller–Hinton agar plates (for S. aureus, E. coli and P. aeruginosa) or Sabouraud agar plates (for C. albicans). The Petri dishes were incubated at 37 ± 1°C for 16–24 h for bacteria and at 28 ± 1°C for 44–48 h for C. albicans. MBC and MFC values were recorded as the lowest concentrations of the tested compounds that produced no visible colony growth on the agar surface (17).
Experimental part
Chemistry
Synthesis of 7-ethyl-3-methyl-8-{[(4-methyl-5-sulfanyl-4H 1,2,4-triazol-3-yl)methyl]sulfanyl}-3,7-dihydro-1H-purine-2,6-dione (2). A mixture of 3.0 g (0.01 mol) of 2-[(7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-8-yl)sulfanyl]acetohydrazide (1) (18), 0.7 g (0.01 mol) of N-methylisothiocyanate, 20 mL of H2O, and 20 mL of propan-2-ol was refluxed for 70 minutes, cooled, the precipitate was filtered, washed with H2O, and crystallized from aqueous dioxane. Calcd for C12H15N7O2S2 %: C, 40.78; H, 4.28; N, 27.74. Found %: C, 40.59; H, 4.12; N, 27.68. M.p. 273-275 °C, yield 54% (1.92 g). 1H NMR spectrum (400 MHz, DMSO-d6, δ, ppm): 13.55 (1H, s, SH), 11.10 (1H, s, N1H), 4.51 (2H, s, H-11), 4.14 (2H, q, J = 6.9 Hz, H-9), 3.45 (3H, s, H–3), 3.23 (3H, s, H-12), 1.19 (3H, t, J = 7.0 Hz, H-10).
7-Ethyl-8-{[(4-ethyl-5-sulfanyl-4H-1,2,4-triazol-3-yl)methyl]sulfanyl}-3-methyl-3,7-dihydro-1H-purine-2,6-dione (3). Obtained similarly to compound 2 (refluxed for 2 h and crystallized from aqueous propan-2-ol). Calcd for C13H17N7O2S2 %: C, 42.49; H, 4.66; N, 26.68. Found %: C, 42.66; H, 4.74; N, 26.75. M.p. 217-218 °C, yield 35% (1.3 g). 1H NMR spectrum (400 MHz, DMSO-d6, δ, ppm): 13.55 (1H, s, SH), 11.10 (1H, s, N1H), 4.55 (2H, s, H-11), 4.12 (2H, q, J = 6.8 Hz, H-9), 4.03 (2H, q, J = 7.4 Hz, H-12), 3.30 (3H, s, H-3), 1.25-1.17 (6H, m, H-10, 13).
Synthesis of 2-(2-[(7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetra hydro-1H-purin-8-yl)sulfanyl]acetyl)-N-phenylhydrazine-1-car bothioamide (4). A mixture of 3.0 g (0.01 mol) of compound 1, 1.5 g (0.011 mol) of N-phenyl isothiocyanate, 20 mL of H2O, and 20 mL of propan-2-ol was refluxed for 45 minutes, cooled, the precipitate was filtered, washed with H2O, and crystallized from aqueous dioxane. Calcd for C17H19N7O3S2 %: C, 47.1; H, 4.42; N, 22.62. Found %: C, 47.26; H, 4.55; N, 22.57. M.p. 180–181 °C, yield 67% (2.92g). 1H NMR spectrum (400 MHz, DMSO-d6, δ, ppm): 11.08 (1H, s, N1H), 10.32 (1H, s, NHCO), 9.71 (1H, s, NHCS), 9.42 (1H, s, NHPh), 7.44–7.12 (5H, m, H-12-16), 4.14 (2H, q, J = 7.1 Hz, H-9), 4.08 (2H, s, H-11), 3.30 (3H, s, H-3), 1.22 (3H, t, J = 7.1 Hz, H-10).
Synthesis of 7-ethyl-3-methyl-8-{[(4-phenyl-5-sulfanyl-4H-1,2,4-triazol-3-yl)methyl]sulfanyl}-3,7-dihydro-1H-purine-2,6-dione (5). A solution of 8.66 g (0.02 mol) of compound 4, 1.2 g (0.03 mol) of NaOH in a mixture of 100 mL H2O and 30 mL propan-2-ol was refluxed for 30 minutes, cooled, diluted with water to 250 mL, filtered, and the filtrate neutralized with acetic acid. The precipitate was filtered, washed with H2O, and crystallized from aqueous dioxane. Calcd for C17H17N7O2S2 %: C, 49.14; H, 4.12; N, 23.6. Found %: C, 49.01; H, 4.03; N, 23.72. M.p. 255-256 °C, yield 88% (7.3 g). 1H NMR spectrum (400 MHz, DMSO-d6, δ, ppm): 13.82 (1H, s, SH), 11.10 (1H, s, N1H), 7.45-7.37 (5H, m, H-12-16), 4.32 (2H, s, H-11), 4.03 (2H, q, J = 7.1 Hz, H-9), 3.31 (3H, s, H-3), 1.18 (3H, t, J = 6.4 Hz, H-10).
General method for synthesis of S-substituted 5-{[(7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-8-yl)sulfanyl]methyl}-4-phenyl-4H-1,2,4-triazol-3-yl sulfanyl derivatives (6–15). A solution of 1.04 g (2.5 mmol) of compound 5, 0.12 g (3.0 mmol) of NaOH, 3.0 mmol of the corresponding halogen derivative in 30 mL of water and 10 mL of propan-2-ol was refluxed for 20–30 minutes, cooled, the precipitate filtered, washed with H2O, and crystallized from aqueous propan-2-ol (10, 13–15) or aqueous dioxane (6–9, 11, 12).
7-Ethyl-3-methyl-8-({[5-(2-methylpropylsulfanyl)-4-phenyl-4H-1,2,4-triazol-3-yl]methyl}sulfanyl)-3,7-dihydro-1H-purine-2,6-dione(6). Calcd for C21 H25 N7 O2 S2 %: C, 53.48; H, 5.34; N, 20.79. Found %: C, 53.61; H, 5.21; N, 20.71. M.p. 195-196 °C, yield 59% (0.7 g). 1H NMR spectrum (400 MHz, DMSO-d6, δ, ppm): 11.10 (1H, s, N1H), 7.48–7.37 (5H, m, H-12-16), 4.49 (2H, s, H-11), 4.02 (2H, q, J = 7.0 Hz, H-9), 3.31 (3H, s, H-3), 2.93 (2H, d, J = 6.4 Hz, H-17), 1.85-1.75 (1H, m, H-18), 1.12 (3H, t, J = 7.1 Hz, H-10), 0.84 (6H, d, J = 6.5 Hz, H-19, 20).
8-({[5-[(Cyclohexylmethyl)sulfanyl]-4-phenyl-4H-1,2,4-triazol-3-yl]methyl}sulfanyl)-7-ethyl-3-methyl-3,7-dihydro-1H-purine-2,6-dione(7). Calcd for C24H29N7O2S2 %: C, 56.34; H, 5.71; N, 19.16. Found %: C, 56.2; H, 5.6; N, 19.07. M.p. 192-193 °C, yield 78% (1.0 g). 1H NMR (400 MHz, DMSO-d6, δ, ppm): 11.10 (1H, s, N1H), 7.48-7.37 (5H, m, H-12-16), 4.49 (2H, s, H-11), 4.02 (2H, q, J = 7.0 Hz, H-9), 3.32 (3H, s, H-3), 2.90 (2H, d, J = 6.8 Hz, H-17), 1.65-1.40 (6H, m, H-19, 20, 23), 1.11 (3H, t, J = 6.6 Hz, H-10), 1.08–1.00 (3H, m, H-18, 22), 0.88–0.79 (2H, m, H-21).
7-Ethyl-3-methyl-8-({[5-[(phenylmethyl)sulfanyl]-4-phenyl-4H-1,2,4-triazol-3-yl]methyl}sulfanyl)-3,7-dihydro-1H-purine-2,6-dione (8). Calcd for C24H23N7O2S2 %: C, 57.01; H, 4.59; N, 19.39. Found %: C, 57.16; H, 4.76; N, 19.46. M.p. 241-242 °C, yield 59 % (0.75 g). 1H NMR (400 MHz, DMSO-d6, δ, ppm): 11.10 (1H, s, N1H), 7.50–7.15 (10H, m, H-12-16, 18–22), 4.50 (2H, s, H-11), 4.29 (2H, s, H-17), 4.00 (2H, q, J = 6.1 Hz, H-9), 3.35 (3H, s, H-3), 1.16 (3H, t, J = 6.2 Hz, H-10).
7-Ethyl-3-methyl-8-({[4-phenyl-5-[(prop-2-en-1-yl)sulfanyl]-4H-1,2,4-triazol-3-yl]methyl}sulfanyl)-3,7-dihydro-1H-purine-2,6-dione(9). Calcd for C20H21N7O2S2 %: C, 52.73; H, 4.65; N, 21.52. Found %: C, 52.62; H, 4.58; N, 21.38. M.p. 211–212 °C, yield 88% (1.0 g). 1H NMR (400 MHz, DMSO-d6, δ, ppm): 11.10 (1H, s, N1H), 7.47–7.36 (5H, m, H-12-16), 5.85-5.76 (1H, m, H-18), 5.17–5.01 (2H, m, H-17), 4.49 (2H, s, H-11), 4.04 (2H, q, J = 6.8 Hz, H-9), 3.67 (2H, d, J = 7.0 Hz, H-19), 3.30 (3H, s, H-3), 1.14 (3H, t, J = 6.6 Hz, H-10).
7-Ethyl-8-({[5-[(2-hydroxyethyl)sulfanyl]-4-phenyl-4H-1,2,4-triazol-3-yl]methyl}sulfanyl)-3-methyl-3,7-dihydro-1H-purine-2,6-dione(10). Calcd for C19 H21 N7 O3 S2 %: C, 49.66; H, 4.61; N, 21.34 Found %: C, 49.79; H, 4.48; N, 21.43. M.p. 233–235 °C, yield 78% (0.9 g). 1H NMR (400 MHz, DMSO-d6, δ, ppm): 11.11 (1H, s, N1H), 7.48–7.37 (5H, m, H-12-16), 4.99 (1H, t, J = 5.6 Hz, OH), 4.50 (2H, s, H-11), 4.00 (2H, q, J = 6.9 Hz, H-9), 3.55 (2H, q, J = 5.9 Hz, H-18), 3.37–3.31 (5H, m, H-3, 17), 1.13 (3H, t, J = 6.7 Hz, H-10).
2-({3-[({7-Ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro-1H purin-8-yl}sulfanyl)methyl]-4-phenyl-4H-1,2,4-triazol-5-yl}sulfa nyl)acetamide (11). Calcd for C19 H18 N8 O3 S2 %: C, 48.29; H, 4.27; N, 23.71.Found %: C, 48.19; H, 4.2; N, 23.63. M.p. 258–259 °C, yield 76% (0.9 g). 1H NMR (400 MHz, DMSO-d6, δ, ppm): 11.10 (1H, s, N1H), 7.60 (1H, s, NH), 7.49–7.40 (5H, m, H-12-16), 7.18 (1H, s, NH), 4.50 (2H, s, H-11), 4.02 (2H, q, J = 7.2 Hz, H-9), 3.82 (2H, s, H-17), 3.32 (3H, s, H-3), 1.13 (3H, t, J = 6.9 Hz, H-10).
2-({3-[({7-Ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro-1H purin-8-yl}sulfanyl)methyl]-4-phenyl-4H-1,2,4-triazol-5-yl}sulfa nyl)acetonitrile (12). Calcd for C19 H16 N8 O2 S2 %: C, 50.21; H, 3.99; N, 24.65.Found %: C, 50.35; H, 4.11; N, 24.73. M.p. 248–249 °C, yield 53% (0.6 g). 1H NMR (400 MHz, DMSO-d6, δ, ppm): 11.10 (1H, s, N1H), 7.51-7.42 (5H, m, H-12-16), 4.53 (2H, s, H-11), 4.23 (2H, s, H-17), 4.03 (2H, q, J = 7.3 Hz, H-9), 3.33 (3H, s, H-3), 1.16 (3H, t, J = 7.2 Hz, H-10).
Methyl 2-({3-[({7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahyd ro-1H-purin-8-yl}sulfanyl)methyl]-4-phenyl-4H-1,2,4-triazol-5-yl} sulfanyl)acetate (13). Calcd for C20 H21 N7 O4 S2 %: C, 49.27; H, 4.34; N, 20.11.Found %: C, 49.14; H, 4.26; N, 20.0. M.p. 233–234 °C, yield 49% (0.6 g). 1H NMR spectrum (400 MHz, DMSO-d6, δ, ppm): 11.11 (1H, s, N1H), 7.51-7.39 (5H, m, H-12-16), 4.51 (2H, s, H-11), 4.05–3.99 (4H, m, H-9, 17), 3.58 (3H, s, H-18), 3.32 (3H, s, H-3), 1.14 (3H, t, J = 7.05 Hz, H-10).
Propyl 2-({3-[({7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro--1H-purin-8-yl}sulfanyl)methyl]-4-phenyl-4H-1,2,4-triazol-5-yl} sulfanyl)acetate (14). Calcd for C22 H25 N7 O4 S2 %: C, 51.25; H, 4.89; N, 19.02. Found %: C, 51.39; H, 4.76; N, 18.97. M.p. 170–171 °C, yield 54% (0.7 g). 1H NMR spectrum (400 MHz, DMSO-d6, δ, ppm): 11.10 (1H, s, N1H), 7.51–7.39 (5H, m, H-12-16), 4.50 (2H, s, H-11), 4.05-3.93 (6H, m, H-17, 19, 9), 3.32 (3H, s, H-3), 1.56–1.47 (2H, m, H-20), 1.14 (3H, t, J = 6.8 Hz, H-10), 0.79 (3H, t, J = 7.2 Hz, H-21). Mass spectrum (EI, 70 eV), m/z (Irel, %): 515 (M+·, 10.3), 326 (16.3), 292 (7.8), 291 (25.8), 290 (100), 248 (11.4), 239 (8.1), 232 (7.3), 230 (17.5), 226 (5.1), 225 (18.2), 202 (15.0), 190 (8.1), 117 (5.7), 99 (12.2), 77 (10.0).
Propan-2-yl 2-({3-[({7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-8-yl}sulfanyl)methyl]-4-phenyl-4H-1,2,4-triazol-5-yl}sulfanyl)acetate (15). Calcd for C22H25N7O4S2 %: C, 51.25; H, 4.89; N, 19.02. Found %: C, 51.24; H, 4.98; N, 19.08. M.p. 205–206 °C, yield 70% (0.9 g). 1H NMR spectrum (400 MHz, DMSO-d6, δ, ppm): 11.10 (1H, s, N1H), 7.51–7.39 (5H, m, H-12-16), 4.86–4.80 (1H, m, H-18), 4.51 (2H, s, H-11), 4.02 (2H, q, J = 7.3 Hz, H-9), 3.93 (2H, s, H-17), 3.32 (3H, s, H-3), 1.16-1.10 (9H, m, H-10, 19, 20).
Results and discussion
In accordance with this, we used the previously synthesized18) 2-[(7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-8-yl)sulfanyl]acetohydrazide (1) as the starting synthon for further structural modification. To construct the 1,2,4-triazole nucleus, we studied the reaction of hydrazide 1 with N-substituted isothiocyanates (Fig. 1). It was established that the direction of the reaction depends on the structure of the radical bound to the nitrogen atom.
1. Scheme of 5-({[7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-8-yl]sulfanyl}methyl)-4-R-4H-1,2,4-triazole-3-thiol synthesis ![Scheme of 5-({[7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-8-yl]sulfanyl}methyl)-4-R-4H-1,2,4-triazole-3-thiol synthesis](https://pl-master.mdcdn.cz/media/cache/media_object_image_small/media/image/78e399aff9c335e812805cf0ed0c0b58.png)
As shown in the scheme, brief boiling of hydrazide 1 with an equivalent amount of methyl or ethyl isothiocyanate in a water-propan-2-ol mixture leads to the addition of alkyl isothiocyanate with further cyclization into 3-sulfanyl-1,2,4-triazole derivatives (2, 3). In the case of phenyl isothiocyanate under similar conditions, cyclization does not occur, and only the addition product is formed – 2-(2-[(7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-8-yl)sulfanyl]acetyl)-N-phenylhydrazine-1-carbothioamide (4). Heating of compound 4 in an aqueous-alcoholic NaOH solution results in its intramolecular cyclization into 7-ethyl-3-methyl-8-{[(4-phenyl-5-sulfanyl-4H-1,2,4-triazol-3-yl)methyl]sulfanyl}-3,7-dihydro-1H-purine-2,6-dione 5. In our opinion, the nucleophilicity of the nitrogen atom in carbothioamide 4 is reduced due to the conjugation of its electron pair with the π-electrons of the benzene nucleus, which prevents the formation of the triazole nucleus in one stage.
The structure of 3-sulfanyl-1,2,4-triazoles 2, 3, 5 is unambiguously confirmed by the low-field singlets of the SH group protons at 13.55, 13.55 and 13.82 ppm, respectively. In the 1H NMR spectrum of carbothioamine 4, three single-proton singlets of NH groups of the hydrazinocarbothioamide fragment of the molecule are clearly registered. Signals of protons of other groups are registered in the corresponding field, with appropriate shape and intensity.
The presence of the SH group in the triazole nucleus provides ample opportunities for further structural modifications. We have established that the interaction of N-phenyl-substituted 3-sulfanyltriazole 5 with alkyl halides, 2-chloroethanol, and chloroacetic acid derivatives in an aqueous-alcoholic NaOH solution for 20–30 minutes leads exclusively to the formation of corresponding sulfur-substituted compounds (6-15) (Fig. 2).
2. Scheme of synthesis of S-substituted 5-{[(7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-8-yl)sulfanyl]methyl}-4-phenyl-4H-1,2,4-triazol-3-yl sulfanyl derivatives (6–15) ![Scheme of synthesis of S-substituted 5-{[(7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-8-yl)sulfanyl]methyl}-4-phenyl-4H-1,2,4-triazol-3-yl sulfanyl derivatives (6–15)](https://pl-master.mdcdn.cz/media/cache/media_object_image_small/media/image/02d8db199d85ea1e96133456cefd76f9.png)
Evidence of S-alkylation is provided by the absence of SH group proton signals at 13.82 ppm, characteristic of the initial synthon's spectrum, and by the presence of a single-proton singlet of the N1H group of the xanthine moiety at 11.10 ppm, observed for all synthesized compounds and independent of the substituent structure at positions 3 and 4 of the triazole ring. The mass spectrum (Fig. 3) of propyl 2-({3-[({7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-8-yl}sulfanyl)methyl]-4-phenyl-4H-1,2,4-triazol-5-yl}sulfanyl)acetate (14) is consistent with the proposed structure. The molecular ion peak at m/z 515 (M+·) corresponds to the calculated molecular mass and confirms the molecular composition of the synthesized compound. The fragmentation pattern observed under electron impact conditions does not contradict the proposed structure but was not interpreted mechanistically. The absence of SH proton signals in the 1H NMR spectra provides clear evidence for S-alkylation.
Fig. 3. EI mass spectrum (70 eV) of propyl 2-({3-[({7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-8-yl}sulfanyl)methyl]-4-phenyl-4H-1,2,4-triazol-5-yl}sulfanyl)acetate (14) ![Fig. 3. EI mass spectrum (70 eV) of propyl 2-({3-[({7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-8-yl}sulfanyl)methyl]-4-phenyl-4H-1,2,4-triazol-5-yl}sulfanyl)acetate (14)](https://pl-master.mdcdn.cz/media/image/3883263f90e63f196aac0a9660f313e7.png?version=1777395877)
Antibacterial and antifungal activity
Analysis of the results from studying the antimicrobial and antifungal activity of the synthesized compounds indicates that most of the studied substances showed weak activity, while selected derivatives demonstrated moderate activity against S. aureus and/or C. albicans (Tab. 1). The most active compounds were benzyl (8) - and allyl(9)sulfanyl-substituted derivatives, as well as the acetamide (11) and acetonitrile (12) derivatives, which at a concentration of 50 μg/mL inhibit the growth of Staphylococcus aureus. It should also be noted that compounds 11 and 12 exert notable antifungal effects against C. albicans, similar to the action of ketoconazole (the reference standard). Their minimum inhibitory concentration is 25 μg/mL, and the minimum fungicidal concentration is 50 μg/mL.
Tab. 1. Results of antimicrobial and antifungal activity tests for synthesized compounds 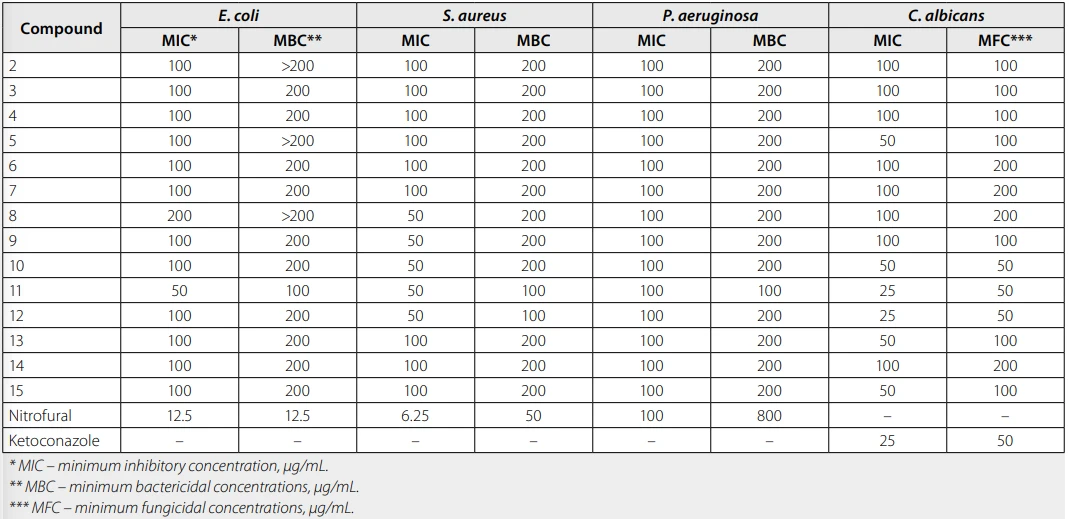
These findings extend our previous work on 2-[(7-ethyl-3-methyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-8-yl)sulfanyl]acetohydrazide derivatives, which displayed similar MIC ranges (50–100 µg/mL) against ATCC strains, where hydrazone modifications improved antifungal effects18). Recent studies on 1,3-dimethyl-3,7-dihydro-1H-purine-2,6-dione-4H-1,2,4-triazole hybrids report MIC values of 12.5–50 µg/mL against S. aureus and C. albicans, confirming that polar amide/nitrile groups at the triazole C–3 position boost antifungal selectivity (14). Compared to these analogs, compounds 11 and 12 demonstrate competitive antifungal potency within the xanthine scaffold, supporting further S-substituent optimization.
Conclusion
A series of novel 7-ethyl-3-methyl-3,7-dihydro-1H-purine-2,6-dione derivatives bearing 1,2,4-triazole substituents at the 8-position were synthesized with yields of 35-88% and characterized by 1H NMR spectroscopy, elemental analysis, and mass spectrometry (compound 14).
Antimicrobial screening against standard bacterial and fungal strains revealed that most compounds exhibited weak activity (MIC ≥ 100 μg/mL). Notably, compounds 11 and 12 displayed moderate antifungal activity against C. albicans (MIC 25 μg/mL), comparable to that of ketoconazole (MIC 25 μg/mL).
Structure-activity relationship analysis underscored the impact of S-substitution and side-chain modification on biological activity within this xanthine-triazole framework. These results highlight the promise of such hybrid molecules as a basis for further optimization and development of novel agents targeting antimicrobial resistance.
Conflict of interest: none.
Acknowledgements: The authors used ChatGPT (OpenAI) as a tool for language editing and stylistic improvement of the manuscript. All content was critically reviewed and validated by the authors. The authors take full responsibility for the accuracy, integrity, and final interpretation of the work.
Sources
1. Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet. 2022; 399(10325), 629–655.
2. GBD 2021 Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance 1990–2021: a systematic analysis with forecasts to 2050. Lancet. 2024; 404(10459), 1199–1226.
3. World Bank Group. Antimicrobial Resistance (AMR). https://www.worldbank.org/en/topic/health/brief/antimicrobial-resistance-amr (27. 09. 2024).
4. Tokuda M, Shintani M. Microbial evolution through horizontal gene transfer by mobile genetic elements. Microb. Biotechnol. 2024; 17(1), e14408.
5. Abebe GM. The Role of Bacterial Biofilm in Antibiotic Resistance and Food Contamination, International Journal of Microbiology. 2020; 1705814, 1–10.
6. da Silva Dantas A. Antimicrobial resistance. Mol. Microbiol. 2022; 117, 959–960.
7. Kok M, Maton L, van der Peet M, Hankemeier T, van Hasselt JGC. Unraveling antimicrobial resistance using metabolomics. Drug Discovery Today. 2022; 27(6), 1774–1783.
8. Jiang B, Lai Y, Xiao W, Zhong T, Liu F, Gong J, Huang J. Microbial extracellular vesicles contribute to antimicrobial resistance. PLoS Pathog. 2024; 20(5), e1012143.
9. Rathi B, Gupta S, Kumar P, Kesarwani V, Dhanda RS, Kushwaha SK, Yadav M. Anti-biofilm activity of caffeine against uropathogenic E. coli is mediated by curli biogenesis. Sci Rep. 2022; 12(1), 18903.
10. Kovalenko VM. COMPENDIUM 2019 – medicinal products. Kyiv: Morion; 2019 [in Ukrainian].
11. Li H, Kang WT, Zheng Y, He Y, Zhong R, Fang S, Wen W, Liu S, Lin S. Development of xanthone derivatives as effective broad-spectrum antimicrobials: Disrupting cell wall and inhibiting DNA synthesis. Sci Adv. 2025; 11(10), eadt4723.
12. Ivanchenko DG, Romanenko NI, Kornienko VI, Polishchuk NN, Sharapova TA. Synthesis and Properties of 8-Substituted 1-(2-Oxopropyl)Theobromine Derivatives. Chem. Nat. Compd. 2019; 55(3), 509–512.
13. Borowiecki P, Wińska P, Bretner M, Gizińska M, Koronkiewicz M, Staniszewska M. Synthesis of novel proxyphylline derivatives with dual Anti-Candida albicans and anticancer activity. Eur. J. Med. Chem. 2018; 150, 307–333.
14. Saeed S, Shahzadi I, Zahoor AF, Al-Mutairi AA, Kamal S, Faisal S, Irfan A, Al-Hussain SA, Muhammed MT, Zaki MEA. Exploring theophylline-1,2,4-triazole tethered N-phenylacetamide derivatives as antimicrobial agents: unraveling mechanisms via structure-activity relationship, in vitro validation, and in silico insights. Front. Chem. 2024; 12, 1372378.
15. Strzelecka M, Świątek P. 1,2,4-Triazoles as Important Antibacterial Agents. Pharmaceuticals. 2021; 14(3), 224.
16. Kumari M, Tahlan S, Narasimhan B, Ramasamy K, Lim SM, Shah SAA, Mani V, Kakkar S. Synthesis and biological evaluation of heterocyclic 1,2,4-triazole scaffolds as promising pharmacological agents. BMC Chemistry. 2021; 15, 5.
17. Komykhov SA, Bondarenko AA, Musatov VI, Diachkov MV, Gorobets NYu, Desenko SM. (5S,7R)-5-Aryl-7-methyl-4,5,6,7-tetrahydro-[1, 2, 4]triazolo[1,5-a]pyrimidin-7-ols as products of three-component condensation. Chem. Heterocycl. Comp. 2017; 53, 378–380.
18. Romanenko MI, Dolhikh OP, Ivanchenko DH, Aleksandrova KV, Polishchuk NM. Synthesis, physical-chemical and biological properties of derivatives of 3-methyl-7-ethylxanthinyl-8-thioacetic acid hydrazide. Curr. Issues Pharm. Med. Sci. 2018; 11(3), 264–269 [in Ukrainian].
Labels
Pharmacy Clinical pharmacology
Article was published inCzech and Slovak Pharmacy

2026 Issue 1-
All articles in this issue
- Již 155 let pomáháme rozvíjet farmacii
- Idiopathic interstitial lung diseases – treatment options
- Synthesis and antimicrobial properties of 2-[(7-ethyl-3-methylxanthin-8-yl)sulfanyl]acetohydrazide derivatives
- Drug unavailability increases off-label prescribing in the pediatric population in Slovakia
- Pharmacokinetic changes and approach to drug dosing in patients with obesity and after bariatric surgery
- Artificial intelligence in pharmacy services – practical applications
- Effective communication in the pharmacy
- Where did history of the pharmacy go?
- Pelargonium sidoides DC. vs. Pelargonium reniforme (Andrews) Curtis – active constituents and medicinal effects
- Sinusitis self-treatment
- Ocular manifestations of allergies – options for self-treatment
- Skin hydration: How to increase the water content in the skin with dermocosmetics?
- Střípky z XXVII. sympozia klinické farmacie René Macha
- Odborné akce pořádané Českou farmaceutickou společností České lékařské společnosti Jana Evangelisty Purkyně, z. s., v roce 2026
- Fixní kombinace antihypertenziv, adherence a rizika záměny v klinické praxi
- Czech and Slovak Pharmacy
- Journal archive
- Current issue
- Online only
- About the journal
Most read in this issue- Idiopathic interstitial lung diseases – treatment options
- Pharmacokinetic changes and approach to drug dosing in patients with obesity and after bariatric surgery
- Synthesis and antimicrobial properties of 2-[(7-ethyl-3-methylxanthin-8-yl)sulfanyl]acetohydrazide derivatives
- Sinusitis self-treatment
Login#ADS_BOTTOM_SCRIPTS#Forgotten passwordEnter the email address that you registered with. We will send you instructions on how to set a new password.
- Career

